Abstract
The minireview is devoted to the analysis of the influence of soil pollution with heavy metals, polyaromatic hydrocarbons (PAHs), and the polychlorinated biphenyls (PCBs) on the distribution of antibiotics resistance genes (ARGs) in soil microbiomes. It is shown that the best understanding of ARGs distribution process requires studying the influence of pollutants on this process in natural microbiocenoses. Heavy metals promote co-selection of genes determining resistance to them together with ARGs in the same mobile elements of a bacterial genome, but the majority of studies focus on agricultural soils enriched with ARGs originating from manure. Studying nonagricultural soils would clear mechanisms of ARGs transfer in natural and anthropogenically transformed environments and highlight the role of antibiotic-producing bacteria. PAHs make a considerable shift in soil microbiomes leading to an increase in the number of Actinobacteria which are the source of antibiotics formation and bear multiple ARGs. The soils polluted with PAHs can be a selective medium for bacteria resistant to antibiotics, and the level of ARGs expression is much higher. PCBs are accumulated in soils and significantly alter the specific structure of soil microbiocenoses. In such soils, representatives of the genera Acinetobacter, Pseudomonas, and Alcanivorax dominate, and the ability to degrade PCBs is connected to horizontal gene transfer (HGT) and high level of genomic plasticity. The attention is also focused on the need to study the properties of the soil having an impact on the bioavailability of pollutants and, as a result, on resistome of soil microorganisms.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The development of antibiotic resistance is considered a major threat to the mankind. The large-scale use and misuse of antibiotics in medicine and agriculture have led to the selection of strains resistant to several or even all known classes of clinically relevant antibiotics. The threat had been long recognized by major international organizations, and World Health Organization takes efforts to increase public awareness on the issue (WHO 2015; Pires et al. 2017). Taking into account the severity of the human health and economic consequences, action plans are proposed by scientific organizations (Fuentes et al. 2017), and measures are taken by governments of many countries (WHO 2015; Xiao and Li 2016; Assiri and Banjar 2017).
Though the global efforts to struggle with the antibiotic resistance are focused mainly on the clinically relevant strains of pathogenic bacteria, it should be noted that the origin of antibiotic resistance is in the environment, rather than in human or animal organisms. It has been shown that the ARGs existed before the onset of the antibiotic era, and ARGs of pathogenic bacteria are identical to those found in environmental strains (D'Costa et al. 2011). It has been recently shown that horizontal gene transfer (HGT) took place both in the past and quite recently, and there is direct evidence that the source of ARGs is antibiotic-producing actinobacteria (Jiang et al. 2017).
It is now well established that the environments such as soil, water bodies, plant and animal-associated, and artificial ecosystems harbor microorganisms with ARGs and capabilities for their mobilization (Surette and Wright 2017). The sum of all ARGs in a particular environment is often referred to as “resistome”
The community-level studies, including the resistome studies based on isolation of ARGs from the diversity of environmental strains, always rely on the local conditions that influence community composition and functions. Such conditions include abiotic factors, biotic factors, and various disturbances (Luzuriaga et al. 2012). Bacterial communities are particularly sensitive to anthropogenic stress, which leads to their possible use in bioindication (Gorovtsov et al. 2017). However, this sensitivity makes it quite difficult to predict and control the processes linked to antibiotic resistance in complex natural environments, especially in soils. Thus, the interactions between the anthropogenic impact on the soils and the soil resistome remain mostly unclear. Still, there is a growing body of research dedicated to such interactions.
The present review aims to analyze the available data on the influence of pollution on the soil resistome, with particular attention paid to heavy metals, PAHs, and PHBs. This study is aimed to highlight the significance of major anthropogenic pollutants in co-selection of ARGs in soils that are not affected by resistant strains originating from clinical or veterinary practices. Heavy metals, PAHs, and PCBs are among the substances that are introduced into the environment in large quantities from industrial and urban sources. To date, only a few studies link the persistent organic pollutants, including PAHs and PCBs, to the selection of ARGs in the soil. The present review is an attempt to summarize and analyze the available scarce data.
The influence of heavy metals on the emergence of antibiotic resistance in polluted soils
Heavy metals have been used for different purposes for thousands of years. Their adverse effects on human health are well known, but in some areas, the exposure to heavy metals continues to grow (Järup 2003). Heavy metal contamination has been studied extensively for decades, but the vast majority of the papers consider either the issues of potential health risks linked to their accumulation in agricultural products (Blanco et al. 2017; Marrugo-Negrete et al. 2017) or the influence of pollution on natural ecosystems functioning and stability (Ding et al. 2018; Wang et al. 2017).
However, there is a significant indirect consequence of heavy metal pollution, that is the selection and accumulation of ARGs in the microbial communities of the polluted soils.
The first report of the link between resistance to antibiotics and heavy metals appeared in the 1960s when this threat was only starting to be recognized (Novick and Morse 1967). By now, the mechanisms of co-resistance to several agents have been well established. Co-resistance development in microorganisms is caused by location of two or more different resistance genes on the same plasmid, transposon, or integron (Chapman 2003). It has been shown that the antibiotic resistance genes are not an intrinsic trait of such genetic structures and that the acquisition of ARGs by pre-existing plasmids and transposons is a recent event (Mindlin et al. 2005).
It is quite natural to assume that the R-plasmids and transposons comprise a multi-tool, developed by microorganisms to cope with environmental threats, and that the core of such genetic structures remain stable, while the “weaponry” that is included changes in response to changing environment. Still, the exact pathways and conditions leading to the transfer of ARGs cassettes to R-plasmids remain largely unclear. One of the proposed mechanisms includes the class I integrons, which are widespread and act as recruiters of ARGs from environmental strains (Gillings 2014). It is noted that class I integrons are prevalent in metal-contaminated environments (Poole 2017). Thus, the process of ARGs transfer is enhanced, and the resistant strains may be selected by the local heterogeneity of metal concentrations. It should be noted that the majority of the studies of ARGs transfer in metal-contaminated environments were performed in aquatic ecosystems (Rosewarne et al. 2010; Su et al. 2014). The cause of this high attention to aquatic ecotopes is that water bodies are well-recognized sources of clinically relevant antibiotic-resistant bacteria (ARB) spreading (Guo et al. 2017; Hsu et al. 2017).
However, the soil comprises an environment with more uneven properties, especially at the microscale level, which is the real niche for soil microorganisms. The levels of heavy metals capable of influencing the bacteria are dependent on the processes of adsorption-desorption of metals on clay minerals, interactions with humic substances, microaggregate stability, and many other factors (Dror et al. 2017). This leads to a view of contaminated soil as a highly heterogeneous matrix with myriads of hot spots for the selection of resistant strains.
The most recent findings of the linkage of heavy metal contamination of soils and antibiotic resistance genes are summarized in Table 1.
The analysis of recent data on co-resistance to antibiotics and heavy metals in soil bacterial communities has shown that most of the studies are focused on agricultural soils. The most frequent objects of study are the farmland soils amended with composts produced from manure, or less frequently, with wastewater sludge.
The search for metal-antibiotic co-resistance in manure-amended soil is due to extensive use of both heavy metals and antibiotics in animal husbandry. The livestock feeds are often supplemented with trace elements, but most of them do not absorb in the intestines which leads to their accumulation in the manure (Xiong et al. 2010). The prophylactic use of antibiotics remains a widespread practice in animal husbandry despite the criticism and concerns for its role in antibiotic resistance development. Even the therapeutic use of antimicrobials poses an environmental threat (Cabello and Godfrey 2016). This makes the agricultural soils affected by animal waste the most apparent object for the research of co-resistance. As for the wastewater sludge, it is a well-documented source of ARB (Xie et al. 2016) and can contain relatively high concentrations of heavy metals, precipitated during the water treatment.
There is only limited evidence and few papers dealing with antibiotic resistance in soils, contaminated with heavy metals from other sources. However, the development of antibiotic resistance in microbial communities without obvious source of antibiotics (like sewage sludge or manure) is an issue of great importance. The intentional applications of wastewater treatment sludge or organic fertilizers take place on specific sites and therefore can be monitored and controlled. The unintentional metal pollution, originating from industrial sources and spreading with air or groundwater, is much more widespread and hard to prevent.
It has been shown that the microbial communities of soils amended with copper and that had not been previously subject to other agricultural or industrial discharges still developed antibiotic resistance via co-selection (Berg et al. 2005). There is an urgent need to study the non-agricultural soils, which are subject to metal pollution, including the urban soils, soils of mining areas, soils along the major highways, etc. The studies of soils which had not been in direct contact with antibiotics produced artificially can contribute to our knowledge of the process of gene transfer from the natural pool of ARGs present in environmental strains.
Another major concern is the existing gap between the studies of the ARGs in polluted soils and the studies of the environmental fate of the antibiotics. The reviews of antibiotic resistance tend to focus on the ARGs and the mechanisms of HGT but do not pay attention to the antibiotic substances themselves and their interaction with the environment. There is significant data available on the interaction of antibiotics with soil inorganic and organic matter (Pils and Laird 2007), and the current knowledge of processes of sorption, desorption, and degradation of antibiotics in soil was summarized in a recent review (Hashmi et al. 2017). The origin of this gap is the complete difference in methods utilized in the studies of the ARGs and soil-antibiotic interactions, which are difficult to be combined in a single study. However, a joint research could bring new insights into the development of antibiotic resistance in soil environment.
Most of the studies focused on the ARGs and microbial communities leave the basic soil properties aside, though they can have a significant influence on the development of resistance. The data on soil properties, if present, are taken into supplementary materials and are almost never used in the discussion of the results. At the same time, it has been shown that clay minerals may be a significant source of DNA for soil bacteria, adsorbing up to 70 μg of DNA per dry gram of soil.(Gardner and Gunsch 2017). Lv et al. (2017) have shown that montmorillonite drastically reduces antimicrobial activity of tetracycline, which could lead to selection of antibiotic-resistant bacteria. Kaolinite had a less pronounced effect. This data leads to the conclusion that the physical properties of soils, such as particle size composition, should not be underestimated in research of antibiotic resistance development.
To conclude, the soils as a major pool of environmental ARGs and an optimal environment for HGT and selection of resistant strains should receive proper attention from the researchers. The focus of these studies should be moved from farmland soils, and wastewater treatment sludge-amended soils to a broader range of soils subject to atmospheric metal pollution and include undisturbed soils as controls. Finally, the studies of ARGs and metal resistance genes (MRGs) distribution in metal-polluted soils should come along with the studies of soil properties significantly influencing all aspects of soil microbial communities functioning.
The influence of PAHs on the emergence of antibiotic resistance in polluted soils
PAHs are ubiquitous molecules of predominantly anthropogenic origin that pose a considerable threat to humans and environment. The primary sources of anthropogenic PAHs include coal pyrolysis, liquid fossil fuels, and biomass combustion (Bosch et al. 2015). Many of the PAHs possess genotoxic and mutagenic effects. Some of them are established or suspected human carcinogens. The mechanism of PAH action is based on metabolic activation of their molecules with cytochrome P450 leading to formation of highly reactive electrophilic species binding with DNA (Binková and Šrám 2004). This mechanism leads to high level of toxicity to both eukaryotic and prokaryotic organisms.
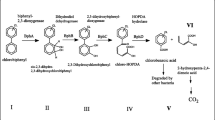
The PAH contamination has been shown to influence the microbial community significantly, leading to major shifts in phylum-level community composition in the affected soils (Yang et al. 2014). The dominant phylum in most of the PAH-contaminated soils is Proteobacteria (Yu et al. 2017). However, a significant growth of Actinobacteria abundance has been shown in batch experiments after contamination (Muangchinda et al. 2017; Zhu et al. 2017). The representatives of Actinobacteria are known for their capabilities to degrade PAHs which is best known for such genera as Arthrobacter and Rhodococcus. However, there is enough evidence for the existence of PAH-degrading strains in Streptomyces genus (Chaudhary et al. 2011; Balachandran et al. 2012). At the same time, the members of Streptomyces genus are the main source of antibiotic production and have been identified as bearing multiple antibiotic resistance genes (D'costa et al. 2006).
These findings have led to concern that PAH-contaminated soils could be a selective environment for antibiotic-resistant bacteria and that the ARGs could be further transferred to pathogenic species. The screening of major scientific information databases has shown that the number of recent studies linking the PAHs contamination with antibiotic resistance is several times lower in comparison to research of heavy metal contamination. This difference may be due to complicated methods of isolation and quantification of PAHs in environmental objects that need labor- and time-consuming methods and advanced analytical instruments.
In this section of present review, we shall summarize the available findings in this area. Some of the examples of the coincidence of PAH tolerance and degradation and antibiotic resistance in soil bacteria are given in Table 2.
The examples listed in the table show that most frequently, the antibiotic resistance developed in the representatives of Proteobacteria and Actinobacteria phyla. Among the Proteobacteria, the genus Pseudomonas was mentioned in the majority of studies, which corresponds to their remarkable capability to degrade organic pollutants.
The study of diesel-contaminated soils (Arenic anthrosols) in Bălan, Romania, has shown that there was a significant correlation between heavy metal tolerance and antibiotic resistance as well as between antibiotic resistance and hydrocarbon degradation ability in the isolated strains of bacteria. Most of the identified species belong to Pseudomonas genus, including Pseudomonas corrugata, Pseudomonas fluorescens, Pseudomonas putida, Pseudomonas veronii, Pseudomonas mandelii, and Pseudomonas syringae. Some representatives of Actinobacteria, including Rhodococcus erythropolis and Dietzia psychralcaliphila were also isolated (Máthé et al. 2012).
The level of expression of ARGs in PAH-contaminated soil was found to be significantly higher. It was also influenced by the presence of willow plants due to rhizosphere effects, and the ARG expression levels measured by Illumina mRNA sequencing were higher in the rhizosphere regardless of contamination level (Yergeau et al. 2014). The increased expression of ARGs in the rhizosphere is a worrying finding, as phytoremediation is a widely used practice in the recultivation of PAH-polluted soils (Bisht et al. 2014).
It should be noted that after entering the soil, PAHs can interact with soil particles and are not always bioaccessible. Sun et al. (2015) found that there is no correlation between total content of phenanthrene in soil and the ARGs abundance, but significant correlations were found for the bioaccessible portion of the contaminant. The bioaccessibility of PAHs in soil is a subject of many separate studies and can not be covered in detail in this review.
However, some of the mechanisms of PAH interaction with soil should be mentioned. PAHs comprise nonpolar hydrophobic compounds that may interact with soil particles through hydrophobic partitioning or via chemical or physical bond formation. The concentration and composition of soil organic matter are also of great importance. In general, the contaminants in soils with higher organic matter content tend to have lower bioavailability. The same is true for clay content, due to high level of pollutants sorption on the surfaces of soil clay minerals (Rostami and Juhasz 2011). Therefore, the soil properties can significantly alter the effective concentrations of PAHs in soil, which can lead to errors in the estimation of the influence that contamination has on the development of antibiotic resistance.
The exact mechanisms underlying the observed enrichment of ARGs in PAH-contaminated soils are not understood in full details. There is considerable evidence that many of the efflux pumps that discharge organic pollutants from bacterial cells are functionally similar or even homologous to multidrug efflux pumps, determining the antibiotic resistance (Martinez et al. 2009). EmhABC efflux pump has been shown to remove both antibiotics and PAHs, including phenanthrene in Pseudomonas fluorescens (Adebusuyi and Foght 2011). The genes, coding for such pumps, can be inserted to the same mobile genetic elements with the help of intl1 integrons, which is supported by higher abundance of intl1 in contaminated soils (Sun et al. 2015). Hydrocarbons have been shown to increase the level of ROS in the bacterial cell, which can damage DNA and, accordingly, activate its reparation. This may be the reason for the intensification of mutagenesis and HGT processes (Pérez-Pantoja et al. 2013; Sazykin et al. 2016).
It has been shown in a recent study that the contamination of soil with PAHs enriched the abundance of Proteobacteria and ARGs in soil. The identified genes were mostly represented by efflux pump-encoding ARGs associated with aromatic antibiotics and were located at the chromosomes of PAH-tolerant bacteria rather than plasmids, suggesting that the plasmids were not the primary vector of gene transfer (Chen et al. 2017). The contamination of soil with hydrocarbons has been shown to promote not only antibiotic resistance but also to increase production of antibiotic compounds by indigenous soil bacteria (Hemala et al. 2014). This enhanced antibiotic production could contribute to the selective pressure, leading to the enrichment of resistant strains.
However, there are some recent evidence that PAH contamination can even reduce the horizontal transfer of the ARGs. It has been shown that due to non-covalent binding of PAHs and plasmids, the rate of transformation is significantly reduced. This effect was most pronounced for smaller PAHs, namely phenanthrene, less significant for pyrene, and no significant influences on transformation rates was obtained for benzo-a-pyrene (Kang et al. 2015). These findings, supported by the data of Chen et al. (2017), of predominantly chromosomal location of efflux-pump encoding genes lead to a hypothesis that the mechanism of ARG enrichment in PAH-contaminated soils may be different from what has been shown for heavy metal pollution.
If the HGT is suspended in PAH-contaminated environment, the number of ARG copies can still increase with the growth of ARG-bearing PAH-degrading bacteria population. Moreover, the growth of this community will eventually lead to a decrease in bioavailable PAH content due to biodegradation.
After exhausting the pool of available polyaromatic hydrocarbons, the population will inevitably decline due to increasing competition from bacteria that are not able to utilize PAHs. It has been shown in a model experiment by phospholipid fatty acid analysis (PLFA) that the microbial community structure of oil-treated experimental plots became similar to unoiled controls 14 weeks after treatment (MacNaughton et al. 1999). At this stage, the HGT is likely to happen, as the environmental DNA from dead cells serves as an essential source of genetic material for bacteria (Takeuchi et al. 2014).
It may be concluded that our knowledge of PAH interactions with soil resistome remains mostly fragmented and a lot of research has to be done to elucidate the mechanisms and factors underlying the observed findings.
The influence of PCBs on the emergence of antibiotic resistance in polluted soils
PCBs are a class of organochlorine compounds that have been considered as persistent organic pollutants (Lyall et al. 2017). Their usage in many countries was banned decades ago, but due to meager rates of degradation, they are still found in atmospheric precipitation and soils all over the world (Chakraborty et al. 2016). Their distribution in the environment is highly uneven with elevated concentrations found in urban areas, particularly at the electronic waste recycling sites (Jiang et al. 2011). The PCBs in urban areas tend to originate from multiple pyrogenic sources, which has been shown for the soils of eastern London (Vane et al. 2014).
The soil is a major sink for the PCBs which is similar to many other classes of organic pollutants due to their strong affinity to the soil organic matter. However, these substances are semi-volatile which leads to exchange between polluted soil and atmosphere. It has been shown that for some polychlorinated compounds, including PCBs, diurnal variations of this exchange are present, with increased volatilization during the day and deposition at night (Degrendele et al. 2016).
This process can lead to increased persistence of PCBs in the surface soil layers, unlike many other contaminants that are found deeper into the soil profile with time. The process of leaching and deposition of new material on the soil surface eventually leaves the pollutants in the buried soil layers, which is not the case for semi-volatile organic compounds. It should be noted that surface soil layers are characterized by the highest activity of soil microorganisms, and thus, the PCBs comprise a persistent threat to soil microbial communities and ecosystems as a whole.
PCBs have been shown to cause significant shifts in bacterial community composition of affected soils. A recent study has shown that in the soils polluted with PCBs originating from e-waste processing bacteria belonging to Acinetobacter, Pseudomonas, and Alcanivorax, became dominant species (Liu et al. 2015). The representatives of these bacterial genera have been previously shown to degrade PCBs (Field and Sierra-Alvarez 2008) which explains their selective enrichment in contaminated soils.
At the same time, pathogenic strains of Acinetobacter and Pseudomonas are considered the most capable of developing antibiotic resistance, which leads to an assumption that the PCBs pollution can cause co-selection of antibiotic-resistant bacteria in the same way as it was shown for heavy metals and PAHs.
Another finding indicates that commonly used antiseptic triclosan causes overexpression of MexAB-OprM efflux pump, which leads to multi-drug resistance in Pseudomonas aeruginosa (Chuanchuen et al. 2001). Triclosan (5-chloro-2-(2,4-dichlorophenoxy)phenol) is structurally very similar to PCBs and PBDEs (Lee and Chu 2013). Thus, PCBs are likely to cause the development of resistance to antibiotics.
To date, the number of studies on this issue is insufficient. The PCB-degrading bacteria from Psychrobacter genus, isolated from shallow Antarctic sediments, were tolerant to heavy metals and resistant to chloramphenicol and ampicillin (Giudice et al. 2013).
PCB-degrading strain of Pseudomonas putida was shown to have remarkable genomic instability, including the presence of four plasmids, an integrative and conjugative element (ICE) bph-sal (Suenaga et al. 2017). The PCB-degradation genes in Acidovorax were also located on ICE (Ohtsubo et al. 2012).
ICE are recognized as an essential factor of microbial adaptation to environmental conditions (Burrus and Waldor 2004) and have been shown to harbor ARGs in previous studies (Carraro et al. 2015). Rhodococcus species harbor a wide set of conjugative plasmids, including PTA421, PSP6, and PLP3 coding for PCBs degradation genes and PFiD188 plasmid determining chloramphenicol resistance (Fetzner et al. 2007).
These findings indicate that the ability to degrade PCBs is strongly associated with the mechanisms of HGT and a high level of genomic plasticity which makes the PCB-polluted soils the most likely environment for the emergence of antibiotic resistance. Despite this fact, the data on the enrichment of ARGs in PCB-degrading bacteria in soil is currently missing.
Conclusions and perspectives
The development of antibiotic resistance is the most serious challenge of the recent decades. The efforts to cope with this problem should not be limited to the restriction of the uncontrolled use of antibiotics in healthcare and agriculture antibiotic leading to their discharge into the environment. Despite the fact that these aspects of combating antibiotic resistance remain fundamental, attention should also be paid to the study of environmental enrichment and selection of the ARGs.
The study of recent findings on that issue revealed that there are significant gaps in our knowledge of the environmental fate of the ARGs in heavy metal contaminated soils. The research is focused on the farmland soils, where both metals and antibiotics originate from animal feeds. The ARGs start to be enriched in the animal guts, and then enrichment continues in the manure (Xiong et al. 2010) and manure-amended soils (Ji et al. 2012). Much less data is available for the non-agricultural soils polluted by industrial dust reaching the soil with atmospheric precipitation. The studies of such soils could elucidate the mechanisms of ARG transfer from the natural gene pool and the role of antibiotic-producing bacteria in the emergence of new ARGs. The study of co-selection process in the environments that are not affected by an external source of antibiotics is likely to bring new insights into this field.
There is a certain degree of controversy in our knowledge of the antibiotic resistance development in PAH-contaminated soils. On the one hand, there are many examples of coincidence of antibiotic resistance and PAH tolerance is the same strains of bacteria. On the other hand, there are indications of reduced rate of HGT in PAH-contaminated soils (Kang et al. 2015). The proteins of EmhABC family that can pump out both antibiotics and hydrocarbons are studied not sufficiently, especially concerning the regulation of their work. The exact mechanisms of interaction between the gene coding for such efflux pumps other ARGs and intl1 integrons are also not clear enough.
An insufficient amount of data is available for the development of antibiotic resistance in soils, contaminated with persistent organic pollutants, particularly with PCBs. Though the PCB pollution of soils has been studied for decades, and many efforts are taken in the field of biodegradation and bioremediation, they are rarely considered in conjunction with antibiotic resistance. However, many facts support their possible role in antibiotic resistance development, including high genomic plasticity of PCB-degrading strains and the dominance of taxonomic groups known for high rates of ARGs emergence.
Finally, in the studies of antibiotic resistance development in heavy metal, PAH, and PCB contaminated soils, more attention should be paid to the soil properties, including the interactions of the contaminants with soil organic matter, processes of sorption on clay minerals, the issues of bioavailability of the pollutants in the specific soil conditions, etc.
The attention paid to the environmental issues regarding the antibiotic resistance development would help to find and implement adequate measures against spreading the antibiotic-resistant bacteria in soils. At the same time, these studies can increase public awareness of the negative consequences of soil contamination.
Such problems are widely recognized for agricultural soils, as the possibility of contaminants entering the food chain and affecting human health is evident. The contamination of soils that are not used for growing crops, for instance, of urban soils or soils of industrial areas attracts the attention of scientists but not of the general public. The data analyzed in the present review shows that the threat of soil contamination far exceeds the food quality issues and should not be underestimated regardless of the type of land use.
References
Adebusuyi AA, Foght JM (2011) An alternative physiological role for the EmhABC efflux pump in Pseudomonas fluorescens cLP6a. BMC Microbiol 11(1):252. https://doi.org/10.1186/1471-2180-11-252
Assiri AM, Banjar WM (2017) The strategic plan for combating antimicrobial resistance in gulf cooperation council states, KSA perspective. J Infect Public Health 10(5):485–486. https://doi.org/10.1016/j.jiph.2016.09.013
Balachandran C, Duraipandiyan V, Balakrishna K, Ignacimuthu S (2012) Petroleum and polycyclic aromatic hydrocarbons (PAHs) degradation and naphthalene metabolism in Streptomyces sp. (ERI-CPDA-1) isolated from oil-contaminated soil. Bioresour Technol 112:83–90. https://doi.org/10.1016/j.biortech.2012.02.059
Berg J, Tom-Petersen A, Nybroe O (2005) Copper amendment of agricultural soil selects for bacterial antibiotic resistance in the field. Lett Appl Microbiol 40(2):146–151. https://doi.org/10.1111/j.1472-765X.2004.01650.x
Binková B, Šrám RJ (2004) The genotoxic effect of carcinogenic PAHs, their artificial and environmental mixtures (EOM) on human diploid lung fibroblasts. Mutat Res 547(1-2):109–121. https://doi.org/10.1016/j.mrfmmm.2003.12.006
Bisht S, Kumar V, Kumar M, Sharma S (2014) Innoculant technology in Populus deltoides rhizosphere for effective bioremediation of Polyaromatic hydrocarbons (PAHs) in contaminated soil, Northern India. Emir J Food and Agric 26(9):786. https://doi.org/10.9755/ejfa.v26i9.18436
Blanco A, Salazar MJ, Cid CV, Pignata ML, Rodriguez JH (2017) Accumulation of lead and associated metals (Cu and Zn) at different growth stages of soybean crops in lead-contaminated soils: food security and crop quality implications. Environ Earth Sci 76(4):182. https://doi.org/10.1007/s12665-017-6508-x
Bosch C, Andersson A, Kruså M, Bandh C, Hovorková I, Klánová J, Knowles TD, Pancost RD, Evershed RP, Gustafsson O (2015) Source apportionment of polycyclic aromatic hydrocarbons in central European soils with compound-specific triple isotopes (δ13C, Δ14C, and δ2H). Environ Sci Technol 49(13):7657–7665. https://doi.org/10.1021/acs.est.5b01190
Burrus V, Waldor MK (2004) Shaping bacterial genomes with integrative and conjugative elements. Res Microbiol 155(5):376–386. https://doi.org/10.1016/j.resmic.2004.01.012
Cabello FC, Godfrey HP (2016) Even therapeutic antimicrobial use in animal husbandry may generate environmental hazards to human health. Environ Microbiol 18(2):311–313. https://doi.org/10.1111/1462-2920.13247
Campbell JI, Jacobsen CS, Sørensen J (1995) Species variation and plasmid incidence among fluorescent Pseudomonas strains isolated from agricultural and industrial soils. FEMS Microbiol Lett 18(1):51–62. https://doi.org/10.1111/j.1574-6941.1995.tb00163.x
Carraro N, Poulin D, Burrus V (2015) Replication and active partition of integrative and conjugative elements (ICEs) of the SXT/R391 family: the line between ICEs and conjugative plasmids is getting thinner. PLoS Genet 11(6):e1005298. https://doi.org/10.1371/journal.pgen.1005298
Chakraborty P, Zhang G, Li J, Selvaraj S, Breivik K, Jones KC (2016) Soil concentrations, occurrence, sources and estimation of air-soil exchange of polychlorinated biphenyls in Indian cities. Sci Total Environ 562:928–934. https://doi.org/10.1016/j.scitotenv.2016.03.009
Chapman JS (2003) Disinfectant resistance mechanisms, cross-resistance, and co-resistance. Int Biodeterior Biodegrad 51(4):271–276. https://doi.org/10.1016/S0964-8305(03)00044-1
Chaudhary P, Sharma R, Singh SB, Nain L (2011) Bioremediation of PAH by Streptomyces sp. Bull Environ Contam Toxicol 86(3):268–271. https://doi.org/10.1007/s00128-011-0211-5
Chen B, He R, Yuan K, Chen E, Lin L, Chen X, Sha S, Zhong J, Lin L, Yang L, Yang Y, Wang X, Zou S, Luan T (2017) Polycyclic aromatic hydrocarbons (PAHs) enriching antibiotic resistance genes (ARGs) in the soils. Environ Pollut 220(Pt B):1005–1013. https://doi.org/10.1016/j.envpol.2016.11.047
Chuanchuen R, Beinlich K, Hoang TT, Becher A, Karkhoff-Schweizer RR, Schweizer HP (2001) Cross-resistance between triclosan and antibiotics in Pseudomonas aeruginosa is mediated by multidrug efflux pumps: exposure of a susceptible mutant strain to triclosan selects nfxB mutants overexpressing MexCD-OprJ. Antimicrob Agents Chemother 45(2):428–432. https://doi.org/10.1128/AAC.45.2.428-432.2001
D'costa VM, McGrann KM, Hughes DW, Wright GD (2006) Sampling the antibiotic resistome. Science 311(5759):374–377. https://doi.org/10.1126/science.1120800
D'Costa VM, King CE, Kalan L, Morar M, Sung WW, Schwarz C, Froese D, Zazula G, Calmels F, Debruyne R, Golding GB, Poinar HN, Wright GD (2011) Antibiotic resistance is ancient. Nature 477(7365):457–461. https://doi.org/10.1038/nature10388
Degrendele C, Audy O, Hofman J, Kučerik J, Kukučka P, Mulder MD, Přibylová P, Prokeš R, Šáňka M, Schaumann GE, Lammel G (2016) Diurnal variations of air-soil exchange of semivolatile organic compounds (PAHs, PCBs, OCPs, and PBDEs) in a central European receptor area. Environ Sci Technol 50(8):4278–4288. https://doi.org/10.1021/acs.est.5b05671
Ding K, Wu Q, Wei H, Yang W, Séré G, Wang S, Echevarria G, Tang Y, Tao J, Morel JL, Qiu R (2018) Ecosystem services provided by heavy metal-contaminated soils in China. J Soils Sediments 18(2):380–390. https://doi.org/10.1007/s11368-016-1547-6
Dror I, Yaron B, Berkowitz B (2017) Microchemical contaminants as forming agents of anthropogenic soils. Ambio 46(1):109–120. https://doi.org/10.1007/s13280-016-0804-7
Duan M, Gu J, Wang X, Li Y, Li P, Yin Y (2017) Combined effects of compost containing sulfamethazine and zinc on pakchoi (Brassica chinensis L.) growth, soil sulfonamide resistance genes, and microbial communities. Arch Agron Soil Sci. https://doi.org/10.1080/03650340.2017.1342033
Fetzner S, Kolkenbrock S, Parschat K (2007) Catabolic linear plasmids. In: Meinhardt F, Klassen R (eds) Microbial linear plasmids. Springer, Berlin, pp 63–98. https://doi.org/10.1007/7171_2007_091
Field JA, Sierra-Alvarez R (2008) Microbial transformation and degradation of polychlorinated biphenyls. Environ Pollut 155(1):1–12. https://doi.org/10.1016/j.envpol.2007.10.016
Fuentes E, Wickham C, Carbajal C, Lopez C, Jauregui S, Lechler R, Kannan S (2017) Genesis of antibiotic resistance XXVII: action plan for global Union for Antibiotics Research and Development (GUARD) to mitigate AR pandemic (ARP). FASEB J 31:777–779
Gardner CM, Gunsch CK (2017) Adsorption capacity of multiple DNA sources to clay minerals and environmental soil matrices less than previously estimated. Chemosphere 175:45–51. https://doi.org/10.1016/j.chemosphere.2017.02.030
Gillings MR (2014) Integrons: past, present, and future. Microbiol Mol Biol Rev 78(2):257–277. https://doi.org/10.1128/MMBR.00056-13
Giudice AL, Casella P, Bruni V, Michaud L (2013) Response of bacterial isolates from Antarctic shallow sediments towards heavy metals, antibiotics and polychlorinated biphenyls. Ecotoxicology 22(2):240–250. https://doi.org/10.1007/s10646-012-1020-2
Gorovtsov A, Rajput VD, Gorbov S, Vasilchenko N (2017) Bioindication-based approaches for sustainable management of urban ecosystems. In: Singh R, Kumar S (eds) Green technologies and environmental sustainability. Springer, Cham, pp 203–228. https://doi.org/10.1007/978-3-319-50654-8_9
Guo J, Li J, Chen H, Bond P, Yuan Z (2017) Metagenomic analysis reveals wastewater treatment plants as hotspots of antibiotic resistance genes and mobile genetic elements. Water Res 123:468–478. https://doi.org/10.1016/j.watres.2017.07.002
Hashmi MZ, Mahmood A, Kattel DB, Khan S, Hasnain A, Ahmed Z (2017) Antibiotics and antibiotic resistance genes (ARGs) in soil: occurrence, fate, and effects. In: Hashmi M, Kumar V, Varma A (eds) Xenobiotics in the soil environment. Springer International Publishing, Cham, pp 41–54. https://doi.org/10.1007/978-3-319-47744-2_4
Hearn EM, Dennis JJ, Gray MR, Foght JM (2003) Identification and characterization of the emhABC efflux system for polycyclic aromatic hydrocarbons in Pseudomonas fluorescens cLP6a. J Bacteriol 185(21):6233–6240. https://doi.org/10.1128/JB.185.21.6233-6240.2003
Hemala L, Zhang D, Margesin R (2014) Cold-active antibacterial and antifungal activities and antibiotic resistance of bacteria isolated from an alpine hydrocarbon-contaminated industrial site. Res Microbiol 165(6):447–456. https://doi.org/10.1016/j.resmic.2014.05.035
Hsu TTD, Mitsch WJ, Martin JF, Lee J (2017) Towards sustainable protection of public health: the role of an urban wetland as a frontline safeguard of pathogen and antibiotic resistance spread. Ecol Eng. https://doi.org/10.1016/j.ecoleng.2017.02.051
Hu HW, Wang JT, Li J, Shi XZ, Ma YB, Chen D, He JZ (2017) Long-term nickel contamination increases the occurrence of antibiotic resistance genes in agricultural soils. Environ Sci Technol 51(2):790–800. https://doi.org/10.1021/acs.est.6b03383
Järup L (2003) Hazards of heavy metal contamination. Br Med Bull 68(1):167–182. https://doi.org/10.1093/bmb/ldg032
Ji X, Shen Q, Liu F, Ma J, Xu G, Wang Y, Wu M (2012) Antibiotic resistance gene abundances associated with antibiotics and heavy metals in animal manures and agricultural soils adjacent to feedlots in Shanghai; China. J Hazard Mater 235-236:178–185. https://doi.org/10.1016/j.jhazmat.2012.07.040
Jiang Y, Wang X, Zhu K, Wu M, Sheng G, Fu J (2011) Polychlorinated biphenyls contamination in urban soil of shanghai: level, compositional profiles and source identification. Chemosphere 83(6):767–773. https://doi.org/10.1016/j.chemosphere.2011.02.077
Jiang X, Ellabaan MMH, Charusanti P, Munck C, Blin K, Tong Y, Weber T, Sommer MOA, Lee SY (2017) Dissemination of antibiotic resistance genes from antibiotic producers to pathogens. Nat Commun 8:15784. https://doi.org/10.1038/ncomms15784
Kang F, Hu X, Liu J, Gao Y (2015) Noncovalent binding of polycyclic aromatic hydrocarbons with genetic bases reducing the in vitro lateral transfer of antibiotic resistant genes. Environ Sci Technol l49:10340–10348
Knapp CW, Callan AC, Aitken B, Shearn R, Koenders A, Hinwood A (2017) Relationship between antibiotic resistance genes and metals in residential soil samples from Western Australia. Environ Sci Pollut Res Int 24(3):2484–2494. https://doi.org/10.1007/s11356-016-7997-y
Lee DG, Chu KH (2013) Effects of growth substrate on triclosan biodegradation potential of oxygenase-expressing bacteria. Chemosphere 93(9):1904–1911. https://doi.org/10.1016/j.chemosphere.2013.06.069
Lin H, Sun W, Zhang Z, Chapman SJ, Freitag TE, Fu J, Zhang X, Ma J (2016) Effects of manure and mineral fertilization strategies on soil antibiotic resistance gene levels and microbial community in a paddy-upland rotation system. Environ Pollut 211:332–337. https://doi.org/10.1016/j.envpol.2016.01.007
Liu J, He XX, Lin XR, Chen WC, Zhou QX, Shu WS, Huang LN (2015) Ecological effects of combined pollution associated with e-waste recycling on the composition and diversity of soil microbial communities. Environ Sci Technol 49(11):6438–6447. https://doi.org/10.1021/es5049804
Luzuriaga AL, Sánchez AM, Maestre FT, Escudero A (2012) Assemblage of a semi-arid annual plant community: abiotic and biotic filters act hierarchically. PLoS One 7:1–9
Lv G, Li Z, Elliott L, Schmidt MJ, MacWilliams MP, Zhang B (2017) Impact of tetracycline-clay interactions on bacterial growth. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2017.09.029
Lyall K, Croen LA, Sjödin A, Yoshida CK, Zerbo O, Kharrazi M, Windham GC (2017) Polychlorinated biphenyl and organochlorine pesticide concentrations in maternal mid-pregnancy serum samples: association with autism spectrum disorder and intellectual disability. Environ Health Perspect 125:474
MacNaughton SJ, Stephen JR, Venosa AD, Davis GA, Chang YJ, White DC (1999) Microbial population changes during bioremediation of an experimental oil spill. Appl Environ Microbiol 65(8):3566–3574
Marrugo-Negrete J, Pinedo-Hernández J, Díez S (2017) Assessment of heavy metal pollution, spatial distribution and origin in agricultural soils along the Sinú River Basin, Colombia. Environ Res 154:380–388. https://doi.org/10.1016/j.envres.2017.01.021
Martinez JL, Sánchez MB, Martínez-Solano L, Hernandez A, Garmendia L, Fajardo A, Alvarez-Ortega C (2009) Functional role of bacterial multidrug efflux pumps in microbial natural ecosystems. FEMS Microbiol Rev 33(2):430–449. https://doi.org/10.1111/j.1574-6976.2008.00157.x
Máthé I, Benedek T, Táncsics A, Palatinszky M, Lányi S, Márialigeti K (2012) Diversity, activity, antibiotic and heavy metal resistance of bacteria from petroleum hydrocarbon contaminated soils located in Harghita County (Romania). Int Biodeterior Biodegrad 73:41–49. https://doi.org/10.1016/j.ibiod.2012.05.018
Mindlin S, Minakhin L, Petrova M, Kholodii G, Minakhina S, Gorlenko Z, Nikiforov V (2005) Present-day mercury resistance transposons are common in bacteria preserved in permafrost grounds since the Upper Pleistocene. Res Microbiol 156(10):994–1004. https://doi.org/10.1016/j.resmic.2005.05.011
Muangchinda C, Yamazoe A, Polrit D, Thoetkiattikul H, Mhuantong W, Champreda V, Pinyakong O (2017) Biodegradation of high concentrations of mixed polycyclic aromatic hydrocarbons by indigenous bacteria from a river sediment: a microcosm study and bacterial community analysis. Environ Sci Pollut Res Int 24(5):4591–4602. https://doi.org/10.1007/s11356-016-8185-9
Novick RP, Morse SI (1967) In vivo transmission of drug resistance factors between strains of Staphylococcus aureus. J Exp Med 125(1):45–59. https://doi.org/10.1084/jem.125.1.45
Ohtsubo Y, Ishibashi Y, Naganawa H, Hirokawa S, Atobe S, Nagata Y, Tsuda M (2012) Conjugal transfer of polychlorinated biphenyl/biphenyl degradation genes in Acidovorax sp. strain KKS102, which are located on an integrative and conjugative element. J Bacteriol 194(16):4237–4248. https://doi.org/10.1128/JB.00352-12
Pérez-Pantoja D, Nikel PI, Chavarría M, de Lorenzo V (2013) Endogenous stress caused by faulty oxidation reactions fosters evolution of 2,4-dinitrotoluene-degrading bacteria. PLoS Genet 9(8):e1003764. https://doi.org/10.1371/journal.pgen.1003764
Pils JR, Laird DA (2007) Sorption of tetracycline and chlortetracycline on K-and Ca-saturated soil clays, humic substances, and clay-humic complexes. Environ Sci Technol 41(6):1928–1933. https://doi.org/10.1021/es062316y
Pires D, de Kraker MEA, Tartari E, Abbas M, Pittet D (2017) ‘Fight antibiotic resistance—It’s in your hands’: call from the World Health Organization for 5th May 2017. Clin Infect Dis 64(12):1780–1783. https://doi.org/10.1093/cid/cix226
Poole K (2017) At the nexus of antibiotics and metals: the impact of Cu and Zn on antibiotic activity and resistance. Trends Microbiol 25(10):820–832. https://doi.org/10.1016/j.tim.2017.04.010
Pyrchenkova IA, Gafarov AB, Puntus IF, Filonov AE, Boronin AM (2006) Selection and characterization of active psychrotrophic microbial oil-degrading microorganisms. Appl Biochem Microbiol 42(3):263–269. https://doi.org/10.1134/S0003683806030070
Rosewarne CP, Pettigrove V, Stokes HW, Parsons YM (2010) Class 1 integrons in benthic bacterial communities: abundance, association with Tn 402-like transposition modules and evidence for co selection with heavy-metal resistance. FEMS Microbiol Ecol 72(1):35–46. https://doi.org/10.1111/j.1574-6941.2009.00823.x
Rostami I, Juhasz AL (2011) Assessment of persistent organic pollutant (POP) bioavailability and bioaccessibility for human health exposure assessment: a critical review. Crit Rev Environ Sci Technol 41(7):623–656. https://doi.org/10.1080/10643380903044178
Sazykin IS, Sazykina MA, Khmelevtsova LE, Khammami MI, Karchava SK, Zhuravlevа MV, Kudeevskaya EM (2016) Expression of SOD and production of reactive oxygen species in Acinetobacter calcoaceticus caused by hydrocarbons oxidation. Ann Microbiol 66(3):1039–1045. https://doi.org/10.1007/s13213-015-1188-9
Sinegani AAS, Younessi N (2017) Antibiotic resistance of bacteria isolated from heavy metal-polluted soils with different land uses. J Glob Antimicrob Resist 10:247–255. https://doi.org/10.1016/j.jgar.2017.05.012
Stancu MM (2014) Physiological cellular responses and adaptations of Rhodococcus erythropolis IBB Po1 to toxic organic solvents. J Environ Sci (China) 26(10):2065–2075. https://doi.org/10.1016/j.jes.2014.08.006
Stancu MM, Grifoll M (2011) Multidrug resistance in hydrocarbon-tolerant Gram-positive and Gram-negative bacteria. J Gen Appl Microbiol 57(1):1–18. https://doi.org/10.2323/jgam.57.1
Su HC, Pan CG, Ying GG, Zhao JL, Zhou LJ, Liu YS, Tao R, Zhang RQ, He LY (2014) Contamination profiles of antibiotic resistance genes in the sediments at a catchment scale. Sci Total Environ 490:708–714. https://doi.org/10.1016/j.scitotenv.2014.05.060
Suenaga H, Fujihara H, Kimura N, Hirose J, Watanabe T, Futagami T, Goto M, Shimodaira J, Furukawa K (2017) Insights into the genomic plasticity of Pseudomonas putida KF715, a strain with unique biphenyl-utilizing activity and genome instability properties. Environ Microbiol Rep 9(5):589–598. https://doi.org/10.1111/1758-2229.12561
Sun K, Liu J, Li X, Ling W (2014) Isolation, identification, and performance of two pyrene-degrading endophytic bacteria. Acta Ecol Sin 34(4):853–861. https://doi.org/10.5846/stxb201210091393
Sun M, Ye M, Wu J, Feng Y, Wan J, Tian D, Shen F, Liu K, Hu F, Li H Jiang X, Yang L, Kengara FO (2015) Positive relationship detected between soil bioaccessible organic pollutants and antibiotic resistance genes at dairy farms in Nanjing, Eastern China. Environ Pollut 206:421–428. https://doi.org/10.1016/j.envpol.2015.07.022
Surette M, Wright GD (2017) Lessons from environmental antibiotic resistome. Ann Rev Microbiol 71(1):309–329. https://doi.org/10.1146/annurev-micro-090816-093420
Takeuchi N, Kaneko K, Koonin EV (2014) Horizontal gene transfer can rescue prokaryotes from Muller’s ratchet: benefit of DNA from dead cells and population subdivision. G3 (Bethesda) 4(2):325–339. https://doi.org/10.1534/g3.113.009845
Vane CH, Kim AW, Beriro DJ, Cave MR, Knights K, Moss-Hayes V, Nathanail PC (2014) Polycyclic aromatic hydrocarbons (PAH) and polychlorinated biphenyls (PCB) in urban soils of Greater London, UK. Appl Geochem 51:303–314. https://doi.org/10.1016/j.apgeochem.2014.09.013
Wang X, Zhang C, Qiu B, Ashraf U, Azad R, Wu J, Ali S (2017) Biotransfer of Cd along a soil-plant-mealybug-ladybird food chain: a comparison with host plants. Chemosphere 168:699–706. https://doi.org/10.1016/j.chemosphere.2016.11.005
World Health Organization (2015) Global action plan on antimicrobial resistance. WHO Document Production Services, Geneva
Xiao Y, Li L (2016) China’s national plan to combat antimicrobial resistance. Lancet Infect Dis 16(11):1216–1218. https://doi.org/10.1016/S1473-3099(16)30388-7
Xie WY, McGrath SP, Su JQ, Hirsch PR, Clark IM, Shen Q, Zhu YG, Zhao FJ (2016) Long-term impact of field applications of sewage sludge on soil antibiotic resistome. Environ Sci Technol 50(23):12602–12611. https://doi.org/10.1021/acs.est.6b02138
Xiong X, Yanxia L, Wei L, Chunye L, Wei H, Ming Y (2010) Copper content in animal manures and potential risk of soil copper pollution with animal manure use in agriculture. Resour Conserv Recycl 54:985–990. https://doi.org/10.1016/j.resconrec.2010.02.005
Yang S, Wen X, Zhao L, Shi Y, Jin H (2014) Crude oil treatment leads to shift of bacterial communities in soils from the deep active layer and upper permafrost along the China-Russia crude oil pipeline route. PLoS One 9(5):e96552. https://doi.org/10.1371/journal.pone.0096552
Ye M, Sun M, Wan J, Feng Y, Zhao Y, Tian D, Hu F Jiang X (2016) Feasibility of lettuce cultivation in sophoroliplid-enhanced washed soil originally polluted with Cd, antibiotics, and antibiotic-resistant genes. Ecotoxicol Environ Saf 124:344–350. https://doi.org/10.1016/j.ecoenv.2015.11.013
Yergeau E, Sanschagrin S, Maynard C, St-Arnaud M, Greer CW (2014) Microbial expression profiles in the rhizosphere of willows depend on soil contamination. ISME J 8(2):344–358. https://doi.org/10.1038/ismej.2013.163
Yu B, Tian J, Feng L (2017) Remediation of PAH polluted soils using a soil microbial fuel cell: influence of electrode interval and role of microbial community. J Hazard Mater 336:110–118. https://doi.org/10.1016/j.jhazmat.2017.04.066
Zhou B, Wang C, Zhao Q, Wang Y, Huo M, Wang J, Wang S (2016) Prevalence and dissemination of antibiotic resistance genes and coselection of heavy metals in Chinese dairy farms. J Hazard Mater 320:10–17. https://doi.org/10.1016/j.jhazmat.2016.08.007
Zhou Y, Niu L, Zhu S, Lu H, Liu W (2017) Occurrence, abundance, and distribution of sulfonamide and tetracycline resistance genes in agricultural soils across China. Sci Total Environ 599-600:1977–1983. https://doi.org/10.1016/j.scitotenv.2017.05.152
Zhu F, Storey S, Ashaari MM, Clipson N, Doyle E (2017) Benzo (a) pyrene degradation and microbial community responses in composted soil. Environ Sci Pollut Res Int 24(6):5404–5414. https://doi.org/10.1007/s11356-016-8251-3
Acknowledgements
This study was funded by the Ministry of Education and Science of the Russian Federation (grant № 6.2379.2017/PCh), Russian Foundation for Basic Research (grant № 17-04-00787 A), President of Russian Federation (grant № NSh-3464.2018.11).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible editor: Diane Purchase
Rights and permissions
About this article
Cite this article
Gorovtsov, A.V., Sazykin, I.S. & Sazykina, M.A. The influence of heavy metals, polyaromatic hydrocarbons, and polychlorinated biphenyls pollution on the development of antibiotic resistance in soils. Environ Sci Pollut Res 25, 9283–9292 (2018). https://doi.org/10.1007/s11356-018-1465-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-1465-9