Abstract
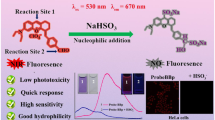
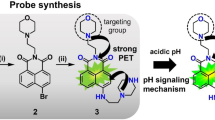
Modification of classic fluorophore to possess the emission transitions between aggregation-induced emission (AIE) and intrinsic emission offers reliable approach to the design of ratiometric fluorescent sensors. In this study, a proton acceptor benzimidazole was integrated with BODIPY to form three compounds, BBI-1/2/3, which demonstrated the AIE (~595 nm, Iagg) in neutral aqueous medium and intrinsic BODIPY emission (~510 nm, Iint) in acidic medium. All the three showed the ratiometric pH sensing behavior in a dual excitation/dual emission mode, yet BBI-3 displayed still the dual emission ratiometric pH sensing ability. The pH-dependent emission ratio Iint/Iagg of the three were fully reversible, and no interference was observed from normal abundant chemical species in live cells. Their different pKa (BBI-1, pKa 4.4; BBI-2, pKa 2.7; BBI-3, pKa 3.6) suggested that the substituents on benzimidazole moiety were essential to govern their functioning pH range. The ratiometric imaging of BBI-1 in A549 cells provided an effective intracellular pH (pHi) calibration formula corresponding to emission ratio of Iint/Iagg. Ratiometric pHi imaging in A549 cells upon small particle exposure confirmed the particle-induced cellular acidification with this formula. Both particle size and the chemical nature of the particle contribute to the observed acidification effect. The synchronization of lysosome disruption to cellular acidification in A549 cells upon crystalline silica exposure was directly observed for the first time with BBI-1, showing the potential application of BBI-1 in the study of silicosis and other related diseases. This study demonstrated that endowing fluorophore with AIE/intrinsic emission transition could be a promising strategy for ratiometric sensor design.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Stern ST, Adiseshaiah PP, Crist RM. Part Fibre Toxicol, 2012, 9: 20
Thibodeau MS, Giardina C, Knecht DA, Helble J, Hubbard AK. Toxicol Sci, 2004, 80: 34–48
Wan Q, Chen S, Shi W, Li L, Ma H. Angew Chem Int Ed, 2014, 53: 10916–10920
Wang X, Meier RJ, Wolfbeis OS. Angew Chem Int Ed, 2013, 52: 406–409
Tantama M, Hung YP, Yellen G. J Am Chem Soc, 2011, 133: 10034–10037
Chen Y, Zhu C, Cen J, Bai Y, He W, Guo Z. Chem Sci, 2015, 6: 3187–3194
Casey JR, Grinstein S, Orlowski J. Nat Rev Mol Cell Biol, 2010, 11: 50–61
Chrétien D, Bénit P, Ha HH, Keipert S, El-Khoury R, Chang YT, Jastroch M, Jacobs HT, Rustin P, Rak M. PLoS Biol, 2018, 16: e2003992
Zhang J, Wei H, Tan J, Qiao W, Guan Y, Zhang J. Sci China Chem, 2018, 61: 328–335
Hong Y, Lam JWY, Tang BZ. Chem Soc Rev, 2011, 40: 5361
Mei J, Leung NLC, Kwok RTK, Lam JWY, Tang BZ. Chem Rev, 2015, 115: 11718–11940
Yu CYY, Xu H, Ji S, Kwok RTK, Lam JWY, Li X, Krishnan S, Ding D, Tang BZ. Adv Mater, 2017, 29: 1606167
Li Q, Li Z. Sci China Chem, 2015, 58: 1800–1809
Chen S, Hong Y, Liu Y, Liu J, Leung CWT, Li M, Kwok RTK, Zhao E, Lam JWY, Yu Y, Tang BZ. J Am Chem Soc, 2013, 135: 4926–4929
Qin AJ, Zhang Y, Han N, Mei J, Sun JZ, Fan WM, Tang BZ. Sci China Chem, 2012, 55: 772–778
Wang ZT, Fang Y, Sun JZ, Qin AJ, Tang BZ. Sci China Chem, 2013, 56: 1187–1190
Sun YQ, Liu J, Zhang H, Huo Y, Lv X, Shi Y, Guo W. J Am Chem Soc, 2014, 136: 12520–12523
Kim S, Tachikawa T, Fujitsuka M, Majima T. J Am Chem Soc, 2014, 136: 11707–11715
Qin W, Baruah M, De Borggraeve WM, Boens N. J Photochem Photobiol A-Chem, 2006, 183: 190–197
Kolemen S, Isik M, Kim GM, Kim D, Geng H, Buyuktemiz M, Karatas T, Zhang XF, Dede Y, Yoon J, Akkaya EU. Angew Chem Int Ed, 2015, 54: 5340–5344
Pettit GR, Thornhill AJ, Moser BR, Hogan F. J Nat Prod, 2008, 71: 1561–1563
Ottenwaelder X, Ruiz-García R, Blondin G, Carasco R, Cano J, Lexa D, Journaux Y, Aukauloo A. Chem Commun, 2004, 6: 504–505
Costantini LM, Gilberti RM, Knecht DA. PLoS ONE, 2011, 6: e14647
Kowada T, Maeda H, Kikuchi K. Chem Soc Rev, 2015, 44: 4953–4972
Daly B, Ling J, de Silva AP. Chem Soc Rev, 2015, 44: 4203–4211
Mukherjee S, Thilagar P. Chem Eur J, 2014, 46: 9052–9062
Zhu X, Liu R, Li Y, Huang H, Wang Q, Wang D, Zhu X, Liu S, Zhu H. Chem Commun, 2014, 50: 12951–12954
Li Z, Zheng M, Guan X, Xie Z, Huang Y, Jing X. Nanoscale, 2014, 6: 5662–5665
Kim HJ, Heo CH, Kim HM. J Am Chem Soc, 2013, 135: 17969–17977
Sevinç G, Küçüköz B, Yilmaz H, Sirikçi G, Yaglioglu HG, Hayvali M, Elmali A. Sensor Actuat B-Chem, 2014, 193: 737–744
Li Z, Li LJ, Sun T, Liu L, Xie Z. Dyes Pigments, 2016, 128: 165–169
Dong Y, Lam JWY, Qin A, Li Z, Sun J, Sung HHY, Williams ID, Tang BZ. Chem Commun, 2007, 127: 40–42
Zhang Z, Xu B, Su J, Shen L, Xie Y, Tian H. Angew Chem Int Ed, 2011, 50: 11654–11657
Shen XY, Yuan WZ, Liu Y, Zhao Q, Lu P, Ma Y, Williams ID, Qin A, Sun JZ, Tang BZ. J Phys Chem C, 2012, 116: 10541–10547
Masuda A, Oyamada M, Nagaoka T, Tateishi N, Takamatsu T. Brain Res, 1998, 807: 70–77
Jaganathan H, Godin B. Adv Drug Deliver Rev, 2012, 64: 1800–1819
Ding M, Chen F, Shi X, Yucesoy B, Mossman B, Vallyathan V. Int Immunopharmacol, 2002, 2: 173–182
Hnizdo E. Occupl Environ Med, 2003, 60: 237–243
Greenberg MI, Waksman J, Curtis J. Disease-a-Month, 2007, 53: 394–416
Fubini B, Hubbard A. Free Radical Biol Med, 2003, 34: 1507–1516
Deshpande A, Narayanan PK, Lehnert BE. Toxicol Sci, 2002, 67: 275–283
Jessop F, Hamilton Jr RF, Rhoderick JF, Fletcher P, Holian A. Toxicol Appl Pharmacol, 2017, 318: 58–68
Huang DM, Hung Y, Ko BS, Hsu SC, Chen WH, Chien CL, Tsai CP, Kuo CT, Kang JC, Yang CS, Mou CY, Chen YC. FASEB J, 2005, 19: 2014–2016
Boya P, Kroemer G. Oncogene, 2008, 27: 6434–6451
Kroemer G, Jäättelä M. Nat Rev Cancer, 2005, 5: 886–897
Merget R, Bauer T, Küpper H, Philippou S, Bauer H, Breitstadt R, Bruening T. Arch Toxicol, 2002, 75: 625–634
Mossman BT, Churg A. Am J Respir Crit Care Med, 1998, 157: 1666–1680
Thibodeau M, Giardina C, Hubbard AK. Toxicol Sci, 2003, 76: 91–101
Sohaebuddin SK, Thevenot PT, Baker D, Eaton JW, Tang L. Part Fibre Toxicol, 2010, 7: 22
Yan X, Ye T, Hu X, Zhao P, Wang X. Sci Rep, 2016, 6: 27875
Acknowledgements
This work was supported by the National Basic Research Program of China (2015CB856300), the National Natural Science Foundation of China (21571099, 21731004) and the Natural Science Foundation of Jiangsu Province (BK20150054).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
11426_2018_9284_MOESM1_ESM.docx
BODIPY-derived ratiometric fluorescent sensors: pH-regulated aggregation-induced emission and imaging application in cellular acidification triggered by crystalline silica exposure
Rights and permissions
About this article
Cite this article
Bai, Y., Liu, D., Han, Z. et al. BODIPY-derived ratiometric fluorescent sensors: pH-regulated aggregation-induced emission and imaging application in cellular acidification triggered by crystalline silica exposure. Sci. China Chem. 61, 1413–1422 (2018). https://doi.org/10.1007/s11426-018-9284-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-018-9284-4