Abstract
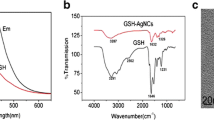
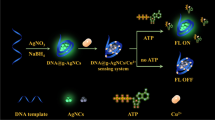
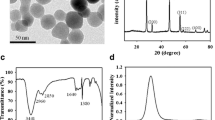
To develop the high-performance fluorescent bio-sensors, the metal nanoparticles were employed as nanoquenchers and attracted reasonable attention in the design of fluorescent biosensors. In this work, silver nanoparticles (AgNPs) were obtained via reduction of Ag+ on FAM-labeled DNA template. For the tight binding between AgNPs and DNA, the template-synthesized AgNPs turned out high quenching efficiency and could be applied as super nanoquenchers to establish the biosensing platform for fluorescent detection. As an example, the template-synthesized DNA-AgNPs conjugates were employed in sensing thiols. By forming S-Ag bonds, thiols interact intensely with AgNPs and replace the FAM-labeled DNA off from the surface of AgNPs, resulting in a fluorescence enhancement. Besides the advantages of lower background and higher signal-to-background ratio (S/B), the conjugates present better stability, making them applicable in complicated biological fluids. To further evidence the feasibility of sensing thiols in real samples, the thiols in human urine were detected. The total amount of free thiols found in human urine was ranging from 229 μM to 302 μM with the proposed sensor. To conclude the reliability, low content of Cys was added and the recovery was 98%–103%.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Rosi NL, Mirkin CA. Nanostructures in biodiagnostics. Chem Rev, 2005, 105: 1547–1562
Lu Y, Liu JW. Smart nanomaterials inspired by biology: Dynamic assembly of error-free nanomaterials in response to multiple chemical and biological stimuli. Acc Chem Res, 2007, 40: 315–323
Storhoff JJ, Lucas AD, Garimella V, Bao YP, Müller UR. Homogeneous detection of unamplified genomic DNA sequences based on colorimetric scatter of gold nanoparticle probes. Nat Biotechnol, 2004, 22: 883–887
Guo WW, Yuan JP, Dong QZ, Wang EK. Highly sequence-dependent formation of fluorescent silver nanoclusters in hybridized DNA duplexes for single nucleotide mutation identification. J Am Chem Soc, 2009, 132: 932–934
Zhang J, Ting BP, Jana NR, Gao, ZQ, Ying JY. Ultrasensitive electrochemical DNA biosensors based on the detection of a highly characteristic solid-state process. Small, 2009, 5: 1414–1417
Maxwell DJ, Taylor JR, Nie SM. Self-assembled nanoparticle probes for recognition and detection of biomolecules. J Am Chem Soc, 2002, 124: 9606–9612
Sun IC, Lee S, Koo H, Kwon IC, Choi K, Ahn CH, Kim K. Caspase sensitive gold nanoparticle for apoptosis imaging in live cells. Bioconjugate Chem, 2010, 21: 1939–1942
Wang H, Wang YX, Jin JY, Yang RH. Gold nanoparticle-based colorimetric and “turn-on” fluorescent probe for mercury(II) ions in aqueous solution. Anal Chem, 2008, 80: 9021–9028
Jin Y, Li HY, Bai JY. Homogeneous selecting of a quadruplexbinding ligand-based gold nanoparticle fluorescence resonance energy transfer assay. Anal Chem, 2009, 81: 5709–5715
Wang H, Li JS, Wang YX, Jin JY, Yang RH, Wang KM, Tan WH. Combination of DNA ligase reaction and gold nanoparticle-quenched fluorescent oligonucleotides: A simple and efficient approach for fluorescent assaying of single-nucleotide polymorphisms. Anal Chem, 2010, 82: 7684–7690
Pihlasalo S, Kirjavainen J, Hänninen P, Härmä H. Ultrasensitive protein concentration measurement based on particle adsorption and fluorescence quenching. Anal Chem, 2009, 81: 4995–5000
Li HX, Rothberg LJ. DNA sequence detection using selective fluorescence quenching of tagged oligonucleotide probes by gold nanoparticles. Anal Chem, 2004, 76: 5414–5417
Oh E, Hong MY, Lee D, Nam SH, Yoon HC, Kim HS. Inhibition assay of biomolecules based on fluorescence resonance energy transfer (FRET) between quantum dots and gold nanoparticles. J Am Chem Soc, 2005, 127: 3270–3271
Sun YG, Xia YN. Shape-controlled synthesis of gold and silver nanoparticles. Science, 2002, 298: 2176–2179
Naik RR, Stringer SJ, Agarwal G, Jones SE, Stone MO. Biomimetic synthesis and patterning of silver nanoparticles. Nat Mater, 2002, 1: 169–172
Kuo PL, Chen WF. Formation of silver nanoparticles under structured amino groups in pseudo-dendritic poly(allylamine) derivatives. J Phys Chem B, 2003, 107: 11267–11272
Shang L, Dong SJ. Facile preparation of water-soluble fluorescent silver nanoclusters using a polyelectrolyte template. Chem Commun, 2008, 1088-1090
Petty JT, Zheng J, Hud NV, Dickson RM. DNA-templated Ag nanocluster formation. J Am Chem Soc, 2004, 126: 5207–5212
Richards CI, Choi S, Hsiang JC, Antoku Y, Vosch T, Bongiorno A, Tzeng YL, Dickson RM. Oligonucleotide-stabilized Ag nanocluster fluorophores. J Am Chem Soc, 2008, 130: 5038–5039
Berti L, Alessandrini A, Facci P. DNA-templated photoinduced silver deposition. J Am Chem Soc, 2005, 127: 11216–11217
Zinchenko AA, Yoshikawa K, Baigl D. DNA-templated silver nanorings. Adv Mater, 2005, 17: 2820–2823
Lakowicz J. Principles of Fluorescence Spectroscopy, 3rd. Springer, 2006
Wang WH, Rusin O, Xu XY, Kim KK, Escobedo JO, Fakayode SO, Fletcher KA, Lowry M, Schowalter CM, Lawrence CM, Fronczek FR, Warner IM, Strongin RM. Detection of homocysteine and cysteine. J Am Chem Soc, 2005, 127: 15949–15958
Tanaka F, Mase N, Barbas CF. Determination of cysteine concentration by fluorescence increase: Reaction of cysteine with a fluorogenic aldehyde. Chem Commun, 2004, 1762-1763
Lee KS, Kim TK, Lee JH, Kim HJ, Hong JI. fluorescence turn-on probe for homocysteine and cysteine in water. Chem Commun, 2008, 6173–6175
Shao N, Jin JY, Cheung SM, Yang RH, Chan WH, Mo T. Spiropyran-based ensemble for visual recognition and quantification of cysteine and homocysteine at physiological levels. Angew Chem Int Ed, 2006, 45: 4944–4948
Sudeep PK, Joseph STS, Thomas KG. Selective detection of cysteine and glutathione using gold nanorods. J Am Chem Soc, 2005, 127: 6516–6517
Chen Z, Luo SL, Liu CB, Cai QY. Simple and sensitive colorimetric detection of cysteine based on ssDNA-stabilized gold nanoparticles. Anal Bioanal Chem, 2009, 395: 489–494
Lu C, Zu YB. Specific detection of cysteine and homocysteine: recognizing one-methylene difference using fluorosurfactant-capped gold nanoparticles. Chem Commun, 2007, 3871-3873
Shang L, Qin CJ, Wang T, Wang M, Wang LX, Dong SJ. Fluorescent conjugated polymer-stabilized gold nanoparticles for sensitive and selective detection of cysteine. J Phys Chem C, 2007, 111: 13414–13417
Kuśmierek K, Glowacki R, Bald E. Analysis of urine for cysteine, cysteinylglycine, and homocysteine by high-performance liquid chromatography. Anal Bioanal Chem, 2006, 385: 855–860
Fermo I, Arcelloni C, Paroni R. High-performance liquid chromatographic method to quantify total cysteine excretion in urine. Anal Biochem, 2002, 307: 181–183
Rafii M, Elango R, Courtney-Martin G, House JD, Fisher L, Pencharz PB. High-throughput and simultaneous measurement of homocysteine and cysteine in human plasma and urine by liquid chromatography-electro spray tandem mass spectrometry. Anal Biochem, 2007, 371: 71–81
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jin, J., Ouyang, X., Li, J. et al. DNA template-synthesized silver nanoparticles: A new platform for high-performance fluorescent biosensing of biothiols. Sci. China Chem. 54, 1266–1272 (2011). https://doi.org/10.1007/s11426-011-4320-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11426-011-4320-0