Abstract
The vine mealybug (VMB), Planococcus ficus (Hemiptera: Pseudococcidae), is a key insect pest of vineyards. While pheromone-based mating disruption (MD) has been successfully tested against a wide range of insect pests, knowledge about its efficacy against key mealybug species, such as P. ficus, is scarce. In this study, a novel MD product, Isonet® PF, was evaluated by testing 300, 400, and 500 dispensers/ha at four study sites located in Northern (Veneto) and Southern (Sicily) Italy. Experiments were carried out over 2 years by monitoring the mealybug populations in wine grape and table grape vineyards managed with and without the application of MD. Pheromone dispensers were periodically collected during the grapevine-growing season, extracted, and analyzed by GC-MS, to determine their pheromone content and the release in mg/ha/day. The results showed that use of the MD dispenser Isonet® PF reduced the percentage of VMB-infested bunches and the number of VMB specimens per bunch compared with the untreated controls. This was recorded over 2 years at all experimental sites. Differences in the incidence of infested bunches among the three tested rates of Isonet® PF were not detected. Overall, the results presented here contribute to optimizing the sex pheromone dosage used in MD control programs against VMB allowing a reduction of broad-spectrum insecticides currently employed to manage this important pest.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Planococcus ficus (Signoret) (Hemiptera: Pseudococcidae), commonly known as vine mealybug (VMB), is an important insect pest of vineyards in various regions worldwide (Daane et al. 2012), including several European countries, with special reference to Italy, Southern France, and Spain, as well as the USA (CA), Mexico (Baja California and Sonora), Argentina, Brazil, and South Africa (da Silva et al. 2016; Le Vieux and Malan 2016; Walton and Pringle 2017; Daane et al. 2018a). Planococcus ficus infestations lead to severe economic losses, both on wine and table grape: VMB populations produce abundant honeydew that can cover vine plants, leading to sooty mold development on foliage and bunches (Chiotta et al. 2010). In addition, P. ficus can transmit various grape pathogens, including the grapevine leafroll-associated virus (GLRaV) (Douglas and Krüger 2008; Tsai et al. 2008, 2010) and the corky-bark disease (Tanne et al. 1989). Notably, it has been reported that heavy infestations by VMB can lead to the destruction of whole vineyards (Walton et al. 2006; Sharon et al. 2016).
The economic importance of this pest highlights the need to develop suitable control strategies for P. ficus. The effective and environmentally sustainable control of this pest is extremely important for ensuring the quality of wine and table grape production, as well as to prevent the further spread of GLRaV (Walton et al. 2004; Franco et al. 2009; Almeida et al. 2013; Sokolsky et al. 2013). Finding adequate pest management tactics against the VMB is a challenging issue. The use of insecticides against VMB populations—including systemic ones (e.g., spirotetramat)—may not show consistent results, since a relevant percentage of the population feeds on roots or is concealed under the bark (Gutierrez et al. 2008; Mansour et al. 2010). Various control strategies have been attempted, ranging from hot water treatments for preparing nursery stock (Haviland et al. 2005) to the use of VMB biological control agents (Fallahzadeh et al. 2011), in particular parasitoids (Güleç et al. 2007; Suma et al. 2012; Mansour et al. 2017a; Mgocheki and Addison 2009; Romano et al. 2018). Within the field of eco-friendly control tools that can be used against insect pests, a prominent role is played by sex pheromone–based mating disruption (MD) (Millar 2007; Miller and Gut 2015). This strategy is species-specific, is safe for human health, and does not lead to acute or chronic toxicity on non-target organisms (Welter et al. 2005; Witzgall et al. 2010). While MD has been successfully tested against a wide range of insect pests (Gut et al. 2004; Lance et al. 2016; Ioriatti et al. 2008), in particular certain moth species (e.g., codling moth, Cydia pomonella (L.)), European grapevine moth, Lobesia botrana (Denis & Schiffermüller), and European grape berry moth, Eupoecilia ambiguella (Hübner) (Miller and Gut 2015; Ioriatti and Lucchi 2016; Lucchi et al. 2018a,b), knowledge about its efficacy against important mealybugs species, such as P. ficus, is limited. Indeed, little research is available on this topic (Walton et al. 2006; Cocco et al. 2014, 2018; Sharon et al. 2016; Mansour et al. 2017b). The studies conducted by da Silva et al. (2010) suggest that biparental mealybug species, including the Planococcus genus, do not rely on facultative parthenogenesis for reproduction (see also Lentini et al. 2018). Furthermore, Waterworth et al. (2011) reported that VMB must mate to produce viable offspring, outlining the potential of MD to control their populations.
The sex pheromone of P. ficus was first identified by Hinkens et al. (2001) as lavandulyl senecioate. Later, Millar et al. (2002) better clarified the VMB sexual communication, showing that the VMB-synthesized (S)-enantiomer was equally attractive for males, if compared to the racemic enantiomer mixture (see also Zada et al. 2003, 2008). Subsequently, various researchers developed monitoring methods based on the use of synthetic VMB sex pheromone (Millar et al. 2002; Walton et al. 2006).
Furthermore, the control of P. ficus through MD has been investigated by testing the VMB sex pheromone in sprayable microencapsulated formulations (Daane et al. 2006; Walton et al. 2006), as well as in reservoir dispensers (Cocco et al. 2014; Sharon et al. 2016). Results obtained with hand-applied dispensers were promising. In addition, they are easy to use for farmers compared to sprayable microencapsulations. However, the appeal of this eco-friendly control tool is limited by the high density of dispensers per hectare; indeed, studies currently available tested MD products at a density higher than 600 units per hectare (Cocco et al. 2014; Sharon et al. 2016). Costs of MD programs for farmers need to be reduced by decreasing either the amount of pheromone per hectare (i.e., costs for material) or the number of dispensers per hectare (i.e., labor costs for deployment) or both.
In this research, a novel hand-applied reservoir dispenser (Isonet PF®, Shin-Etsu Chemical Co. Ltd., Japan) loaded with 180 mg of racemic lavandulyl senecioate was evaluated: three different dispenser densities (i.e., 300, 400, and 500 dispensers/ha) were tested at four study sites located in Northern (Veneto region) and Southern (Sicily region) Italy. Experiments were carried out over 2 years by monitoring the VMB populations in untreated wine and table grape vineyards as well as in vineyards managed with MD. Lastly, Isonet® PF dispensers were periodically collected during the whole grape-growing season, extracted, and analyzed by GC-MS, to monitor content and release of VMB sex pheromone in mg/ha/day.
Materials and methods
Experimental sites
Trials were conducted in 2016 and 2017 in areas typical for Italian grape cultivation, on different wine and table grape varieties. In 2016, three trials were carried out in the area of Acate, Ragusa province, Sicily region, Southern Italy (Table 1). Two trials were performed on early-ripening wine grape varieties, one on Chardonnay and one on Pinot Grigio (expected harvest time: beginning of August), and another trial was carried out on the later-ripening wine grape variety Nero d’Avola (expected harvest time: end of August–beginning of September) (Table 1).
In 2017, a total of four trials were conducted. Again, three trials were performed in the province of Ragusa (Sicily region, Southern Italy): one on the early-ripening wine grape variety Chardonnay, one on the later-ripening wine grape variety Nero d’Avola, and one on the table grape variety Italia. A fourth trial was carried out in Colognola ai Colli, Verona province (Veneto region, Northern Italy), on the wine grape variety Garganega. Details on vineyard location and pest history are found in Table 1. The study sites 1 and 4 are the same vineyard block tested over different years; this also applies to sites 3 and 5. A detailed description of each cultivar is given in Table 2.
Reservoir dispensers (Isonet® PF, Shin-Etsu Chemical Co. Ltd., Japan) were tested at three different application rates: 300, 400, and 500 dispensers/ha. Isonet® PF consists of two parallel polyethylene tubes, one of which contains an aluminum wire that enables their placement on supports and the other is filled with 180 mg of racemic lavandulyl senecioate TGAI. The VMB racemic pheromone amount per hectare tested in our MD trials was 54, 72, and 90 g, corresponding to a dispenser density of 300, 400, and 500 Isonet® PF dispensers/ha, respectively.
Experimental design
A randomized block design is not applicable to the large plots required for studies on MD products (European and Mediterranean Plant Protection Organization 2016), so each treatment (i.e., Isonet® PF at 300, 400, and 500 dispensers/ha, respectively, and untreated control) was applied to a large plot. There were six sampling units per plot, which were distributed in a grid pattern. Each sampling unit was sufficiently large for there to be at least 100 bunches per assessment. Isonet® PF–treated plots were at least 2 ha in size (range 2.01–4.75 ha). Untreated control plots were not as large (range 0.12–0.64 ha), but had a comparable pest history and allowed for assessments on the same number of bunches per sampling unit. In all Isonet® PF–treated plots, the dispensers were deployed before the beginning of the migration of VMB nymphs in spring (mid-April in Sicily, end of April in Veneto).
Male captures
Following the method by Walton et al. (2006), P. ficus males were monitored during both 2016 and 2017 using red Pherocon Delta IIID sticky traps (Trécé Inc., Adair, OK). Traps were baited with 100 μg of racemic synthetic lavandulyl senecioate from Pherobank B.V. (Wijk bij Duurstede, The Netherlands) in rubber septa lures (Suterra Europe). One trap was used per large plot. Trap sticky plates and sex pheromone lures were replaced monthly, and trapped males were counted by direct observation using a stereoscopic microscope (Leica, Germany). Data were analyzed as the total number of males caught before harvest.
Infestation and population density evaluation
The MD efficacy of Isonet® PF was evaluated on 100 bunches per sampling unit by determining the percentage of bunches infested by P. ficus and the number of VMB/bunch at harvest (BBCH 89). Within each of six sampling units, the number of bunches infested by VMB was counted on 100 randomly selected bunches, giving a total of 600 bunches per plot. Then, the percentage of P. ficus–infested bunches was calculated. In addition, the number of VMB per bunch was noted to provide an indication of the damage severity.
Sex pheromone release
In both study years, Isonet® PF dispensers were periodically collected in Sicily during the whole grapevine-growing season, extracted, and analyzed by GC-MS, according to the method by Lucchi et al. (2018c). Isonet® PF residual content in racemic lavandulyl senecioate was estimated based on internal (SEC) standard GC-MS analysis. The latter was done using an Agilent 6890 N gas chromatograph equipped with a 5973 N mass spectrometer (MS). MS settings were as follows: EI mode, 70 eV, mass to charge ratio (m/z) scan between 35 and 400. HP-5 MS capillary column (30 m × ID 0.25 mm × 0.25-μm film thickness, J & W Scientific, Folsom, CA, USA) with helium gas flow (1.0 mL/min) was used for separation. GC temperature program was as follows: initial 50 °C for 5 min, then increasing with 20 °C/min to 300 °C. The injector temperature was set at 150 °C. The GC-MS estimate of the dispenser residual content allowed the calculation of the pheromone release for the duration of the field deployment of the dispenser. Each value was a mean ± SD of four replicates, each obtained extracting and analyzing a single dispenser, except for the value presented on April 22, 2016, obtained as a mean ± SD of 10 analyzed dispensers.
Statistical analysis
Mealybug populations in vineyards are typically clumped in their spatial distribution (Geiger and Daane 2001); in our study, the incidence of P. ficus–infested bunches and number of VMB per bunch were not normally distributed. Data transformation, including ln (x + 1), was not able to normalize the distribution and homogenize the variance (Shapiro-Wilk test, goodness of fit P < 0.001). Thus, non-parametric statistics were used. Differences in male catches, incidence of bunches infested by P. ficus, and the number of VMB per bunch among treatments (i.e., the tested sex pheromone dispenser Isonet® PF at three different densities per hectare and the untreated control), years, and study site were assessed using the Kruskal–Wallis test followed by the Steel–Dwass multiple comparison; a P value of 0.05 was selected as threshold to assess significant differences.
Results
Male captures
VMB male captures in pheromone-baited traps, on wine grape in 2017, were significantly lower in MD plots at all Isonet® PF–tested rates (300, 400, and 500 dispensers/ha) compared with the untreated plots (χ2 = 37.381, d.f. = 3, P < 0.0001). This suggests that Isonet® PF, irrespective of the tested dispenser rate, can be effective in preventing VMB males from finding females (Fig. 1a).
Box plots showing differences in Planococcus ficus (VMB) male total catches among a mating disruption treatments, b study site, and c year. Red box plots indicate the median (line) within each box and the range of dispersion (lower and upper quartiles and outliers) of the median capture parameter. Green lines and blue T-bars indicate mean values and standard errors, respectively
Furthermore, a significant difference in the abundance of trapped VMB males was noted between the trial sites (χ2 = 13.325, d.f. = 4, P = 0.001), with higher catches in Colognola ai Colli over other vineyards (Fig. 1b).
VMB total catches per trap also showed a significant difference between 2016 and 2017 (χ2 = 3.725, d.f. = 1, P = 0.05) (Fig. 1c). In the trials carried out on table grape in 2017, as well as in all experiments conducted in 2016, negligible trap catches were recorded in all treatments. The total number of VMB male catches per trap up to harvest never exceeded 15 individuals in all treatments (Fig. 1). Due to the low number of P. ficus individuals caught during the entire study period, no conclusions can be drawn on the efficacy of Isonet® PF, based on pest monitoring in table grapes.
Incidence of bunches infested by Planococcus ficus
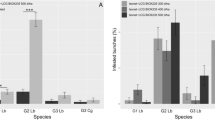
Bunch infestation by P. ficus was statistically different among treatments (χ2 = 65.131, d.f. = 3, P < 0.0001) (Fig. 2a), with significantly higher infestation levels in the untreated control compared with vineyards where Isonet® PF dispensers were deployed at 300 (Z = 6.792, P < 0.0001), 400 (Z = 6.649, P < 0.0001), and 500 units (Z = 5.990, P < 0.0001) per ha, respectively. No significant differences were noted in the incidence of bunches infested by P. ficus among the three tested densities of Isonet® PF dispensers (300 vs. 400 dispensers ha−1: Z = − 0.031, P = 1.000; 400 vs. 500 dispensers ha−1: Z = 1.200, P = 0.626; 300 vs. 500 dispensers ha−1: Z = 1.093, P = 0.693) (Fig. 2a).
Box plots showing the differences in the incidence of Planococcus ficus (VMB)–infested bunches (%) among a different mating disruption treatments, b study site, and c year. Red box plots indicate the median (line) within each box and the range of dispersion (lower and upper quartiles and outliers) of the median infestation parameter. Green lines and blue T-bars indicate mean values and standard errors, respectively
Furthermore, significant differences in the incidence of VMB infestation were detected among the trial sites (χ2 = 29.962, d.f. = 4, P < 0.0001), where Chiaramonte Gulfi showed lower infestation incidence when compared to Colognola ai Colli (Z = − 4.023, P = 0.0005), Acate 1 (Z = − 4.544, P < 0.0001), Acate 2 (Z = − 3.981, P = 0.0007), and Acate 3 (Z = − 4.684, P < 0.0001) (Fig. 2b). Also, the incidence of P. ficus infestation on bunches varied between the two experimental years (χ2 = 43.318, d.f. = 1, P < 0.0001) (Fig. 2c).
Number of vine mealybugs per bunch
The number of VMB per bunch significantly differed among treatments (χ2 = 55.406, d.f. = 3, P < 0.0001) (Fig. 3a), with significantly higher values in the untreated control than in the vineyards managed using Isonet® PF dispensers at 300 (Z = 5.989, P < 0.0001), 400 (Z = 6.640, P < 0.0001), and 500 units (Z = 5.639, P < 0.0001) per ha, respectively. No significant differences were noted in the number of VMB per bunch among the three tested densities of Isonet® PF dispensers (300 vs. 400 dispensers ha−1: Z = − 0.428, P = 0.973; 400 vs. 500 dispensers ha−1: Z = 0.576, P = 0.939; 300 vs. 500 dispensers ha−1: Z = 0.071, P = 0.973) (Fig. 3a).
Box plots showing the differences in number of Planococcus ficus (VMB) per bunch among a different mating disruption treatments, b study site, and c year. Red box plots indicate the median (line) within each box and the range of dispersion (lower and upper quartiles and outliers) of the median infestation parameter. Green lines and blue T-bars indicate mean values and standard errors, respectively
Significant differences were observed in the number of VMB per bunch across the study sites (χ2 = 25.213, d.f. = 4, P < 0.0001), with Chiaramonte Gulfi showing a lower number of VMB/bunch than Colognola ai Colli (Z = − 4.211, P = 0.0002), Acate 1 (Z = − 4.226, P = 0.0002), Acate 2 (Z = − 3.609, P = 0.0029), and Acate 3 (Z = − 4.199, P = 0.0003) (Fig. 3b). Lastly, significant differences in the number of VMB per bunch were found between the two study years (χ2 = 12.801, d.f. = 1, P = 0.0003) (Fig. 3c).
Sex pheromone release
Figure 4 shows the load of racemic lavandulyl senecioate per dispenser in Sicilian study vineyards during the growing season 2016 (Fig. 4a) and 2017 (Fig. 4b). Results highlighted a rather homogenous release rate of the active ingredient during the whole grapevine-growing season.
In 2016, GC-MS analyses outlined a halving of the dispenser load in late July (i.e., from 173.1 ± 1.7 on April 22 to 97.5 ± 1.8 mg/dispenser), declining to 59.9 ± 4.4 mg/dispenser in mid-late September (Fig. 4a).
Furthermore, the 2017 data show a comparable release trend, with a halving of the racemic lavandulyl senecioate content in late July (i.e., from 169.3 ± 2.2 on April 11 to 85.1 ± 6.5 mg/dispenser). Later, the active ingredient content dropped to 41.6 ± 6.3 mg/dispenser on September 21; the residual content was 29.0 ± 2.3 mg/dispenser on November 27 (Fig. 4b).
Discussion
The results show the successful use of MD dispenser Isonet® PF, which led to a significant reduction of VMB-infested bunches (%) and number of VMB per bunch, compared with the untreated vineyards, in both wine and table grape varieties. On the other hand, the strong variation in the abundance of male catches between 2016 and 2017 highlighted the poor reliability of male trapping for evaluating MD efficacy compared to VMB direct sampling on grapes. The history of MD against P. ficus is relatively recent. Walton et al. (2006) firstly tested MD efficacy against VMB. They conducted field assays in California, using a sprayable microencapsulated formulation of racemic lavandulyl senecioate. After three to four applications per year, the authors observed that VMB trap catches were lower, if compared to the control without MD. In addition, as shown in our study, VMB density and crop damage significantly decreased. Moreover, P. ficus females exposed to MD treatments produced a limited number of eggs, if compared to control ones (Walton et al. 2006). Research conducted in California vineyards showed that VMB can be successfully controlled with a pheromone dispenser density of 437 dispensers per hectare (93.75 g of a.i. per hectare) (Langone et al. 2014). Cocco et al. (2014) attempted MD against VMB in Sardinian vineyards. The authors observed no significant differences among treatments in grape damage at harvest due to very low damage rates throughout all treatments in both field trials. However, they observed that hand-applied reservoir dispensers (625 units/ha) releasing multiple plumes of the VMB racemic sex pheromone influenced pest density and modified age structure of the population. In addition, the effect was dose-dependent: MD at a pheromone dose of 62.5 g/ha did not affect VMB populations, while racemic lavandulyl senecioate at 93.8 g/ha reduced P. ficus density and also affected the population’s age structure, showing a lower abundance of ovipositing females (Cocco et al. 2014). Later, Mansour et al. (2017b) showed that deploying 625 Checkmate® pheromone dispensers per hectare (150 mg of a.i.) led to the effective management of VMB populations in Tunisian table grape vineyards. Very recently, Cocco et al. (2018) pointed out that the MD dispenser tested led to a large reduction (i.e., minimum reduction 18.8%; maximum reduction 66.2%, depending on the year) in ovipositing females in MD vineyards, over a 3-year study period. In addition, this study highlighted that MD led to a longer pre-oviposition period by up to 12.5 days (Cocco et al. 2018).
It can be argued that MD on wide areas may affect behavioral traits of non-target arthropods. The use of MD as an eco-friendly IPM strategy has been widely promoted as insect pheromones are usually species-specific and non-toxic (Witzgall et al. 2010; Lucchi and Benelli 2018). However, Shapira et al. (2018) recently showed that managing VMB with MD may affect the abundance of Anagyrusvladimiri Triapitsyn (ex Anagyrus sp. near pseudococci (Girault)), the main parasitoid of P. ficus, outlining the urgent need of further research on this issue.
Interestingly, in the MD experiments reported in this study, all three tested pheromone doses (54, 72, and 90 g/ha, corresponding to 300, 400, and 500 Isonet® PF dispenser/ha, respectively) resulted in a significant reduction of VMB infestation in comparison to the untreated control. No significant differences in the incidence of bunches infested by P. ficus and number of VMB/bunch among the three tested pheromone doses were detected. In addition, a lower amount of VMB racemic pheromone was used, if compared to earlier research, where 93 g/ha (Sharon et al. 2016) or 62.5 and 93.8 g/ha (Cocco et al. 2014) of VMB racemic lavandulyl senecioate had been tested. It is well known that the efficacy of MD products depends on several factors (Neumann 1996; Ioriatti and Lucchi 2016). Among them, a key role is played by the size of the area where MD products are applied, the curve of pheromone release over time, and the pheromone concentration per area, which is to a great extent determined by the type of dispenser. In these MD experiments, the VMB sex pheromone release was rather constant during the grapevine-growing season (Fig. 4). In addition, the residual content of racemic lavandulyl senecioate detected in late November 2017 (Fig. 4b) ensures a good release during the mating period of overwintering females. A constant and long-lasting pheromone release is extremely important for effective MD, especially for VMB, which in Mediterranean countries can develop four generations per year, with males searching for mates until late October (Cocco et al. 2014). It can be assumed that the constant and long-lasting release of pheromone over time by Isonet® dispensers and the large size of the study plots (at least 2 ha) enabled the authors to achieve a reliable MD efficacy during the whole growing season, including with a VMB pheromone dose of only 54 g/ha.
The findings in this research support earlier studies highlighting that MD can represent a reliable control method to significantly reduce P. ficus populations, particularly after one season characterized by low population density (Walton et al. 2006; Cocco et al. 2014; Mansour et al. 2017b). Sharing this theoretical background, Sharon et al. (2016) showed that in presence of high VMB populations, the MD effectiveness achieves a substantial increase only after 2 years of application. In other words, MD needs to be deployed over several seasons to result in acceptable population levels. However, in our field experiment conducted in Veneto region, the MD approach ensured an effective control of VMB, despite the high pest pressure characterizing the study site. In contrast to earlier observations (Lentini et al. 2008), the overwintering population in our study plots was mainly composed of young instars, with negligible presence of mated females (data not shown), which can contribute to VMB population build-up despite MD.
In this study, it was observed that there was a lower VMB infestation in the table grape variety Italia over the other varieties. This difference is probably due to the differences in historic pesticide strategies in the selected vineyards, since table grape vineyards have a lower tolerance threshold and are more subjected to insecticide treatments (Cabras and Angioni 2000; Ravelo-Pérez et al. 2009).
Overall, the results presented here outline a successful MD approach that should be taken into consideration within an IPM program to effectively manage VMB populations. Indeed, if it cannot be used alone as a fully efficient sustainable control option, especially in the case of high mealybug infestations, the use of MD in combination with an effective and safer insecticide could represent the appropriate way to ensure long-term field efficacy to control vine mealybugs in vineyards (Mansour et al. 2018). Contrary to earlier studies, this research provides evidence for successful MD programs for P. ficus control using low pheromone doses per hectare. This can help farmers to reduce costs in terms of both material to be purchased and labor for field application, as well as to adopt area-wide sustainable VMB control strategies for vineyards (Daane et al. 2018b).
References
Almeida RP, Daane KM, Bell VA, Blaisdell GK, Cooper ML, Herrbach E, Pietersen G (2013) Ecology and management of grapevine leafroll disease. Front Microbiol 4:1–13
Cabras P, Angioni A (2000) Pesticide residues in grapes, wine, and their processing products. J Agric Food Chem 48:967–973
Chiotta ML, Ponsone ML, Torres AM, Combina M, Chulze SN (2010) Influence of Planococcus ficus on Aspergillus section Nigri and ochratoxin A incidence in vineyards from Argentina. Lett Appl Microbiol 51:212–218
Cocco A, Lentini A, Serra G (2014) Mating disruption of Planococcus ficus (Hemiptera: Pseudococcidae) in vineyards using reservoir pheromone dispensers. J Insect Sci 14:144
Cocco A, Muscas E, Mura A, Iodice A, Savino F, Lentini A (2018) Influence of mating disruption on the reproductive biology of the vine mealybug, Planococcus ficus (Hemiptera: Pseudococcidae), under field conditions. Pest Manag Sci 74:2806–2816
da Silva EB, Mendel Z, Franco JC (2010) Can facultative parthenogenesis occur in biparental mealybug species? Phytoparasitica 38:19–21
da Silva VP, Galzer ECW, Malausa T, Germain JF, Kaydan MB, Botton M (2016) The vine mealybug Planococcus ficus (Signoret) (Hemiptera: Pseudococcidae) damaging vineyards in Brazil. Neotrop Entomol 45:449–451
Daane KM, Bentley W, Walton V, Malakar-Kuenen R, Millar J et al (2006) New controls investigated for vine mealybug. Calif Agric 60:31–38
Daane KM, Almeida RP, Bell VA, Walker JT, Botton M, Fallahzadeh M, et al (2012) Biology and management of mealybugs in vineyards. In: Bostanian NJ, Vincent C, Isaacs R (eds) Arthropod management in vineyards. Springer, Dordrecht, p 271–307
Daane KM, Middleton MC, Sforza RF, Kamps-Hughes N, Watson GW, Almeida RP et al (2018a) Determining the geographic origin of invasive populations of the mealybug Planococcus ficus based on molecular genetic analysis. PLoS One 13(3):e0193852
Daane KM, Vincent C, Isaacs R, Ioriatti C (2018b) Entomological opportunities and challenges for sustainable viticulture in a global market. Annu Rev Entomol 63:193–214
Douglas N, Krüger K (2008) Transmission efficiency of grapevine leafroll-associated virus 3 (GLRaV-3) by the mealybugs Planococcus ficus and Pseudococcus longispinus (Hemiptera: Pseudococcidae). Eur J Plant Pathol 122:207–212
European and Mediterranean Plant Protection Organization (2016) Efficacy evaluation of plant protection products. Mating disruption pheromones (http://pp1.eppo.int/). First approved in 2008–09. Last update December 2016
Fallahzadeh M, Japoshvili G, Saghaei N, Daane KM (2011) Natural enemies of Planococcus ficus (Hemiptera: Pseudococcidae) in Fars province vineyards, Iran. Biocontrol Sci Tech 21:427–433
Franco JC, Zada A, Mendel Z, Ishaaya I, Horowitz AR (2009) Novel approaches for the management of mealybug pests. In: Ishaaya I, Horowitz AR (eds) Biorational control of arthropod pests. Springer, Dordrecht, pp 233–278
Geiger CA, Daane KM (2001) Seasonal movement and distribution of the grape mealybug (Homoptera: Pseudococcidae); developing a sampling program for San Joaquin Valley vineyards. J Econ Entomol 94:291–301
Güleç G, Kilincer AN, Kaydan MB, Ülgentürk S (2007) Some biological interactions between the parasitoid Anagyrus pseudococci (Girault) (Hymenoptera: Encyrtidae) and its host Planococcus ficus (Signoret) (Hemiptera: Coccoidea: Pseudococcidae). J Pest Sci 80:43–49
Gut LJ, Stelinski LL, Thomson DR, Miller JR (2004) Behaviour-modifying chemicals: prospects and constraints in IPM. In: Koul, Dhaliwal, Cuperus (eds) Integrated pest management: potential, constraints, and challenges. CABI Publishing, Cambridge, pp 73–121
Gutierrez AP, Daane KM, Ponti L, Walton VM, Ellis CK (2008) Prospective evaluation of the biological control of vine mealybug: refuge effects and climate. J Appl Ecol 45:524–536
Haviland DR, Bentley WJ, Daane KM (2005) Hot-water treatments for control of Planococcus ficus (Homoptera: Pseudococcidae) on dormant grape cuttings. J Econ Entomol 98:1109–1115
Hinkens DM, McElfresh JS, Millar JG (2001) Identification and synthesis of the sex pheromone of the vine mealybug, Planococcus ficus. Tetrahedron Lett 42:1619–1621
Ioriatti C, Lucchi A (2016) Semiochemical strategies for tortricid moth control in apple orchards and vineyards in Italy. J Chem Ecol 42(7):571–583
Ioriatti C, Lucchi A, Bagnoli B (2008) Grape area-wide pest management in Italy. In: Koul et al (eds) Area-wide pest management: theory and implementation. CAB International, Wallingford, p 208–225
Lance DR, Leonard DS, Mastro VC, Walters ML (2016) Mating disruption as a suppression tactic in programs targeting regulated lepidopteran pests in US. J Chem Ecol 42:590–605
Langone D, Kurtural K, Daane KM (2014) Mating disruption of vine mealybug. Practical Winery & Vineyard, San Rafael, p 1–3
Le Vieux PD, Malan AP (2016) An overview of the vine mealybug (Planococcus ficus) in South African vineyards and the use of entomopathogenic nematodes as potential biocontrol agent. South Afric J Enol Vitic 34:108–118
Lentini A, Serra G, Ortu S, Delrio G (2008) Seasonal abundance and distribution of Planococcus ficus on grape vine in Sardinia. IOBC/WPRS Bull 36:267–272
Lentini A, Mura A, Muscas E, Nuvoli MT, Cocco A (2018) Effects of delayed mating on the reproductive biology of the vine mealybug, Planococcus ficus (Hemiptera: Pseudococcidae). Bull Entomol Res 108:263–270
Lucchi A, Benelli G (2018) Towards pesticide-free farming? Sharing needs and knowledge promotes integrated pest management. Environ Sci Pollut Res 25:13439–13445
Lucchi A, Sambado P, Juan Royo AB, Bagnoli B, Benelli G (2018a) Lobesia botrana males mainly fly at dusk: video camera-assisted pheromone traps and implications for mating disruption. J Pest Sci 91:1327–1334
Lucchi A, Sambado P, Juan Royo AB, Bagnoli B, Conte G, Benelli G (2018b) Disrupting mating of Lobesia botrana using sex pheromone aerosol devices. Environ Sci Pollut Res 25:22196–22204
Lucchi A, Ladurner E, Iodice A, Savino F, Ricciardi R, Cosci F, Conte G, Benelli G (2018c) Eco-friendly pheromone dispensers – a green route to manage the European grapevine moth? Environ Sci Pollut Res 25:9426–9442
Mansour R, Lebdi KG, Rezgui S (2010) Assessment of the performance of some new insecticides for the control of the vine mealybug Planococcus ficus in a Tunisian vineyard. Entomol Hell 19:21–33
Mansour R, Grissa-Lebdi K, Suma P, Mazzeo G, Russo A (2017a) Scale insects (Hemiptera: Coccoidea) of high economic importance in a Mediterranean area: host plants, bio-ecological characteristics, natural enemies and pest management strategies – a review. Plant Prot Sci 53:1–14
Mansour R, Grissa-Lebdi K, Khemakhem M, Imed C, Trabelsi I, Sabri A, Marti S (2017b) Pheromone-mediated mating disruption of Planococcus ficus (Hemiptera: Pseudococcidae) in Tunisian vineyards: effect on insect population dynamics. Biologia 72(3):333–341
Mansour R, Belzunces L, Suma P, Zappalà L, Mazzeo G, Grissa-Lebdi K, Russo A, Biondi A (2018) Vine and citrus mealybug pest control based on synthetic chemicals. A review. Agron Sustain Dev 38:37
Mgocheki N, Addison P (2009) Interference of ants (Hymenoptera: Formicidae) with biological control of the vine mealybug Planococcus ficus (Signoret) (Hemiptera: Pseudococcidae). Biol Control 49:180–185
Millar JG (2007) Insect pheromones for integrated pest management: promise versus reality. Redia 90:51–55
Millar JG, Daane KM, McElfresh JS, Moreira JA, Malakar-Kuenen R, Guillén M, Bentley WJ (2002) Development and optimization of methods for using sex pheromone for monitoring the mealybug Planococcus ficus (Homoptera: Pseudococcidae) in California vineyards. J Econ Entomol 95:706–714
Miller JR, Gut LJ (2015) Mating disruption for the 21st century: matching technology with mechanism. Environ Entomol 44:427–453
Neumann U (1996) Factors affecting the effectiveness of the mating disruption technique principles and necessities. Acta Hortic 422:241–246
Ravelo-Pérez LM, Hernández-Borges J, Herrera-Herrera AV, Rodríguez-Delgado MÁ (2009) Pesticide extraction from table grapes and plums using ionic liquid based dispersive liquid–liquid microextraction. Anal Bioanal Chem 395:2387–2395
Romano D, Benelli G, Stefanini C, Desneux N, Ramirez-Romero R, Canale A, Lucchi A (2018) Behavioral asymmetries in the mealybug parasitoid Anagyrus sp. near pseudococci: does lateralized antennal tapping predicts male mating success? J Pest Sci 91:341–349
Shapira I, Keasar T, Harari AR, Gavish-Regev E, Kishinevsky M, Steinitz H, Sofer-Arad C, Tomer M, Avraham A, Sharon R (2018) Does mating disruption of Planococcus ficus and Lobesia botrana affect the diversity, abundance and composition of natural enemies in Israeli vineyards? Pest Manag Sci 74:1837–1844. https://doi.org/10.1002/ps.4883
Sharon R, Zahavi T, Sokolsky T, Sofer-Arad C, Tomer M, Kedoshim R, Harari AR (2016) Mating disruption method against the vine mealybug, Planococcus ficus: effect of sequential treatment on infested vines. Entomol Exp Appl 161:65–69
Sokolsky T, Cohen Y, Zahavi T, Sapir G, Sharon R (2013) Potential efficiency of grapevine leafroll disease management strategies using simulation and real spatio-temporal disease infection data. Austr J Grape Wine Res 19:431–438
Suma P, Mansour R, Russo A, La Torre I, Bugila AAA, Franco JC (2012) Encapsulation rates of the parasitoid Anagyrus sp. nr. pseudococci, by three mealybug species (Hemiptera: Pseudococcidae). Phytoparasitica 40(1):11–16
Tanne E, Ben-Dov Y, Raccah B (1989) Transmission of the corky-bark disease by the mealybug Planococcus ficus. Phytoparasitica 17:55–55
Tsai CW, Chau J, Fernandez L, Bosco D, Daane KM, Almeida RPP (2008) Transmission of grapevine leafroll-associated virus 3 by the vine mealybug (Planococcus ficus). Phytopathology 98:1093–1098
Tsai CW, Rowhani A, Golino DA, Daane KM, Almeida RP (2010) Mealybug transmission of grapevine leafroll viruses: an analysis of virus–vector specificity. Phytopathology 100(8):830–834
Walton VM, Pringle KL (2017) Vine mealybug, Planococcus ficus (Signoret) (Hemiptera: Pseudococcidae), a key pest in South African vineyards. A review. South Afric J Enol Vitic 25:54–73
Walton VM, Daane KM, Pringle KL (2004) Monitoring Planococcus ficus in South African vineyards with sex pheromone-baited traps. Crop Prot 23:1089–1096
Walton VM, Daane KM, Bentley WJ, Millar JG, Larsen TE, Malakar-Kuenen R (2006) Pheromone-based mating disruption of Planococcus ficus (Hemiptera: Pseudococcidae) in California vineyards. J Econ Entomol 99:1280–1290
Waterworth RA, Wright IM, Millar JG (2011) Reproductive biology of three cosmopolitan mealybug (Hemiptera: Pseudococcidae) species, Pseudococcus longispinus, Pseudococcus viburni, and Planococcus ficus. Ann Entomol Soc Am 104:249–260
Welter S, Pickel C, Millar J, Cave F, Van Steenwyk R, Dunley J (2005) Pheromone mating disruption offers selective management options for key pests. Calif Agric 59:16–22
Witzgall P, Kirsch P, Cork A (2010) Sex pheromones and their impact on pest management. J Chem Ecol 36:80–100
Zada A, Dunkelblum E, Assael F, Harel M, Cojocaru M, Mendel Z (2003) Sex pheromone of the vine mealybug, Planococcus ficus in Israel: occurrence of a second component in a mass-reared population. J Chem Ecol 29:977–988
Zada A, Dunkelblum E, Assael F, Franco JC, Da Silva EB, Protasov A, Mendel Z (2008) Attraction of Planococcus ficus males to racemic and chiral pheromone baits: flight activity and bait longevity. J Appl Entomol 132:480–489
Acknowledgements
Three anonymous reviewers kindly improved an earlier version of our manuscript. The authors are grateful to Shin-Etsu for kindly providing the tested dispensers. We would like to thank Dr. Mauro Varner, Cantine Mezzacorona (Villa Albius), and Dr. Sergio Rizzo, Cantine Fichera & Torrisi (Chiaramonte Gulfi), for the technical assistance, and Dr. Roma L. Gwynn (Duns, Scotland, UK) for proofreading the manuscript.
Funding
This research was partially funded by the University of Catania, Research Project 2016-18 “Emergent Pests and Pathogens and Relative Sustainable Strategies” - 5A722192113.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Responsible editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lucchi, A., Suma, P., Ladurner, E. et al. Managing the vine mealybug, Planococcus ficus, through pheromone-mediated mating disruption. Environ Sci Pollut Res 26, 10708–10718 (2019). https://doi.org/10.1007/s11356-019-04530-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-04530-6