Abstract
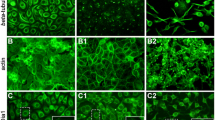
Bleeding is a clinical characteristic of severe dengue and may be due to increased vascular permeability. However, the pathogenesis of severe dengue remains unclear. In this study, we showed that the Rac1-microfilament signal pathway was involved in the process of DENV serotype 2 (DENV2) infection in EAhy926 cells. DENV2 infection induced dynamic changes in actin organization, and treatment with Cytochalasin D or Jasplakinolide disrupted microfilament dynamics, reduced DENV2 entry, and inhibited DENV2 assembly and maturation. Rac1 activities decreased during the early phase and gradually increased by the late phase of infection. Expression of the dominant-negative form of Rac1 promoted DENV2 entry but inhibited viral assembly, maturation and release. Our findings demonstrated that Rac1 plays an important role in the DENV2 life cycle by regulating actin reorganization in EAhy926 cells. This finding provides further insight into the pathogenesis of severe dengue.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Angelini, M.M., Akhlaghpour, M., Neuman, B.W., and Buchmeier, M.J. (2013). Severe acute respiratory syndrome coronavirus nonstructural proteins 3, 4, and 6 induce double-membrane vesicles. MBio doi: 10.1128/mBio.00524-13.
Bass, D.M., Baylor, M., Chen, C., and Upadhyayula, U. (1995). Dansylcadaverine and cytochalasin D enhance rotavirus infection of murine L cells. Virology 212, 429–437.
Bhatt, S., Gething, P.W., Brady, O.J., Messina, J.P., Farlow, A.W., Moyes, C.L., Drake, J.M., Brownstein, J.S., Hoen, A.G., Sankoh, O., Myers, M.F., George, D.B., Jaenisch, T., Wint, G.R., Simmons, C.P., Scott, T.W., Farrar, J.J., and Hay, S.I. (2013). The global distribution and burden of dengue. Nature 496, 504–507.
Bokoch, G.M., Vlahos, C.J., Wang, Y., Knaus, U.G., and Traynor-Kaplan, A.E. (1996). Rac GTPase interacts specifically with phosphatidylinositol 3-kinase. Biochem J 315 ( Pt 3), 775–779.
Chu, J.J., Choo, B.G., Lee, J.W., and Ng, M.L. (2003). Actin filaments participate in West Nile (Sarafend) virus maturation process. J Med Virol 71, 463–472.
Dai, J., Pan, W., and Wang, P. (2011). ISG15 facilitates cellular antiviral response to dengue and west nile virus infection in vitro. Virol J 8, 468.
Edwards, D.C., Sanders, L.C., Bokoch, G.M., and Gill, G.N. (1999). Activation of LIM-kinase by Pak1 couples Rac/Cdc42 GTPase signalling to actin cytoskeletal dynamics. Nat Cell Biol 1, 253–259.
Fang, X., Hu, Z., Shang, W., Zhu, J., Xu, C., and Rao, X. (2012). Genetic polymorphisms of molecules involved in host immune response to dengue virus infection. FEMS Immunol Med Microbiol 66, 134–146.
Frischknecht, F., Moreau, V., Rottger, S., Gonfloni, S., Reckmann, I., Superti-Furga, G., and Way, M. (1999). Actin-based motility of vaccinia virus mimics receptor tyrosine kinase signalling. Nature 401, 926–929.
Garoff, H., Hewson, R., and Opstelten, D.J. (1998). Virus maturation by budding. Microbiol Mol Biol Rev 62, 1171–1190.
Green, A.M., Beatty, P.R., Hadjilaou, A., and Harris, E. (2014). Innate immunity to dengue virus infection and subversion of antiviral responses. J Mol Biol 426, 1148–1160.
Hanley, J.G. (2014). Actin-dependent mechanisms in AMPA receptor trafficking. Front Cell Neurosci 8, 381.
Hishiki, T., Han, Q., Arimoto, K., Shimotohno, K., Igarashi, T., Vasudevan, S.G., Suzuki, Y., and Yamamoto, N. (2014). Interferon-mediated ISG15 conjugation restricts dengue virus 2 replication. Biochem Biophys Res Commun 448, 95–100.
Jimenez-Baranda, S., Gomez-Mouton, C., Rojas, A., Martinez-Prats, L., Mira, E., Ana Lacalle, R., Valencia, A., Dimitrov, D.S., Viola, A., Delgado, R., Martinez, A.C., and Manes, S. (2007). Filamin-A regulates actin-dependent clustering of HIV receptors. Nat Cell Biol 9, 838–846.
Kallewaard, N.L., Bowen, A.L., and Crowe, J.E.. (2005). Cooperativity of actin and microtubule elements during replication of respiratory syncytial virus. Virology 331, 73–81.
Krishna, D., and Le Doux, J.M. (2006). Murine leukemia virus particles activate Rac1 in HeLa cells. Biochem Biophys Res Commun 345, 1184–1193.
Lamaze, C., Chuang, T.H., Terlecky, L.J., Bokoch, G.M., and Schmid, S.L. (1996). Regulation of receptor-mediated endocytosis by Rho and Rac. Nature 382, 177–179.
Lei, S., Tian, Y.P., Xiao, W.D., Li, S., Rao, X.C., Zhang, J.L., Yang, J., Hu, X.M., and Chen, W. (2013). ROCK is involved in vimentin phosphorylation and rearrangement induced by dengue virus. Cell Biochem Biophys 67, 1333–1342.
Li, E., Stupack, D., Bokoch, G.M., and Nemerow, G.R. (1998). Adenovirus endocytosis requires actin cytoskeleton reorganization mediated by Rho family GTPases. J Virol 72, 8806–8812.
Lyman, M.G., and Enquist, L.W. (2009). Herpesvirus interactions with the host cytoskeleton. J Virol 83, 2058–2066.
Monaghan, P., Gold, S., Simpson, J., Zhang, Z., Weinreb, P.H., Violette, S.M., Alexandersen, S., and Jackson, T. (2005). The alpha(v)beta6 integrin receptor for Foot-and-mouth disease virus is expressed constitutively on the epithelial cells targeted in cattle. J Gen Virol 86, 2769–2780.
Morrison, T.G., and McGinnes, L.J. (1985). Cytochalasin D accelerates the release of Newcastle disease virus from infected cells. Virus Res 4, 93–106.
Normile, D. (2013). Tropical medicine. Surprising new dengue virus throws a spanner in disease control efforts. Science 342, 415.
Ott, D.E., Coren, L.V., Kane, B.P., Busch, L.K., Johnson, D.G., Sowder, R.C., Chertova, E.N., Arthur, L.O., and Henderson, L.E. (1996). Cytoskeletal proteins inside human immunodeficiency virus type 1 virions. J Virol 70, 7734–7743.
Pfanzagl, B., Andergassen, D., Edlmayr, J., Niespodziana, K., Valenta, R., and Blaas, D. (2014). Entry of human rhinovirus 89 via ICAM-1 into HeLa epithelial cells is inhibited by actin skeleton disruption and by bafilomycin. Arch Virol 159, 125–140.
Povoa, T.F., Alves, A.M., Oliveira, C.A., Nuovo, G.J., Chagas, V.L., and Paes, M.V. (2014). The pathology of severe dengue in multiple organs of human fatal cases: histopathology, ultrastructure and virus replication. PLoS One 9, e83386.
Ravkov, E.V., Nichol, S.T., Peters, C.J., and Compans, R.W. (1998). Role of actin microfilaments in Black Creek Canal virus morphogenesis. J Virol 72, 2865–2870.
Sanlioglu, S., Benson, P.K., Yang, J., Atkinson, E.M., Reynolds, T., and Engelhardt, J.F. (2000). Endocytosis and nuclear trafficking of adeno-associated virus type 2 are controlled by rac1 and phosphatidylinositol-3 kinase activation. J Virol 74, 9184–9196.
Sodeik, B., Ebersold, M.W., and Helenius, A. (1997). Microtubule- mediated transport of incoming herpes simplex virus 1 capsids to the nucleus. J Cell Biol 136, 1007–1021.
Stefanovic, S., Windsor, M., Nagata, K.I., Inagaki, M., and Wileman, T. (2005). Vimentin rearrangement during African swine fever virus infection involves retrograde transport along microtubules and phosphorylation of vimentin by calcium calmodulin kinase II. J Virol 79, 11766–11775.
Sun, X., and Whittaker, G.R. (2007). Role of the actin cytoskeleton during influenza virus internalization into polarized epithelial cells. Cell Microbiol 9, 1672–1682.
Tan, T.L., Fang, N., Neo, T.L., Singh, P., Zhang, J., Zhou, R., Koh, C.G., Chan, V., Lim, S.G., and Chen, W.N. (2008). Rac1 GTPase is activated by hepatitis B virus replication—involvement of HBX. Biochim Biophys Acta 1783, 360–374.
Tolias, K.F., Cantley, L.C., and Carpenter, C.L. (1995). Rho family GTPases bind to phosphoinositide kinases. J Biol Chem 270, 17656–17659.
Wang, J.L., Zhang, J.L., Chen, W., Xu, X.F., Gao, N., Fan, D.Y., and An, J. (2010). Roles of small GTPase Rac1 in the regulation of actin cytoskeleton during dengue virus infection. PLoS Negl Trop Dis doi: 10.1371/journal.pntd.0000809.
Weir, D.L., Laing, E.D., Smith, I.L., Wang, L.F., and Broder, C.C. (2014). Host cell virus entry mediated by Australian bat lyssavirus G envelope glycoprotein occurs through a clathrin-mediated endocytic pathway that requires actin and Rab5. Virol J 11, 40.
Yang, N., Higuchi, O., Ohashi, K., Nagata, K., Wada, A., Kangawa, K., Nishida, E., and Mizuno, K. (1998). Cofilin phosphorylation by LIM-kinase 1 and its role in Rac-mediated actin reorganization. Nature 393, 809–812.
Zhang, J.L., Wang, J.L., Gao, N., Chen, Z.T., Tian, Y.P., and An, J. (2007). Up-regulated expression of beta3 integrin induced by dengue virus serotype 2 infection associated with virus entry into human dermal microvascular endothelial cells. Biochem Biophys Res Commun 356, 763–768.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at springerlink.bibliotecabuap.elogim.com
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Zhang, J., Wu, N., Gao, N. et al. Small G Rac1 is involved in replication cycle of dengue serotype 2 virus in EAhy926 cells via the regulation of actin cytoskeleton. Sci. China Life Sci. 59, 487–494 (2016). https://doi.org/10.1007/s11427-016-5042-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-016-5042-5