Abstract
Bangladesh is a deltaic country in Asia, and its riverine systems ultimately drain into the Bay of Bengal. Plastic is a severe environmental issue for coastal-marine ecosystems due to the indiscriminate usage and discarding of plastic items in the upstream river that eventually find their route into the Bay of Bengal. Microplastics (MPs) are widespread pollutants in almost all environmental compartments, including aquatic environments. This study aimed to quantify and understand the distribution of microplastics in surface water and sediments of the river Karnaphuli, a tidal confluence river adjacent to the Chattogram seaport city of Bangladesh, a highly inhabited and industrial area on the southeast coast of the Bay of Bengal. A manta trawl net (300-µm mesh size) was used to collect surface water samples, while an Ekman dredge was used to collect sediment samples. The concentrations of microplastics in the surface water of the river Karnaphuli during late monsoon, winter, and early summer were recorded to be 120,111.11, 152,222.22, and 164,444.44 items/km2, respectively, while in sediments, those were recorded to be 103.83, 137.50, and 103.67 items/kg, respectively. A higher abundance of microplastics was observed in downstream surface water (228,888.88 items/km2) and sediments (164.17 items/kg). Smaller sizes (0.3 to 0.5 mm) of microplastics were predominant, fibers or threads were the frequent types, and black was the most common color in the river Karnaphuli. The Fourier transform infrared analysis revealed that polyethylene terephthalate (surface water: 22%, sediments: 19%), polyamide (surface water: 15%, sediments: 13%), polyethylene (surface water: 12%, sediments: 18%), polystyrene (surface water: 13%, sediments: 11%), and alkyd resin (surface water: 13%, sediments: 10%) were the most prevalent polymers in the river Karnaphuli. Moreover, there was a moderate positive correlation between MPs abundance in surface water and sediments. Therefore, improved long-term research (in different seasons with horizontal and vertical monitoring) is necessary in order to accurately determine the flux of microplastics from the river Karnaphuli to the Bay of Bengal.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Plastic production and consumption have expanded significantly from the initial commercial manufacture of plastics in the 1940s (Cole et al. 2011), with around 368 million tonnes of plastic manufactured in 2019 (Plastics Europe 2020). Plastic debris can be dispersed over large distances by oceanic currents, tides, winds, river discharge, and drift (Ng and Obbard 2006; Barnes et al. 2009; Martinez et al. 2009), including islands of the mid-ocean (Ivar do Sul et al. 2009; Rey et al. 2021), the sea around Antarctica (Barnes et al. 2010; Leistenschneider et al. 2021), and the deeper Atlantic region (Lozano and Mouat 2009; Reineccius and Waniek 2022). As a consequence, one of the most prominent types of anthropogenic litter found in the marine ecosystem is plastic debris (Gregory and Ryan 1997; Barnes et al. 2009; Thushari and Senevirathna 2020).
Microplastics (MPs), typically described as bits of plastic in the dimensional range of 1 µm and 5 mm, are one subclass of plastic that has sparked significant concern (Arthur et al. 2009; Fendall and Sewell 2009; Napper et al. 2021). MPs are classed as either primary (directly released from the source, e.g., resin pellets, cosmetic scrubbers) or secondary (produced by the breakdown of larger plastic products, e.g., plastic bottles, toys) and can come from a variety of sources such as household waste and industrial waste (Andrady 2011; Cole et al. 2011; Hidalgo-Ruz et al. 2012). Additionally, various chemicals are utilized as plasticizers and flame retardants to improve the performance of plastic objects. These chemicals may be released into the environment by MPs, raising several environmental issues (e.g., causing toxicity to aquatic biota) (Liu et al. 2019). Furthermore, MPs adsorb hydrophobic pollutants such as polychlorinated biphenyls (PCBs), dichlorodiphenyldichloroethylene (DDE), and dichlorodiphenyltrichloroethane (DDT) (Laist 1997; Teuten et al. 2009), impacting aquatic habitats and the organisms within them.
Many pieces of evidence indicate that MPs are ubiquitous, and their long-term existence may pose a substantial threat to the health of aquatic ecosystems (Murray and Cowie 2011; Farrell and Nelson 2013; Setälä et al. 2014; Kühn et al. 2015; Gall and Thompson 2015; Biginagwa et al. 2016). MPs have been reported to affect aquatic creatures through toxicological effects, tissue inflammation, physical blockages, digestive impairment, and functioning as a potential affluent for the transfer of other hazardous components (Rosenkranz et al. 2009; Moos et al. 2012; Besseling et al. 2013; Lambert and Wagner 2018). MPs have been found in a variety of marine environments, including beach sediments (Costa et al. 2010; Martins and Sobral 2011; Jayasiri et al. 2013; Lee et al. 2013; Besley et al. 2017; Karthik et al. 2018), estuaries (Leslie et al. 2013), in surface, shallow, and deep water (Collignon et al. 2012; Hidalgo-Ruz et al. 2012; Ivar do Sul et al. 2013; Cutroneo et al. 2022). MPs have also been found in the sediments and surface water of various rivers throughout the world (Wang et al. 2017a, b; Ta et al. 2020; Wu et al. 2020; Chauhan et al. 2021; He et al. 2021; Napper et al. 2021).
Bangladesh is a deltaic country in Asia with 257 active rivers that eventually drain into the Bay of Bengal. Besides, oceanic currents also contribute to the relocation of MPs from other portions of the ocean. MPs are assumed to have entered the Bay of Bengal by draining water and sediments from various sources (Hossain et al. 2021). As a result, considering the indiscriminate usage and dumping of plastic items in aquatic surroundings that finally find their way into the Bay of Bengal, plastic is a serious environmental issue for coastal-marine ecosystems. Moreover, the widespread incidence of MPs in the world’s oceans (which also serve as a sink for other harmful chemicals) can cause adverse ecological effects.
Variations in spatial–temporal patterns may significantly influence the incidence and distribution of MPs in tidal confluence rivers. Furthermore, temporal changes owing to precipitation may have a significant effect on the abundance of MPs in the aquatic environment (Lima et al. 2014, 2015). As a result, combining the study of MPs’ spatial and temporal distributions would be beneficial in obtaining full information on MPs’ sources and transit patterns (Lebreton et al. 2017). There have been a few studies in Bangladesh on MPs contamination for fish (Hossain et al. 2019; Ghosh et al. 2021), penaeid shrimp (Hossain et al. 2020), and sandy beaches (Hossain et al. 2021). However, to the best of our knowledge, no study has evaluated the abundance and distribution of MPs in the tidal confluence river of Bangladesh. Thus, we examined the spatial and temporal distribution of MPs in Karnaphuli river, a major tidal river system in the southeast coast of the Bay of Bengal, Bangladesh.
Materials and methods
Study area
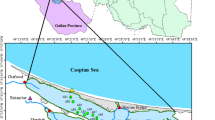
Chattogram is a coastal seaport city and is known as Bangladesh’s industrial capital, located in the southeast coast of the Bay of Bengal. The Karnaphuli is a transboundary river basin distributed between India and Bangladesh. This river was chosen due to its exposure to intense industrial contaminants and anthropogenic pressures, such as proximity to harbors, fishing ports, and large cities. This study focused on a 9-km stretch of the river Karnaphuli in Chattogram, Bangladesh (Fig. 1).
Surface water sample collection
Triplicate samples were taken from five sampling stations (S1, S2, S3, S4, and S5) during September 2019 (late monsoon in Bangladesh), January 2020 (winter), and March 2020 (early summer) (Table S1). Surface water samples (water depth 16 cm) were collected using a manta net (300-µm mesh, 60-cm opening width). MPs sampling from surface water was performed according to Kovač Viršek et al. (2016) with slight modifications. In brief, the manta net was launched from the port and starboard side of the research vessel, approximately 3–4 m away from the vessel. The action started to move in one straight direction with a ~ 2 knots speed for 15 min. Then, the research vessel was stopped, and the manta net was taken out of the water. The net was rinsed thoroughly from the outer side of the net with clean water from the vessel water reservoir. The manta net was rinsed in the mouth to the cod-end direction to concentrate all particles adhered to the net into the cod-end. After that, the cod-end was removed carefully, and the sample in the cod-end was sieved through a 300-μm mesh sieve. The cod-end was rinsed thoroughly from the outer side, and the rest of the sample was poured through the sieve. With the use of a funnel, the sieve was rinsed into a glass jar using 70% ethanol. The use of 70% ethanol is important to preserve the sample. Besides, in the step of visual inspection of the sample, ethanol helps to discolor the organisms; hence, colorful plastics become easier to find (Kovač Viršek et al. 2016). Then, the samples were processed in the laboratory of the Interdisciplinary Institute for Food Security (IIFS), Bangladesh Agricultural University, Bangladesh.
Separation of MPs from water samples
Separation of MPs (< 5 mm) from water samples was done according to Masura et al. (2015) with slight modifications. Briefly, water samples were filtered with a sieve (0.3 mm). Sieved samples were taken in 500-mL beakers and dried in an oven (Genlab OV/200/F/DI, England) at 90 °C for 24 h. Then 20 mL of 0.05 M ferrous sulfate (FeSO4) solution was poured into each beaker. Afterward, 20 mL of 30% hydrogen peroxide (H2O2) was mixed and kept mixtures at room temperature for about 5 min. Then the mixtures were heated at 75 °C on a hotplate (AM4, Velp Scientifica, Italy) until gas bubbles appeared. Six grams of salt (NaCl) was added per 20 mL of the sample during heating to enhance the density of the wet peroxide oxidation (WPO) solution (~ 5 M). Afterward, the WPO solution was carefully poured into the density separator with 700 mL of filtered ZnCl2 (1.5 g cm−3) solution (Coppock et al. 2017), covered with aluminum foil, and kept overnight to settle down. We use the ZnCl2 solution because ZnCl2 was estimated as an effective and comparatively inexpensive floatation media, enabling the floatation of dense polymers (Coppock et al. 2017). With the help of a vacuum pump, the solution obtained from the density separator was filtered with cellulose nitrate filter paper (47 mm diameter and 5 μm pore size) (Prata et al. 2019). Then the filtrate was examined under the microscope (Olympus CX41 with camera DP22, Japan) to identify potential MPs. The main result of MPs samples is calculated as the number of MPs particles per sample (Kovač Viršek et al. 2016). These data were further normalized as per km2. The formula for normalization is MPs particles per sample divided by the sampling area, where the sampling area is determined by multiplying the sampling distance (1 km) by the width of the manta net opening (60 cm).
Sediment sample collection and separation of MPs from sediment samples
Sediment samples were collected by an Ekman dredge with three replicates per station during September 2019 (late monsoon in Bangladesh), January 2020 (winter), and March 2020 (early summer) (Table S1) and stored in glass jars. The separation of MPs from sediment samples was performed according to Masura et al. (2015). In brief, wet sediment samples (400 g) were taken in a 1-L beaker and oven-dried (Genlab OV/200/F/DI, England) at 90 °C for 24 h. Afterward, 300 mL of filtered ZnCl2 (1.5 g cm−3) solution (Coppock et al. 2017) was added and stirred for 10 min. Then all the floating particles were filtered with a 0.3-mm sieve and collected into a beaker (500 mL). After that, 20 mL of 0.05 M ferrous sulfate (FeSO4) solution was poured into each beaker. Then 20 mL of 30% H2O2 was added and mixed for 5 min. The mixtures were heated at 75 °C on a hotplate (AM4, Velp Scientifica, Italy) until gas bubbles were observed. In this stage, to enhance the density of the WPO solution (~ 5 M), 6 g of salt (NaCl) was added per 20 mL of the sample. Then the WPO solution was carefully poured into the density separator with 700 mL of filtered ZnCl2 (1.5 g cm−3) solution (Coppock et al. 2017), covered with aluminum foil, and left overnight to settle down. With the help of a vacuum pump, the solution from the density separator was filtered with cellulose nitrate filter paper (47 mm diameter and 5 μm pore size) (Prata et al. 2019). Afterward, potential MPs in the filters were categorized under the microscope (Olympus CX41 with camera DP22, Japan).
Identification and categorization of MPs
The definition of a plastic item was based on the criteria put forth by Norén (2007): (i) no cellular or organic structural features should be visible within the plastic particle or fiber; (ii) if the particle is a fiber, it should be similarly thick, not taper toward the ends, and have a three-dimensional bend (not entirely straight fibers which direct a biologically derived particle); (iii) clear and homogeneously colored particles; and (iv) if it is not clear that the particle or fiber is colored, for example, if it is whitish or translucent, it should be carefully inspected under a microscope at high magnification and with fluorescence microscopy to rule out an organic origin. Only particles that met the aforementioned requirements were classified as plastic particles after careful examination of the particles. Other items like algae fragments, animal shells, or other parts found on the filters were ignored throughout the detection period.
A fluorescence microscope (Olympus CX41 with camera DP22, Japan) at 4 × to 100 × magnification was used to identify potential MPs and measured digitally with cellSens imaging software. The MPs were identified visually (Hidalgo-Ruz et al. 2012) and analyzed based on their length, shape, and color (Lusher et al. 2013). Besides, the MPs were characterized into fragments, films, fibers, foams, pellets, and microbeads (Kovač Viršek et al. 2016; Murphy et al. 2016; CLEAR 2017; Calcutt et al. 2018). MPs were also categorized into different colors, such as red, white, blue, black, pink, green, orange, and translucent (Bellas et al. 2016; Murphy et al. 2016; Naji et al. 2019). By merely touching suspected plastic particles with a hot needle, the hot point melting test was made to confirm that the particles were plastic (De Witte et al. 2014; Devriese et al. 2015; Vandermeersch et al. 2015; Bellas et al. 2016).
Moreover, MPs were further analyzed (about 20% of the candidate MPs were selected randomly) to determine their chemical composition with a Fourier transform infrared (FTIR) spectrophotometer. FTIR spectrophotometer (Shimadzu IR Prestige 21™, Japan) was used to investigate the chemical composition of different particles. The particles were randomly selected and distributed on a KBr crystal (Parvin et al. 2021). The spectral range was set at 4000–400 cm−1, using the IR solution Agent software with a match threshold > 70% (Tanaka and Takada 2016; Blettler et al. 2017). A SpectraBase™ database from John Wiley & Sons, Inc. has been used to detect the absorption bands of polymers. Additionally, the spectra were compared with the existing literature (Noda et al. 2007; Murphy et al. 2016; Jung et al. 2018).
Quality assurance and quality control
Several control measures were employed strictly during the study. Laboratory coats made of natural fibers, nitrile gloves, and face masks were worn to prevent plastic contamination during sample collection and processing. All the glassware, containers, filtration units, and other necessary instruments were rinsed three times with filtered (45 μm) clean water before use. Samples were wrapped with aluminum foil to prevent air-borne contamination. Procedural blank tests with three replicates (per season) were performed at the same time without any dried sediments or water samples to cross-check the air-borne contamination in the research laboratory (Wu et al. 2020). In brief, 20 mL of 0.05 M ferrous sulfate (FeSO4) solution and 20 mL of 30% hydrogen peroxide (H2O2) were poured into a 500-mL glass beaker. Then 6 g of salt (NaCl) was added, and the solution was poured into the density separator with 700 mL of filtered ZnCl2 (1.5 g cm−3) solution and kept overnight without any covering/foil paper. Afterward, the solution in the density separator was filtered through a 5-μm cellulose nitrate filter. This filter was examined under the microscope, and any particles identified were tested using FTIR. We did not find any potential candidate MPs in the filter, except for a few fibers. We could fairly exclude laboratory contamination because FTIR confirmed that the detected fibers were rayon fibers from clothing, not fishing gear (Nakano et al. 2021).
Statistical analysis
In this study, a descriptive analysis was performed to get mean ± standard deviation (SD), maximum and minimum values. Two-way ANOVA was executed to determine the mean differences among the tested samples in different sampling time points and sampling locations, followed by Tukey’s HSD post hoc comparisons. Before the analyses, research data were further verified for normality and homoscedasticity assumptions employing Shapiro–Wilk’s test and Levene’s test, respectively, without any transformation. The type, shape, size, and color of MPs throughout the study period were determined in frequency percentage (%). Statistical analyses were performed using the SPSS software (version 22, IBM, USA), and variations between the mean values at p < 0.05 were considered significant.
Results
MPs in surface water
The mean concentrations of MPs in surface water of the river Karnaphuli during September 2019, January 2020, and March 2020 were found to be 120,111.11, 152,222.22, and 164,444.44 items/km2, respectively, and varied significantly (p < 0.001) among the three sampling time points (Table 1, Fig. 2a). Besides, the mean MPs concentrations in surface water of S1, S2, S3, S4, and S5 were measured as 65,740.74, 102,222.22, 148,888.89, 182,222.22, and 228,888.88 items/km2, respectively. The MPs values among the sampling stations varied significantly (p < 0.001) (Table 1, Fig. 2b). However, significant differences were not observed in the abundance of MPs in the months and sampling stations (Months*Stations; p > 0.05) (Table 1). Throughout the investigation, MPs from surface water varied from 30,000 to 270,000, with a mean value of 145,592.59 ± 63,739.88 items/km2 (Table S1).
In the present study, identified MPs were categorized into three sizes, including 0.3 to 0.5 mm, 0.5 to 1 mm, and 1 to 5 mm. The largest proportion of the MPs recorded in surface water was 0.3 to 0.5 mm in size, followed by 0.5 to 1 mm and 1 to 5 mm (Fig. 3). In temporal occurrence, the largest proportion of MPs (52%) was 0.3 to 0.5 mm recorded in March 2020 (Fig. 3a). In the case of spatial occurrence, the largest proportion of MPs (50%) was 0.3 to 0.5 mm observed in S4 (Fig. 3b). Furthermore, six different types of MPs (fibers, fragments, foams, films, pellets, and microbeads) were found in the studied samples. Fibers (55%) were the prevalent type of MPs detected in surface water in March 2020. The least number of microbeads (1%) was noted in March 2020, whereas the least number of pellets (1%) was recorded in both January and March 2020 (Table 2). In spatial occurrence, fibers (54%) were prevalent in S3, where pellets (0%) were not found (Table 3).
In the current study, different colored MPs were recorded from the river Karnaphuli, such as black, purple, white, blue, green, translucent, red, pink, and brown. In the case of temporal occurrence, the predominant color of MPs in surface water was black (32%), as found in September 2019. The least number of pink (3%) colored MPs was detected in January 2020, while the least number of brown (3%) colored MPs was detected in both September 2019 and March 2020 (Table 2). In the case of station-wise distribution, black (30%) colored MPs were prevalent in S5, whereas brown (2%) were the least dominant in S1 (Table 3).
MPs in sediments
Mean MPs concentrations in sediments of the river Karnaphuli were recorded to be 103.83, 137.50, and 103.67 items/kg in September 2019, January 2020, and March 2020, respectively, and varied significantly (p < 0.05) (Table 1, Fig. 2c). Mean MPs concentrations in sediments at S1, S2, S3, S4, and S5 were 59.44, 75.83, 135.28, 140.28, and 164.17 items/kg, respectively, and differed significantly among the sampling stations (p < 0.001) (Table 1, Fig. 2d). In addition, significant variations were also found in MPs abundance at different months and sampling stations (Months*Stations; p < 0.05) (Table 1). The range of MPs recorded from sediments throughout the investigation was 10 to 255 items/kg, with a mean value of 115.00 ± 60.68 items/kg (Table S1).
In the current investigation, smaller sizes of MPs (0.3 to 0.5 mm) were found in large numbers in sediments as compared to the other size categories. The highest proportion of a small-sized category (47%) was found in January 2020 (Fig. 3a). In spatial occurrence, the highest proportion of a small-sized category (48%) was reported for S1 (Fig. 3b). Furthermore, fibers (86%) were the dominant type of MPs detected in sediments in March 2020. However, foams (0%) were not found in January 2020, whereas pellets (0%) were not reported in September 2019 and March 2020 (Table 2). In spatial occurrence, fibers were dominant in S5 (78%) (Table 3). In the case of temporal events, the prevalent color of MPs in surface water was black (32%), found in January 2020, while the least dominant color, brown (2%), was also recorded in January 2020 (Table 2). In the case of station-wise distribution, black (44%) was dominant in S1, whereas green (0%) was not found (Table 3). The types of MPs detected in the river Karnaphuli have been shown in Fig. 4.
Polymer composition of separated MPs
There were 4010 items and 2070 items found for the MPs in the collected surface water and sediment samples, respectively, of which 802 items of surface water and 414 items of sediment samples (> 300 µm) were analyzed with FTIR. Polyethylene terephthalate was the predominant polymer in both sediments (19%) and surface water (22%) samples (Fig. 5). Polyamide (15%), alkyd resin (13%), polystyrene (13%), polyethylene (12%), urethane alkyd (10%), cellophane (5%), polyvinylidene fluoride (3%), and polyether urethane (3%) were also observed in water samples. However, 2% were non-plastics in water samples, and the remaining 2% belonged to unidentified material (Fig. 5a). In the case of sediment samples, polyethylene (18%), polyamide (13%), polystyrene (11%), alkyd resin (10%), urethane alkyd (9%), polyvinylidene fluoride (8%), polyether urethane (5%), and cellophane (4%) were also found. Around 2% were non-plastics, and the remaining 1% belonged to unidentified material in sediment samples (Fig. 5b).
Correlation of MPs abundance between surface water and sediments
There was a moderate positive correlation of MPs abundance between surface water and sediments of the Karnaphuli river (r: 0.639, p < 0.001) (Fig. 6). The linear regression equation was y = 0.0006x + 26.469 and the coefficient of determination (R2) value was 0.408, according to the regression analysis. Therefore, the number of MPs was high in sediments, while MPs were prevalent in surface water.
Discussion
Because there is no established standard for measuring MPs, the concentration unit has not been defined in published literature (Jiang et al. 2019). Some researchers used items/kg for sediments and items/km2 for waterways, while others used items/m2 for both sediments and waters. In addition, some researchers employed items/L or items/m3 for water. As concentration levels in different units cannot be compared (Jiang et al. 2019), we found it difficult to compare the research results to other published data. However, the amounts of MPs contamination in the current investigation were compared to studies mentioning similar concentration units. In the present study, MPs recorded from the surface water of the river Karnaphuli were 30,000 to 270,000 items/km2, with an average value of 145,592.59 items/km2, comparable to other reported studies.
MPs in the backwater area of Xiangxi river, China (55,000 to 34,200,000 items/km2), and the surface water of the Yangtze river (162,000 to 4,250,000 items/km2, with an average abundance of 2,800,000 items/km2) were observed to be higher, as reported by Zhang et al. (2017) and He et al. (2021), respectively, than the current measured abundance. Furthermore, Eriksen et al. (2013) recorded 43,000 to 466,000 particles/km2 with an average of 43,000 particles/km2 in the surface water samples from the Laurentian Great Lakes, which is also greater than the current study. According to published reports, the abundance of MPs is related to geographical location, population density, and urbanization (Wang et al. 2017a, b; Wen et al. 2018; Nakano et al. 2021). A higher abundance of MPs was observed in downstream surface water (station 5) of the Karnaphuli river. This may be due to waste materials coming from upstream, adjacent channels (Boalkhali and Murai Channels), power stations, and other industries mixed with the river. Besides, a larger number of MPs in surface water were found in early summer (March 2020) than in late monsoon and winter. Reasons for these variations in the abundance of MPs include loadings from the sources such as household waste, garments washing, agricultural and industrial waste, tourist activity, fishing, navigation, and transportation at bridges on the river Karnaphuli; geographic features including Bangladesh is a low lying country, and monsoon brings heavy rainfall here; and hydrodynamic circumstances including the velocities of the emitted particles due to water level rise and fall (Peng et al. 2017; Gray et al. 2018; Bordós et al. 2019; Kataoka et al. 2019).
In the current investigation, MPs recorded in the sediments of the river Karnaphuli were 10 to 255 items/kg, with an average value of 115.00 items/kg. According to Jiang et al. (2019), MPs recorded from the Tibet Plateau sediments fluctuated from 50 to 195 items/kg, which is more or less similar to the current study. Moreover, MPs were recorded as 0.9 to 298.1 items/kg in sediment samples in a coastal metropolis of Australia (Su et al. 2020), supporting the present findings. MPs in the sediments of the Chao Phraya river, Bangkok (2290 items/kg), and Beijiang river, China (178 to 544 items/kg), were found to be higher than the current measured concentrations, as documented by Ta et al. (2020) and Wang et al. (2017a), respectively. Heavy rainfall during the monsoon might increase the river flow (Zhao et al. 2019) and dilute the MPs concentration in the waters (Yan et al. 2019). So, the amount of rainfall, currents, and anthropogenic events (release of industrial waste, fishing, etc.) in the research area may have caused temporal fluctuations in MPs abundance in surface water and sediments. However, at station 5 (near the Shah Amanat Bridge), all the waste materials from adjacent channels and industries are mixed with the river. So, it was obvious that it showed high MPs flux in that particular station’s surface water and sediments. Moreover, several aspects may promote the huge spatial variability of MPs contamination in the samples of this study, such as upstream input, fisheries, navigation, and agricultural and industrial activities in the river (Peng et al. 2018; Kiessling et al. 2019).
In comparison to the other groups, smaller sizes of MPs (0.3 to 0.5 mm) were predominant in sediments and surface water in the current investigation. Wu et al. (2020) also found a large quantity of 0.1 to 1 mm-sized MPs in the Maozhou river’s sediments and surface water. The most common size of MPs reported in the Saigon river was less than 50–250 µm (Lahens et al. 2018), whereas Baldwin et al. (2016) recorded 72% of 0.35 to 0.99 mm-sized MPs in 29 lakes globally. Furthermore, MPs of small sizes (< 1 mm) were found in both trawling and filtering water samples taken from the Yangtze river (He et al. 2021). However, MPs of a smaller size can readily penetrate the various food chains of an aquatic environment and, therefore, potentially influence it (Jeong et al. 2016). As we detected, MPs from surface water and sediments, pelagic and benthic fish, bivalves, crustaceans, etc., may be affected by MPs pollution. MPs may affect aquatic organisms by causing physiological injury, obstructing the digestive tract, disrupting eating and reproductive behavior, decreasing the offspring’s survival rate, and impairing immunological function (Jabeen et al. 2018; Prokić et al. 2019; Strungaru et al. 2019).
Fibers/threads were the most common MPs in the river Karnaphuli’s sediments and surface water, followed by fragments, films, foams, microbeads, and pellets. These types of MPs are suspected of having come from daily-used plastic items such as water bottles, containers, toys, food packaging, and cosmetic scrubbers. In addition, thread MPs were also found in significant quantities in the inland freshwaters of Wuhan, China (Wang et al. 2017b) and two typical estuaries in Bohai Bay, China (Wu et al. 2019). Furthermore, Chauhan et al. (2021) discovered fibers/threads as prevalent MPs in both the sediments and surface water of the Alaknanda river, whereas Napper et al. (2021) found fibers/threads to be the most common kind in the surface water of the Ganga. On the other hand, Wu et al. (2020) found fragment type to be prominent in both water and sediments from the river Maozhou and reported packing industry might be the critical source of this type of MPs. Domestic trash is a significant source of MPs fibers/threads, which are regularly discharged during garment and other home items cleaning (Browne et al. 2011; Kalčíková et al. 2017). Discarding damaged or unusable fishing nets and ropes were found along the Karnaphuli river, which might be another reason for the higher amount of fibers/threads. Furthermore, microbeads from cosmetic items such as facial cleansers (Fendall and Sewell 2009) and pellets from personal care products and medications may be other sources of MPs in the riverine ecosystem (Kalčíková et al. 2017).
Color is often thought to be one of the significant crucial factors influencing MPs ingestion by aquatic organisms, as specific colors may attract predators whenever they resemble the color of their prey (Kühn et al. 2015; Abayomi et al. 2017). In this study, black and purple were the most abundant colors in sediments and surface water of the river Karnaphuli. Moreover, Hossain et al. (2021) speculated that colored MPs might be generated by synthetic and organic compounds, necessitating more extensive research. Colors might differ between regions due to their sources, such as discharge from the residents, industries, and adjacent channels.
Polyethylene terephthalate, polyamide, polyethylene, polystyrene, and alkyd resin were the major polymers in sediments and surface water of the river Karnaphuli, which is more or less similar to other reported findings (Jiang et al. 2019; Ta et al. 2020). Besides, polyethylene and polypropylene were the significant polymers in the identified MPs from the Yangtze river water (He et al. 2021), which does not support the present findings. Furthermore, polyamide and polyethylene terephthalate were revealed in the fish gut documented by Hossain et al. (2019) for the northern Bay of Bengal. However, polyethylene terephthalate is the main component for garments, drinking water bottles, maximum colored fibers, as well as several transparent fragments (Wang et al. 2017a, b). A variety of disposable products, including disposable bags, kitchen utensils, and cutlery, are found in the study area; the majority of them are composed of inexpensive and low-weight polyethylene. Moreover, paint particles (mainly alkyd resin) were estimated as a significant contributor to MPs particles in the surface water of the Incheon/Kyeonggi coastal region (Chae et al. 2015). The boats, ships, and trawlers may release paint particles in the river Karnaphuli. In addition, the discarding of damaged fishing gears, nets, ropes, floats, and fish baskets/bags may increase polyamide particles in the study area (Pruter 1987; Hossain et al. 2019).
In the current study, surface water and sediments had a moderately positive correlation in terms of MPs abundance. As a result, MPs were numerous in sediments, whereas they were common in surface water. According to Browne et al. (2007), high-density plastics typically sink, deposit in the sediments, and may be consumed by deposit feeders. In contrast, low-density plastics float at the surface and may be uptaken by filter feeders/planktivores. However, vertical transport may occur due to biofouling (the development of a biofilm on the MPs), turbulence, and freshwater inflow (Kooi et al. 2017; Melkebeke et al. 2020).
Conclusions
This research presented a preliminary investigation of MPs contamination in the river Karnaphuli of Bangladesh in Asia. MPs were identified in all the surface water and sediment samples at five sampling stations during late monsoon, winter, and early summer. Throughout the investigation, MPs from surface water varied from 30,000 to 270,000 items/km2, with a mean value of 145,592.59 items/km2. In the case of sediment samples, the range of MPs was 10 to 255 items/kg with a mean value of 115.00 items/kg. Smaller sizes (0.3 to 0.5 mm) of MPs were predominant; fibers or threads were the frequent types, and black was the most common color in the river Karnaphuli. Besides, there was a moderate positive correlation between MPs abundance in surface water and sediments of the Karnaphuli river. Polyethylene terephthalate (surface water: 22%; sediments: 19%), polyamide (surface water: 15%; sediments: 13%), polyethylene (surface water: 12%; sediments: 18%), polystyrene (surface water: 13%; sediments: 11%), and alkyd resin (surface water: 13%; sediments: 10%) were the major types of polymers. In conclusion, it is essential to implement an effective management strategy for reducing, reusing, and recycling plastic materials in this region. Long-term research with a broad temporal and spatial distribution is necessary to determine the flux of MPs from the river Karnaphuli to the Bay of Bengal.
Data availability
The data that support the findings of this study are available from the corresponding author upon request.
References
Abayomi OA, Range P, Al-Ghouti MA, Obbard JP, Almeer SH, Ben-Hamadou R (2017) Microplastics in coastal environments of the Arabian Gulf. Mar Pollut Bull 124:181–188. https://doi.org/10.1016/j.marpolbul.2017.07.011
Andrady AL (2011) Microplastics in the marine environment. Mar Pollut Bull 62(8):1596–1605. https://doi.org/10.1016/j.marpolbul.2011.05.030
Arthur C, Baker J, Bamford H (2009) Proceedings of the international research workshop on the occurrence, effects and fate of microplastic marine debris. Sept 9–11, 2008. NOAA Technical Memorandum NOS-OR&R-30. marinedebris.noaa.gov/file/2192/download?token=5dvqb-YY. Accessed 1 Oct 2019
Baldwin AK, Corsi SR, Mason SA (2016) Plastic debris in 29 great lakes tributaries: relations to watershed attributes and hydrology. Environ Sci Technol 50(19):10377–10385. https://doi.org/10.1021/acs.est.6b02917
Barnes DKA, Galgani F, Thompson RC, Barlaz M (2009) Accumulation and fragmentation of plastic debris in global environments. Phil Trans R Soc B: Biol Sci 364:1985–1998. https://doi.org/10.1098/rstb.2008.0205
Barnes DKA, Walters A, Gonçalves L (2010) Macroplastics at sea around Antarctica. Mar Environ Res 70(2):250–252. https://doi.org/10.1016/j.marenvres.2010.05.006
Bellas J, Martínez-Armental J, Martínez-Cámara A, Besada V, Martínez-Gómez C (2016) Ingestion of microplastics by demersal fish from the Spanish Atlantic and Mediterranean coasts. Mar Pollut Bull 109:55–60. https://doi.org/10.1016/j.marpolbul.2016.06.026
Besley A, Vijver MG, Behrens P, Bosker T (2017) A standardized method for sampling and extraction methods for quantifying microplastics in beach sand. Mar Pollut Bull 114(1):77–83. https://doi.org/10.1016/j.marpolbul.2016.08.055
Besseling E, Wegner A, Foekema EM, Heuvel-Greve MJVD, Koelmans AA (2013) Effects of microplastic on fitness and PCB bioaccumulation by the lugworm Arenicola marina (L.). Environ Sci Technol 47(1):593–600. https://doi.org/10.1021/es302763x
Biginagwa FJ, Mayoma BS, Shashoua Y, Syberg K, Khan FR (2016) First evidence of microplastics in the African Great Lakes: recovery from Lake Victoria Nile perch and Nile tilapia. J Great Lakes Res 42(1):146–149. https://doi.org/10.1016/j.jglr.2015.10.012
Blettler MCM, Ulla MA, Rabuffetti AP, Garello N (2017) Plastic pollution in freshwater ecosystems: macro-, meso-, and microplastic debris in a floodplain lake. Environ Monit Assess 189:581. https://doi.org/10.1007/s10661-017-6305-8
Bordós G, Urbányi B, Micsinai A, Kriszt B, Palotai Z, Szabó I, Hantosi Z, Szoboszlay S (2019) Identification of microplastics in fish ponds and natural freshwater environments of the Carpathian basin, Europe. Chemosphere 216:110–116. https://doi.org/10.1016/j.chemosphere.2018.10.110
Browne MA, Galloway T, Thompson R (2007) Microplastic–an emerging contaminant of potential concern? Integr Environ Assess Manag 3(4):559–561. https://doi.org/10.1002/ieam.5630030412
Browne MA, Crump P, Niven SJ, Teuten E, Tonkin A, Galloway T, Thompson R (2011) Accumulation of microplastic on shorelines worldwide: sources and sinks. Environ Sci Technol 45:9175–9179. https://doi.org/10.1021/es201811s
Calcutt J, Nussbaumer A, Sluka R (2018) Guidelines for sampling microplastics on sandy beaches. A Rocha Int Mar Conserv Team. https://www.arocha.org/wp-content/uploads/2018/01/Microplastic-sampling-protocol.pdf
Chae D, Kim I, Kim S, Song YK, Shim WJ (2015) Abundance and distribution characteristics of microplastics in surface seawaters of the Incheon/Kyeonggi coastal region. Arch Environ Contam Toxicol 69:269–278. https://doi.org/10.1007/s00244-015-0173-4
Chauhan JS, Semwal D, Nainwal M, Badola N, Thapliyal P (2021) Investigation of microplastic pollution in river Alaknanda stretch of Uttarakhand. Environ Dev Sustain. https://doi.org/10.1007/s10668-021-01388-y
CLEAR (2017) How to analyze plastics from a surface trawl. Spotter’s guide to plastic pollution. Civic Laboratory for Environmental Action Research (CLEAR). https://civiclaboratory.nl
Cole M, Lindeque P, Halsband C, Galloway TS (2011) Microplastics as contaminants in the marine environment: a review. Mar Pollut Bull 62:2588–2597. https://doi.org/10.1016/j.marpolbul.2011.09.025
Collignon A, Hecq JH, Galgani F, Voisin P, Collard F, Goffart A (2012) Neustonic microplastic and zooplankton in the North Western Mediterranean Sea. Mar Pollut Bull 64(4):861–864. https://doi.org/10.1016/j.marpolbul.2012.01.011
Coppock RL, Cole M, Lindeque PK, Queirós AM, Galloway TS (2017) A small-scale, portable method for extracting microplastics from marine sediments. Environ Pollut 230:829–837. https://doi.org/10.1016/j.envpol.2017.07.017
Costa MF, do sul Ivar JA, Silva-Cavalcanti JS, Araújo MCB, Spengler A, Tourinho PS (2010) On the importance of size of plastic fragments and pellets on the strandline: a snapshot of a Brazilian beach. Environ Monit Assess 168:299–304. https://doi.org/10.1007/s10661-009-1113-4
Cutroneo L, Capello M, Domi A, Consani S, Lamare P, Coyle P, Bertin V, Dornic D, Reboa A, Geneselli I, Anghinolfi M (2022) Microplastics in the abyss: a first investigation into sediments at 2443-m depth (Toulon, France). Environ Sci Pollut Res 29:9375–9385. https://doi.org/10.1007/s11356-021-17997-z
De Witte B, Devriese L, Bekaert K, Hoffman S, Vandermeersch G, Cooreman K, Robbens J (2014) Quality assessment of the blue mussel (Mytilus edulis): comparison between commercial and wild types. Mar Pollut Bull 85(1):146–155. https://doi.org/10.1016/j.marpolbul.2014.06.006
Devriese LI, van der Meulen MD, Maes T, Bekaert K, Paul-Pont I, Frère L, Robbens J, Vethaak AD (2015) Microplastic contamination in brown shrimp (Crangon crangon, Linnaeus 1758) from coastal waters of the Southern North Sea and Channel area. Mar Pollut Bull 98:179–187. https://doi.org/10.1016/j.marpolbul.2015.06.051
do Sul Ivar JA, Spengler A, Costa MF (2009) Here, there and everywhere. Small plastic fragments and pellets on beaches of Fernando de Noronha (Equatorial Western Atlantic). Mar Pollut Bull 58(8):1236–1238. https://doi.org/10.1016/j.marpolbul.2009.05.004
do Sul Ivar JA, Costa MF, Barletta M, Cysneiros FJA (2013) Pelagic microplastics around an archipelago of the equatorial Atlantic. Mar Pollut Bull 75:305–309. https://doi.org/10.1016/j.marpolbul.2013.07.040
Eriksen M, Mason S, Wilson S, Box C, Zellers A, Edwards W, Farley H, Amato S (2013) Microplastic pollution in the surface waters of the Laurentian Great Lakes. Mar Pollut Bull 77:177–182. https://doi.org/10.1016/j.marpolbul.2013.10.007
Farrell P, Nelson K (2013) Trophic level transfer of microplastic: Mytilus edulis (L.) to Carcinus maenas (L.). Environ Pollut 177:1–3. https://doi.org/10.1016/j.envpol.2013.01.046
Fendall LS, Sewell MA (2009) Contributing to marine pollution by washing your face: microplastics in facial cleansers. Mar Pollut Bull 58(8):1225–1228. https://doi.org/10.1016/j.marpolbul.2009.04.025
Gall SC, Thompson RC (2015) The impact of debris on marine life. Mar Pollut Bull 92(1–2):170–179. https://doi.org/10.1016/j.marpolbul.2014.12.041
Ghosh GC, Akter SM, Islam RM, Habib A, Chakraborty TK, Zaman S, Kabir AHME, Shipin OV, Wahid MA (2021) Microplastics contamination in commercial marine fish from the Bay of Bengal. Reg Stud Mar Sci 44:101728. https://doi.org/10.1016/j.rsma.2021.101728
Gray AD, Wertz H, Leads RR, Weinstein JE (2018) Microplastic in two South Carolina Estuaries: occurrence, distribution, and composition. Mar Pollut Bull 128:223–233. https://doi.org/10.1016/j.marpolbul.2018.01.030
Gregory MR, Ryan PG (1997) Pelagic plastics and other seaborne persistent synthetic debris: a review of Southern Hemisphere perspectives. In: Coe JM, Rogers DB (eds) Marine debris. Springer series on environmental management. Springer, New York, NY, pp 49–66. https://doi.org/10.1007/978-1-4613-8486-1_6
He D, Chen X, Zhao W, Zhu Z, Qi X, Zhou L, Chen W, Wan C, Li D, Zou X, Wu N (2021) Microplastics contamination in the surface water of the Yangtze River from upstream to estuary based on different sampling methods. Environ Res 196:110908. https://doi.org/10.1016/j.envres.2021.110908
Hidalgo-Ruz V, Gutow L, Thompson RC, Thiel M (2012) Microplastics in the marine environment: a review of the methods used for identification and quantification. Environ Sci Technol 46:3060–3075. https://doi.org/10.1021/es2031505
Hossain MS, Sobhan F, Uddin MN, Sharifuzzaman SM, Chowdhury SR, Sarker S, Chowdhury MSN (2019) Microplastics in fishes from the Northern Bay of Bengal. Sci Total Environ 690:821–830. https://doi.org/10.1016/j.scitotenv.2019.07.065
Hossain MS, Rahman MS, Uddin MN, Sharifuzzaman SM, Chowdhury SR, Sarker S, Chowdhury MSN (2020) Microplastic contamination in penaeid shrimp from the Northern Bay of Bengal. Chemosphere 238:124688. https://doi.org/10.1016/j.chemosphere.2019.124688
Hossain MB, Banik P, Nur AU, Rahman T (2021) Abundance and characteristics of microplastics in sediments from the world’s longest natural beach, Cox’s Bazar, Bangladesh. Mar Pollut Bull 163:111956. https://doi.org/10.1016/j.marpolbul.2020.111956
Jabeen K, Li B, Chen Q, Su L, Wu C, Hollert H, Shi H (2018) Effects of virgin microplastics on goldfish (Carassius auratus). Chemosphere 213:323–332. https://doi.org/10.1016/j.chemosphere.2018.09.031
Jayasiri HB, Purushothaman CS, Vennila A (2013) Quantitative analysis of plastic debris on recreational beaches in Mumbai, India. Mar Pollut Bull 77(1–2):107–112. https://doi.org/10.1016/j.marpolbul.2013.10.024
Jeong CB, Won EJ, Kang HM, Lee MC, Hwang DS, Hwang UK, Zhou B, Souissi S, Lee SJ, Lee JS (2016) Microplastic size-dependent toxicity, oxidative stress induction, and p-JNK and p-p38 activation in the monogonont rotifer (Brachionus koreanus). Environ Sci Technol 50(16):8849–8857. https://doi.org/10.1021/acs.est.6b01441
Jiang C, Yin L, Li Z, Wen X, Luo X, Hu S, Yang H, Long Y, Deng B, Huang L, Liu Y (2019) Microplastic pollution in the rivers of the Tibet Plateau. Environ Pollut 249:91–98. https://doi.org/10.1016/j.envpol.2019.03.022
Jung MR, Horgen FD, Orski SV, Rodriguez CV, Beers KL, Balazs GH, Jones TT, Work TM, Brignac KC, Royer SJ, Hyrenbach KD, Jensen BA, Lynch JM (2018) Validation of ATR FT-IR to identify polymers of plastic marine debris, including those ingested by marine organisms. Mar Pollut Bull 127:704–716. https://doi.org/10.1016/j.marpolbul.2017.12.061
Kalčíková G, Alič B, Skalar T, Bundschuh M, Gotvajn AŽ (2017) Wastewater treatment plant effluents as source of cosmetic polyethylene microbeads to freshwater. Chemosphere 188:25–31. https://doi.org/10.1016/j.chemosphere.2017.08.131
Karthik R, Robin RS, Purvaja R, Ganguly D, Anandavelu I, Raghuraman R, Hariharan G, Ramakrishna A, Ramesh R (2018) Microplastics along the beaches of southeast coast of India. Sci Total Environ 645:1388–1399. https://doi.org/10.1016/j.scitotenv.2018.07.242
Kataoka T, Nihei Y, Kudou K, Hinata H (2019) Assessment of the sources and inflow processes of microplastics in the river environments of Japan. Environ Pollut 244:958–965. https://doi.org/10.1016/j.envpol.2018.10.111
Kiessling T, Knickmeier K, Kruse K, Brennecke D, Nauendorf A, Thiel M (2019) Plastic pirates sample litter at rivers in Germany – riverside litter and litter sources estimated by school children. Environ Pollut 245:545–557. https://doi.org/10.1016/j.envpol.2018.11.025
Kooi M, van Nes EH, Scheffer M, Koelmans AA (2017) Ups and downs in the ocean: effects of biofouling on vertical transport of microplastics. Environ Sci Technol 51(14):7963–7971. https://doi.org/10.1021/acs.est.6b04702
Kovač Viršek M, Palatinus A, Koren Š, Peterlin M, Horvat P, Kržan A (2016) Protocol for microplastics sampling on the sea surface and sample analysis. J vis Exp 118:55161
Kühn S, Bravo Rebolledo EL, van Franeker JA (2015) Deleterious effects of litter on marine life. In: Bergmann M, Gutow L, Klages M (eds) Marine anthropogenic litter. Springer, Cham, pp 75–116. https://doi.org/10.1007/978-3-319-16510-3_4
Lahens L, Strady E, Kieu-Le TC, Dris R, Boukerma K, Rinnert E, Gasperi J, Tassin B (2018) Macroplastic and microplastic contamination assessment of a tropical river (Saigon River, Vietnam) transversed by a developing megacity. Environ Pollut 236:661–671. https://doi.org/10.1016/j.envpol.2018.02.005
Laist DW (1997) Impacts of marine debris: entanglement of marine life in marine debris including a comprehensive list of species with entanglement and ingestion records. In: Coe JM, Rogers DB (eds) Marine debris. Springer series on environmental management. Springer, New York, NY, pp 99–119. https://doi.org/10.1007/978-1-4613-8486-1_10
Lambert S, Wagner M (2018) Microplastics are contaminants of emerging concern in freshwater environments: an overview. In: Wagner M, Lambert S (eds) Freshwater microplastics. The handbook of environmental chemistry 58:1–23. Springer, Cham. https://doi.org/10.1007/978-3-319-61615-5_1
Lebreton LCM, van der Zwet J, Damsteeg JW, Slat B, Andrady A, Reisser J (2017) River plastic emissions to the world’s oceans. Nat Commun 8:15611. https://doi.org/10.1038/ncomms15611
Lee J, Hong S, Song YK, Hong SH, Jang YC, Jang M, Heo NW, Han GM, Lee MJ, Kang D, Shim WJ (2013) Relationships among the abundances of plastic debris in different size classes on beaches in South Korea. Mar Pollut Bull 77(1–2):349–354. https://doi.org/10.1016/j.marpolbul.2013.08.013
Leistenschneider C, Burkhardt-Holm P, Mani T, Primpke S, Taubner H, Gerdts G (2021) Microplastics in the Weddell Sea (Antarctica): a forensic approach for discrimination between environmental and vessel-induced microplastics. Environ Sci Technol 55(23):15900–15911. https://doi.org/10.1021/acs.est.1c05207
Leslie HA, van Velzen MJM, Vethaak AD (2013) Microplastic survey of the Dutch environment. Novel data set of microplastics in North Sea sediments, treated wastewater effluents and marine biota. IVM Institute for Environmental Studies (Report number R-13/11. pp. 30). https://science.vu.nl/en/Images/IVM_report_Microplastic_in_sediment_STP_Biota_2013_tcm296-409860.pdf
Lima ARA, Costa MF, Barletta M (2014) Distribution patterns of microplastics within the plankton of a tropical estuary. Environ Res 132:146–155. https://doi.org/10.1016/j.envres.2014.03.031
Lima ARA, Barletta M, Costa MF (2015) Seasonal distribution and interactions between plankton and microplastics in a tropical estuary. Estuar Coast Shelf Sci 165:213–225. https://doi.org/10.1016/j.ecss.2015.05.018
Liu F, Liu G, Zhu Z, Wang S, Zhao F (2019) Interactions between microplastics and phthalate esters as affected by microplastics characteristics and solution chemistry. Chemosphere 214:688–694. https://doi.org/10.1016/j.chemosphere.2018.09.174
Lozano RL, Mouat J (2009) Marine litter in the north-east Atlantic region: assessment and priorities for response. KIMO International. https://qsr2010.ospar.org/media/assessments/p00386_Marine_Litter_in_the_North-East_Atlantic_with_addendum.pdf
Lusher AL, McHugh M, Thompson RC (2013) Occurrence of microplastics in the gastrointestinal tract of pelagic and demersal fish from the English Channel. Mar Pollut Bull 67:94–99. https://doi.org/10.1016/j.marpolbul.2012.11.028
Martinez E, Maamaatuaiahutapu K, Taillandier V (2009) Floating marine debris surface drift: convergence and accumulation toward the South Pacific subtropical gyre. Mar Pollut Bull 58:1347–1355. https://doi.org/10.1016/j.marpolbul.2009.04.022
Martins J, Sobral P (2011) Plastic marine debris on the Portuguese coastline: a matter of size? Mar Pollut Bull 62(12):2649–2653. https://doi.org/10.1016/j.marpolbul.2011.09.028
Masura J, Baker J, Foster G, Arthur C (2015) Laboratory methods for the analysis of microplastics in the marine environment: recommendations for quantifying synthetic particles in waters and sediments. NOAA Technical Memorandum NOS-OR&R-48. https://marinedebris.noaa.gov/sites/default/files/publications-files/noaa_microplastics_methods_manual.pdf
Melkebeke MV, Janssen C, Meester SD (2020) Characteristics and sinking behavior of typical microplastics including the potential effect of biofouling: implications for remediation. Environ Sci Technol 54(14):8668–8680. https://doi.org/10.1021/acs.est.9b07378
Moos NV, Burkhardt-Holm P, Köhler A (2012) Uptake and effects of microplastics on cells and tissue of the blue mussel Mytilus edulis L. after an experimental exposure. Environ Sci Technol 46:11327–11335. https://doi.org/10.1021/es302332w
Murphy F, Ewins C, Carbonnier F, Quinn B (2016) Wastewater treatment works (WwTW) as a source of microplastics in the aquatic environment. Environ Sci Technol 50:5800–5808. https://doi.org/10.1021/acs.est.5b05416
Murray F, Cowie PR (2011) Plastic contamination in the decapod crustacean Nephrops norvegicus (Linnaeus, 1758). Mar Pollut Bull 62(6):1207–1217. https://doi.org/10.1016/j.marpolbul.2011.03.032
Naji A, Nuri M, Amiri P, Niyogi S (2019) Small microplastic particles (S-MPPs) in sediments of mangrove ecosystem on the northern coast of the Persian Gulf. Mar Pollut Bull 146:305–311. https://doi.org/10.1016/j.marpolbul.2019.06.033
Nakano H, Arakawa H, Tokai T (2021) Microplastics on the sea surface of the semi-closed Tokyo Bay. Mar Pollut Bull 162:111887. https://doi.org/10.1016/j.marpolbul.2020.111887
Napper IE, Baroth A, Barrett AC, Bhola S, Chowdhury GW, Davies BFR, Duncan EM, Kumar S, Nelms SE, Niloy MNH, Nishat B, Maddalene T, Thompson RC, Koldewey H (2021) The abundance and characteristics of microplastics in surface water in the transboundary Ganges River. Environ Pollut 274:116348. https://doi.org/10.1016/j.envpol.2020.116348
Ng KL, Obbard JP (2006) Prevalence of microplastics in Singapore’s coastal marine environment. Mar Pollut Bull 52:761–767. https://doi.org/10.1016/j.marpolbul.2005.11.017
Noda I, Dowrey AE, Haynes JL, Marcott C (2007) Group frequency assignments for major infrared bands observed in common synthetic polymers. Physical properties of polymers handbook. Springer, New York, pp 395–406. https://doi.org/10.1007/978-0-387-69002-5_22
Norén F (2007) Small plastic particles in coastal Swedish waters. KIMO Sweden Report. pp. 11
Parvin F, Jannat S, Tareq SM (2021) Abundance, characteristics and variation of microplastics in different freshwater fish species from Bangladesh. Sci Total Environ 784:147137. https://doi.org/10.1016/j.scitotenv.2021.147137
Peng G, Zhu B, Yang D, Su L, Shi H, Li D (2017) Microplastics in sediments of the Changjiang Estuary, China. Environ Pollut 225:283–290. https://doi.org/10.1016/j.envpol.2016.12.064
Peng G, Xu P, Zhu B, Bai M, Li D (2018) Microplastics in freshwater river sediments in Shanghai, China: a case study of risk assessment in mega-cities. Environ Pollut 234:448–456. https://doi.org/10.1016/j.envpol.2017.11.034
Plastics Europe (2020) Plastics – the facts 2020. An analysis of European plastics production, demand and waste data. https://plasticseurope.org/knowledge-hub/plastics-the-facts-2020/
Prata JC, da Costa JP, Duarte AC, Rocha-Santos T (2019) Methods for sampling and detection of microplastics in water and sediment: a critical review. TrAC-Trends Anal Chem 110:150–159. https://doi.org/10.1016/j.trac.2018.10.029
Prokić MD, Radovanović TB, Gavrić JP, Faggio C (2019) Ecotoxicological effects of microplatics: examination of biomarkers, current state and future perspectives. TrAC, Trends Anal Chem 111:37–46. https://doi.org/10.1016/j.trac.2018.12.001
Pruter AT (1987) Sources, quantities and distribution of persistent plastics in the marine environment. Mar Pollut Bull 18(6):305–310. https://doi.org/10.1016/S0025-326X(87)80016-4
Reineccius J, Waniek JJ (2022) First long-term evidence of microplastic pollution in the deep subtropical Northeast Atlantic. Environ Pollut 305:119302. https://doi.org/10.1016/j.envpol.2022.119302
Rey SF, Franklin J, Rey SJ (2021) Microplastic pollution on island beaches, Oahu, Hawai’i. PLoS One 16(2):e0247224. https://doi.org/10.1371/journal.pone.0247224
Rosenkranz P, Chaudhry Q, Stone V, Fernandes TF (2009) A comparison of nanoparticle and fine particle uptake by Daphnia magna. Environ Toxicol Chem 28(10):2142–2149. https://doi.org/10.1897/08-559.1
Setälä O, Fleming-Lehtinen V, Lehtiniemi M (2014) Ingestion and transfer of microplastics in the planktonic food web. Environ Pollut 185:77–83. https://doi.org/10.1016/j.envpol.2013.10.013
Strungaru SA, Jijie R, Nicoara M, Plavan G, Faggio C (2019) Micro (nano) plastics in freshwater ecosystems: abundance, toxicological impact and quantification methodology. Trends Anal Chem 110:116–128. https://doi.org/10.1016/j.trac.2018.10.025
Su L, Sharp SM, Pettigrove VJ, Craig NJ, Nan B, Du F, Shi H (2020) Superimposed microplastic pollution in a coastal metropolis. Water Res 168:115140. https://doi.org/10.1016/j.watres.2019.115140
Ta AT, Babel S, Haarstrick A (2020) Microplastics contamination in a high population density area of the Chao Phraya River, Bangkok. J Eng Technol Sci 52(4):534–545. https://doi.org/10.5614/j.eng.technol.sci.2020.52.4.6
Tanaka K, Takada H (2016) Microplastic fragments and microbeads in digestive tracts of planktivorous fish from urban coastal waters. Sci Rep 6:34351. https://doi.org/10.1038/srep34351
Teuten EL, Saquing JM, Knappe DRU, Barlaz MA, Jonsson S, Björn A, Rowland SJ, Thompson RC, Galloway TS, Yamashita R, Ochi D, Watanuki Y, Moore C, Viet PH, Tana TS, Prudente M, Boonyatumanond R, Zakaria MP, Akkhavong K, Ogata Y, Hirai H, Iwasa S, Mizukawa K, Hagino Y, Imamura A, Saha M, Takada H (2009) Transport and release of chemicals from plastics to the environment and to wildlife. Phil Trans R Soc B: Biol Sci 364:2027–2045. https://doi.org/10.1098/rstb.2008.0284
Thushari GGN, Senevirathna JDM (2020) Plastic pollution in the marine environment. Heliyon 6(8):e04709. https://doi.org/10.1016/j.heliyon.2020.e04709
Vandermeersch G, Van Cauwenberghe L, Janseen CR, Marques A, Granby K, Fait G, Kotterman MJJ, Diogène J, Bekaert K, Robbens J, Devriese L (2015) A critical view on microplastic quantification in aquatic organisms. Environ Res 143:46–55. https://doi.org/10.1016/j.envres.2015.07.016
Wang J, Peng J, Tan Z, Gao Y, Zhan Z, Chen Q, Cai L (2017a) Microplastics in the surface sediments from the Beijiang River littoral zone: composition, abundance, surface textures and interaction with heavy metals. Chemosphere 171:248–258. https://doi.org/10.1016/j.chemosphere.2016.12.074
Wang W, Ndungu AW, Li Z, Wang J (2017b) Microplastics pollution in inland freshwaters of China: a case study in urban surface waters of Wuhan, China. Sci Total Environ 575:1369–1374. https://doi.org/10.1016/j.scitotenv.2016.09.213
Wen X, Du C, Xu P, Zeng G, Huang D, Yin L, Yin Q, Hu L, Wan J, Zhang J, Tan S, Deng R (2018) Microplastic pollution in surface sediments of urban water areas in Changsha, China: abundance, composition, surface textures. Mar Pollut Bull 136:414–423. https://doi.org/10.1016/j.marpolbul.2018.09.043
Wu N, Zhang Y, Zhang X, Zhao Z, He J, Li W, Ma Y, Niu Z (2019) Occurrence and distribution of microplastics in the surface water and sediment of two typical estuaries in Bohai Bay, China. Environ Sci: Processes & Impacts 21(7):1143–1152. https://doi.org/10.1039/C9EM00148D
Wu P, Tang Y, Dang M, Wang S, Jin H, Liu Y, Jing H, Zheng C, Yi S, Cai Z (2020) Spatial-temporal distribution of microplastics in surface water and sediments of Maozhou River within Guangdong-Hong Kong-Macao Greater Bay Area. Sci Total Environ 717:135187. https://doi.org/10.1016/j.scitotenv.2019.135187
Yan M, Nie H, Xu K, He Y, Hu Y, Huang Y, Wang J (2019) Microplastic abundance, distribution and composition in the Pearl River along Guangzhou city and Pearl River estuary, China. Chemosphere 217:879–886. https://doi.org/10.1016/j.chemosphere.2018.11.093
Zhang K, Xiong X, Hu H, Wu C, Bi Y, Wu Y, Zhou B, Lam PKS, Liu J (2017) Occurrence and characteristics of microplastic pollution in Xiangxi Bay of Three Gorges Reservoir, China. Environ Sci Technol 51:3794–3801. https://doi.org/10.1021/acs.est.7b00369
Zhao S, Wang T, Zhu L, Xu P, Wang X, Gao L, Li D (2019) Analysis of suspended microplastics in the Changjiang Estuary: implications for riverine plastic load to the ocean. Water Res 161:560–569. https://doi.org/10.1016/j.watres.2019.06.019
Acknowledgements
This work was supported by a research grant from the “Marine Fisheries Research Strengthening and Infrastructural Development Project” under the Marine Station, Bangladesh Fisheries Research Institute, Cox’s Bazar, Bangladesh. The authors are thankful to Md. Ashraful Islam Sarker and Kazi Shahrukh Elahi for their help in sampling and technical assistance. The authors are also grateful to the anonymous reviewers for their constructive comments and suggestions.
Author information
Authors and Affiliations
Contributions
Kaniz Fatema analyzed data and drafted the manuscript. Turabur Rahman and Md. Helal Uddin collected and analyzed the sample. Md Jakiul Islam, Kizar Ahmed Sumon, Shanur Jahedul Hasan, Md. Mahfuzul Haque, and Hisayuki Arakawa provided technical and editorial assistance. S. M. Abe Kawsar performed chemical analysis (FTIR). Harunur Rashid conceived and supervised the study and edited the manuscript.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
All names in the author list have been involved throughout the study and writing.
Consent for publication
This manuscript was approved by all authors. Moreover, this work has original research that has not been published previously and is not under consideration for publication elsewhere, in whole or in part.
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible Editor: Ester Heath
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Kaniz Fatema and Turabur Rahman are equally contributing first authors.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fatema, K., Rahman, T., Islam, M.J. et al. Microplastics pollution in the river Karnaphuli: a preliminary study on a tidal confluence river in the southeast coast of Bangladesh. Environ Sci Pollut Res 30, 38853–38868 (2023). https://doi.org/10.1007/s11356-022-24998-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-24998-z