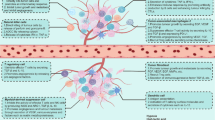
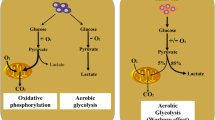
Abstract
Mounting evidence has revealed that the therapeutic efficacy of immunotherapies is restricted to a small portion of cancer patients. A deeper understanding of how metabolic reprogramming in the tumor microenvironment (TME) regulates immunity remains a major challenge to tumor eradication. It has been suggested that metabolic reprogramming in the TME may affect metabolism in immune cells and subsequently suppress immune function. Tumor cells compete with infiltrating immune cells for nutrients and metabolites. Notably, the immunosuppressive TME is characterized by catabolic and anabolic processes that are critical for immune cell function, and elevated inhibitory signals may favor cancer immune evasion. The major energy sources that supply different immune cell subtypes also undergo reprogramming. We herein summarize the metabolic remodeling in tumor cells and different immune cell subtypes and the latest advances underlying the use of metabolic checkpoints in antitumor immunotherapies. In this context, targeting both tumor and immune cell metabolic reprogramming may enhance therapeutic efficacy.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Abu-Elheiga, L., Brinkley, W.R., Zhong, L., Chirala, S.S., Woldegiorgis, G., and Wakil, S.J. (2000). The subcellular localization of acetyl-coa carboxylase 2. Proc Natl Acad Sci USA 97, 1444–1449.
Ahluwalia, G.S., Grem, J.L., Hao, Z., and Cooney, D.A. (1990). Metabolism and action of amino acid analog anti-cancer agents. Pharmacol Ther 46, 243–271.
Al-Khami, A.A., Rodriguez, P.C., and Ochoa, A.C. (2016). Metabolic reprogramming of myeloid-derived suppressor cells (MDSC) in cancer. Oncoimmunology 5, e1200771.
Bahrambeigi, S., and Shafiei-Irannejad, V. (2020). Immune-mediated antitumor effects of metformin; targeting metabolic reprogramming of T cells as a new possible mechanism for anti-cancer effects of metformin. Biochem Pharmacol 174, 113787.
Bensinger, S.J., Bradley, M.N., Joseph, S.B., Zelcer, N., Janssen, E.M., Hausner, M.A., Shih, R., Parks, J.S., Edwards, P.A., Jamieson, B.D., et al. (2008). LXR signaling couples sterol metabolism to proliferation in the acquired immune response. Cell 134, 97–111.
Berod, L., Friedrich, C., Nandan, A., Freitag, J., Hagemann, S., Harmrolfs, K., Sandouk, A., Hesse, C., Castro, C.N., Bähre, H., et al. (2014). De novo fatty acid synthesis controls the fate between regulatory t and t helper 17 cells. Nat Med 20, 1327–1333.
Bola, B.M., Chadwick, A.L., Michopoulos, F., Blount, K.G., Telfer, B.A., Williams, K.J., Smith, P.D., Critchlow, S.E., and Stratford, I.J. (2014). Inhibition of monocarboxylate transporter-1 (MCT1) by AZD3965 enhances radiosensitivity by reducing lactate transport. Mol Cancer Ther 13, 2805–2816.
Brooks, G.A. (2009). Cell-cell and intracellular lactate shuttles. J Physiol 587, 5591–5600.
Byersdorfer, C.A., Tkachev, V., Opipari, A.W., Goodell, S., Swanson, J., Sandquist, S., Glick, G.D., and Ferrara, J.L.M. (2013). Effector T cells require fatty acid metabolism during murine graft-versus-host disease. Blood 122, 3230–3237.
Calcinotto, A., Filipazzi, P., Grioni, M., Iero, M., De Milito, A., Ricupito, A., Cova, A., Canese, R., Jachetti, E., Rossetti, M., et al. (2012). Modulation of microenvironment acidity reverses anergy in human and murine tumor-infiltrating T lymphocytes. Cancer Res 72, 2746–2756.
Cha, J.H., Yang, W.H., Xia, W., Wei, Y., Chan, L.C., Lim, S.O., Li, C.W., Kim, T., Chang, S.S., Lee, H.H., et al. (2018). Metformin promotes antitumor immunity via endoplasmic-reticulum-associated degradation of PD-L1. Mol Cell 71, 606–620.e7.
Chakraborty, P., Vaena, S.G., Thyagarajan, K., Chatterjee, S., Al-Khami, A., Selvam, S.P., Nguyen, H., Kang, I., Wyatt, M.W., Baliga, U., et al. (2019). Pro-survival lipid sphingosine-1-phosphate metabolically programs T cells to limit anti-tumor activity. Cell Rep 28, 1879–1893.e7.
Chang, C.H., Qiu, J., O’Sullivan, D., Buck, M.D., Noguchi, T., Curtis, J.D., Chen, Q., Gindin, M., Gubin, M.M., van der Windt, G.J.W., et al. (2015). Metabolic competition in the tumor microenvironment is a driver of cancer progression. Cell 162, 1229–1241.
Chatterjee, S., Chakraborty, P., Daenthanasanmak, A., Iamsawat, S., Andrejeva, G., Luevano, L.A., Wolf, M., Baliga, U., Krieg, C., Beeson, C.C., et al. (2019). Targeting PIM kinase with PD1 inhibition improves immunotherapeutic antitumor T-cell response. Clin Cancer Res 25, 1036–1049.
Chi, H. (2012). Regulation and function of mTOR signalling in T cell fate decisions. Nat Rev Immunol 12, 325–338.
Chirasani, S.R., Leukel, P., Gottfried, E., Hochrein, J., Stadler, K., Neumann, B., Oefner, P.J., Gronwald, W., Bogdahn, U., Hau, P., et al. (2013). Diclofenac inhibits lactate formation and efficiently counteracts local immune suppression in a murine glioma model. Int J Cancer 132, 843–853.
Dang, C.V., Le, A., and Gao, P. (2009). MYC-induced cancer cell energy metabolism and therapeutic opportunities. Clin Cancer Res 15, 6479–6483.
DeBerardinis, R.J., Mancuso, A., Daikhin, E., Nissim, I., Yudkoff, M., Wehrli, S., and Thompson, C.B. (2007). Beyond aerobic glycolysis: Transformed cells can engage in glutamine metabolism that exceeds the requirement for protein and nucleotide synthesis. Proc Natl Acad Sci USA 104, 19345–19350.
Durham, N.M., Nirschl, C.J., Jackson, C.M., Elias, J., Kochel, C.M., Anders, R.A., and Drake, C.G. (2014). Lymphocyte activation gene 3 (LAG-3) modulates the ability of CD4 T-cells to be suppressed in vivo. PLoS ONE 9, e109080.
Ecker, C., Guo, L., Voicu, S., Gil-de-Gómez, L., Medvec, A., Cortina, L., Pajda, J., Andolina, M., Torres-Castillo, M., Donato, J.L., et al. (2018). Differential reliance on lipid metabolism as a salvage pathway underlies functional differences of T cell subsets in poor nutrient environments. Cell Rep 23, 741–755.
Eleftheriadis, T., Pissas, G., Karioti, A., Antoniadi, G., Antoniadis, N., Liakopoulos, V., and Stefanidis, I. (2013). Dichloroacetate at therapeutic concentration alters glucose metabolism and induces regulatory T-cell differentiation in alloreactive human lymphocytes. J Basic Clin Physiol Pharmacol 24, 271–276.
Fantin, V.R., St-Pierre, J., and Leder, P. (2006). Attenuation of LDH-A expression uncovers a link between glycolysis, mitochondrial physiology, and tumor maintenance. Cancer Cell 9, 425–434.
Franchi, L., Monteleone, I., Hao, L.Y., Spahr, M.A., Zhao, W., Liu, X., Demock, K., Kulkarni, A., Lesch, C.A., Sanchez, B., et al. (2017). Inhibiting oxidative phosphorylation in vivo restrains TH17 effector responses and ameliorates murine colitis. J Immunol 198, 2735–2746.
Fredholm, B.B., IJzerman, A.P., Jacobson, K.A., Klotz, K.N., Linden, J. (2001). International union of pharmacology. XXV. Nomenclature and classification of adenosine receptors. Pharmacol Rev 53: 527–552.
Ge, J., Wang, J., Wang, H., Jiang, X., Liao, Q., Gong, Q., Mo, Y., Li, X., Li, G., Xiong, W., et al. (2020). The BRAF V600E mutation is a predictor of the effect of radioiodine therapy in papillary thyroid cancer. J Cancer 11, 932–939.
Gerriets, V.A., Kishton, R.J., Johnson, M.O., Cohen, S., Siska, P.J., Nichols, A.G., Warmoes, M.O., de Cubas, A.A., MacIver, N.J., Locasale, J.W., et al. (2016). Foxp3 and Toll-like receptor signaling balance Treg cell anabolic metabolism for suppression. Nat Immunol 17, 1459–1466.
Goetze, K., Walenta, S., Ksiazkiewicz, M., Kunz-Schughart, L.A., and Mueller-Klieser, W. (2011). Lactate enhances motility of tumor cells and inhibits monocyte migration and cytokine release. Int J Oncol 39, 453–463.
Görgün, G., Calabrese, E., Soydan, E., Hideshima, T., Perrone, G., Bandi, M., Cirstea, D., Santo, L., Hu, Y., Tai, Y.T., et al. (2010). Immunomodulatory effects of lenalidomide and pomalidomide on interaction of tumor and bone marrow accessory cells in multiple myeloma. Blood 116, 3227–3237.
Gottfried, E., Kunz-Schughart, L.A., Ebner, S., Mueller-Klieser, W., Hoves, S., Andreesen, R., Mackensen, A., and Kreutz, M. (2006). Tumor-derived lactic acid modulates dendritic cell activation and antigen expression. Blood 107, 2013–2021.
Guilbaud, E., Gautier, E.L., and Yvan-Charvet, L. (2019). Macrophage origin, metabolic reprogramming and IL-1 signaling: Promises and pitfalls in lung cancer. Cancers 11, 298.
Hameed, S., Bhattarai, P., and Dai, Z. (2018). Nanotherapeutic approaches targeting angiogenesis and immune dysfunction in tumor microenvironment. Sci China Life Sci 61, 380–391.
Harun, N.O., Zou, J., Zhang, Y.A., Nie, P., and Secombes, C.J. (2008). The biological effects of rainbow trout (Oncorhynchus mykiss) recombinant interleukin-8. Dev Comp Immunol 32, 673–681.
Hassanein, M., Qian, J., Hoeksema, M.D., Wang, J., Jacobovitz, M., Ji, X., Harris, F.T., Harris, B.K., Boyd, K.L., Chen, H., et al. (2015). Targeting SLC1a5-mediated glutamine dependence in non-small cell lung cancer. Int J Cancer 137, 1587–1597.
Ho, P.C., Bihuniak, J.D., Macintyre, A.N., Staron, M., Liu, X., Amezquita, R., Tsui, Y.C., Cui, G., Micevic, G., Perales, J.C., et al. (2015). Phosphoenolpyruvate is a metabolic checkpoint of anti-tumor T cell responses. Cell 162, 1217–1228.
Husain, Z., Huang, Y., Seth, P., and Sukhatme, V.P. (2013). Tumor-derived lactate modifies antitumor immune response: Effect on myeloid-derived suppressor cells and NK cells. J Immunol 191, 1486–1495.
Imhann, F., Bonder, M.J., Vich Vila, A., Fu, J., Mujagic, Z., Vork, L., Tigchelaar, E.F., Jankipersadsing, S.A., Cenit, M.C., Harmsen, H.J.M., et al. (2016). Proton pump inhibitors affect the gut microbiome. Gut 65, 740–748.
Jiang, Z., Liu, Z., Li, M., Chen, C., and Wang, X. (2019). Increased glycolysis correlates with elevated immune activity in tumor immune microenvironment. EBioMedicine 42, 431–442.
Jiralerspong, S., Palla, S.L., Giordano, S.H., Meric-Bernstam, F., Liedtke, C., Barnett, C.M., Hsu, L., Hung, M.C., Hortobagyi, G.N., and Gonzalez-Angulo, A.M. (2009). Metformin and pathologic complete responses to neoadjuvant chemotherapy in diabetic patients with breast cancer. J Clin Oncol 27, 3297–3302.
Johnson, M.O., Wolf, M.M., Madden, M.Z., Andrejeva, G., Sugiura, A., Contreras, D.C., Maseda, D., Liberti, M.V., Paz, K., Kishton, R.J., et al. (2018). Distinct regulation of Th17 and Th1 cell differentiation by glutaminase-dependent metabolism. Cell 175, 1780–1795.e19.
Kelly, B., and O’Neill, L.A.J. (2015). Metabolic reprogramming in macrophages and dendritic cells in innate immunity. Cell Res 25, 771–784.
Kim, J., Tchernyshyov, I., Semenza, G.L., and Dang, C.V. (2006). Hif-1-mediated expression of pyruvate dehydrogenase kinase: A metabolic switch required for cellular adaptation to hypoxia. Cell Metab 3, 177–185.
Kim, J., Gao, P., Liu, Y.C., Semenza, G.L., and Dang, C.V. (2007). Hypoxia-inducible factor 1 and dysregulated c-Myc cooperatively induce vascular endothelial growth factor and metabolic switches hexokinase 2 and pyruvate dehydrogenase kinase 1. Mol Cell Biol 27, 7381–7393.
Kishton, R.J., Sukumar, M., and Restifo, N.P. (2017). Metabolic regulation of T cell longevity and function in tumor immunotherapy. Cell Metab 26, 94–109.
Kolev, M., Dimeloe, S., Le Friec, G., Navarini, A., Arbore, G., Povoleri, G. A., Fischer, M., Belle, R., Loeliger, J., Develioglu, L., et al. (2015). Complement regulates nutrient influx and metabolic reprogramming during Th1 cell responses. Immunity 42, 1033–1047.
Kouidhi, S., Elgaaied, A.B., and Chouaib, S. (2017). Impact ofmetabolism on T-cell differentiation and function and cross talk with tumor microenvironment. Front Immunol 8, 270.
Kunisada, Y., Eikawa, S., Tomonobu, N., Domae, S., Uehara, T., Hori, S., Furusawa, Y., Hase, K., Sasaki, A., and Udono, H. (2017). Attenuation of CD4+CD25+ regulatory T cells in the tumor microenvironment by metformin, a type 2 diabetes drug. EBioMedicine 25, 154–164.
Le, A., Cooper, C.R., Gouw, A.M., Dinavahi, R., Maitra, A., Deck, L.M., Royer, R.E., Vander Jagt, D.L., Semenza, G.L., and Dang, C.V. (2010). Inhibition of lactate dehydrogenase a induces oxidative stress and inhibits tumor progression. Proc Natl Acad Sci USA 107, 2037–2042.
Lee, S.Y., Lee, S.H., Yang, E.J., Kim, E.K., Kim, J.K., Shin, D.Y., and Cho, M.L. (2015). Metformin ameliorates inflammatory bowel disease by suppression of the STAT3 signaling pathway and regulation of the between Th17/Treg balance. PLoS ONE 10, e0135858.
Li, B., and Simon, M.C. (2013). Molecular pathways: Targeting Myc-induced metabolic reprogramming and oncogenic stress in cancer. Clin Cancer Res 19, 5835–5841.
Li, J., and Dong, C. (2018). Nobel goes to immune checkpoint—Innovative cancer treatment by immunotherapy. Sci China Life Sci 61, 1445–1450.
Li, L., Liu, X., Sanders, K.L., Edwards, J.L., Ye, J., Si, F., Gao, A., Huang, L., Hsueh, E.C., Ford, D.A., et al. (2019). TLR8-mediated metabolic control of human Treg function: A mechanistic target for cancer immunotherapy. Cell Metab 29, 103–123.e5.
Lin, C., and Zhang, J. (2019). Chimeric antigen receptor engineered innate immune cells in cancer immunotherapy. Sci China Life Sci 62, 633–639.
Liu, D., Chang, C., Lu, N., Wang, X., Lu, Q., Ren, X., Ren, P., Zhao, D., Wang, L., Zhu, Y., et al. (2017). Comprehensive proteomics analysis reveals metabolic reprogramming of tumor-associated macrophages stimulated by the tumor microenvironment. J Proteome Res 16, 288–297.
Luo, X., Cheng, C., Tan, Z., Li, N., Tang, M., Yang, L., and Cao, Y. (2017). Emerging roles of lipid metabolism in cancer metastasis. Mol Cancer 16, 76.
Ma, X., Bi, E., Lu, Y., Su, P., Huang, C., Liu, L., Wang, Q., Yang, M., Kalady, M.F., Qian, J., et al. (2019). Cholesterol induces CD8+ T cell exhaustion in the tumor microenvironment. Cell Metab 30, 143–156.e5.
Macintyre, A.N., Gerriets, V.A., Nichols, A.G., Michalek, R.D., Rudolph, M.C., Deoliveira, D., Anderson, S.M., Abel, E.D., Chen, B.J., Hale, L. P., et al. (2014). The glucose transporter Glut1 is selectively essential for CD4 T cell activation and effector function. Cell Metab 20, 61–72.
MacIver, N.J., Michalek, R.D., and Rathmell, J.C. (2013). Metabolic regulation of T lymphocytes. Annu Rev Immunol 31, 259–283.
McGranahan, N., Rosenthal, R., Hiley, C.T., Rowan, A.J., Watkins, T.B.K., Wilson, G.A., Birkbak, N.J., Veeriah, S., Van Loo, P., Herrero, J., et al. (2017). Allele-specific HLA loss and immune escape in lung cancer evolution. Cell 171, 1259–1271.e11.
Mineharu, Y., Kamran, N., Lowenstein, P.R., and Castro, M.G. (2014). Blockade of mTOR signaling via rapamycin combined with immunotherapy augments antiglioma cytotoxic and memory T-cell functions. Mol Cancer Ther 13, 3024–3036.
Mondanelli, G., Ugel, S., Grohmann, U., and Bronte, V. (2017). The immune regulation in cancer by the amino acid metabolizing enzymes ARG and IDO. Curr Opin Pharmacol 35, 30–39.
Morello, S., Pinto, A., Blandizzi, C., and Antonioli, L. (2016). Myeloid cells in the tumor microenvironment: Role of adenosine. Oncoimmunology 5, e1108515.
Motz, G.T., and Coukos, G. (2013). Deciphering and reversing tumor immune suppression. Immunity 39, 61–73.
Nabe, S., Yamada, T., Suzuki, J., Toriyama, K., Yasuoka, T., Kuwahara, M., Shiraishi, A., Takenaka, K., Yasukawa, M., and Yamashita, M. (2018). Reinforce the antitumor activity of CD8+ T cells via glutamine restriction. Cancer Sci 109, 3737–3750.
Nayak-Kapoor, A., Hao, Z., Sadek, R., Dobbins, R., Marshall, L., Vahanian, N.N., Jay Ramsey, W., Kennedy, E., Mautino, M.R., Link, C.J., et al. (2018). Phase Ia study of the indoleamine 2,3-dioxygenase 1 (IDO1) inhibitor navoximod (GDC-0919) in patients with recurrent advanced solid tumors. J Immunother Cancer 6, 61.
Okkenhaug, K., Graupera, M., and Vanhaesebroeck, B. (2016). Targeting PI3K in cancer: Impact on tumor cells, their protective stroma, angiogenesis, and immunotherapy. Cancer Discov 6, 1090–1105.
Orillion, A., Damayanti, N.P., Shen, L., Adelaiye-Ogala, R., Affronti, H., Elbanna, M., Chintala, S., Ciesielski, M., Fontana, L., Kao, C., et al. (2018). Dietary protein restriction reprograms tumor-associated macrophages and enhances immunotherapy. Clin Cancer Res 24, 6383–6395.
Otvos, B., Silver, D.J., Mulkearns-Hubert, E.E., Alvarado, A.G., Turaga, S. M., Sorensen, M.D., Rayman, P., Flavahan, W.A., Hale, J.S., Stoltz, K., et al. (2016). Cancer stem cell-secreted macrophage migration inhibitory factor stimulates myeloid derived suppressor cell function and facilitates glioblastoma immune evasion. Stem Cells 34, 2026–2039.
Pantziarka, P., Sukhatme, V., Bouche, G., Melhuis, L., and Sukhatme, V.P. (2016). Repurposing drugs in oncology (ReDO)—Diclofenac as an anti-cancer agent. Ecancermedicalscience 10, 610.
Papaioannou, N.E., Beniata, O.V., Vitsos, P., Tsitsilonis, O., and Samara, P. (2016). Harnessing the immune system to improve cancer therapy. Ann Transl Med 4, 261.
Parra-Bonilla, G., Alvarez, D.F., Al-Mehdi, A.B., Alexeyev, M., and Stevens, T. (2010). Critical role for lactate dehydrogenase a in aerobic glycolysis that sustains pulmonary microvascular endothelial cell proliferation. Am J Physiol Lung Cell Mol Physiol 299, L513–L522.
Patsoukis, N., Bardhan, K., Chatterjee, P., Sari, D., Liu, B., Bell, L.N., Karoly, E.D., Freeman, G.J., Petkova, V., Seth, P., et al. (2015). PD-1 alters T-cell metabolic reprogramming by inhibiting glycolysis and promoting lipolysis and fatty acid oxidation. Nat Commun 6, 6692.
Pearce, E.L., Poffenberger, M.C., Chang, C.H., and Jones, R.G. (2013). Fueling immunity: Insights into metabolism and lymphocyte function. Science 342, 1242454.
Peng, M., Mo, Y., Wang, Y., Wu, P., Zhang, Y., Xiong, F., Guo, C., Wu, X., Li, Y., Li, X., et al. (2019). Neoantigen vaccine: An emerging tumor immunotherapy. Mol Cancer 18, 128.
Pinheiro, C., Reis, R.M., Ricardo, S., Longatto-Filho, A., Schmitt, F., and Baltazar, F. (2010). Expression of monocarboxylate transporters 1, 2, and 4 in human tumours and their association with CD147 and CD44. J Biomed Biotech 2010, 1–7.
Previte, D.M., Martins, C.P., O’Connor, E.C., Marre, M.L., Coudriet, G.M., Beck, N.W., Menk, A.V., Wright, R.H., Tse, H.M., Delgoffe, G.M., et al. (2019). Lymphocyte activation gene-3 maintains mitochondrial and metabolic quiescence in naive CD4+ T cells. Cell Rep 27, 129–141.e4.
Püschel, F., Favaro, F., Redondo-Pedraza, J., Lucendo, E., Iurlaro, R., Marchetti, S., Majem, B., Eldering, E., Nadal, E., Ricci, J.E., et al. (2020). Starvation and antimetabolic therapy promote cytokine release and recruitment of immune cells. Proc Natl Acad Sci USA 117, 9932–9941.
Raychaudhuri, B., Rayman, P., Ireland, J., Ko, J., Rini, B., Borden, E.C., Garcia, J., Vogelbaum, M.A., and Finke, J. (2011). Myeloid-derived suppressor cell accumulation and function in patients with newly diagnosed glioblastoma. Neuro Oncol 13, 591–599.
Reilley, M.J., Bailey, A., Subbiah, V., Janku, F., Naing, A., Falchook, G., Karp, D., Piha-Paul, S., Tsimberidou, A., Fu, S., et al. (2017). Phase I clinical trial of combination imatinib and ipilimumab in patients with advanced malignancies. J Immunother Cancer 5, 35.
Ren, D., Hua, Y., Yu, B., Ye, X., He, Z., Li, C., Wang, J., Mo, Y., Wei, X., Chen, Y., et al. (2020). Predictive biomarkers and mechanisms underlying resistance to PD1/PD-L1 blockade cancer immunotherapy. Mol Cancer 19, 19.
Rodriguez, P.C., Quiceno, D.G., Zabaleta, J., Ortiz, B., Zea, A.H., Piazuelo, M.B., Delgado, A., Correa, P., Brayer, J., Sotomayor, E.M., et al. (2004). Arginase i production in the tumor microenvironment by mature myeloid cells inhibits T-cell receptor expression and antigen-specific T-cell responses. Cancer Res 64, 5839–5849.
Rosenberg, S.A., Yang, J.C., Sherry, R.M., Kammula, U.S., Hughes, M.S., Phan, G.Q., Citrin, D.E., Restifo, N.P., Robbins, P.F., Wunderlich, J.R., et al. (2011). Durable complete responses in heavily pretreated patients with metastatic melanoma using T-cell transfer immunotherapy. Clin Cancer Res 17, 4550–4557.
Sancho, P., Barneda, D., and Heeschen, C. (2016). Hallmarks of cancer stem cell metabolism. Br J Cancer 114, 1305–1312.
Scharping, N.E., and Delgoffe, G.M. (2016). Tumor microenvironment metabolism: A new checkpoint for anti-tumor immunity. Vaccines 4, 46.
Scheffel, M.J., Scurti, G., Wyatt, M.M., Garrett-Mayer, E., Paulos, C.M., Nishimura, M.I., and Voelkel-Johnson, C. (2018). N-acetyl cysteine protects anti-melanoma cytotoxic T cells from exhaustion induced by rapid expansion via the downmodulation of Foxo1 in an Akt-dependent manner. Cancer Immunol Immunother 67, 691–702.
Schmidt, K.M., Hellerbrand, C., Ruemmele, P., Michalski, C.W., Kong, B., Kroemer, A., Hackl, C., Schlitt, H.J., Geissler, E.K., and Lang, S.A. (2017). Inhibition of mTORC2 component RICTOR impairs tumor growth in pancreatic cancer models. Oncotarget 8, 24491–24505.
Shang, S., Ji, X., Zhang, L., Chen, J., Li, C., Shi, R., Xiang, W., Kang, X., Zhang, D., Yang, F., et al. (2019). Macrophage ABHD5 suppresses NF-κB-dependent matrix metalloproteinase expression and cancer metastasis. Cancer Res 79, 5513–5526.
Simon-Molas, H., Arnedo-Pac, C., Fontova, P., Vidal-Alabró, A., Castaño, E., Rodríguez-García, A., Navarro-Sabaté, À., Lloberas, N., Manzano, A., and Bartrons, R. (2018). PI3K-Akt signaling controls PFKFB3 expression during human T-lymphocyte activation. Mol Cell Biochem 448, 187–197.
Sinclair, L.V., Rolf, J., Emslie, E., Shi, Y.B., Taylor, P.M., and Cantrell, D. A. (2013). Control of amino-acid transport by antigen receptors coordinates the metabolic reprogramming essential for T cell differentiation. Nat Immunol 14, 500–508.
Song, M., Sandoval, T.A., Chae, C.S., Chopra, S., Tan, C., Rutkowski, M. R., Raundhal, M., Chaurio, R.A., Payne, K.K., Konrad, C., et al. (2018a). IRE1a-XBP1 controls T cell function in ovarian cancer by regulating mitochondrial activity. Nature 562, 423–428.
Song, Y.J., Kim, A., Kim, G.T., Yu, H.Y., Lee, E.S., Park, M.J., Kim, Y.J., Shim, S.M., and Park, T.S. (2018b). Inhibition oflactate dehydrogenase A suppresses inflammatory response in RAW 264.7 macrophages. Mol Med Report 19, 629–637.
Souto-Carneiro, M.M., Klika, K.D., Abreu, M.T., Meyer, A.P., Saffrich, R., Sandhoff, R., Jennemann, R., Kraus, F.V., Tykocinski, L.O., Eckstein, V., et al. (2020). Proinflammatory profile of autoimmune CD8+ T cells relies on increased LDHA activity and aerobic glycolysis. Arthritis Rheumatol doi: https://doi.org/10.1002/art.41420.
Staron, M.M., Gray, S.M., Marshall, H.D., Parish, I.A., Chen, J.H., Perry, C.J., Cui, G., Li, M.O., and Kaech, S.M. (2014). The transcription factor FoxO1 sustains expression of the inhibitory receptor PD-1 and survival of antiviral CD8+ T cells during chronic infection. Immunity 41, 802–814.
Starska, K., Forma, E., Józwiak, P., Brys, M., Lewy-Trenda, I., Brzeziñska-Blaszczyk, E., and Krzeslak, A. (2015). Gene and protein expression of glucose transporter 1 and glucose transporter 3 in human laryngeal cancer—The relationship with regulatory hypoxia-inducible factor-1α expression, tumor invasiveness, and patient prognosis. Tumor Biol 36, 2309–2321.
Sukumar, M., Liu, J., Ji, Y., Subramanian, M., Crompton, J.G., Yu, Z., Roychoudhuri, R., Palmer, D.C., Muranski, P., Karoly, E.D., et al. (2013). Inhibiting glycolytic metabolism enhances CD8+ T cell memory and antitumor function. J Clin Invest 123, 4479–4488.
Sun, Y., Tian, T., Gao, J., Liu, X., Hou, H., Cao, R., Li, B., Quan, M., and Guo, L. (2016). Metformin ameliorates the development of experimental autoimmune encephalomyelitis by regulating T helper 17 and regulatory T cells in mice. J Neuroimmunol 292, 58–67.
Tang, T., Yang, L., Cao, Y., Wang, M., Zhang, S., Gong, Z., Xiong, F., He, Y., Zhou, Y., Liao, Q., et al. (2020). LncRNA AATBC regulates pinin to promote metastasis in nasopharyngeal carcinoma. Mol Oncol doi: https://doi.org/10.1002/1878-0261.12703.
Tatematsu, K., Kuroda, S. (2018). Automated single-cell analysis and isolation system: A paradigm shift in cell screening methods for biomedicines. In: Gu, J., Wang, X., eds. Single Cell Biomedicine. Advances in Experimental Medicine and Biology. Singapore: Springer. 1068, 7–17.
Tennant, D.A., Durán, R.V., and Gottlieb, E. (2010). Targeting metabolic transformation for cancer therapy. Nat Rev Cancer 10, 267–277.
Thorpe, L.M., Yuzugullu, H., and Zhao, J.J. (2015). PI3K in cancer: Divergent roles of isoforms, modes of activation and therapeutic targeting. Nat Rev Cancer 15, 7–24.
Thurnher, M., and Gruenbacher, G. (2015). T lymphocyte regulation by mevalonate metabolism. Sci Signal 8, re4.
Ti, D., Niu, Y., Wu, Z., Fu, X., and Han, W. (2018). Genetic engineering of T cells with chimeric antigen receptors for hematological malignancy immunotherapy. Sci China Life Sci 61, 1320–1332.
Tian, M., Shi, Y., Liu, W., and Fan, J. (2019). Immunotherapy of hepatocellular carcinoma: strategies for combinatorial intervention. Sci China Life Sci 62, 1138–1143.
Troncone, M., Cargnelli, S.M., Villani, L.A., Isfahanian, N., Broadfield, L. A., Zychla, L., Wright, J., Pond, G., Steinberg, G.R., and Tsakiridis, T. (2017). Targeting metabolism and AMP-activated kinase with metformin to sensitize non-small cell lung cancer (NSCLC) to cytotoxic therapy: Translational biology and rationale for current clinical trials. Oncotarget 8, 57733–57754.
van der Mijn, J.C., Panka, D.J., Geissler, A.K., Verheul, H.M., and Mier, J. W. (2016). Novel drugs that target the metabolic reprogramming in renal cell cancer. Cancer Metab 4, 14.
Vella, S., Conti, M., Tasso, R., Cancedda, R., and Pagano, A. (2012). Dichloroacetate inhibits neuroblastoma growth by specifically acting against malignant undifferentiated cells. Int J Cancer 130, 1484–1493.
Wang, D., Tang, L., Wu, Y., Fan, C., Zhang, S., Xiang, B., Zhou, M., Li, X., Li, Y., Li, G., et al. (2020a). Abnormal X chromosome inactivation and tumor development. Cell Mol Life Sci 77, 2949–2958.
Wang, D., Zeng, Z., Zhang, S., Xiong, F., He, B., Wu, Y., Li, W., Tang, L., Wei, F., Xiang, B., et al. (2020b). Epstein-Barr virus-encoded miR-BART6-3p inhibits cancer cell proliferation through the LOC553103-STMN1 axis. FASEB J 34, 8012–8027.
Wang, H., Franco, F., Tsui, Y.C., Xie, X., Trefny, M.P., Zappasodi, R., Mohmood, S.R., Fernández-García, J., Tsai, C.H., Schulze, I., et al. (2020c). CD36-mediated metabolic adaptation supports regulatory T cell survival and function in tumors. Nat Immunol 21, 298–308.
Wang, R., Dillon, C.P., Shi, L.Z., Milasta, S., Carter, R., Finkelstein, D., McCormick, L.L., Fitzgerald, P., Chi, H., Munger, J., et al. (2011). The transcription factor Myc controls metabolic reprogramming upon T lymphocyte activation. Immunity 35, 871–882.
Wang, T., Liu, G., and Wang, R. (2014). The intercellular metabolic interplay between tumor and immune cells. Front Immunol 5, 358.
Wang, Y., Wu, Y., Wang, Y., Fu, A., Gong, L., Li, W., and Li, Y. (2017). Bacillus amyloliquefaciens SC06 alleviates the oxidative stress of IPEC-1 via modulating Nrf2/Keap1 signaling pathway and decreasing ROS production. Appl Microbiol Biotechnol 101, 3015–3026.
Warburg, O. (1956). On the origin of cancer cells. Science 123, 309–314.
Wherry, E.J., Ha, S.J., Kaech, S.M., Haining, W.N., Sarkar, S., Kalia, V., Subramaniam, S., Blattman, J.N., Barber, D.L., and Ahmed, R. (2007). Molecular signature of CD8+ T cell exhaustion during chronic viral infection. Immunity 27, 670–684.
Williams, N.C., and O’Neill, L.A.J. (2018). A role for the Krebs cycle intermediate citrate in metabolic reprogramming in innate immunity and inflammation. Front Immunol 9, 141.
Won, W.J., Deshane, J.S., Leavenworth, J.W., Oliva, C.R., and Griguer, C. E. (2019). Metabolic and functional reprogramming of myeloid-derived suppressor cells and their therapeutic control in glioblastoma. Cell Stress 3, 47–65.
Wu, P., Mo, Y., Peng, M., Tang, T., Zhong, Y., Deng, X., Xiong, F., Guo, C., Wu, X., Li, Y., et al. (2020). Emerging role of tumor-related functional peptides encoded by lncRNA and circRNA. Mol Cancer 19, 22.
Yan, S., Zhang, Y., and Sun, B. (2019). The function and potential drug targets of tumour-associated Tregs for cancer immunotherapy. Sci China Life Sci 62, 179–186.
Yang, W., Bai, Y., Xiong, Y., Zhang, J., Chen, S., Zheng, X., Meng, X., Li, L., Wang, J., Xu, C., et al. (2016). Potentiating the antitumour response of CD8+ T cells by modulating cholesterol metabolism. Nature 531, 651–655.
Yu, H., Pan, J., Guo, Z., Yang, C., and Mao, L. (2019). CART cell therapy for prostate cancer: Status and promise. Onco Targets Ther Volume 12, 391–395.
Zhang, L., Sun, S., Wang, Y., Mo, Y., Xiong, F., Zhang, S., Zeng, Z., Xiong, W., Li, G., Chen, H., et al. (2020). Gossypol induces apoptosis of multiple myeloma cells through the Jun-Jnk pathway. Am J Cancer Res 10, 870–883.
Zhang, Q., Wang, H., Mao, C., Sun, M., Dominah, G., Chen, L., and Zhuang, Z. (2018). Fatty acid oxidation contributes to IL-1β secretion in M2 macrophages and promotes macrophage-mediated tumor cell migration. Mol Immunol 94, 27–35.
Zhang, Y., Kurupati, R., Liu, L., Zhou, X.Y., Zhang, G., Hudaihed, A., Filisio, F., Giles-Davis, W., Xu, X., Karakousis, G.C., et al. (2017). Enhancing CD8+ T cell fatty acid catabolism within a metabolically challenging tumor microenvironment increases the efficacy of melanoma immunotherapy. Cancer Cell 32, 377–391.e9.
Zhao, J., Guo, C., Xiong, F., Yu, J., Ge, J., Wang, H., Liao, Q., Zhou, Y., Gong, Q., Xiang, B., et al. (2020). Single cell RNA-seq reveals the landscape of tumor and infiltrating immune cells in nasopharyngeal carcinoma. Cancer Lett 477, 131–143.
Zhao, L., Ji, G., Le, X., Wang, C., Xu, L., Feng, M., Zhang, Y., Yang, H., Xuan, Y., Yang, Y., et al. (2017). Long noncoding RNA LINC00092 acts in cancer-associated fibroblasts to drive glycolysis and progression of ovarian cancer. Cancer Res 77, 1369–1382.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (81903138, 81972776, 81803025, 81772928, 81702907, 81772901, 81672993, 81672683), and the Natural Science Foundation of Hunan Province (2019JJ50778, 2018SK21210, 2018SK21211, 2018JJ3704, 2018JJ3815).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Compliance and ethics
The author(s) declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Fan, C., Zhang, S., Gong, Z. et al. Emerging role of metabolic reprogramming in tumor immune evasion and immunotherapy. Sci. China Life Sci. 64, 534–547 (2021). https://doi.org/10.1007/s11427-019-1735-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-019-1735-4