Abstract
Purpose
The main objectives of this research are to decipher the stoichiometric characteristics of carbon (C), nitrogen (N), and phosphorus (P) in soils from the alpine ecosystem and to obtain information about nutrient limitation on plants and microbes.
Materials and methods
The soils were sampled along an altitudinal gradient (2000 to 4300 m above sea level) from the eastern slope of Gongga Mountain in eastern Tibetan Plateau. In total of 102 soil samples in profiles and 27 soil microbial biomass (SMB) samples from five vegetation zones were collected to analyze the concentrations of C, N, and P as well as their ratios. The concentrations of C and N were measured using an automated C/N analyzer, total P was detected by inductively coupled plasma-atomic emission spectrometer, and the concentrations of microbial biomass C, N, and P were measured by the chloroform fumigation-extraction method. Soil P fractions were extracted by modified Hedley sequential extraction method.
Results and discussion
The concentrations of C, N, and P in the soils and SMB varied spatially, whereas the variation of their ratios was constrained. The C:N:P ratios were 556:22:1 for the O horizon, 343:16:1 for the A horizon, 154:7:1 for the B horizon, and 63:3:1 for the C horizon, indicating a significant decrease with depth. The mean ratio in the SMB was 51:6.6:1. Microbial biomass C, N, and P were important components of soil nutrients, especially the microbial biomass P which accounted for 40.8 % of soil available P. The C:P and N:P were higher in the soils of broadleaf-coniferous and coniferous forests, whereas the ratios in the SMB were higher in the broadleaf forest. The ratios of C and N to available P in the soils decreased significantly with altitude.
Conclusions
The local climate, vegetation succession, and soil development in the high mountain resulted in the soil nutrient cycling different from that in other terrestrial ecosystems. Among the different vegetation zones, the P-limitation of plants and microbial communities might be possible in the soils of lower land forests in the long term.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Whether a similar stoichiometric ratio of carbon (C), nitrogen (N), and phosphorus (P) exists across terrestrial ecosystems has been explored to understand their biogeochemical processes and nutrient limitation (Elser et al. 2000; McGroddy et al. 2004; Mooshammer et al. 2012; Beermann et al. 2015). Unlike marine ecosystems, terrestrial ecosystems are more complex due to the various conditions (e.g., topography, vegetation, human intervene, etc.), and hence, result in a large spatial heterogeneity of biogenic element distribution and their ratios. Aponte et al. (2010) concluded that the season, vegetation type, and soil depth were the main factors affecting the stoichiometry of C, N, and P in the soils of Mediterranean forests. Li et al. (2012) found that the variation of C:N:P ratios in the soils of subtropical China was closely related to landscape and land use. The research of Tian et al. (2010) showed that the variations of C:P and N:P in Chinese soils were attributed to different climatic characteristics, soil orders, soil depth, and weathering stages. In contrast, Cleveland and Liptzin (2007) reported a well-constrained C:N:P ratio in soil microbial biomass (SMB). Kirkby et al. (2011) obtained a constant stoichiometric ratio of C, N, and organic P in the stable portion of soil organic materials across a wide range of global soils. These researchers suggested that the stoichiometric ratio of C, N, and P in SMB may be homeostatic, whereas the ratio in soils exhibits a marked spatial heterogeneity in terrestrial ecosystems. To date, there has been limited research concerning the stoichiometry of C, N, and P in alpine ecosystems where the storage of nutrients is increasingly important with global warming and increasing N deposition. Mountain soils are an important pool of nutrients. Due to the hasher climatic conditions at high altitudes, the nutrients in mountain soils are facing high vulnerability, which not only modulates climatic warming through C dynamics in soils (Hagedorn et al. 2010) but also affects ecosystem functioning (e.g., nutrient limitation). Therefore, the research on biogeochemical cycling of C, N, and P in alpine soils is important for local and regional ecological stability.
The stoichiometry of C, N, and P is a powerful tool to decipher their coupling mechanisms and nutrient limitation in terrestrial ecosystems (Aponte et al. 2010; Kirkby et al. 2011; Ågren et al. 2012; Ostrowska and Porębska 2015). Many researchers have attempted to establish the critical value of foliar N:P to reveal nutrient limitation in terrestrial ecosystems (Koerselman and Meuleman 1996; Bennett and Adams 2001; Güsewell 2004), although some debates still exist in species-specific ecosystems (Drenovsky and Richards 2004; Kerkhoff et al. 2005; Craine et al. 2008). The research of potential nutrient limitation by the stoichiometry of C, N, and P in soils of alpine ecosystems is far from complete. The high mountains are characterized by large altitudinal gradients, which form special climate conditions and vegetation distribution. Climate, plant community, and soil development are regarded as major factors controlling soil nutrient availability by changing soil physiochemical properties and litter quality (Binkley et al. 2000; De Kovel et al. 2000; Chen et al. 2008). Furthermore, microbes, an important component in soils, regulate key processes of nutrient cycling through litterfall decomposition, mineralization, and immobilization (Ushio et al. 2010). On the other hand, the development and activity of microbes also respond to nutrient levels in soils as well as the environmental variation (Wan et al. 2015). Therefore, the stoichiometry of C, N, and P in soils and SMB from alpine ecosystems can improve our understanding of the nutrient availability and their potential limitation.
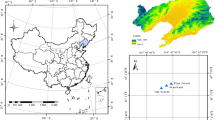
Gongga Mountain (29° 20′–30° 20′ N, 101° 30′–102° 15′ E), with the peak of 7556 m a.s.l. (above sea level), is located at the eastern border of the Tibetan Plateau (Thomas 1999). The eastern slope of Mt. Gongga is characterized by high mountain-deep valley (1100–7556 m a.s.l.), a transitional seasonal climate and typical pristine forests, which offers an ideal area to investigate the stoichiometry of C, N, and P. Previous research found that the soil development on the eastern slope of Mt. Gongga was in the early stage (He and Tang 2008). In the present study, five vegetation zones along an altitudinal gradient (2000–4300 m a.s.l.) were selected on the eastern slope of Mt. Gongga, including broadleaf forest (BLF), broadleaf and coniferous forest (BCF), coniferous forest (CF), shrub, and meadow (Fig. 1). The objectives of this work are (1) to quantify the spatial distribution of C, N, and P concentrations as well as their ratios in the soils and SMB; (2) to identify the possible factors influencing the stoichiometry of C, N, and P in the soils; and (3) to provide evidence for nutrient limitation among different vegetation zones using the C:N:P stoichiometry.
2 Materials and methods
2.1 Study area
Soil sampling was conducted on the eastern slope of Mt. Gongga within the Hailuogou Glacier valley (Fig. 1), where the climate is typical temperate monsoon. The main components of parent materials are residual deposit and slope wash deriving from weathered Cenozoic feldspar granite and Permian quartz schist. There are many debris flow gullies and deposits developed by frequent debris flow (Zhang et al. 2006). The geographical features, meteorology, and vegetation distribution with altitude are summarized in Table 1.
2.2 Field sampling
After preliminary survey on topography, altitude gradients and vegetation, the sampling sites were selected to include all vegetation zones (Table 1). The soil sampling was conducted in September 2010 at nine altitudes: 2032, 2772, 2856, 3090, 3544, 3614, 3896, 4015, and 4221 m a.s.l. (Fig. 1). At each site, three soil profiles were hand-dug until the bedrock was reached. Four soil units were divided according to the primary aspects. The O horizon represents the soils with brown color and decomposition/semi-decomposition organic matter; the A horizon represents the soils with dark brown color and humus; the B horizon represents the soils with illuvial and/or eluvial materials; and the C horizon is the soil parent materials. Due to the heterogeneous development of the soils, the B horizon at the altitude of 2856 m a.s.l. and the O horizon at the altitude of 4221 m a.s.l. were absent. In total, 102 soil samples including 24 samples from the O horizon, 27 samples from the A horizon, 24 samples from the B horizon, and 27 samples from the C horizon were collected.
The soil samples of the SMB were also collected in the five vegetation zones (at the same sites to the bulk soil samples, respectively), and the surface soils (0–15 cm) were used to analyze the concentrations of microbial biomass C, N, and P (MBC, MBN, and MBP). In total, nine sites and 27 soil samples (three replicates at each site) for the SMB analysis were obtained (Fig. 1).
Each soil sample was put into a polyethylene plastic bag and brought to the laboratory in an icebox. The soil samples were sieved to <2 mm to remove the large rocks and plant remains. The wet soil samples for the measurements of MBC, MBN, and MBP as well as soil moisture were immediately treated after the sieving. The other soil samples for physical and chemical analysis were stored at 4 °C, and then air-dried for analysis of the concentrations of soil organic carbon (SOC), N, and P.
2.3 Chemical analysis
The SOC and N concentrations were measured using a FlashEA1112 elemental analyzer linked to a Thermo DeltaPlus Advantage mass spectrometer after the soils were treated by 5 % HCl to remove carbonates. The standard reference material (GSS-11) was used during the measurement of SOC and N, and the standard deviations were <10 % of the certified value. The soils for analyzing total P concentration were digested with HNO3-HF-HClO4, and the P concentration was measured using an American Leeman Labs Profile inductively coupled plasma-atomic emission spectrometer. Standard solution SPEXTM from the USA was used as the standard. Quality control was assured by the analysis of duplicate samples, blanks, and reference materials (GSD-9 and GSD-11, Chinese geological reference materials). The precision was good with variability in repeated analysis of samples and reference materials below 5 %. Recovery was 90 ± 6 % (error expressed as 95 % confidence interval) for the reference materials.
The concentrations of MBC, MBN, and MBP were measured by the chloroform fumigation-extraction method (Brookes et al. 1985; Vance et al. 1987). For the measurements of MBC and MBN, 10 g of the moist soil samples were extracted using 40 mL of 0.5 M K2SO4 for 30 min by shaking at 300 rpm. Meanwhile, the other soil subsamples (10 g) were fumigated with chloroform for 24 h in a vacuum desiccator, followed by the same extraction procedure as the unfumigated samples. The concentrations of C and N in the extracts were measured by the C/N analyzer. The concentrations of MBC and MBN were estimated as the difference in K2SO4-extractable dissolved organic C and N between fumigated and unfumigated soils using the correction factors: KC = 0.45 for C and KN = 0.40 for N (Jonasson et al. 1996; Rinnan et al. 2008). For the measurement of MBP, 5 g of the moist soil samples were extracted by 100 mL of 0.025 N HCl and 0.03 N NH4F for 30 min by shaking at 300 rpm. The other soil subsamples (5 g) were fumigated with chloroform for 24 h in a vacuum desiccator, followed by the same extraction procedure as the unfumigated samples. The concentration of MBP in the extracts was measured using the Molybdate colorimetric method. It was estimated as the difference of available P between the fumigated and unfumigated soils using a correction factor KP = 0.40 (Brookes et al. 1982).
The soil P fractions in the A horizon were detected by the modified Hedley sequential extraction method (Tiessen and Moir 1993). This method uses a sequence of increasingly strong reagents to successively remove more recalcitrant fractions of inorganic (Pi) and organic (Po) P. The available P is defined as the sum of resin-extractable Pi (resin-Pi), HCO3 − extractable Pi (HCO3 − Pi), and HCO3 − extractable Po (HCO3 − Po).
2.4 Statistical analysis
One-way ANOVA (Fisher test, p < 0.05) was used to identify the significant differences in the mean concentrations of C, N, and P and their ratios at each soil horizon among different vegetation zones. In order to discuss the effects of vegetation types on the stoichiometry of C, N and P, the ANOVA was also applied to analyze the altitudinal differences of the stoichiometric ratios of C, N, and P in the soils and SMB. The results showed the same trend as the data compiled based on the form of vegetation zones. Thus, the variation of the stoichiometric ratios of C, N, and P in soils among different vegetation zones was presented in the text (Fig. 2). Linear fit was applied to establish the relationship between elements and their ratios with altitude (or different vegetation zones). All statistical analysis in this study was performed using the software package Origin 8.0 for Windows.
The concentrations and atomic ratios of SOC, N, and P in the soils. The different letters represent the significant difference of the means at each soil horizon among the vegetation zones (p < 0.05), whereas the same letter (or no letter) indicates insignificant difference. The error bars represent the standard error
3 Results
3.1 The concentrations and ratios of SOC, N, and P in soils
As shown in Table 2, the concentrations of SOC and N in the soil profiles decreased significantly with depth, whereas the variation of P concentrations was little. Among the vegetation zones, the concentrations of SOC at each soil horizon were generally higher in the CF zone, followed by the BCF, shrub, and BLF zones, and the lowest concentration was observed in the meadow zone (Fig. 2). Except in the O horizon, the N concentrations were not significantly different among the vegetation zones. The concentrations of P varied insignificantly in the C horizon indicating a uniform background of P in the soils of the eastern slope of Mt. Gongga. However, the P concentrations in other horizons were markedly higher in the soils of shrub and meadow zones than of other zones.
The atomic ratios of C:N, C:P, and N:P varied in a constrained magnitude compared with their concentrations (Table 2). These ratios were significantly higher in the O horizon than in other horizons, respectively, and the ratios of C:P and N:P showed a marked decreasing trend with depth. Specifically, the C:N:P ratio was 556:22:1 for the O horizon, 343:16:1 for the A horizon, 154:7:1 for the B horizon, and 63:3:1 for the C horizon, with the mean of 279:12:1 in the soils. Among the vegetation zones, the C:N at each soil horizon was notably higher in the CF zone (Fig. 2). The ratios of C:P and N:P were significantly higher in the BCF and CF zones followed by the shrub and BLF zones, and the lowest was observed in the meadow zone.
In the A horizon, the available P (Av_P) concentrations varied between 2.5 and 15.9 mmol/kg with the mean of 7.6 mmol/kg (Table 2). The atomic ratios of C:Av_P and N:Av_P in the A horizon were 1870 and 91.6 on average, respectively. Spatially, both ratios of C:Av_P and N:Av_P in the A horizon showed a marked decreasing trend with altitude (or from lower-land to higher-land vegetation zones) (Fig. 3).
3.2 The concentrations and ratios of C, N, and P in the SMB
The concentrations of MBC, MBN, and MBP were much lower than those in the soils (Table 2). Among the vegetation zones, the concentrations of MBC were significantly higher in the BLF, BCF, and CF zones than in the shrub and meadow zones (Fig. 4). The MBN concentrations did not show marked difference among the vegetation zones, whereas they decreased gradually with altitude. The MBP concentrations increased from the BLF to the shrub, but decreased considerably in the meadow zone.
The atomic ratio of C:N:P in the SMB was 51:6.6:1 on average (Table 2). The relatively high C:N in the SMB was observed in the BCF and CF zones, although the difference was not significant among the vegetation zones (Fig. 4). The similar variation of C:P and N:P was observed in the SMB among the vegetation zones, and these ratios were significantly higher in the BLF zone.
4 Discussion
4.1 Possible factors affecting the stoichiometry of C, N, and P in the soils
Many researchers have reported the stoichiometry of C, N, and P in different terrestrial ecosystems (Table 3). Compared with these results, our analysis showed much higher C:P in the O and A horizons, but comparable N:P with global forest soils and grassland soils. For the ratios in the SMB, a little lower C:P was observed on the eastern slope of Mt. Gongga, whereas the N:P was similar to those of Chinese and global soils. The difference of C:N:P stoichiometry between the alpine and other terrestrial ecosystems is mainly attributed to the complex conditions in alpine ecosystems, which are currently experiencing strong climatic warming, more precipitation and anthropogenic impacts (Hagedorn et al. 2010). Soil formation, changes of vegetation cover and atmospheric N deposition during last decades have intensive impact on nutrient biogeochemical process in mountain soils even within a short distance.
4.1.1 Climate
Climate plays an important role in soil development and consequently nutrient cycling and availability (Oleksyn et al. 2003; Dijkstra et al. 2012). An increase in temperature can enhance N availability to plants with increased microbial activity in soils (Melillo et al. 2002). In the alpine ecosystem, the temperature markedly decreased with increasing altitude (Table 1). Correspondingly, the concentrations of MBC and MBN were higher in the forests of the lower altitude, indicating the sensitivity of microbes to temperature and then their effect on nutrient cycling. However, the concentrations of C, N, and P in the soils of Mt. Gongga were not significantly higher in the lower land forests. This could be attributed to the influence of precipitation as well as the soil moisture. The higher precipitation and soil moisture can increase soil weathering, and then lead to rapid release of P from parent materials.Footnote 1 Under the high precipitation and soil moisture (Table 1), the higher ratios of C:P and N:P were observed in the soils of the BCF and CF zones (Fig. 2). On the one hand, the higher precipitation increases organic matter production, and thus leads to higher organic matter content (high SOC and N concentrations) in the soils. On the other hand, the high P leaching or low P concentrations in decomposers may exist under the higher precipitation and humid conditions (Manzoni et al. 2010). In addition, the higher soil moisture can increase the accessibility of nutrients, enhance microbial growth, and consequently, induce N and P immobilization in microbial biomass and plants (Nielsen et al. 2009; Aponte et al. 2010).
4.1.2 Plant species
Plants regulate the soil stoichiometry through nutrient uptake, litterfall inputs, and root exudates. Plants can control their homeostatic stoichiometry under the stable environment but not in the variable environment (Sardans et al. 2012). Meanwhile, the physiological strategies of plants to optimize use of potentially limiting nutrients, such as nutrient resorption, leaf turnover rate, and nutrient investment in biomass, can induce various changes of C, N, and P in soils (McGroddy et al. 2004). Thus, the difference of plant communities may change nutrient distribution in soils. On the eastern slope of Mt. Gongga, five different vegetation zones have formed along the large altitude gradients (Fig. 1). The stoichiometric variation of C, N, and P in the soils, especially for the significantly lower P in the BLF, BCF, and CF zones (Fig. 2), evidenced the “pumping” effects of plants. The large amounts of biomass and production have been observed in the lower land forests of Mt. Gongga, especially in the BCF zone (Luo et al. 2000), confirming that the plants indeed modify the nutrient distribution in the soils through the biomass production. Furthermore, Shen et al. (2004) reported that the richness of plant species on the eastern slope of Mt. Gongga decreased with the increasing altitude. The diversity of plant species in the lower land forests could increase the competition of organisms for P, which was evident from the relatively higher ratios of C:P and N:P in the soils.
Another effect of plants on the C:N:P stoichiometry in soils is related to the quality and quantity of litterfall inputs from different plant species. The inputs of litterfall can modify community structure and microbial biomass (Yeates and Saggar 1998; Ehlers et al. 2010) and then the decomposition processes (Rutigliano et al. 2004; Aponte et al. 2010). The quantity of litterfall can change the contents of soil organic matter and total soil N, and subsequently, influence the microbial nutrient level (Kara et al. 2008; Rinnan et al. 2008). The deciduous trees provide nutrient richer litterfall to microbial growth. Luo et al. (2003) found much more litterfall inputs in the BLF zones (3810 kg/hm2/year) than in the CF zones (2810 kg/hm2/year), and they also observed higher N and P concentrations returned from litterfall in the BLF zones than in the CF zones. Combined with the higher temperature at the lower altitude, the higher litterfall inputs are conducive to microbial decomposition processes through high energy supply. Our data supported this assertion. Compared with the CF zone, the C:P and N:P ratios in the BLF zone were higher in the SMB but lower in the soils (Figs. 2 and 4), which suggested a high transformation rate of P by microbes.
The higher ratios of C:P and N:P in the soils of BCF and CF zones are also related to the root exudes including acid phosphatases, low-molecular-weight organic acid, and proton (Wu et al. 2013b), which can acidify the soils and then modify the soil nutrient solubility. The coniferous trees have been found to secret much more acid exudes than broadleaf trees (Raulund-Rasmussen and Vejre 1995). In the CF zone, the soil pH is markedly lower than that in other zones (Table 1), indicating that the soil acidification by root exudes would increase soil weathering and P solubility. These exudes have been reported to improve nutrient mobility and availability in soils, and then increase their uptake by organisms (Wang et al. 2008; Carvalhais et al. 2011; Richardson et al. 2011).
4.1.3 Soil development
Soil development (e.g., substrate age and weathering intensity) can change nutrient availability and then the C:N:P ratios in soils (Frizano et al. 2002; Reich and Oleksyn 2004). Tian et al. (2010) found that soil C:N ratios increased significantly with soil weathering intensity, and the stronger the soil weathering the higher C:P and N:P ratios. Although there is a similar parent material in the soils on the eastern slope of Mt. Gongga, the soil development is not consistent along the large altitudinal gradients, which was well observed by the soil physicochemical properties in profiles (Table 2). The strongest weathering of the soils should occur in the forests of lower altitude due to the abundant precipitation and the complex plant communities (Table 1), and correspondingly, we observed a notably higher ratios of C:P and N:P in the BCF and CF zones (Fig. 2). Meanwhile, the soil physicochemical properties (e.g., soil organic matter, pH, and soil texture) on the eastern slope of Mt. Gongga have been largely changed with the soil development (Tables 1 and 2). This not only modified indirectly the stoichiometry of C, N, and P in the soils, but also affected microbial development and then the nutrient mineralization (Hassink 1994). However, compared with the BCF and CF zones, the much lower C:P and N:P ratios were observed in the BLF zone. This was not an issue of the P weathering due to the same concentrations in the soils of the three zones (Fig. 2). On the one hand, the faster remineralization of SOC and N to CO2 and N2, respectively, could happen in the BLF zone than in the BCF and CF zones under the better climate conditions, and thus, removed their pools in the soils. On the other hand, the C and N in the soils may be taken up faster by the broadleaf trees in the BLF.
4.1.4 Microbial effect
Compared with the concentrations of C, N, and P in the A horizon, the concentrations of MBC, MBN, and MBP accounted for the corresponding concentrations in the soils of 0.8 %, 2.0 %, and 8.4 %, respectively, which was similar to the mean estimates of 1.2 %, 2.6 %, and 8.0 % globally (Xu et al. 2013). Meanwhile, the proportion of MBP to the available P in the A horizon was 40.8 %. This suggested that soil microbial biomass was an important pool of P in the soils, and microorganisms were actively involved in the P biogeochemical cycling as well as the P supply for plants (Turner et al. 2013; Mooshammer et al. 2014). The decrease of microbial population can release available P for plant growth, whereas its increase will be associated with strong competition for this grow-limiting resource (Lipson et al. 1999; Schmidt et al. 2007). The ratio of C:Po is often used to estimate the mineralization potential of organic P, and the mineralization occurs at C:Po below 200, whereas the immobilization process dominates when it is up to 300 (ChacÓn et al. 2005). This ratio varied between 92.5 and 804 with the mean of 460 in the A horizon, which indicated that the uptake and immobilization of P by microbes dominated in the soils. Compared with other vegetation zones, the relatively lower C:Po ratios were observed in the lower land forests (BLF and BCF), which suggested that the organic P mineralization was more favored in these zones under the strong microbial activity discussed above. Additionally, we also analyzed the soil microbial C:N:P ratios in May 2010 (beginning of growing season) with the mean of 48:9:1 (unpublished data), which was distinctly different from the mean of 51:6.6:1 in September 2010 (late growing season). This discrepancy confirmed that microbes did compete with plants for soil nutrients in the growing season.
4.2 Implication for nutrient limitation
Researchers have proposed the critical N:P ratios to indicate N- or P-limitation for terrestrial ecosystems using different plant tissues. For example, the critical N:P ratio in plant leaf has been applied to indicate N-limitation (N:P < 14) and P-limitation (N:P > 16) (Aerts and Chapin Iii 1999; Reich and Oleksyn 2004), and Güsewell (2004) suggested N:P < 10 and > 20 to correspond to N- and P-limited biomass production, respectively, by short-term fertilization experiment. On the eastern slope of Mt. Gongga, the foliar N:P of dominant plants in different vegetation zones decreased prominently with altitude, and the relatively high values were observed in the low altitude (N:P > 17.0 and > 20 on 2362 m and 1750 m a.s.l., respectively, unpublished data). According to the definition above, the P-limitation is possible in the lower land forests. However, the foliar N:P ratio is not simply a function of soil availability, and the variation of climate, tree species and growth rates, soil order, and others can change the foliar chemistry and then the ratio (Güsewell 2004; Townsend et al. 2007; Elser et al. 2010). Soils, especially the surface soils, have the most active organism-environment interaction and are highly involved in the biogeochemical cycling of nutrients in terrestrial ecosystems (Tian et al. 2010; Izquierdo et al. 2013). Therefore, the nutrient limitation of ecosystems should be roundly investigated by the C:N:P ratios in soils and SMB.
On the eastern slope of Mt. Gongga, the C:N was significantly higher in the soils of the CF zone (C:N > 20) despite the soil horizon, and the ratios of C:P and N:P were much higher in the BCF and CF zones (Fig. 2). Meanwhile, the ratios of C:Av_P and N:Av_P in the A horizon (Table 2), lower than those in Chinese soils (Tian et al. 2010), decreased notably with altitude (Fig. 4). These evidences suggested that with the pedogenesis P becomes ecologically scarce. In the Mediterranean oak forest soils, Aponte et al. (2010) found the higher proportion of N (C:N = 7.6) and the lower fraction of P (C:P = 93.8) indicating the P-limitation in the soils. Izquierdo et al. (2013) reported that the N:P of mineral soils increased significantly during pedogenesis and pointed to P as the primary limiting nutrient in the forest ecosystem. Furthermore, tree species are responsible for the variation of nutrient stoichiometry in soils (Wan et al. 2015). Huang et al. (2013) found that the P limitation increased during forest succession on the Dinghu Mountain, southeastern China. However, our results suggested the possible P limitation in the soils of BCF and CF zones. This difference was attributed to the complex conditions in the alpine ecosystem. On the one hand, the climatic conditions are markedly different between the southeastern and southwestern China. As mentioned above, the abundant precipitation was observed around 3000 m a.s.l. on the eastern slope of Mt. Gongga corresponding to the BCF and CF zones (Table 1), which modulated the nutrient availability and microbial biomass. On the other hand, the differences of plant species and diversity can change nutrient-use efficiency and competition for nutrients. Compared with the Dinghu Mountain (pine forests, mixed pine and broadleaf forests, and monsoon evergreen broadleaf forests), the vegetation composition is much more complicated on the Mt. Gongga (Fig. 1).
The C:N:P ratios in the SMB showed different patterns from those in the soils, which were related to the microbial homeostasis (Mooshammer et al. 2014). The direct evidence was that there was no significant difference of the C:P and N:P ratios among the vegetation zones except the BLF zone (Fig. 4). Compared with the results of Cleveland and Liptzin (2007) (mean microbial biomass N:P ratio of 6.9), our results showed a similar N:P ratio of 6.6 in the SMB. This seemed to indicate little nutrient limitation to microorganisms on the eastern slope of Mt. Gongga. However, the obviously higher ratios of C:P and N:P in the SMB of the BLF zone suggested the potential P-limitation for plants.
5 Conclusions
The concentrations of C, N, and P in the soils and SMB exhibited spatial heterogeneity on the eastern slope of Mt. Gongga, whereas the variations of their ratios were constrained. The soil stoichiometric ratios of C, N, and P on the eastern slope of Mt. Gongga were different from those in other regions of the world. The local climate, vegetation variation, and soil development played a key role in the soil nutrient stoichiometry. Microbial biomass C, N, and P were important components of soil nutrients, especially the microbial biomass P content which accounted for 40.8 % of the soil available P. The ratios of C and N to the available P also increased with the decreasing altitude. According to the C:N:P stoichiometry, the P-limitation of plants was possible in the soils of broadleaf-coniferous and coniferous forests, whereas the limitation of microbial biomass existed in the soils of broadleaf forests. Our data would provide supplementary information for the stoichiometry of C, N, and P in global terrestrial ecosystems.
Notes
Zhou J (2014) Weathering, pedogenesis and changes of soil phosphorus speciation of Hailuogou Glacier foreland chronosequence. (unpublished dissertation)
References
Aerts R, Chapin Iii FS (1999) The mineral nutrition of wild plants revisited: A re-evaluation of processes and patterns. In: Fitter AH, Raffaelli DG (eds) Advances in Ecological Research, vol Volume 30. Academic Press, pp 1–67
Ågren GI, Wetterstedt JÅM, Billberger MFK (2012) Nutrient limitation on terrestrial plant growth—modeling the interaction between nitrogen and phosphorus. New Phytol 194:953–960
Aponte C, Marañón T, García L (2010) Microbial C, N, and P in soils of Mediterranean oak forests: influence of season, canopy cover and soil depth. Biogeochemistry 101:77–92
Beermann F, Teltewskoi A, Fiencke C, Pfeiffer EM, Kutzbach L (2015) Stoichiometric analysis of nutrient availability (N, P, K) within soils of polygonal tundra. Biogeochemistry 122:211–227
Bennett LT, Adams MA (2001) Response of a perennial grassland to nitrogen and phosphorus additions in sub-tropical, semi-arid Australia. J Arid Environ 48:289–308
Binkley D, Giardina C, Bashkin MA (2000) Soil phosphorus pools and supply under the influence of Eucalyptus saligna and nitrogen-fixing Albizia facaltaria. For Ecol Manag 128:241–247
Brookes PC, Powlson DS, Jenkinson DS (1982) Measurement of microbial biomass phosphorus in soil. Soil Biol Biochem 14:319–329
Brookes PC, Landman A, Pruden G, Jenkinson DS (1985) Chloroform fumigation and the release of soil nitrogen: a rapid direct extraction method to measure microbial biomass nitrogen in soil. Soil Biol Biochem 17:837–842
Carvalhais LC, Dennis PG, Fedoseyenko D, Hajirezaei MR, Borriss R, von Wirén N (2011) Root exudation of sugars, amino acids, and organic acids by maize as affected by nitrogen, phosphorus, potassium, and iron deficiency. J Plant Nutr Soil Sci 174:3–11
ChacÓn N, Dezzeo N, MuÑoz B, RodrÍGuez JM (2005) Implications of soil organic carbon and the biogeochemistry of iron and aluminum on soil phosphorus distribution in flooded forests of the lower Orinoco River, Venezuela. Biogeochemistry 73:555–566
Chen CR, Condron LM, Xu ZH (2008) Impacts of grassland afforestation with coniferous trees on soil phosphorus dynamics and associated microbial processes: a review. For Ecol Manag 255:396–409
Cleveland C, Liptzin D (2007) C:N:P stoichiometry in soil: is there a “Redfield ratio” for the microbial biomass? Biogeochemistry 85:235–252
Craine JM, Morrow C, Stock WD (2008) Nutrient concentration ratios and co-limitation in South African grasslands. New Phytol 179:829–836
De Kovel CGF, Van Mierlo AEM, Wilms YJO, Berendse F (2000) Carbon and nitrogen in soil and vegetation at sites differing in successional age. Plant Ecol 149:43–50
Dijkstra FA, Pendall E, Morgan JA, Blumenthal DM, Carrillo Y, LeCain DR, Follett RF, Williams DG (2012) Climate change alters stoichiometry of phosphorus and nitrogen in a semiarid grassland. New Phytol 196:807–815
Drenovsky R, Richards J (2004) Critical N:P values: predicting nutrient deficiencies in desert shrublands. Plant Soil 259:59–69
Ehlers K, Bakken LR, Frostegård Å, Frossard E, Bünemann EK (2010) Phosphorus limitation in a Ferralsol: impact on microbial activity and cell internal P pools. Soil Biol Biochem 42:558–566
Elser JJ, Sterner RW, Gorokhova E, Fagan WF, Markow TA, Cotner JB, Harrison JF, Hobbie SE, Odell GM, Weider LW (2000) Biological stoichiometry from genes to ecosystems. Ecol Lett 3:540–550
Elser JJ, Fagan WF, Kerkhoff AJ, Swenson NG, Enquist BJ (2010) Biological stoichiometry of plant production: metabolism, scaling and ecological response to global change. New Phytol 186:593–608
Frizano J, Johnson AH, Vann DR, Scatena FN (2002) Soil phosphorus fractionation during forest development on landslide scars in the Luquillo Mountains, Puerto Ricol. Biotropica 34:17–26
Gao SH, Peng JW (1993) Research of climate in the Gongga Mountain. In: Chen FB, Gao SH (eds) Studies on the alpine ecology and environment of Gongga Mountain. University of Science and Technology Press, Chengdu, pp 80–86
Griffiths BS, Spilles A, Bonkowski M (2012) C:N:P stoichiometry and nutrient limitation of the soil microbial biomass in a grazed grassland site under experimental P limitation or excess. Ecol Process 1:6
Güsewell S (2004) N:P ratios in terrestrial plants: variation and functional significance. New Phytol 164:243–266
Hagedorn F, Mulder J, Jandl R (2010) Mountain soils under a changing climate and land-use. Biogeochemistry 97:1–5
Hassink J (1994) Effect of soil texture on the size of the microbial biomass and on the amount of C and N mineralized per unit of microbial biomass in Dutch grassland soils. Soil Biol Biochem 26:1573–1581
He L, Tang Y (2008) Soil development along primary succession sequences on moraines of Hailuogou Glacier, Gongga Mountain, Sichuan, China. Catena 72:259–269
Huang WJ, Liu JX, Wang YP, Zhou GY, Han TF, Li Y (2013) Increasing phosphorus limitation along three successional forests in southern China. Plant Soil 364:181–191
Izquierdo J, Houlton B, Huysen T (2013) Evidence for progressive phosphorus limitation over long-term ecosystem development: examination of a biogeochemical paradigm. Plant Soil 1–13
Jonasson S, Michelsen A, Schmidt I, Nielsen E, Callaghan T (1996) Microbial biomass C, N and P in two arctic soils and responses to addition of NPK fertilizer and sugar: implications for plant nutrient uptake. Oecologia 106:507–515
Kara Ö, Bolat İ, Çakıroğlu K, Öztürk M (2008) Plant canopy effects on litter accumulation and soil microbial biomass in two temperate forests. Biol Fertil Soils 45:193–198
Kerkhoff AJ, Enquist BJ, Elser JJ, Fagan WF (2005) Plant allometry, stoichiometry and the temperature-dependence of primary productivity. Glob Ecol Biogeogr 14:585–598
Kirkby CA, Kirkegaard JA, Richardson AE, Wade LJ, Blanchard C, Batten G (2011) Stable soil organic matter: a comparison of C:N:P:S ratios in Australian and other world soils. Geoderma 163:197–208
Koerselman W, Meuleman AF (1996) The vegetation N:P ratio: a new tool to detect the nature of nutrient limitation. J Appl Ecol 3(6):1441–1450
Li Y, Wu JS, Liu SL, Shen JL, Huang DY, Su YR, Wei WX, Syers JK (2012) Is the C:N:P stoichiometry in soil and soil microbial biomass related to the landscape and land use in southern subtropical China? Global Biogeochem Cy 26, GB4002
Lipson DA, Schmidt SK, Monson RK (1999) Links between microbial population dynamics and nitrogen availability in an alpine ecosystem. Ecology 80:1623–1631
Luo J, Yang Z, Yang QW (2000) A study on the biomass and production of forest on the Gongga Mountain. Acta Phytoecological Sin 24:191–196
Luo J, Chen GW, Chen BR, Li W (2003) Characteristic of forests litterfall along vertical spectrum on the Gongga Mountain. Mountain Research 21:287–292
Manzoni S, Trofymow JA, Jackson RB, Porporato A (2010) Stoichiometric controls on carbon, nitrogen, and phosphorus dynamics in decomposing litter. Ecol Monogr 80:89–106
McGroddy ME, Daufresne T, Hedin LO (2004) Scaling of C:N:P stoichiometry in forests wordwide: implications of terrestrial Redfield-type ratios. Ecology 85:2390–2401
Melillo J, Steudler P, Aber J, Newkirk K, Lux H, Bowles F, Catricala C, Magill A, Ahrens T, Morrisseau S (2002) Soil warming and carbon-cycle feedbacks to the climate system. Science 298:2173–2176
Mooshammer M, Wanek W, Schnecker J, Wild B, Leitner S, Hofhansl F, Blochl A, Hammerle I, Frank A, Fuchslueger L, Keibinger K, Zechmeister-Boiltenstern S, Richter A (2012) Stoichiometric controls of nitrogen and phosphorus cycling in decomposing beech leaf litter. Ecology 93:770–782
Mooshammer M, Wanek W, Zechmeister-Boltenstern S, Richter A (2014) Stoichiometric imbalances between terrestrial decomposer communities and their resources: mechanisms and implications of microbial adaptations to their resources. Front Microbiol 5:22
Nielsen PL, Andresen LC, Michelsen A, Schmidt IK, Kongstad J (2009) Seasonal variations and effects of nutrient applications on N and P and microbial biomass under two temperate heathland plants. Appl Soil Ecol 42:279–287
Oleksyn J, Reich PB, Zytkowiak R, Karolewski P, Tjoelker MG (2003) Nutrient conservation increases with latitude of origin in European Pinus sylvestris populations. Oecologia 136:220–235
Ostrowska A, Porębska G (2015) Assessment of the C/N ratio as an indicator of the decomposability of organic matter in forest soils. Ecol Indic 49:104–109
Raulund-Rasmussen K, Vejre H (1995) Effect of tree species and soil properties on nutrient immobilization in the forest floor. Plant Soil 168–169:345–352
Reich PB, Oleksyn J (2004) Global patterns of plant leaf N and P in relation to temperature and latitude. Proc Natl Acad Sci U S A 101:11001–11006
Richardson AE, Lynch JP, Ryan PR, Delhaize E, Smith FA, Smith SE, Harvey PR, Ryan MH, Veneklaas EJ, Lambers H (2011) Plant and microbial strategies to improve the phosphorus efficiency of agriculture. Plant Soil 349:121–156
Rinnan R, Michelsen A, Jonasson S (2008) Effects of litter addition and warming on soil carbon, nutrient pools and microbial communities in a subarctic heath ecosystem. Appl Soil Ecol 39:271–281
Rutigliano F, D’ascoli R, Virzo De Santo A (2004) Soil microbial metabolism and nutrient status in a Mediterranean area as affected by plant cover. Soil Biol Biochem 36:1719–1729
Sardans J, Rivas-Ubach A, Peñuelas J (2012) The C:N:P stoichiometry of organisms and ecosystems in a changing world: a review and perspectives. Perspect Plant Ecol 14:33–47
Schmidt SK, Costello EK, Nemergut DR, Cleveland CC, Reed SC, Weintraub MN, Meyer AF, Martin AM (2007) Biogeochemical consequences of rapid microbial turnover and seasonal succession in soil. Ecology 88:1379–1385
Shen ZH, Liu ZL, Wu J (2004) Altitudinal pattern of flora on the eastern slope of Mt. Gongga. Biodivers Sci 12:89–98
Thomas A (1999) Overview of the geoecology of the Gongga Shan Range, Sichuan Province, China. Mt Res Dev 19:17–30
Tian H, Chen G, Zhang C, Melillo J, Hall C (2010) Pattern and variation of C:N:P ratios in China’s soils: a synthesis of observational data. Biogeochemistry 98:139–151
Tiessen H, Moir J (1993) Characterization of available phosphorus by sequential extraction. In: Carter MR (ed) Soil sampling and methods of analysis Canadian Society of Soil Science. Lewis, Boca Raton, FL, pp 75–86
Townsend AR, Cleveland CC, Asner GP, Bustamante MM (2007) Controls over foliar N:P ratios in tropical rain forests. Ecology 88:107–118
Turner BL, Lambers H, Condron LM, Cramer MD, Leake JR, Richardson AE, Smith SE (2013) Soil microbial biomass and the fate of phosphorus during long-term ecosystem development. Plant Soil 367:225–234
Ushio M, Kitayama K, Balser TC (2010) Tree species effects on soil enzyme activities through effects on soil physicochemical and microbial properties in a tropical montane forest on Mt. Kinabalu, Borneo. Pedobiologia 53:227–233
Vance ED, Brookes PC, Jenkinson DS (1987) An extraction method for measuring soil microbial biomass C. Soil Biol Biochem 19:703–707
Wan XH, Huang ZQ, He ZM, Yu ZP, Wang MH, Davis MR, Yang YS (2015) Soil C:N ratio is the major determinant of soil microbial community structure in subtropical coniferous and broadleaf forest plantations. Plant Soil 387:103–116
Wang X, Tang C, Guppy CN, Sale PWG (2008) Phosphorus acquisition characteristics of cotton (Gossypium hirsutum L.), wheat (Triticum aestivum L.) and white lupin (Lupinus albus L.) under P deficient conditions. Plant Soil 312:117–128
Wu YH, Li W, Zhou J, Cao Y (2013a) Temperature and precipitation variations at two meteorological stations on eastern slope of Gongga Mountain, SW China, in the past two decades. J Mt Sci:43–53
Wu YH, Zhou J, Yu D, Sun SQ, Luo J, Bing HJ, Sun HY (2013b) Phosphorus biogeochemical cycle research in mountainous ecosystems. J Mt Sci 10:43–53
Xu XF, Thornton PE, Post WM (2013) A global analysis of soil microbial biomass carbon, nitrogen and phosphorus in terrestrial ecosystems. Glob Ecol Biogeogr 22:737–749
Yeates GW, Saggar S (1998) Comparison of soil microbial properties and fauna under tussock-grassland and pine plantation. J R Soc N Z 28:523–535
Zhang B, He Y, Miao G, Ding C (2006) Synthetical evaluation of soil structure under subalpine forests on east slope of Gongga Mountain. Mountain Research 24:504–509
Acknowledgments
This work is supported by National Natural Science Foundation of China (41402313 and 41272200) and Chinese Academy of Sciences (CAS “Light of West China” Program and SDS-135-1201-04).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Chengrong Chen
Rights and permissions
About this article
Cite this article
Bing, H., Wu, Y., Zhou, J. et al. Stoichiometric variation of carbon, nitrogen, and phosphorus in soils and its implication for nutrient limitation in alpine ecosystem of Eastern Tibetan Plateau. J Soils Sediments 16, 405–416 (2016). https://doi.org/10.1007/s11368-015-1200-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-015-1200-9