Abstract
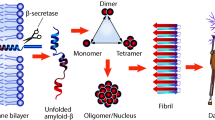
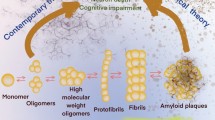
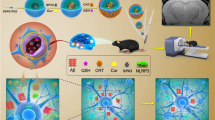
Amyloid-β peptide (Aβ) aggregates, particularly Aβ oligomers, are established biomarker and toxic species in Alzheimer’s disease (AD). Early detection and disaggregation of Aβ aggregates are of great importance for the treatment of AD due to the unavailability of therapy at the advanced stages of the disease. A multitalented agent, 2-{2-[(1H-benzoimidazol-2-yl)methoxy] phenyl}benzothiazole (BPB), is designed by merging two β-sheet targeting groups into one molecule to detect and inhibit the Aβ aggregation. BPB can quantitatively measure the β-sheet level of soluble Aβ oligomers and specifically distinguish the aggregates of Aβ40 and Aβ42 by unique luminescence spectrum. Animal tests demonstrate that BPB can efficiently penetrate the blood brain barrier and precisely stain Aβ plaques in the brain; more importantly, it can differentiate the blood of APP transgenic mice from that of normal ones. In addition to the diagnostic potential, BPB also suppresses the generation of ROS, protects the neurons from neurotoxicity, and disaggregates the Aβ aggregates in brain homogenates of APP transgenic mice induced by metal ions or self-assembly. In view of its detective ability toward Aβ oligomers and inhibition to Aβ-related neurotoxicity, BPB may be developed into a sensitive probe for screening blood samples in the early diagnosis of AD as well as an effective inhibitor for diminishing Aβ aggregates in the treatment of the disease.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Kepp KP. Chem Rev, 2012, 112: 5193–5239
Vinters HV. Annu Rev Pathol Mech Dis, 2015, 10: 291–319
Gao N, Sun H, Dong K, Ren J, Duan T, Xu C, Qu X. Nat Commun, 2014, 5: 3422–3431
Faller P, Hureau C, La Penna G. Acc Chem Res, 2014, 47: 2252–2259
Selkoe DJ. Science, 2012, 337: 1488–1492
Bateman RJ, Xiong C, Benzinger TLS, Fagan AM, Goate A, Fox NC, Marcus DS, Cairns NJ, Xie X, Blazey TM, Holtzman DM, Santacruz A, Buckles V, Oliver A, Moulder K, Aisen PS, Ghetti B, Klunk WE, McDade E, Martins RN, Masters CL, Mayeux R, Ringman JM, Rossor MN, Schofield PR, Sperling RA, Salloway S, Morris JC. N Engl J Med, 2012, 367: 795–804
Tentolouris-Piperas V, Ryan NS, Thomas DL, Kinnunen KM. Brain Res, 2017, 1655: 23–32
Selkoe DJ. Nat Med, 2011, 17: 1060–1065
Zhu L, Ploessl K, Kung HF. Chem Soc Rev, 2014, 43: 6683–6691
Johnson KA, Fox NC, Sperling RA, Klunk WE. Cold Spring Harb Perspect Med, 2012, 2: a006213
Selkoe DJ, Hardy J. EMBO Mol Med, 2016, 8: 595–608
Guo JL, Lee VMY. Nat Med, 2014, 20: 130–138
Ratnayaka JA, Serpell LC, Lotery AJ. Eye, 2015, 29: 1013–1026
Clark LF, Kodadek T. Alzheimer’s Res Ther, 2013, 5: 18–25
Wood H. Nat Rev Neurol, 2016, 12: 678
Telpoukhovskaia MA, Orvig C. Chem Soc Rev, 2013, 42: 1836–1846
Chen T, Wang X, He Y, Zhang C, Wu Z, Liao K, Wang J, Guo Z. Inorg Chem, 2009, 48: 5801–5809
Santos MJ, Quintanilla RA, Toro A, Grandy R, Dinamarca MC, Godoy JA, Inestrosa NC. J Biol Chem, 2005, 280: 41057–41068
Cheng S, Hou J, Zhang C, Xu C, Wang L, Zou X, Yu H, Shi Y, Yin Z, Chen G. Sci Rep, 2015, 5: 10535–10549
Wang X, Wang X, Zhang C, Jiao Y, Guo Z. Chem Sci, 2012, 3: 1304–1312
Yang T, Wang X, Zhang C, Ma X, Wang K, Wang Y, Luo J, Yang L, Yao C, Wang X. Chem Commun, 2016, 52: 2245–2248
Trott O, Olson AJ. J Comput Chem, 2010, 31: 455–461
van de Waterbeemd H, Gifford E. Nat Rev Drug Discov, 2003, 2: 192–204
Clark DE, Pickett SD. Drug Discov Today, 2000, 5: 49–58
Rodriguez-Rodriguez C, Sanchez de Groot N, Rimola A, Alvarez-Larena A, Lloveras V, Vidal-Gancedo J, Ventura S, Vendrell J, Sodupe M, Gonzalez-Duarte P. J Am Chem Soc, 2009, 131: 1436–1451
Savelieff MG, DeToma AS, Derrick JS, Lim MH. Acc Chem Res, 2014, 47: 2475–2482
Zhang X, Tian Y, Li Z, Tian X, Sun H, Liu H, Moore A, Ran C. J Am Chem Soc, 2013, 135: 16397–16409
Gao N, Qu XG. Sci Sin Chim, 2018, 48: 941–955
Economou NJ, Giammona MJ, Do TD, Zheng X, Teplow DB, Buratto SK, Bowers MT. J Am Chem Soc, 2016, 138: 1772–1775
Bibl M, Esselmann H, Mollenhauer B, Weniger G, Welge V, Liess M, Lewczuk P, Otto M, Schulz JB, Trenkwalder C, Kornhuber J, Wiltfang J. J Neurochem, 2007, 103: 467–474
Urraca JL, Aureliano CSA, Schillinger E, Esselmann H, Wiltfang J, Sellergren B. J Am Chem Soc, 2011, 133: 9220–9223
Wang J, Wang Y, Hu X, Zhu C, Ma Q, Liang L, Li Z, Yuan Q. Anal Chem, 2019, 91: 823–829
Chen Y, Liu L. Adv Drug Deliver Rev, 2012, 64: 640–665
Haass C, Selkoe DJ. Nat Rev Mol Cell Biol, 2007, 8: 101–112
Hickey JL, Lim SC, Hayne DJ, Paterson BM, White JM, Villemagne VL, Roselt P, Binns D, Cullinane C, Jeffery CM, Price RI, Barnham KJ, Donnelly PS. J Am Chem Soc, 2013, 135: 16120–16132
Kochi A, Eckroat TJ, Green KD, Mayhoub AS, Lim MH, Garneau-Tsodikova S. Chem Sci, 2013, 4: 4137–4145
Ma X, Hua J, Wang K, Zhang H, Zhang C, He Y, Guo Z, Wang X. Inorg Chem, 2018, 57: 13533–13543
Barnham KJ, Bush AI. Chem Soc Rev, 2014, 43: 6727–6749
Atrián-Blasco E, Del Barrio M, Faller P, Hureau C. Anal Chem, 2018, 90: 5909–5915
Yang L, Sun J, Xie W, Liu Y, Liu J. J Mater Chem B, 2017, 5: 5954–5967
Chen Q, Yang L, Zheng C, Zheng W, Zhang J, Zhou Y, Liu J. Nanoscale, 2014, 6: 6886–6897
Hong S, Beja-Glasser VF, Nfonoyim BM, Frouin A, Li S, Ramakrishnan S, Merry KM, Shi Q, Rosenthal A, Barres BA, Lemere CA, Selkoe DJ, Stevens B. Science, 2016, 352: 712–716
Tanzi RE. Nat Neurosci, 2005, 8: 977–979
Fu AKY, Hung KW, Yuen MYF, Zhou X, Mak DSY, Chan ICW, Cheung TH, Zhang B, Fu WY, Liew FY, Ip NY. Proc Natl Acad Sci USA, 2016, 113: E2705–E2713
Tan MS, Tan L, Jiang T, Zhu XC, Wang HF, Jia CD, Yu JT. Cell Death Dis, 2014, 5: e1382
Yu G, Yin S, Liu Y, Shuai Z, Zhu D. J Am Chem Soc, 2003, 125: 14816–14824
Li M, Howson SE, Dong K, Gao N, Ren J, Scott P, Qu X. J Am Chem Soc, 2014, 136: 11655–11663
Ono K, Condron MM, Teplow DB. Proc Natl Acad Sci USA, 2009, 106: 14745–14750
Zhao DS, Chen YX, Liu Q, Zhao YF, Li YM. Sci Sin Chim, 2012, 42: 226–228
Lv G, Sun A, Wei P, Zhang N, Lan H, Yi T. Chem Commun, 2016, 52: 8865–8868
Chan HN, Xu D, Ho SL, Wong MS, Li HW. Chem Sci, 2017, 8: 4012–4018
Yin Z, Wang S, Shen B, Deng C, Tu Q, Jin Y, Shen L, Jiao B, Xiang J. Anal Chem, 2019, 91: 3539–3545
Acknowledgements
This work was supported by the National Natural Science Foundation of China (21877059, 31570809, 21731004), the National Basic Research Program of China (2015CB856300), the Natural Science Foundation of Jiangsu Province (BK20150054), and the Research Foundation of the Chinese State Key Laboratory of Coordination Chemistry (SKLCC1912).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest The authors declare that they have no conflict of interest.
Supplementary Information
11426_2019_9594_MOESM1_ESM.pdf
A β-Sheet-targeted Theranostic Agent for Diagnosing and Preventing Aggregation of Pathogenic Peptides in Alzheimer’s Disease
Rights and permissions
About this article
Cite this article
Ma, X., Wang, Y., Hua, J. et al. A β-sheet-targeted theranostic agent for diagnosing and preventing aggregation of pathogenic peptides in Alzheimer’s disease. Sci. China Chem. 63, 73–82 (2020). https://doi.org/10.1007/s11426-019-9594-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-019-9594-y