Abstract
Purpose
The purpose of this paper is to study the responses of soil biological parameters as indicator of ecological status on PAH-contaminated soil.
Materials and methods
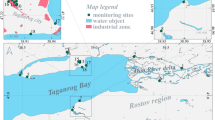
Studies are conducted on the soils and natural grassy vegetation of monitoring plots subjected to Novocherkassk power station (NPS) emissions. Monitoring plots were established at different distances from the NPS (1.0–20.0 km).
Results and discussion
The level of polycyclic aromatic hydrocarbons (PAHs) around NPS is the highest at the monitoring plot located at distance 1.6 km to the northwest through the prevailing wind direction. Gradually, decrease of PAHs was observed while increasing the distance from the NPS through the prevailing wind direction. Calculation of correlations between PAH level and biological activity parameters of soils showed lack of dependence with total and every PAH content in all 12 studied monitoring plots. The most significant correlations were found between PAH content and enzyme activity in the monitoring plots situated through the prevailing wind direction from NPS.
Conclusions
The main pollution source in the studied area is NPS. It was found that contamination of soil by PAHs has a direct dependence on the activity of all biological communities in chernozems, as well as the activities of dehydrogenase and the phytotoxicity of soils. Inverse correlations have been revealed between the PAH contamination and abundance of soil bacteria.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Polycyclic aromatic hydrocarbons (PAHs) are one of the most significant environmental contaminants with mutagenic and carcinogenic properties to all living organisms (Tsibart and Gennadiev 2013; The state of food and agriculture, 1998). Sixteen PAH compounds are obligatory controlled all over the world that is regulated by legislation because of their carcinogenicity, mutagenicity, and toxicity (Jian 2004; Wenzl et al. 2006). These compounds are very persistent and hydrophobic in environmental objects especially during soil contamination because of complicated soil matrix structure (Antizar-Ladislao et al. 2006).
Soil has a high ability to PAH accumulation that causes a high availability of these compounds to plants (Gabov et al. 2010; Gennadiev and Tsibart 2013). The changes in microbial community structure in technogenic-polluted soil may be used as tools for predicting and monitoring natural degradation (Jain et al. 2005). Soil microbial community structure in the technogenic pollution conditions has the main significance in remediation can also be used for search for the most effective and appropriate pathways of bioremediation (Demmerová et al. 2005; Pérez-Leblic et al. 2012).
PAHs presence in soils and plants is connected with the increased level of the technogenic pressure. PAH accumulation in a soil surface is involved in the processes of distribution in the soil-plant system and migration in a soil profile that is depended on soil enzyme and microbial activity (García et al. 1997; Pascual et al. 2000) and on sorption properties of a soil matrix (Tsibart and Gennadiev 2013). BaP background content in plants and the majority of mineral soils fluctuate within 0.1–5.0 μg/kg whereas for some chernozems and peaty soils are characterized by more high level of BaP—15.0–20.0 μg/kg. That is caused by the increasing contents of highly condensed organic substances and specific structure of soil microbial community. The parameters of biological activity interaction of soils in technogenic areas are important object for control of BaP environmental pollution (Sastre and Vicente 2003).
Several microbiological parameters have been used to define the soil ecological status and the effects of PAH contamination on soils, such as dehydrogenase activity, urease activities, soil bacterial abundance, and integrated biological indices (IBIs) (Jain et al. 2005; Liu et al. 2014; Wu et al. 2014; Hadibarata and Kristanti 2012; Rodríguez-Morgado et al. 2015).
Soil enzymes are the catalysts of important metabolic processes including the decomposition of organic pollutants and the detoxification of xenobiotics (Margesin et al. 2000a, b). Soil enzymes activity considered to be sensitive to pollution and have the further advantage to be used as a bioassay instrument. Parameters of soil enzyme activity are used as indicators for measuring the soil pollution level. Dehydrogenase and urease are the most important soil enzymes and frequently used test for determining the influence of the various pollutants including PAHs on the biological parameters of soil (Shen et al. 2005). Several researchers investigated the effect of PAHs on urease and dehydrogenase activity (Lipińska et al. 2015). Margesin et al. (2000a) proved a positive influence of naphthalene and phenanthrene on urease activities and gave the correlation for urease activity and phenanthrene content (Margesin et al. 2000a, b). Some researchers show the positive correlation between soil biological activity and PAHs presence (Margesin et al. 2000a, b; Shen et al. 2005; Demmerová et al. 2005), some of them show the negative correlation (Unbehaun et al. 2000; Baran et al. 2004), but all articles acquaint with possibilities of using soil microbiological parameters as indicator of pollution by PAHs.
The present study is aimed to research the biological activity of the soil in the emission zone of Novocherkassk power station (Russia) polluted by PAHs in 2015.
2 Materials and methods
2.1 Experimental area
The objects of the study are soils of the joint area to the Novocherkassk power station (NPS). The NPS is one of the largest thermal power stations in the south of Russia. It is classified among the enterprises of I hazardous class. The NPS is located in the Rostov region at 7.5 km to the southeast of the Novocherkassk city. The NPS is the main plant polluting the atmospheric air and the soil in Rostov region. The dynamics of pollutant emissions from the NPS during the last 50 years of continuous exploitation was 240–260 thousand tons annually till 1994 and 130–150 thousand tons annually after 1995 (Ecological Bulletin of the Don Region 2015). The coals delivered to the NPS are enriched with a wide range of PAHs, toxic elements, and radionuclides (Minkina et al. 2014; Sushkova et al. 2016a, b, c). Most pollutants (90%) fall to the Novocherkassk city. This is related to the location of the NPS with direction to the city: the nonobservance of the 10-km sanitary zone (7.5 km from the Novocherkassk, close to the settlements of Donskoi and Krivyanskaya) and the prevailing (mainly southeastern and northwestern) wind directions.
Monitoring plots were established at different distances (1–20 km) from the NPS in 2000. The main direction from the contamination source through the residential zones of the Novocherkassk city and the Krivyanskaya village was determined in accordance with the prevailing winds (Fig. 1). Monitoring plots were located on virgin or no-till fallow areas and not subject to the sanitary-protection zone of the NPS. Soil samples were taken from a depth of 0- to 20-cm, because the major part of PAHs are accumulated in the surface soil layer.
2.2 Soil sampling
In this study, 12 surface soil samples (0–20 cm) were taken around the Novocherkassk power station (Minkina et al. 2013). Field-moist samples from both sampling batches were manually and gently crumbled and sieved (<2 mm) to remove root material according to GOST 17.4.4.02–84 (GOST 17.4.1.02.-83 2004) requirements. The samples were transported to the laboratory for analysis at the same day of sampling and refrigerated at 4 °C for no longer than 72 h prior to analysis. Samples were analyzed for their physicochemical properties, PAHs content, and microbiological properties.
The soil microbiota and plants are among the major co-receptors of NPS; especially, the soil-microbial biomass serves as a pool of nutrients and is a sensitive indicator of microbial changes in soils (Atlas 1984).
2.3 Soil analyses
2.3.1 Soil physicochemical properties
Physicochemical analyses were conducted on air-dried samples stored at room temperature and from which crop residues, root fragments, and rock larger than 2 mm in diameter had been removed. Selected soil physicochemical properties were determined according to the Methodological guidelines for the integrated monitoring of soil fertility of agricultural lands (2003). Physicochemical and agrochemical properties of the studied soils are represented in Table 1. Most of the area in the affected zone of the NPS occupied by Haplic Chernozem (plots 1, 4, 5, 7, 9, and 10), Phaeozems Haplic (plots 3, 6, 8, and 11), and Fluvisols Umbric (plots 2 and 12) also occur in the Tuzlov River floodplain of the studied zone (Table 2) (IUSS Working Group WRB 2015).
The Co and MCS have thick humus horizons (70–100 cm), relatively high content of humus (4.1–5.0%), and high cation exchange capacity (CEC) (31.2–47.6 cmol(+)/kg), including a high content of exchangeable calcium (76–90% of total exchangeable cations), and neutral or weak alkaline reaction (pHwater 7.4–7.7) (GOST 26423-85 1985). According to particle size distribution, they belong to heavy loamy and light clayey varieties developed on calcareous loess-like rocks. The climatic index of biological productivity (Bc) is 90–100 under natural conditions and 170–175 under optimum wetting conditions. The sufficient amount of heat and precipitation forms soils with high natural fertility and the enrichment with carbonates from the parent rocks favors the development of high buffering properties. The alluvial soil has light texture, thinner humus horizon (40–60 cm), lower humus content (lower than 3.1%), and lower CEC (10.6 cmol(+)/kg) with a relatively high content of exchangeable calcium.
2.3.2 Soil microbiological analysis
Dehydrogenase activity (ADG, EC 1.1.1) was determined according to Kazeev and Kolesnikov (2012) of the standard methods of ecology and soil biology (Khaziev 2005). Moist soil (1 g) was treated with 1 mL of 1% glucose solution and 1 mL of 1% TTC (2,3,5-triphenyltetrazolium chloride) in purified water (pH, 6.6) for 24 h in darkness at 30 °C. The thiamine triphosphate (TTP) formed extracted with an ethanol TTP was measured by spectrophotometry method at 540 nm using the extracting solution as a blank. Data was expressed as mg TTP 10 g−1 dry soil 24 h−1.
Urease activity (AUR, EC 3.5.1.5) was measured according to Kazeev and Kolesnikov (2012). Five mL of 3% urea substrate solution was added to 1 g of dry soil, and subsequently the samples were incubated for 24 h at 30 °C. The released ammonium was determined by spectrophotometry method at 400 nm. Results were expressed as mg NH3 10 g−1 dry soil 24 h−1.
The abundance of soil bacteria (AB) was determined by the Zvyagintsev method of luminescence microscopy (Kazeev and Kolesnikov 2012; Zvyagintsev 1991). Results were expressed as 109 CFU g−1 dry soil.
2.3.3 PAH determination methods
PAHs are extracted from the soils of the objects under study by the standard method using for the removal of the interfering soil components by saponification (Directive document 52.10.556-95 2002; Sushkova et al. 2015, 2016b, c).
A 1-g portion of the prepared soil was put into a pear-shaped flask for rotary evaporator; 20 mL of 2% KOH solution in ethanol was added, and the mixture was refluxed on a water bath for 3 h. The saponification of lipids and gummy soil components occurred during the refluxing, which increased the recovery of PAHs and reduced the amount of coextracted substances in the extract. The supernatant decanted into an Erlenmeyer flask, and 15 mL of n-hexane and 5 mL of distilled water added for the better separation of the layers. The mixture was shaken on a rotary shaker for 10 min and transferred into a dividing funnel. The hexane layer was poured into a separate vessel. The residue in the flask was extracted twice more in a similar way. The combined hexane extract was washed with distilled water to neutral pH (using litmus as an indicator), transferred into a dark vessel with a close lid, and desiccated by adding 5 g of anhydrous Na2SO4. After exposure at +5 °C for 8 h, the desiccated extract was decanted into a dry round-bottomed flask and evaporated to dry on a rotary evaporator at a bath temperature of 40 °C. The dry residue dissolved in 1 mL of acetonitrile.
The content of BaP in the test samples was determined by the external standard method (Procedure of measurements… 2008). The content of BaP in the soil calculated from the equation
where C s is the content of BaP in the soil sample (μg/kg); S st and S i are the BaP peak areas for the standard solution and the sample, respectively; C st is the concentration of the standard BaP solution (μg/kg); k is the recovery factor of BaP from the sample; V is the volume of the acetonitrile extract (mL); and m is the mass of the sample (g).
Data were processed using STATISTICA 11.0. All results presented as averages of three replicates.
During monitoring of PAH contamination in the NPS emission zone in 2015, it determined 15 priority PAHs in the soil samples: benzo(a)pyrene, anthracene, fluoranthene, pyrene, dibenz(a,h)anthracene, acenaphthylene, biphenyl, acenaphthene, benzo(b)fluoranthene, benzo(k)fluoranthene, benzo(a)anthracene, naphthalene, fluorene, phenanthrene, and benz(g,h,i)perylene. The total PAH content in soil samples counted as a summary of 15 PAH content.
3 Results
The maximum of PAH pollution of monitoring plots was observed in the northwest direction from the NPS at a distance of 1.6 km and consists 1600.1 ± 14.7 μg/kg. Location of this monitoring plot belongs to the northwest direction the main prevailing wind direction from NPs. It was observed in a maximum level of soil contamination at a distance of 1.0–1.2 km in the northwest direction by all of PAHs, include main marker of pollution benzo(a)pyrene gradually decreasing with distance from the enterprise (Fig. 2A). The received data was conformed by previous research of PAH accumulation by soil in NPS zone (Sushkova et al. 2015, 2016b, c). The level of PAH pollution of monitoring plots in the northwest direction decreasing at the distance 5.0nw and 15.0nw up to 741.0 ± 8.6, 383.1 ± 4.6 μg/kg, respectively (Fig. 2A).
The content of total PAHs in monitoring plot 10 (20.0 km to the northwest) 20.0nw increased up to 759.6 ± 12.4 μg/kg compare to plot 15.0nw. The soils on the most remote monitoring plot 10 (20.0 km to the northwest) occupy a special place in the description of the affected zone of NPS. The obtained data indicate the presence of additional sources of pollutant emission near the plot: exhausts from motor vehicles on the M-4 Don highway, which passes at 350 m from the sampling site. The plot is located within the V-shaped area enclosed by two highways (the Rostov−Moscow highway from the northwest and the Rostov−Novocherkassk highway from the southeast), which results in the contamination of soils with vehicle exhausts containing PAHs. In addition, this plot is apparently subjected to the plumes from the Novocherkassk dumps, as well as combustion products formed at the stove heating of houses in the village Grushevskaya. Nonetheless, the accumulation of the pollutant in the soil due to the additional emission sources is significantly lower than that caused by the NPS.
The abrupt decrease in the content of PAHs in the soil of monitoring plot 5 (1.2nw) reached up to 764.3 ± 7.4 compared to plot 1, 6nw (by more than 2.1 times) indicates that the distribution area of the densest plume containing the maximum amount of pollutant is about 5 km to the northwest, and the maximum fallouts are observed at a distance of about 1.6 km (Sushkova et al. 2017) (Fig. 2B). The areas located around the NPS up to 3 km in the northern/northeastern and southwestern directions are less contaminated. The level of contamination of all soils located around NPS mostly depends on soil physicochemical and agrochemical properties.
It should be noted that the soils of the plots located at 1–2 km to the north and northeast of the plant are almost not subjected to the impact of polluting emissions. During the entire period of observations, the content of PAHs in the 0- to 20-cm layer of soils on these plots is located at a short distance from the plant (1–2 km) (Fig. 2B). An exception is the soil on plot 1, which is the closest to predominant wind direction plots, where the concentrations of PAHs in the 0- to 20-cm layer reached 812.5 ± 21.4 μg/kg (Fig. 2B).
The analysis of changes in the biological activity parameters of soils showed uncertain tendencies depending on the distance from and orientation with respect to the NPS (Figs. 3, 4, and 5). According to the scale developed by Zvyagintsev (1978), the chernozems and the meadow-chernozemic soils (Table 1) are considered as highly enriched with dehydrogenases (>30 mg TTP 10 g−1 dry soil 24 h−1), and the alluvial soils are considered as enriched with them (10–30 mg TTP 10 g−1 dry soil 24 h−1) (Zvyagintsev 1991). From the activity of urease, the soils on all plots, except the alluvial meadow soil (plot 2) considered as medium enriched (10–20 mg NH3 10 g−1 dry soil 24 h−1). According to the bacterial abundance, the soils on all plots can be considered as medium enriched, except plot 11 assessed as rich (8.1 109 CFU g−1 dry soil) (Fig. 3).
In chernozems, an increase in the activity of dehydrogenases and urease and the phytotoxicity (root length) with approaching the NPS was observed on the plots in the northwestern direction. A decrease in the abundance of soil bacteria in chernozems was observed in the near impact zone of the NPs: 4.1 109 CFU g−1 dry soil at a distance of 1.2 km (plot 5) compared to 4.6 109 CFU g−1 dry soil at 15 km. Similar tendencies traced for the meadow-chernozemic soils: when the distance in the northwestern direction from the NPS increased, the activity of dehydrogenases decreased (by 25–30%), as well as the bacterial abundance (plots 4, 6). A decrease in soil phytotoxicity was observed with increasing distance from the NPS: the radish root length was 14.4 and 19.1 mm at 2.0 and 5.0 km, respectively (Fig. 4).
The enzymatic activity of alluvial soils significantly varied depending on their location with respect to the NPS: the activities of dehydrogenases and urease in the alluvial soil on plot 2 in the southwestern direction were lower than those on plot 11 in the southern direction by 30–70% (Fig. 3B), and the radish root length increased by 20% (Fig. 4B). Thus, the differences in biological activity parameters depend not only on the distance from the NPS, but also on the type of soil. The biological activity of alluvial soil significantly differs from those of the other soils: the activity of dehydrogenases is lower by 2–3.5 times, the activity of urease is lower by 1.5–2 times, and the abundance of soil bacteria is lower by 2–2.5 times.
4 Discussion
To substantiate the source of PAH intake into the studied soils, the measured PAHs are divided into the two groups: low-molecular (containing three and less aromatic rings) and high-molecular (containing four and more aromatic rings). This approach allows to identify the main components that determine the toxicological activity of the environment medium. Content of low- and high-molecular PAHs in the studied soil shows that the level of high-molecular PAHs exceeds the level low-molecular PAHs in all monitoring plots situated through the prevailing wind direction from NPS (Fig. 6A). This fact explains that the main source of contamination of studied soils situated at the prevailing wind direction is technogenic emissions of the factory working at the low-quality coal.
PAH content in the soil of monitoring plots situated around NPS have exceeds level of low-molecular PAHs compare to high-molecular PAHs, exclude monitoring plot situated most close to the prevailing wind direction (plot 5, 1.2 km to the west) and monitoring plot situated the most closest to factory from the southeastern side (plot 12, 1.0 km to the southeast) (Fig. 6B). Plot 12 contamination by high-molecular PAHs is caused by the spread of the prevailing winds to the southeast also. Despite that, total level of all PAHs does not exceed the level of contamination in the northwest direction (Fig. 6B).
Calculation of correlations between PAH level and biological activity parameters of soils showed lack of dependence with total and every PAH content in 12 studied monitoring plots (Fig. 7). It caused the different nature of contamination of all 12 monitoring plots and different relations between high- and low-molecular PAH content in soils (Table 2) (Tobiszewski and Namiesnik 2012; Lipińska et al. 2015).
The most significant correlations were found between PAH content and biological activity parameters in the monitoring plots situated through the prevailing wind direction from NPS. Level of dehydrogenases has high positive correlation with technogenic accumulated biphenyl, acenaphthene, and negative correlation with anthracene content in studied soil. The low-molecular PAH content of soil influenced activity of dehydrogenases positively. Low-molecular PAHs are the most suitable substratum for enzyme activity, could be an easy power source for dehydrogenases in chernozem soil. Anthracene content in studied soils does not exceed 21.2 ± 0.4 μg/kg it is found a probability of negative influence at dehydrogenase activity.
Urease activity of monitoring plots has a positive correlation with 12 PAHs detected in the studied soils (Margesin et al. 2000a, b; Shen et al. 2005; Demmerová et al. 2005) exclude biphenyl, benzo(a)anthracene, and naphthalene. The activity of soil urease strongly depends on high content of 12 priority PAHs in technogenic polluted soil. Inverse correlation for urease activity from biphenyl, benzo(a)anthracene, and naphthalene are caused by their low-molecular structure. Only benzo(a)anthracene has four aromatic rings and can be attributed to the group of high-molecular compound. This fact allows that urease activity of studied soils increases with high level of PAHs. For urease activity, a negative correlation with anthracene is observed. The present PAH compound has negative correlation with both of studied enzymes in the affected soils. The influence of PAH level on the soil phytotoxicity (root length) showed the same correlations with urease activity. This fact proves the reliability of received data. It means the particular stimulation of plants growth and enzyme activity under technogenic pollution by PAHs by adapting of soil micro-biota to extreme organic pollution (Table 2).
The abundance of soil bacteria has a negative correlation with PAH level exclude anthracene in affected zone. Anthracene is a special compound for all biological soil activity parameters. It has no relations with abundance of soil bacteria and negatively influences on dehydrogenase, urease and soil phytotoxicity (root length) in the long-term contaminated area while other PAHs has positive correlations with above-listed parameters.
5 Conclusions
The most subjected to technogenic pollution in 2015 were monitoring plots situated through the prevailing wind direction from NPS. It was established that ratio of low- and high-molecular PAH content in soils of monitoring plots is the indicator of technogenic pollution soils. In the soil of monitoring plots located though the predominant wind direction prevails the content of high-molecular PAHs, but in the soils of monitoring plots located around NPS prevails the content of low-molecular PAHs. Soil contamination by PAHs in the affected zone has negative influence at the abundance of soil bacteria. It was observed that 12 from 15 determined PAHs in soil has positive correlation with biological activity parameters of soil, like activity of dehydrogenases, activity of urease and and phytotoxicity level (root length) only for soils of monitoring plots located in the predominant wind direction (1.6 nw, 5.0 nw, 15.0 nw, 20.0 nw). Other monitoring plots located around have a lack of dependence with total and every PAH content because of different relations between high- and low-molecular PAH content in soils.
Thus, in Haplic Chernozems, an increase of the dehydrogenases, urease activity and the phytotoxicity (root length) is observed on the plots in the northwestern predominant wind direction. Relations between PAH concentration and biological activity parameters of soils showed high dependence with total and individual PAH content in 12 studied monitoring plots.
References
Antizar-Ladislao B, Lopez-Real J, Beck A (2006) Degradation of polycyclic aromatic hydrocarbons (PAHs) in an aged coal tar contaminated soil under in-vessel composting conditions. Environ Pollut 141(3):459–468
Atlas RM (1984) Use of microbial diversity measurements to assess environmental stress. Current Perspectives in Microbial Ecology ASM:540–545
Baran S, Bielińska JE, Oleszczuk P (2004) Enzymatic activity in an airfield soil polluted with polycyclic aromatic hydrocarbons. Geoderma 118(3–4):221–232
Demmerová K, Mackova M, Speváková V, Beranová K, Kochánkova L, Lovecká P, Ryslavá E, Macek T (2005) Two approaches to biological decontamination of groundwater and soil polluted by aromatics-characterization of microbial populations. Int Microbiol 8(3):1–12
Directive document 52.10.556-95 (2002) Methodical Instructions. Definition of Polluting Substances in Sediments and Suspension. Roshydromet. Moscow, 22p (in Russian)
Ecological Bulletin of the Don Region (2015) The State of the Environment and Natural Resources in Rostov oblast in 2014 Government of Rostov Region. Rostov-on-Don, 374p (in Russian)
Gabov D, Beznosikov V, Kondratenok B, Yakovleva E (2010) Polycyclic aromatic hydrocarbons in the soils of technogenic landscapes. Geochem Int 48(6):569–579
García C, Hernández T, Costa F (1997) Potential use of dehydrogenase activity as an index of microbial activity in degraded soils. Commun Soil Sci Plant Anal 1–2:123–134
Gennadiev A, Tsibart A (2013) Pyrogenic polycyclic aromatic hydrocarbons in soils of reserved and anthropogenically modified areas: factors and features of accumulation. Eurasian Soil Sci 46(1):28–36
GOST 17.4.1.02.-83 (2004) Nature protection. Soils. Classification of chemical compounds for pollution control. Moscow [in Russian]
GOST 26423-85 (1985) Soils Methods for determination of specific electric conductivity, pH, and dissolved solids. Moscow (in Russian)
Hadibarata T, Kristanti RA (2012) Identification of metabolites from benzo[a]pyrene oxidation by ligninolytic enzymes of polyporus sp. S133. J Environ Manag 111:115–119
Jain RK, Kapur M, Labana S, Lal B, Sarma PM, Bhattacharya D, Thakur IS (2005) Microbial diversity: application of microorganisms for the biodegradation of xenobiotics. Curr Sci 89(1):101–112
Jian Y (2004) Photomutagenicity of 16 polycyclic aromatic hydrocarbons from the US EPA priority pollutant list. Mutat Res 557:99–108
Kazeev K Sh, Kolesnikov SI (2012) Biodiagnostics of soils: methodology and methods of study, Rostov-on-Don (in Russian). 132p
Khaziev F Kh (2005) Methods of soil enzymology. Nauka, Moscow, 217p (in Russian)
Lipińska A, Wyszkowska J, Kucharski J (2015) Diversity of organotrophic bacteria, activity of dehydrogenases and urease as well as seed germination and root growth Lepidium sativum, Sorghum saccharatum and Sinapis alba under the influence of polycyclic aromatic hydrocarbons. Environ Sci Pollut Res 22(23):18519–18530
Liu R, Zhao L, Jin C, Xiao N, Jadeja RN, Sun T (2014) Enzyme responses to phytoremediation of PAH-contaminated soil using Echinacea purpurea (L.) Water Air Soil Pollut 225(12):2230–2237
Margesin R, Walder G, Schinner F (2000a) The impact of hydrocarbon remediation (diesel oil and polycyclic aromatic hydrocarbons) on enzyme activities. Acta Biotechnol 20:313–333
Methodological guidelines for the integrated monitoring of soil fertility of agricultural lands (2003) Moscow, 24p (in Russian)
Minkina T, Sushkova S, Mandzhieva S, Tjurina I, Filonova O (2013) Monitoring researches of the benzo[a]pyrene content in soils under the influence of the technogenic zone. Middle East Journal of Scientific Research 17(1):44–49
Minkina T, Pinsky D, Mandzhieva S, Bauer T, Sushkova S, Kushnareva A (2014) Effect of an attendant anion on the balance of cations in the soil-solution system with an ordinary Chernozem as an example. Eurasian Soil Sci 47(8):772–780
Pascual A, García C, Hernández T, Moreno JL, Ros M (2000) Soil microbial activity as a biomarker of degradation and reclamation processes. Soil Biol Biochem 32(13):1877–1883
Pérez-Leblic MI, Turmero A, Hernández M, Hernández AJ, Pastor J, Ball AS, Arias ME (2012) Influence of xenobiotic contaminants on landfill soil microbial activity and diversity. J Environ Manag 95(Suppl):S285–S290
Procedure of measurements benz(a)pyrene content in soils, sediments and sludges by highly effective liquid chromatography method (2008) Certificate 27–08: Moscow, 27p (in Russian)
Rodríguez-Morgado B, Gómez I, Parrado J, García C, Hernández T, Tejada M (2015) Accelerated degradation of PAHs using edaphic biostimulants obtained from sewage sludge and chicken feathers. J Hazard Mater 300:235–242
Sastre MA, Vicente MC (2003) Contamination and environmental impact on soil biological activity. In: Lobo MC, Ibáñez JJ (eds) Preserving soil quality and soil biodiversity. Instituto Madrileño de Investigación Agraria y Alimentaria (IMIA), Madrid, pp 99–117
Shen G, Lu Y, Zhou Q, Hong J (2005) Interaction of polycyclic aromatic hydrocarbons and heavy metals on soil enzyme. Chemosphere 61(8):1175–1182
Sushkova S, Minkina T, Mandzhieva S, Borisenko N, Vasilyeva G, Kızılkaya R, Aşkın T (2015) Approbation of express-method for benzo[a]pyrene extraction from soils in the technogenic emission zone territories. Eurasian J Soil Sc 4(1):15–21
Sushkova SN, Minkina TM, Mandzhieva SS, Vasilyeva GK, Borisenko NI, Turina IG, Bolotova OV, Varduni TV, Kızılkaya R (2016a) New alternative method of benzo[a]pyreneextractionfrom soils and its approbation in soil under technogenic pressure. J Soils Sediments 16(4):1323–1329
Sushkova S, Minkina T, Turina I, Mandzhieva S, Batuer T, Zamulina I, Kızılkaya R (2016b) Benzo[a]pyrene contamination in Rostov region of Russian Federation: a 10-year retrospective of soil monitoring under the effect of long-term technogenic pollution. Eurasian J Soil Sci 5(2):155–165
Sushkova S, Minkina T, Turina I, Mandzhieva S, Bauer T, Kizilkaya R, Zamulina I (2016c) Monitoring of benzo[a]pyrene content in soils under the effect of long-term technogenic pollution. J Geochem Explor, SI: Soil Pollution. doi:10.1016/j.gexplo.2016.02.009
Sushkova SN, Minkina TM, Mandzhieva SS, Deryabkina IG, Vasil’eva GK, Kızılkaya R (2017) 10.1007/s11368-017-1755-8 Dynamics of benzo[α]pyrene accumulation in soils under the influence of aerotechnogenic emissions. Eurasian Soil Sci 50(1):95-105
The state of food and agriculture (1998) FAO Agriculture Series no. 31. Rome, Italy
Tobiszewski M, Namiesnik J (2012) PAH diagnostic ratios for the identification of pollution emission sources. Environ Pollut 162:110–119
Tsibart A, Gennadiev A (2013) Polycyclic aromatic hydrocarbons in soils: sources, behavior, and indication significance. Eurasian Soil Sci 46(7):728–741
Unbehaun H, Dittler B, Kühne G, Wagenführ A (2000) The impact of hydrocarbon remediation (diesel oil and polycyclic aromatic hydrocarbons) on enzyme activities and microbial properties of soil. Acta Biotechnol 20(3–4):313–333
Wenzl T, Simon R, Kleiner J, Anklam E (2006) Analytical methods for polycyclic aromatic hydrocarbons (PAHs) in food and the environment needed for new food legislation in the European Union. Trend Anal Chem 25(7):716–725
Wu B, Lan T, Lu D, Liu Z (2014) Ecological and enzymatic responses to petroleum contamination. Environmental Sciences: Processes and Impacts 16(6):1501–1509
Zvyagintsev DG (1978) Biological activity of soils and scale for assessment of some parameters. Pochvovedenie 6:705–713 (in Russian)
Zvyagintsev DG (ed) (1991) Methods of soil microbiology and biochemistry, Moscow State University. Moscow, 432p (in Russian)
Acknowledgements
This research was supported by a project of the Ministry Education and Science of Russia, no. 5.948.2017/PCh, Grant of President of Russian Federation no. MK-3476.2017.5, RFBR no. 16-35-60051, 16-35-00347. Analytical work was carried out on the equipment of centers for collective use of Southern Federal University “High Technology,” and “Biotechnology, Biomedical and Environmental Monitoring.”.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Claudio Bini
Rights and permissions
About this article
Cite this article
Sushkova, S.N., Minkina, T., Deryabkina (Turina), I. et al. Influence of PAH contamination on soil ecological status. J Soils Sediments 18, 2368–2378 (2018). https://doi.org/10.1007/s11368-017-1755-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-017-1755-8