Abstract
The development of environmentally sustainable control strategies to fight insect pests is a key challenge nowadays. Pheromone-mediated mating disruption (MD) is based on the release of synthetic sex attractants into a crop, interfering with mate finding of a given pest species. However, a limited number of research items have been published on the optimization of MD strategies against the European grapevine moth, Lobesia botrana, as well as on the use of biodegradable dispensers to reduce waste production in vineyards, despite the high economic importance of this pest. Therefore, the present study evaluated the efficacy of the MD products Isonet® L TT and the biodegradable Isonet® L TT BIO, applied at various densities, in reducing L. botrana damage on grapevine in comparison to an untreated control and the reference MD product Isonet® L. Experiments were conducted in three different areas of grapevine cultivation, located in Central and Northern Italy, over three different years. Our MD approach allowed a reliable control of the three generations of L. botrana during the whole grape growing season, leading to a significant reduction in the infested flower clusters and bunches, as well as in the number of nests per flower cluster and bunch, if compared to the untreated control. The performances of Isonet® L TT BIO, Isonet® LTT, and Isonet® L did not differ in terms of infested flower clusters/bunches, as well as nests per flower cluster/bunch. This was confirmed in all experimental sites over 3 years of field experiments. Overall, the present research provides useful information for the optimization of MD programs against L. botrana, highlighting the interesting potential of biodegradable pheromone dispensers that can be easily applied at low densities in vineyards, reducing the use of chemical pesticides to control moth pests.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Currently, about 1.8 billion people are involved in agricultural activities worldwide, and most of them rely on pesticides to protect crops and livestock (Aktar et al. 2009; Alavanja 2009). Nowadays, the European Commission Directives are directed towards a significant reduction in pesticide use in the short to medium term (Hillocks 2012), to produce residue-free foods and reduce the toxicological impact of pesticides on human health and the environment (Hicks et al. 2017; Silver et al. 2017). Therefore, growing research attention is devoted to the development of environmentally friendly and sustainable strategies to control insect pests of agricultural importance (Todd et al. 2015; González-Chang et al. 2016; Holland et al. 2016; Benelli et al. 2017; Benelli 2018; Athanassiou et al. 2018). Besides classical biological control programs, the manipulation of insect chemical ecology has also been considered to develop novel, effective, and eco-friendly control tools (Witzgall et al. 2010; Kaplan 2012; Pérez-Staples et al. 2013).
In this scenario, a prominent role is played by pheromone-mediated mating disruption, which is based on the release of synthetic sex attractants into a crop, thus interfering with mate finding of a given pest species (Cardé 1990; Cardé and Minks 1995; Suckling 2000; Miller et al. 2006). In Lepidoptera, mate finding is generally routed by female sex pheromones, which mediate scramble competition among males for access to females (Tcheslavskaia et al. 2005; Witzgall et al. 2008; Lance et al. 2016). Moth females release small amounts of their sex pheromone and the males detect these plumes relying on their highly sensitive neurosensory structures (Cardé and Haynes 2004; Cardé and Willis 2008). Since moths strongly rely on sex pheromones to find their mates, dispensers releasing synthetic sexual pheromones can be efficaciously exploited in mating disruption programs to suppress pest reproduction in selected areas. This can be achieved by both non-competitive and competitive mechanisms, the first covering camouflage, desensitization, and sensory imbalance, the latter mainly due to false-plume following (Miller et al. 2006; Miller and Gut 2015). Notably, up to now, no negative effects on non-target organisms have been observed, making this method compatible with modern integrated pest management strategies (Welter et al. 2005; Miller et al. 2006; Witzgall et al. 2010; Ioriatti et al. 2012; Ioriatti and Lucchi 2016).
Concerning insect pests of vineyards, pheromone mating disruption was proven to be a reliable and effective tool for the control of the European grapevine moth, Lobesia botrana (Denis & Schiffermüller) (Lepidoptera: Tortricidae) (Ioriatti et al. 2008, 2011; Cooper et al. 2014). In mating disruption programs, a major issue to deal with—to allow large-scale use—is the optimization of the dispensers’ performances, their comparative assessment of efficacy, and their cost-effectiveness, which is linked to the time required for field application. In particular, a reduced number of pheromone dispensers in the field allows a strong reduction in the time required for their deployment, thus in labor costs (Gut et al. 2004; De Lame et al. 2010). Moreover, the development of biodegradable pheromone dispensers will also allow to reduce operational costs in the field (potentially no removal and plastic disposal at the end of the season required), as well as environmental pollution (Guerrini et al. 2017).
However, while the optimization of the abovementioned features has been considered in researches on other insect pest species (e.g., Meissner et al. 2000; Funes et al. 2016; McGhee et al. 2016; Sharon et al. 2016, Vacas et al. 2016), limited research has been done on L. botrana (Hummel 2017), despite the high economic importance of this pest. Most importantly, to the best of our knowledge, the use of biodegradable dispensers for L. botrana mating disruption programs has not yet been considered, with the unique exception of Ecodian (Isagro) dispensers—composed by Mater Bi® (Novamont, Novara) and cellulose—that have been tested with partial success (Anfora et al. 2008), without achieving substantial commercial interest.
On this basis, Shin-Etsu Chemical Co. (Japan) and CBC (Europe) S.r.l, (Italy) developed the two new pheromone dispensers for the mating disruption of L. botrana, namely Isonet® LTT and Isonet® L TT BIO. Both products consist of two parallel capillary tubes filled with the main component [i.e., (7E,9Z)-7,9-dodecadien-1-yl acetate] of L. botrana sexual pheromone blend, joined, and sealed at the ends. The gap in the middle allows each dispenser to form a loop that can be easily and quickly deployed by placing the dispenser over the end of spurs or by looping it around cordons, instead of twisting it around cordons as required for the commercially available reference product Isonet® L. Furthermore, both products can be applied at a lower rate than the conventional reference product Isonet® L (200–250 vs. 500 dispensers/ha, respectively). Notably, Isonet® LTT and Isonet® L TT BIO differ in the material of which the dispensers are made, which is polyethylene for Isonet® L TT and biodegradable polymers for Isonet® LTT BIO.
The research herein reported is aimed at evaluating the efficacy of the mating disruption products Isonet® L TT and the biodegradable Isonet® L TT BIO in reducing European grapevine moth (L. botrana) damage on grape in comparison to an untreated control and the reference mating disruption product Isonet® L. The trials were conducted in three different areas of grapevine cultivation, one located in Tuscany (Central Italy) and two in Emilia Romagna (Northern Italy) over three different years (2014, 2015, and 2016). Each year, the impact of the mating disruption products on the three generations of L. botrana was evaluated by determining the percentage of infested bunches and the number of nests per bunch. Furthermore, the tested dispensers were periodically collected during the grapevine growing season, extracted and analyzed by GC-MS. By evaluating their residual content of (7E,9Z)-7,9-dodecadien-1-yl acetate, we estimated the pheromone release in mg/ha/day during the whole grapevine growing season.
Materials and methods
Experimental sites
All experiments were conducted in areas representative for grapevine cultivation in Italy. Three trials were carried out in the area of Bolgheri, Livorno province, Tuscany region, Central Italy, an area representative for high-value grapevine cultivation in Italy, while additional two trials were conducted in Emilia-Romagna region, Northern Italy, respectively, one in Ravenna province (Campiano) and one in Forlì-Cesena province (Villafranca di Forlì). Details on the location of the study vineyards can be found in Table 1, and a detailed description of the characteristics of the crop in Table 2.
Experimental design of mating disruption trials
Since a randomized block design does not apply to large plots required for studies on mating disruption products (European and Mediterranean Plant Protection Organization 2016), each treatment was applied to 1 large plot, and 10 subplots, big enough to allow for assessments on at least 100 flower clusters/bunches per subplot (32–40 plants), were selected within each large plot (=treatment). All mating disruption products, both test and reference items, were deployed before the beginning of the first flight of the target pest in spring. Details on the size of the plots and the date of application of the MD products in the different trials can be found in Table 3. The reference product Isonet® L, applied at a rate of 500 dispensers per ha, was included in 4 out of 5 trials. Both Isonet® L TT and Isonet® L TT BIO were tested at 200 dispensers per ha in 2014 and at 250 dispensers per ha in 2015 and 2016.
Crop damage and L. botrana population density evaluation
In all trials, crop damage caused by L. botrana was assessed at the end of the 1st generation (=G1, BBCH 69–71), at the end of the 2nd generation (=G2, BBCH 79–81), and at harvest (=G3, BBCH 89). To assess the method effectiveness, we considered the following variables: (i) number of male captures per trap (Trap Test Isagro®, 1 trap per sampling site) per week, (ii) rate of infested flower clusters (G1) or bunches (G2 and G3), (iii) number of nests per flower cluster (G1) or bunch (G2 and G3).
Within each subplot and at each damage assessment, the number of flower clusters (G1) or bunches (G2 and G3) damaged by L. botrana was counted on 100 flower clusters/bunches per subplot at G1 and G2, respectively, and on 50 bunches per subplot at G3. The percentage of L. botrana-damaged flower clusters or bunches at each assessment was then calculated. Furthermore, at each assessment, the number of L. botrana nests per flower cluster (G1) or bunch (G2 and G3) was noted.
In detail, G1 infestation was measured through on-site surveys on non-destructively sampled inflorescences. As to the two carpophagous generations (G2 and G3), an estimate of the infested bunches was made on samples collected in the vineyards and carefully dissected. This is necessary above all for the compact-bunch varieties, such as Sangiovese, Pinot, and Chardonnay, for which a mere field inspection would often lead to a marked underestimation of the infestation level.
Pheromone release of the tested dispensers
For all tested dispensers, by evaluating the residual content of (7E,9Z)-7,9-dodecadien-1-yl acetate, we estimated the pheromone release in mg/ha/day during the whole grapevine growing season. Groups of Isonet® L, Isonet® L TT, and Isonet® L TT BIO dispensers (n = 5 per group) were periodically collected during the grapevine growing season and stored at − 30 °C until chemical analysis. The dispenser residual content in (7E,9Z)-7,9-dodecadien-1-yl acetate was measured based on internal (SEC) standard GC-MS analysis. The analysis was achieved on an Agilent 6890 N gas chromatograph equipped with a 5973 N mass spectrometer (MS). MS settings were as follows: EI mode, 70 eV, mass to charge ratio (m/z) scan between 35 and 400. HP-5 MS capillary column (30 m x ID 0.25 mm × 0.25 μm film thickness, J & W Scientific, Folsom, CA, USA) with He gas flow (1.0 ml/min) was used for separation. GC temperature program was as follows: initial 50 °C for 5 min, then increasing with 20 °C/min to 300 °C. The injector temperature was 150 °C. The GC-MS estimate of the dispenser residual content, allowed us to calculate the pheromone release during the field exposure of the dispenser, as mg/ha/day. Each value was a mean of 5 replicates.
Statistical analysis
Differences in the incidence of infested flower clusters or bunches (%) and nests per flower cluster or bunch (n) among treatments (i.e., tested pheromone dispensers and untreated control), years, and study site were assessed using non-parametric tests (Kruskal–Wallis test followed by Steel–Dwass multiple comparison) at the 5% significance level, since data did not show homogeneity of variance (Shapiro-Wilk test, P < 0.05). All statistical analysis was performed using JMP® 9 (SAS Institute).
Results
First-generation trials
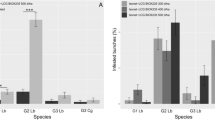
Figure 1 summarizes the field efficacy of mating disruption against the first generation of L. botrana. Isonet® L TT BIO, Isonet® L TT, and Isonet® L led to a significant reduction in the percentage of infested flower clusters if compared to the untreated control (Z = 5.756, P < 0.0001; Z = 5.156, P < 0.0001; Z = 4.811, P < 0.0001, respectively), while no significant differences were noted among the efficacy of the three tested dispensers (Fig. 1a). Furthermore, also the number of nests per flower cluster was significantly lower in Isonet® L TT BIO, Isonet® L TT, and Isonet® L than in the untreated control (Z = 5.681, P < 0.0001; Z = 5.238, P < 0.0001; Z = 4.792, P < 0.0001, respectively), while no significant differences were noted among the efficacy of the three tested dispensers (Fig. 1a).
Field efficacy of mating disruption against the first generation of the European grapevine moth (EGVM) Lobesia botrana. Experiments were carried out over three different years and geographical sites. Box plots of infested flower clusters (%) and nests per flower cluster (n) of EGVM showing the effect of (a) the tested dispenser used for mating disruption, (b) the year, and (c) the geographical site. Box plots indicate the median (solid line) within each box and the range of dispersion (lower and upper quartiles and outliers) of the median infestation parameter
Both the percentage of infested flower clusters and number of nests per flower cluster varied significantly among the years (Fig. 1b). Concerning infested flower clusters (%), EGVM incidence was higher in 2014 and 2016 than in 2015 (Z = 4.534, P < 0.0001; Z = − 2.728, P = 0.018), while no significant differences were noted between 2014 and 2016. The number of nests per flower cluster followed the same trend (Z = 4.561, P < 0.0001; Z = − 2.574, P = 0.027) (Fig. 1b).
The experimental site also played a significant role, showing varying L. botrana infestation levels (Fig. 1c). Concerning infested flower clusters (%), EGVM incidence was highest in Campiano (RA, Emilia Romagna), followed by Bolgheri (LI, Tuscany) and Villafranca di Forlì (FC, Emilia Romagna), with significant differences among them (Z = 7.398, P < 0.0001; Z = − 4.669, P < 0.0001; Z = − 7.711, P < 0.001, respectively). A comparable trend was observed concerning the number of nests per flower cluster (Z = 7.141, P < 0.0001; Z = − 4.899, P < 0.0001; Z = − 7741, P < 0.0001, respectively) (Fig. 1c).
Second generation trials
Mating disruption achieved significant results also in controlling the second generation of L. botrana, as shown in Fig. 2. In this generation as well, Isonet® L TT BIO, Isonet® L TT, and Isonet® L significantly reduced the percentage of infested bunches compared to the untreated control (Z = 6.608, P < 0.0001; Z = 6.236, P < 0.0001; Z = 5.597, P < 0.0001, respectively), with no significant differences among the three tested dispensers (Fig. 2a). Also, the number of nests per bunch was significantly lower in the Isonet® LTT BIO-, Isonet® LTT-, and Isonet® L-treated plots than in the untreated control (Z = 6.189, P < 0.0001; Z = 5.936, P < 0.0001; Z = 6.012, P < 0.0001, respectively), and no significant differences were observed among the three dispensers (Fig. 2a).
Field efficacy of mating disruption against the second generation of the European grapevine moth (EGVM) Lobesia botrana. Experiments were carried out over three different years and geographical sites. Box plots of infested bunches (%) and nests per bunch (n) of EGVM showing the effect of (a) the tested dispenser used for mating disruption, (b) the year, and (c) the geographical site. Box plots indicate the median (solid line) within each box and the range of dispersion (lower and upper quartiles and outliers) of the median infestation parameter
Infested bunches (%) and nests per bunch (n) varied significantly among the years (Fig. 2b). L. botrana infested bunches were significantly more abundant in 2014 over 2015 and 2016 (Z = − 4.126, P = 0.0001; Z = − 4.993, P = 0.018), while no significant differences were noted between 2015 and 2016. The number of nests per bunch followed the same trend (Z = − 4.722, P < 0.0001; Z = − 5.554, P < 0.0001) (Fig. 2b).
Significantly different infestation levels of L. botrana were found in mating disruption tests carried out in the three geographical sites (Fig. 2c). The percentage of EGVM infested bunches was significantly higher in Campiano (RA) than in Bolgheri (LI) and Villafranca di Forlì (FC) (Z = 6.956, P < 0.0001; Z = −7.588, P < 0.0001, respectively), while no significant differences were found between the latter two sites. A comparable trend was observed concerning the number of nests per bunch (Z = 5.958, P < 0.0001; Z = − 7.650, P < 0.0001, respectively) (Fig. 2c).
Third generation trials
The third generation of EGVM was effectively controlled by the application of mating disruption dispensers, irrespective of the type of dispenser tested (Fig. 3). Isonet® L TT BIO, Isonet® L TT, and Isonet® L resulted in a significant reduction in the percentage of infested bunches in comparison to the untreated control (Z = 4.783, P < 0.0001; Z = 4.271, P < 0.0001; Z = 3.470, P = 0.029, respectively), and no significant differences emerged among the three tested dispensers (Fig. 3a). The same trend was observed for the number of nests per bunch: significantly lower values were recorded in plots treated with Isonet® L TT BIO, Isonet® L TT, and Isonet® L than in untreated control plots (Z = 5.014, P < 0.0001; Z = 4.379, P < 0.0001; Z = 3.612, P = 0.0017, respectively), with differences among treated plots not being significant (Fig. 3a).
Field efficacy of mating disruption against the third generation of the European grapevine moth (EGVM) Lobesia botrana. Experiments were carried out over three different years and geographical sites. Box plots of infested bunches (%) and nests per bunch (n) of EGVM showing the effect of (a) the tested dispenser used for mating disruption, (b) the year, and (c) the geographical site. Box plots indicate the median (solid line) within each box and the range of dispersion (lower and upper quartiles and outliers) of the median infestation parameter
Infested bunches (%) and nests per bunch (n) varied significantly among the years (Fig. 3b). The percentage of L. botrana infested bunches was significantly higher in 2014 than in 2015 and 2016 (Z = − 5.554, P < 0.0001; Z = − 4.608, P < 0.0001), while no significant differences were noted between 2015 and 2016. The number of nests per bunch followed the same trend (Z = − 5.213, P < 0.0001; Z = − 4.112, P < 0.0001) (Fig. 3b).
Also at harvest, significantly different EGVM infestation levels were observed in the mating disruption trials carried out in the three geographical sites (Fig. 3c). Percent EGVM infestation was significantly higher in Campiano (RA) than in Bolgheri (LI) and Villafranca di Forlì (FC) (Z = 9.356, P < 0.0001; Z = − 7.671, P < 0.0001, respectively), with the latter two sites differing from each other (Z = 4.959, P < 0.0001). A comparable trend was observed concerning the number of nests per bunch (Z = 9.355, P < 0.0001; Z = − 7.639, P < 0.0001, and Z = 4.433, P < 0.0001, respectively) (Fig. 3c).
In all mating disrupted vineyards, L. botrana males were not captured by Trap Test Isagro® during the whole grape growing seasons, providing a further evidence of proper (7E,9Z)-7,9-dodecadien-1-yl acetate dispersion within the tested fields. Lastly, Fig. 4 shows the continuous release (mg/ha/day) of synthetic (7E,9Z)-7,9-dodecadien-1-yl acetate, by the three mating disruption products Isonet® L, Isonet® L TT, and Isonet® L TT BIO.
Discussion
The development of effective and environmentally sustainable control strategies against agricultural insect pests is a crucial challenge nowadays, considering that more than two million tons of pesticides are employed each year in agricultural activities worldwide (De et al. 2014), of which more than 400,000 tons are currently used in European countries (Eurostat 2016). In this framework, the frequent overuse of insecticides rapidly led to the development of resistance in targeted insects (Bourguet et al. 2000; Franck et al. 2007; Thomas and Read 2016; Grégoire et al. 2017), including moth pests (Reyes et al. 2007; Zhao et al. 2002, Zhao et al. 2006).
Furthermore, the third generation of the European grapevine moth, which is the most dangerous for late grapevine varieties, is difficult to control, since farmers are experiencing a lack of authorized reliable pesticides characterized by short pre-harvest interval, to avoid residues in grapes and wine. Insecticides commonly applied close to harvest against this target pest contain toxins from Bacillus thuringiensis subsp. kurstaki and aizawai, acting as microbial disruptors of insect midgut membranes, or emamectin benzoate as active ingredient (Muccinelli 2017).
Therefore, developing eco-friendly and reliable control tools is crucial. Our results highlighted the high efficacy of the mating disruption programs carried out against L. botrana populations in Northern and Central Italian vineyards. The approach proposed enables to minimize the use of chemical pesticides, since it is based on the deployment of different dispensers releasing multiple plumes of (7E,9Z)-7,9-dodecadien-1-yl acetate — the main sex pheromone component of L. botrana females (Ioriatti and Lucchi 2016; Lance et al. 2016).
Notably, our mating disruption approach testing the efficacy of Isonet® L TT BIO, Isonet® L TT over the standard product Isonet® L allowed a reliable control of the three generations of this moth pest during the whole growing season. The field efficacy of the tested approach was validated in three different geographic sites over a study period of 3 years. As expected, we observed significant differences among experimental sites, mostly due to different pest population sizes in early season in the tested vineyards. In particular, concerning the first generation, we detected a high incidence of L. botrana damage to grapes in Campiano (Emilia Romagna, Northern Italy), highlighting the presence of a larger pest population, if compared to the other sites. Thus, in this context, random encounters between mates may occur, leading to a decreasing efficacy of mating disruption (Millar 2007). In these scenarios, an effective strategy can be the integration of mating disruption with low-impact microbial insecticides (e.g., B. thuringiensis-based ones), since it is well recognized that mating disruption gives its best efficacy on starting pest populations characterized by medium-low densities (Ioriatti and Lucchi 2016).
Regarding experiments conducted against the first generation of L. botrana, we noticed a significant reduction in the number of infested flower clusters, and number of nests per flower cluster as well, if compared to the untreated control. Besides, when mating disruption tests were conducted against the second and third generation of L. botrana, a strong reduction in the number of infested bunches and number of nests per bunch was achieved. Earlier, Anfora et al. (2008) observed a significant field efficacy of mating disruption carried out against L. botrana using Ecodian® dispensers, showing a reduction in the overall attractiveness of traps lured with calling females and monitoring baits. However, the authors tested 1600 dispensers/ha (Anfora et al. 2008), while in the present study, the biodegradable dispenser was tested at 200–250 dispensers/ha, still allowing an adequate release of synthetic (7E,9Z)-7,9-dodecadien-1-yl acetate, and achieving a substantial reduction of the incidence of L. botrana damage on grapes.
As a general trend, the efficacy of Isonet® L TT BIO, Isonet® L TT, and Isonet® L was comparable. Indeed, the performances of all the tested dispensers did not differ in terms of infested flower clusters/bunches and nests per flower cluster/bunch. This was noted in all experimental sites over 3 years of field experiments. As indicated by the curves showing the release of (7E,9Z)-7,9-dodecadien-1-yl acetate over time (Fig. 4), the three dispensers tested here can protect treated vineyards from L. botrana infestation during the whole growing season, ensuring a continuous release of sex pheromone plumes.
To our mind, there are three practical implications arising from these findings. First, the comparable field performances of Isonet® L TT BIO and Isonet® L TT vs. Isonet® L allow reducing the number of pheromone dispensers needed per hectare (200–250 vs. 500 dispensers/ha), thus direct costs for buying them.
Second, a lower number of sex pheromone dispensers has a direct impact on farmers’ economy, reducing labor cost to apply the dispensers in the vineyards. Indeed, the time needed to apply sex pheromone dispensers is 3 h/ha for Isonet® L, while it drops to 1–1.5 h/ha using Isonet® L TT or Isonet® L TT BIO, due to the lower number of required dispensers per hectare. When designing this study, we considered that testing a lower number of dispensers per hectare can lead to reduced efficacy of mating disruption, as earlier outlined by several authors studying the effective rate of mating disruption dispensers per hectare in the fight against other moth pests of economic importance, such as Cydia pomonella (L.) (e.g., Epstein et al. 2006; Stelinski et al. 2006b; Patanita 2007; Grieshop et al. 2010). However, the present results showed that this was not the case, since the tested numbers of dispensers allowed a reliable control of the three L. botrana generations in all the experimental sites.
Third, the comparable efficacy of the biodegradable dispenser Isonet® L TT BIO over the widely adopted non-biodegradable Isonet® L ones contributes reducing waste disposal - which nowdays represents a serious environmental concern (Rochman et al. 2013; Vegter et al. 2014; Jambeck et al. 2015) - because they are made of more eco-friendly materials prepared from natural resources (Ashori 2008; Boghossian and Wegner 2008; Castellano et al. 2008; Scarascia-Mugnozza et al. 2012; Bledzki et al. 2015). Our results also support earlier findings by other authors, focusing on the use of biodegradable dispensers for mating disruption of insect pests of agricultural importance, including the grape berry moth, Paralobesia viteana (Clemens) (Teixeira et al. 2010; Jenkins and Isaacs 2008), the codling moth, C. pomonella (Angeli et al. 2007), the Oriental fruit moth, Grapholita molesta (Busck) (Frédérique et al. 2007; Stelinski et al. 2005, 2006a, 2007), the light brown apple moth, Epiphyas postvittana (Walker) (Brockerhoff et al. 2012; Suckling et al. 2012), the Asiatic rice borer, Chilo suppressalis (Walker) (Vacas et al. 2009a), the California red scale, Aonidiella aurantii Maskell (Vacas et al. 2009b, 2010, 2012), the grub beetle, Dasylepida ishigakiensis Niijima et Kinoshita (Arakaki et al. 2017), and the Oriental beetle, Anomala orientalis Waterhouse (Behle et al. 2008).
Overall, the present research provides useful information for the optimization of eco-friendly mating disruption programs against L. botrana populations, highlighting the interesting potential of biodegradable pheromone dispensers that can be easily applied at low density in vineyards of high economic value, reducing the use of chemical pesticides to control moth pests.
References
Aktar W, Sengupta D, Chowdhury A (2009) Impact of pesticides use in agriculture: their benefits and hazards. Int Toxicol 2(1):1–12
Alavanja MC (2009) Introduction: pesticides use and exposure, extensive worldwide. Rev Environ Health 24(4):303–310
Anfora G, Baldessari M, De Cristofaro A, Germinara GS, Ioriatti C, Reggiori F, Vitagliano S, Angeli G (2008) Control of Lobesia botrana (Lepidoptera: Tortricidae) by biodegradable Ecodian sex pheromone dispensers. J Econ Entomol 101(2):444–450. https://doi.org/10.1093/jee/101.2.444
Angeli G, Anfora G, Baldessari M, Germinara GS, Rama F, De Cristofaro A, Ioriatti C (2007) Mating disruption of codling moth Cydia pomonella with high densities of Ecodian sex pheromone dispensers. J Appl Entomol 131(5):311–318. https://doi.org/10.1111/j.1439-0418.2007.01172.x
Arakaki N, Nagayama A, Kijima K, Yasui H, Tsujii N, Tanaka S, Ueda M, Kanayama S, Kawaguchi K, Jitsuno S, Oike M, Watanabe K, Wakamura S, Mochizuki F, Saguchi R, Fujii T, Naito T (2017) Ground-surface application of pheromones through a mini-dispenser for mating disruption of the white grub beetle Dasylepida ishigakiensis (Coleoptera: Scarabaeidae). Appl Entomol Zool 52(1):159–164. https://doi.org/10.1007/s13355-016-0462-z
Ashori A (2008) Wood–plastic composites as promising green-composites for automotive industries! Bioresour Technol 99(11):4661–4667. https://doi.org/10.1016/j.biortech.2007.09.043
Athanassiou CG, Kavallieratos NG, Benelli G, Losic D, Usha Rani P, Desneux N (2018) Nanoparticles for pest control: current status and future perspectives. J Pest Sci 91:1–15. https://doi.org/10.1007/s10340-017-0898-0
Behle RW, Cossé AA, Dunlap C, Fisher J, Koppenhöfer AM (2008) Developing wax-based granule formulations for mating disruption of oriental beetles (Coleoptera: Scarabaeidae) in turfgrass. J Econ Entomol 101(6):1856–1863. https://doi.org/10.1603/0022-0493-101.6.1856
Bledzki AK, Franciszczak P, Osman Z, Elbadawi M (2015) Polypropylene biocomposites reinforced with softwood, abaca, jute, and kenaf fibers. Ind Crop Prod 70:91–99. https://doi.org/10.1016/j.indcrop.2015.03.013
Benelli G (2018) Plant-borne compounds and nanoparticles: challenges for medicine, parasitology and entomology – GREEN-NANO-PEST & DRUGS. Environ Sci Poll Res. https://doi.org/10.1007/s11356-017-9960-y
Benelli G, Pavela R, Maggi F, Petrelli R, Nicoletti M (2017) Commentary: making green pesticides greener? The potential of plant products for nanosynthesis and pest control. J Clust Sci 28:3–10
Boghossian E, Wegner LD (2008) Use of flax fibres to reduce plastic shrinkage cracking in concrete. Cement Concr Composit 30(10):929–937. https://doi.org/10.1016/j.cemconcomp.2008.09.003
Bourguet D, Genissel A, Raymond M (2000) Insecticide resistance and dominance levels. J Econ Entomol 93(6):1588–1595. https://doi.org/10.1603/0022-0493-93.6.1588
Brockerhoff EG, Suckling DM, Kimberley M, Richardson B, Coker G, Gous S, Kerr JL, Cowan DM, Lance DR, Strand T, Zhang A (2012) Aerial application of pheromones for mating disruption of an invasive moth as a potential eradication tool. PLoS One 7(8):e43767. https://doi.org/10.1371/journal.pone.0043767
Cardé RT (1990) Principles of mating disruption. Behavior-modifying chemicals for pest management: applications of pheromones and other attractants. Marcel Dekker, New York, pp 47–71
Cardé RT, Minks AK (1995) Control of moth pests by mating disruption: successes and constraints. Annu Rev Entomol 40(1):559–585. https://doi.org/10.1146/annurev.en.40.010195.003015
Cardé RT, Haynes KF (2004) Structure of the pheromone communication channel in moths. In: Cardé R T, and Millar J G (eds) Advances in insect chemical ecology. Cambridge University Press, Cambridge, pp. 283–332
Cardé RT, Willis MA (2008) Navigational strategies used by insects to find distant, wind-borne sources of odor. J Chem Ecol 34(7):854–866. https://doi.org/10.1007/s10886-008-9484-5
Castellano S, Mugnozza GS, Russo G, Briassoulis D, Mistriotis A, Hemming S, Waaijenberg D (2008) Plastic nets in agriculture: a general review of types and applications. Appl Eng Agric 24(6):799–808. https://doi.org/10.13031/2013.25368
Cooper M, Varela LG, Smith RJ, Whitmer DR, Simmons GA, Lucchi A, Broadway R, Steinhauer R (2014) Growers, scientists and regulators collaborate on European grapevine moth program. Calif Agric 4:125–133
De A, Bose R, Kumar A, Mozumdar S (2014) Targeted delivery of pesticides using biodegradable polymeric nanoparticles. Springer Briefs in Molecular Science. Springer, New Delhi
De Lame FM, Epstein D, Gut LJ, Goldfarb H, Miller JR (2010) Effect of varying dispenser point source density on mating disruption of Grapholita molesta (Lepidoptera: Tortricidae). J Econ Entomol 103(4):1299–1305. https://doi.org/10.1603/EC09239
Epstein DL, Stelinski LL, Reed TP, Miller JR, Gut LJ (2006) Higher densities of distributed pheromone sources provide disruption of codling moth (Lepidoptera: Tortricidae) superior to that of lower densities of clumped sources. J Econ Entomol 99(4):1327–1333. https://doi.org/10.1093/jee/99.4.1327
Eurostat (2016) Pesticide sales statistics (http://ec.europa.eu/eurostat/statistics-explained/index.php/Pesticide_sales_statistics), accessed October 2016
European and Mediterranean Plant Protection Organization (2016) Efficacy evaluation of plant protection products. Mating disruption pheromones (http://pp1.eppo.int/). First approved in 2008–09. Last update December 2016
Grégoire JC, Miret JAJ, González-Cabrera J, Heimbach U, Lucchi A, Gardi C, Erdos Z, Koufakis I (2017) Protocol for the evaluation of data concerning the necessity of the application of insecticide1 active substances to control a serious danger to plant health which cannot be contained by other available means, including non-chemical methods. EFSA Supporting Publications, 14(4), 29 March 2017. https://doi.org/10.2903/sp.efsa.2017.EN-1201
Franck P, Reyes M, Olivares J, Sauphanor B (2007) Genetic architecture in codling moth populations: comparison between microsatellite and insecticide resistance markers. Mol Ecol 16(17):3554–3564. https://doi.org/10.1111/j.1365-294X.2007.03410.x
Frédérique M, Miller JR, Atterholt CA, Gut LJ (2007) Development and evaluation of an emulsified paraffin wax dispenser for season-long mating disruption of Grapholita molesta in commercial peach orchards. J Econ Entomol 100(4):1316–1327
Funes H, Zerba E, Gonzalez-Audino P (2016) Monolithic dispensers for pheromones and their use in mating disruption of the ambrosia beetle Megaplatypus mutatus in poplar plantations. Agric Forest Entomol 18(1):52–58. https://doi.org/10.1111/afe.12133
González-Chang M, Wratten SD, Lefort MC, Boyer S (2016) Food webs and biological control: a review of molecular tools used to reveal trophic interactions in agricultural systems. Food Webs 9:4–11. https://doi.org/10.1016/j.fooweb.2016.04.003
Grieshop MJ, Brunner JF, Jones VP, Bello NM (2010) Recapture of codling moth (Lepidoptera: Tortricidae) males: influence of lure type and pheromone background. J Econ Entomol 103(4):1242–1249. https://doi.org/10.1603/EC09282
Guerrini S, Borreani G, Voojis H (2017) Biodegradable materials in agriculture: case histories and perspectives. In: Malinconico M (ed) Soil degradable bioplastics for a sustainable modern agriculture, green chemistry and sustainable technology. Springer-Verlag Berlin Heidelberg. https://doi.org/10.1007/978-3-662-54130-2_3
Gut LJ, Stelinski LL, Thomson DR, Miller JR (2004) Behaviour-modifying chemicals: prospects and constraints in IPM. In: Koul, Dhaliwal, Cuperus (eds) Integrated pest management: potential, constraints, and challenges. CABI Publishing, Cambridge, pp 73–121
Hicks S, Wang M, Doraiswamy V, Fry K, Wohlford EM (2017) Neurodevelopmental delay diagnosis rates are increased in a region with aerial pesticide application. Front Pediatr 5:116. https://doi.org/10.3389/fped.2017.00116
Hillocks RJ (2012) Farming with fewer pesticides: EU pesticide review and resulting challenges for UK agriculture. Crop Protect 31(1):85–93. https://doi.org/10.1016/j.cropro.2011.08.008
Holland JM, Bianchi FJ, Entling MH, Moonen AC, Smith BM, Jeanneret P (2016) Structure, function and management of semi-natural habitats for conservation biological control: a review of European studies. Pest Manag Sci 72(9):1638–1651. https://doi.org/10.1002/ps.4318
Hummel HE (2017) A brief review on Lobesia botrana mating disruption by mechanically distributing and releasing sex pheromones from biodegradable mesofiber dispensers. Biochem Mol Biol J 3:1
Ioriatti C, Lucchi A (2016) Semiochemical strategies for tortricid moth control in apple orchards and vineyards in Italy. J Chem Ecol 42(7):571–583. https://doi.org/10.1007/s10886-016-0722-y
Ioriatti C, Anfora G, Tasin M, De Cristofaro A, Witzgall P, Lucchi A (2011) Chemical ecology and management of Lobesia botrana (Lepidoptera: Tortricidae). J Econ Entomol 104(4):1125–1113. https://doi.org/10.1603/EC10443
Ioriatti C, Lucchi A, Bagnoli B (2008) Grape areawide pest management in Italy. In: Koul O, Cuperus G W, and Elliott N (eds) Areawide pest management: Theory and implementation. CABI, , pp. 208–225
Ioriatti C, Lucchi A, Varela LG (2012) Grape berry moths in Western European vineyards and their recent movement into the new world. In: Bostanian NJ, Vincent C, Isaacs R (eds) “Arthropod management in vineyards: pests, approaches, and future directions”. Springer, Dordrecht, pp. 339–359
Jambeck JR, Geyer R, Wilcox C, Siegler TR, Perryman M, Andrady A, Narayan R, Law KL (2015) Plastic waste inputs from land into the ocean. Science 347(6223):768–771. https://doi.org/10.1126/science.1260352
Jenkins PE, Isaacs R (2008) Mating disruption of Paralobesia viteana in vineyards using pheromone deployed in SPLAT-GBM™ wax droplets. J Chem Ecol 34(8):1089–1095. https://doi.org/10.1007/s10886-008-9497-0
Kaplan I (2012) Attracting carnivorous arthropods with plant volatiles: the future of biocontrol or playing with fire? Biol Control 60(2):77–89. https://doi.org/10.1016/j.biocontrol.2011.10.017
Lance DR, Leonard DS, Mastro VC, Walters ML (2016) Mating disruption as a suppression tactic in programs targeting regulated lepidopteran pests in US. J Chem Ecol 42(7):590–605. https://doi.org/10.1007/s10886-016-0732-9
McGhee PS, Miller JR, Thomson DR, Gut LJ (2016) Optimizing aerosol dispensers for mating disruption of codling moth, Cydia pomonella L. J Chem Ecol 42(7):612–616. https://doi.org/10.1007/s10886-016-0724-9
Meissner HE, Atterholt CA, Walgenbach JF, Kennedy GG (2000) Comparison of pheromone application rates, point source densities, and dispensing methods for mating disruption of tufted apple bud moth (Lepidoptera: Tortricidae). J Econ Entomol 93(3):820–827. https://doi.org/10.1603/0022-0493-93.3.820
Millar JG (2007) Insect pheromones for integrated pest management: promise versus reality. Redia 90:51–55
Miller JR, Gut LJ, De Lame FM, Stelinski LL (2006) Differentiation of competitive vs. non-competitive mechanisms mediating disruption of moth sexual communication by point sources of sex pheromone (part 2): case studies. J Chem Ecol 32(10):2115–2143. https://doi.org/10.1007/s10886-006-9136-6
Miller JR, Gut LJ (2015) Mating disruption for the 21st century: matching technology with mechanism. Environ Entomol 44(3):427–453. https://doi.org/10.1093/ee/nvv052
Muccinelli M (2017) Prontuario dei fitofarmaci. Edagricole (http://www.prontuariomuccinelli.it/), accessed October 2017
Patanita MI (2007) Biothecnical methods for the control of main pests of walnut. Rev Ciências Agr 30:518–526
Pérez-Staples D, Shelly TE, Yuval B (2013) Female mating failure and the failure of ‘mating’ in sterile insect programs. Entomol Exp Appl 146(1):66–78. https://doi.org/10.1111/j.1570-7458.2012.01312.x
Reyes M, Franck P, Charmillot PJ, Ioriatti C, Olivares J, Pasqualini E, Sauphanor B (2007) Diversity of insecticide resistance mechanisms and spectrum in European populations of the codling moth, Cydia pomonella. Pest Manag Sci 63(9):890–902. https://doi.org/10.1002/ps.1421
Rochman CM, Browne MA, Halpern BS, Hentschel BT, Hoh E, Karapanagioti HK, Rios-Mendoza LM, Takada H, Teh S, Thompson RC (2013) Policy: classify plastic waste as hazardous. Nature 494(7436):169–171. https://doi.org/10.1038/494169a
Rochman CM, Cook AM, Koelmans AA (2016) Plastic debris and policy: using current scientific understanding to invoke positive change. Environ Toxicol Chem 35(7):1617–1626. https://doi.org/10.1002/etc.3408
Scarascia-Mugnozza G, Sica C, Russo G (2012) Plastic materials in European agriculture: actual use and perspectives. J Agric Eng 42(3):15–28
Sharon R, Zahavi T, Sokolsky T, Sofer-Arad C, Tomer M, Kedoshim R, Harari AR (2016) Mating disruption method against the vine mealybug, Planococcus ficus: effect of sequential treatment on infested vines. Entomol Exp Appl 161(1):65–69. https://doi.org/10.1111/eea.12487
Silver MK, Shao J, Zhu B, Chen M, Xi Y, Kaciroti N, Lozoff B, Meeker JD (2017) Prenatal naled and chlorpyrifos exposure is associated with deficits in infant motor function in a cohort of Chinese infants. Environ Int 106:248–256. https://doi.org/10.1016/j.envint.2017.05.015
Stelinski LL, Gut LJ, Mallinger RE, Epstein D, Reed TP, Miller JR (2005) Small plot trials documenting effective mating disruption of oriental fruit moth by using high densities of wax-drop pheromone dispensers. J Econ Entomol 98(4):1267–1274. https://doi.org/10.1603/0022-0493-98.4.1267
Stelinski LL, Miller JR, Ledebuhr R, Gut LJ (2006a) Mechanized applicator for large-scale field deployment of paraffin-wax dispensers of pheromone for mating disruption in tree fruit. J Econ Entomol 99(5):1705–1710. https://doi.org/10.1093/jee/99.5.1705
Stelinski LL, Gut LJ, Miller JR (2006b) Orientational behaviors and EAG responses of male codling moth after exposure to synthetic sex pheromone from various dispensers. J Chem Ecol 32(7):1527–1538. https://doi.org/10.1007/s10886-006-9067-2
Stelinski LL, Miller JR, Ledebuhr R, Siegert P, Gut LJ (2007) Season-long mating disruption of Grapholita molesta (Lepidoptera: Tortricidae) by one machine application of pheromone in wax drops (SPLAT-OFM). J Pest Sci 80(2):109–117. https://doi.org/10.1007/s10340-007-0162-0
Suckling DM (2000) Issues affecting the use of pheromones and other semiochemicals in orchards. Crop Prot 19(8):677–683. https://doi.org/10.1016/S0261-2194(00)00090-9
Suckling DM, Brockerhoff EG, Stringer LD, Butler RC, Campbell DM, Mosser LK, Cooperband MF (2012) Communication disruption of Epiphyas postvittana (Lepidoptera: Tortricidae) by using two formulations at four point source densities in vineyards. J Econ Entomol 105(5):1694–1701. https://doi.org/10.1603/EC12130
Tcheslavskaia KS, Thorpe KW, Brewster CC, Sharov AA, Leonard DS, Reardon RC, Mastro VC, Sellers P, Roberts EA (2005) Optimization of pheromone dosage for gypsy moth mating disruption. Entomol Exp Appl 115(3):355–361. https://doi.org/10.1111/j.1570-7458.2005.00266.x
Teixeira LAF, Mason K, Mafra-Neto A, Isaacs R (2010) Mechanically-applied wax matrix (SPLAT-GBM) for mating disruption of grape berry moth (Lepidoptera: Tortricidae). Crop Prot 29(12):1514–1520. https://doi.org/10.1016/j.cropro.2010.08.013
Thomas MB, Read AF (2016) The threat (or not) of insecticide resistance for malaria control. Proc Natl Acad Sci 113(32):8900–8902. https://doi.org/10.1073/pnas.1609889113
Todd JH, Barratt BI, Tooman L, Beggs JR, Malone LA (2015) Selecting non-target species for risk assessment of entomophagous biological control agents: evaluation of the PRONTI decision-support tool. Biol Control 80:77–88. https://doi.org/10.1016/j.biocontrol.2014.09.014
Vacas S, Alfaro C, Navarro-Llopis V, Zarzo M, Primo J (2009a) Study on the optimum pheromone release rate for attraction of Chilo suppressalis (Lepidoptera: Pyralidae). J Econ Entomol 102(3):1094–1100. https://doi.org/10.1603/029.102.0330
Vacas S, Alfaro C, Navarro-Llopis V, Primo J (2009b) The first account of the mating disruption technique for the control of California red scale, Aonidiella aurantii Maskell (Homoptera: Diaspididae) using new biodegradable dispensers. Bull Entomol Res 99(4):415–423. https://doi.org/10.1017/S0007485308006470
Vacas S, Alfaro C, Navarro-Llopis V, Primo J (2010) Mating disruption of California red scale, Aonidiella aurantii Maskell (Homoptera: Diaspididae), using biodegradable mesoporous pheromone dispensers. Pest Manag Sci 66(7):745–751. https://doi.org/10.1002/ps.1937
Vacas S, Vanaclocha P, Alfaro C, Primo J, Verdú MJ, Urbaneja A, Navarro-Llopis V (2012) Mating disruption for the control of Aonidiella aurantii Maskell (Hemiptera: Diaspididae) may contribute to increased effectiveness of natural enemies. Pest Manag Sci 68(1):142–148. https://doi.org/10.1002/ps.2239
Vacas S, Navarro I, Primo J, Navarro-Llopis V (2016) Mating disruption to control the striped rice stem borer: pheromone blend, dispensing technology and number of releasing points. J Asia-Pacif Entomol 19(2):253–259. https://doi.org/10.1016/j.aspen.2016.02.001
Vegter AC, Barletta M, Beck C, Borrero J, Burton H, Campbell ML, Costa MF, Eriksen M, Eriksson C, Estrades A, Gilardi KVK, Hardesty BD, Ivar do Sul JA, Lavers JL, Lazar B, Lebreton L, Nichols WJ, Ribic CA, Ryan PG, Schuyler QA, Smith SDA, Takada H, Townsend KA, Wabnitz CCC, Wilcox C, Young LC, Hamann M (2014) Global research priorities to mitigate plastic pollution impacts on marine wildlife. Endang Species Res 25(3):225–247. https://doi.org/10.3354/esr00623
Welter S, Pickel C, Millar J, Cave F, Van Steenwyk R, Dunley J (2005) Pheromone mating disruption offers selective management options for key pests. Calif Agric 59(1):16–22. https://doi.org/10.3733/ca.v059n01p16
Witzgall P, Stelinski L, Gut L, Thomson D (2008) Codling moth management and chemical ecology. Annu Rev Entomol 53(1):503–522. https://doi.org/10.1146/annurev.ento.53.103106.093323
Witzgall P, Kirsch P, Cork A (2010) Sex pheromones and their impact on pest management. J Chem Ecol 36(1):80–100. https://doi.org/10.1007/s10886-009-9737-y
Zhao JZ, Li YX, Collins HL, Gusukuma-Minuto L, Mau RFL, Thompson GD, Shelton AM (2002) Monitoring and characterization of diamondback moth (Lepidoptera: Plutellidae) resistance to spinosad. J Econ Entomol 95(2):430–436. https://doi.org/10.1603/0022-0493-95.2.430
Zhao JZ, Collins HL, Li YX, Mau RFL, Thompson GD, Hertlein M, Andaloro JT, Boykin R, Shelton AM (2006) Monitoring of diamondback moth (Lepidoptera: Plutellidae) resistance to spinosad, indoxacarb, and emamectin benzoate. J Econ Entomol 99(1):176–181. https://doi.org/10.1093/jee/99.1.176
Acknowledgements
The authors would like to thank Shin-Etsu® Chemicals for providing the tested mating disruption products. Furthermore, we are grateful to the staff of Guado al Tasso, Marchesi Antinori (Bolgheri), Coop. Braccianti Campiano (Ravenna), and Az. Agricola Schiumarini (Forlì-Cesena) for kindly allowing fieldwork in their farms.
Funding
This study was funded by BIOCONVITO P.I.F. “Artigiani del Vino Toscano” (Regione Toscana, Italy).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests. The mention of trade names or commercial products in this article does not imply recommendation or endorsement by the authors’ institutions.
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Lucchi, A., Ladurner, E., Iodice, A. et al. Eco-friendly pheromone dispensers—a green route to manage the European grapevine moth?. Environ Sci Pollut Res 25, 9426–9442 (2018). https://doi.org/10.1007/s11356-018-1248-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-1248-3