Abstract
Mosquitoes are responsible for the transmission of many pathogens and parasites, which cause serious diseases in humans and animals. Currently, botanical products have been suggested as alternative tools in the fight against arthropod vectors. In this study, the essential oil (EO) extracted from Zingiber cernuum was tested as larvicide and oviposition deterrent on six mosquito species of public health relevance, including malaria and Zika virus vectors. The EO showed high toxicity on third instar larvae of Anopheles stephensi (LC50 = 41.34 μg/ml), Aedes aegypti (LC50 = 44.88 μg/ml), Culex quinquefasciatus (LC50 = 48.44 μg/ml), Anopheles subpictus (LC50 = 51.42 μg/ml), Aedes albopictus (LC50 = 55.84 μg/ml), and Culex tritaeniorhynchus (LC50 = 60.20 μg/ml). In addition, low doses of Z. cernuum EO reduced oviposition rates in six mosquito species. The acute toxicity of Z. cernuum EO on four mosquito predators was scarce; LC50 ranged from 3119 to 11,233 μg/ml. Overall, our results revealed that the Z. cernuum EO can be considered for the development of effective and environmental-friendly mosquito larvicides and oviposition deterrents.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Arthropods are dangerous vectors of important life-threatening and debilitating diseases. Among them, mosquitoes (Diptera: Culicidae) belonging to the genera Anopheles, Aedes, and Culex act as vectors of pathogens and parasites causing malaria, filariasis, Japanese encephalitis, dengue and dengue hemorrhagic fever, yellow fever, chikungunya, and, very recently, Zika virus (Benelli and Mehlhorn 2016; Ward and Benelli 2017). Several efforts have been made to improve the control of mosquito vectors. Besides the efficacy at low doses, the use of synthetic insecticides can lead to high costs, concerns for environmental sustainability, harmful effects on human health and other non-target populations, and development of insecticide resistance in the targeted pests (Benelli 2015a; Naqqash et al. 2016).
While chemical insecticides are usually based on a single active ingredient, plant-derived pesticides consist of a combination of molecules which can act concertedly on both behavioral and physiological processes (Jain et al. 2001; Pavela 2015). Thus, there is very little chance of resistance development in the treated arthropods. In this scenario, the identification of effective and eco-friendly biopesticides is crucial for the successful management of arthropod vectors (Lucia et al. 2007; Cheng et al. 2003, 2004, 2008, 2009a; Govindarajan and Benelli 2016a, b).
Essential oils (EOs) are complex mixtures of volatile organic compounds produced as secondary metabolites in plants. They include blends of terpenes, sesquiterpenes, and oxygenated compounds, such as alcohols, esters, ethers, aldehydes, ketones, lactones, phenols, and phenol ethers (Chericoni et al. 2004; Astani et al. 2010; Almeida et al. 2011). Plant EOs and their constituents have been proposed as effective pesticides, since they are able to evoke acute toxicity and oviposition and feeding deterrence, as well as repellency (Barnard 1999). The insecticidal properties of EO are widely documented (e.g., Pathak and Dixit 1988; Araujo et al. 2003; Traboulsi et al. 2002; Ansari et al. 2000a, b, 2005; Koul et al. 2008; Rattan 2010; Pavela 2008a, b; Urzúa et al. 2010; Benelli 2015b). To the best of our knowledge, any development of resistance to EOs and their constituents has been reported (Sharma et al. 1992; Cavalcanti et al. 2004; Pavela and Benelli 2016).
A growing number of researchers focused on the effectiveness of plant EOs against young mosquito instars (Pavela 2015). Good examples include Lippia sidoides (Carvalho et al. 2003), Tagetes patula (Dharmagadda et al. 2005), Pinus kesiya (Govindarajan et al. 2016a), Cordia leucomalloides and C. curassavica (Santos et al. 2006), Blumea mollis (Senthilkumar et al. 2008), Piper klotzschianum (Do Nascimento et al. 2013), Tetradium glabrifolium (Liu et al. 2015), Chloroxylon swietenia (Kiran et al. 2006), Origanum majorana (El-Akhal et al. 2014), Thymus vulgaris (El-Akhal et al. 2015), T. magnus (Park et al. 2012), T. transcaspicus (Dargahi et al. 2014), Cinnamomum osmophloeum (Cheng et al. 2009b), Clausena excavata (Cheng et al. 2009c), Toddalia asiatica (Liu et al. 2013), Saussurea lappa (Liu et al. 2012), Ipomoea carica (Thomas et al. 2004), and Zingiber officinalis (Pushpanathan et al. 2008). Pitasawata et al. (2007) and Champakaew et al. (2007) reported that Curcuma zedoaria EO showed larvicidal toxicity on Aedes aegypti, while the Zingiber zerumbet rhizome EO had larvicidal and pupicidal activity on anopheline mosquitoes (Tewtrakul et al. 1998). Sutthanont et al. (2010) recommended the use of this EO as mosquito larvicide. Kamaraj et al. (2010) reported that the hexane extract of Z. zerumbet had larval toxicity against Culex quinquefasciatus.
The family of Zingiberaceae represents a key source of herbal preparations and phytoconstituents of interest for current pharmacology, parasitology, and entomology (Burkill 1966; Negi et al. 1999; Scartezzini and Speroni 2000; Youko et al. 2000; Patricia et al. 2003; Jirovetz et al. 2003; Bendjeddou et al. 2003; Nguefack et al. 2004; Govindarajan et al. 2016b). The Zingiber officinale rhizome EO is one of the most studied Zingiberaceae EOs. It contains monoterpenoids and sesquiterpenoids. Main molecules are α-zingiberene, α-curcumene, β-bisabolene, and β-sesquiphellandrene (Menon 2007; Rana et al. 2008; Padmakumari et al. 2009), while Z. zerumbet EO shows a high content of the monocyclic sesquiterpene ketone zerumbone, as well as an oxygenated humulene derivative (Srivastava et al. 2000; Bhuiyan et al. 2009).
Zingiber cernuum Dalzell (Zingiberaceae), commonly known as curved-stem ginger, is widely found in the evergreen forests of Western Ghats, India. It is a large perennial herb, 1–2 m tall, with curved stem. The leaves are 15–30 cm in length, narrow-elliptic, and long-pointed. The flowers are borne in spikes 5–10 cm long, directly from the rootstock, rising just above the ground. Bracts are 2–3 cm long, greenish-yellow. The sepal cup is shortly three-lobed. Stamen is single, with a short filament. The style is threadlike. The capsules are 1 cm long, smooth with red, channeled seeds. The flowers are yellow colored, variegated with red, with the lib broad and three-lobed (Sanjay 2015). From a phytochemical point of view, Z. cernuum is extremely scarcely studied. Kasarkar and Kulkarni (2011) recently identified flavonoids and tannins in Z. cernuum extracts, which showed antioxidant activity (Sanjay 2015). This species is rich in iron and manganese and showed low amounts of molybdenum, sulfur, and nitrate in rhizome and leaves (Kasarkar and Kulkarni 2011). To the best of our knowledge, the composition and mosquitocidal bioactivity of Z. cernuum EO have not been explored.
In this research, we analyzed the chemical composition of Z. cernuum EO using gas chromatography-mass spectroscopy analysis. Furthermore, we studied the larvicidal and oviposition deterrent activity of the Z. cernuum EO on six mosquito species, the malaria vectors Anopheles stephensi and Anopheles subpictus; the dengue and Zika virus vectors Aedes aegypti and Aedes albopictus; the filariasis, West Nile virus, and St. Louis encephalitis vector Culex quinquefasciatus; and the Japanese encephalitis vector C. tritaeniorhynchus. In addition, to assess the biosafety of Z. cernuum EO-based treatments in the aquatic environment, we investigated the toxicity of Z. cernuum EO on four non-target enemies of mosquito young instars, the insects Anisops bouvieri and Diplonychus indicus and the fishes Poecilia reticulata and Gambusia affinis.
Materials and methods
Extraction and GC-MS analysis of the Z. cernuum essential oil
Fresh rhizomes of Z. cernuum were collected during May 2016 in the Munnar mountains (India 10° 05′ 21″ N 77° 03′ 35″ E, 1700 m a.s.l.). Four hundred grams of fresh rhizomes of Z. cernuum was hydrodistillated for 3 h using a modified Clevenger-type apparatus; then, the Z. cernuum EO was dried over anhydrous sodium sulfate and stored in the dark at 5 °C. GC and GC-MS analyses were carried out as recently described by Govindarajan and Benelli (2016a). The constituents of the Z. cernuum EO were identified by comparison of their mass spectra and retention indices (Table 1) with the ones indexed in the Wiley library, as well as those available in the literature (Adams 2007).
Larvicidal and oviposition deterrence assays
The six mosquito species were reared as described by Govindarajan and Benelli (2016a). Early third instar larvae and adults were used to evaluate the larvicidal potential and oviposition deterrence, respectively. The larvicidal activity of the Z. cernuum EO was studied following the method by WHO (2005). Various doses of the Z. cernuum EO were dissolved in 1 ml dimethyl sulfoxide (DMSO) and then diluted in 249 ml of filtered tap water. Control was 1 ml of DMSO diluted in 249 ml of water. Within each replicate, 20 early third instar larvae were tested (WHO 2005); n = 5 per each dose.
In oviposition deterrent experiments, Z. cernuum EO was evaluated at a dose range of 40–250 μg/ml in DMSO. DMSO diluted in water served as a control. We followed the method by Xue et al. (2001). Twenty gravid females (5–7 days old) of each mosquito species were released in the bioassay cage (60 × 60 × 45 cm). After 24 h, the number of eggs laid in treated and control bowls was counted using a stereomicroscope (Olympus, Japan).
Toxicity on non-target organisms
Toxicity on the four predators was assessed following Sivagnaname and Kalyanasundaram (2004) with minor modifications by Govindarajan and Benelli (2016a). The Z. cernuum EO was evaluated at doses of even 50 × LC50 values calculated for mosquito larvae studied in the paragraph above, 10 replicates per each dose, plus 4 control replicates (where no EO was added to the water). The mortality of non-target species was assessed 48 h post-treatment.
Data analysis
All data were analyzed using the SPSS Statistical Software Package version 16.0. LC50 and LC90 were estimated following the method by Finney (1971). The oviposition activity index (OAI) was calculated as indicated by Kramer and Mulla (1979):
Effective repellency (ER %) due to Z. cernuum EO was estimated following Xue et al. (2001). In non-target assays, the suitability index (SI) was calculated as described by Deo et al. (1988).
Results
GC and GC-MS of Z. cernuum essential oil
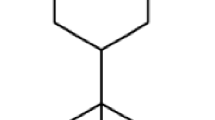
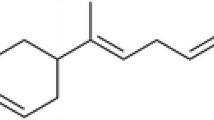
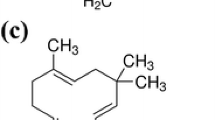
The yield of the Z. cernuum rhizome EO was 1.8 ml/kg of rhizome fresh weight. Table 1 showed a total of 28 compounds representing 96.2% of the Z. cernuum EO. The major constituents of Z. cernuum EO were δ-3-carene, trans-caryophyllene, and α-humulene (Fig. 1). The other 25 compounds ranged from 0.8 to 5.8%.
Larvicidal and oviposition deterrent activity
The Z. cernuum EO showed acute toxicity on third instar larvae of Anopheles stephensi, Aedes aegypti, Culex quinquefasciatus, Anopheles subpictus, Aedes albopictus, and Culex tritaeniorhynchus, with LC50 of 41.34, 44.88, 48.44, 51.42, 55.84, and 60.20 μg/ml, respectively (Table 2). No mortality was detected in the control.
The results obtained from the oviposition deterrence experiments testing Z. cernuum EO on the six mosquito species are reported in Table 3. The mean number of eggs laid in sites treated with the Z. cernuum EO tested at the highest doses (i.e., 200–250 μg/ml) was 44.5, 41.5, and 36.7 eggs per bowl for Anopheles stephensi, Aedes aegypti, and Culex quinquefasciatus, respectively, and 56.2, 52.8, and 47.5 eggs per bowl for Anopheles subpictus, Aedes albopictus, and Culex tritaeniorhynchus, respectively. Significant differences (P < 0.05; P < 0.01) were detected comparing these values to the respective controls (Table 3). The range of OAI achieved by Z. cernuum EO tested against the six mosquito vectors at 200 and 250 μg/ml ranged from −0.79 to −0.84 (Table 3).
Toxicity on non-target predators
Z. cernuum EO toxicity on A. bouvieri, D. indicus, P. reticulata, and G. affinis was reported in Table 4 . LC50 values were 3119, 5273, 10,363, and 11,233 μg/ml, respectively. PSF indicated that the Z. cernuum EO showed scarce toxicity on A. bouvieri, D. indicus, P. reticulata, and G. affinis (Table 5). Survival and swimming activity of the non-target water bugs and fishes were not affected by the exposure to Z. cernuum EO LC50 and LC90 estimated on the six mosquito species.
Discussion
GC and GC-MS of Z. cernuum essential oil
Our results showed that 28 compounds were identified in the Z. cernuum EO, with δ-3-carene, trans-caryophyllene, and α-humulene as main components. This highlighted a quite surprising composition, if compared to EOs extracted from other Zingiber species. Indeed, several studies have been conducted on the EOs from other Zingiber species, such as Z. officinale (Foko et al. 2011), Z. cassumunar (Jantan et al. 2003), Z. zerumbet (Tewtrakul et al. 1998), Z. piperitum (Kamsuk et al. 2006), Z. limonella (Somanabandhu et al. 1992), Z. armatum (Tiwary et al. 2007), and Z. monophyllum (Pavela and Govindarajan 2017). Campbell et al. (2011) pointed out that several mono- and sesquiterpenes, including trans-caryophyllene, α-terpineol, β-pinene, germacrene-D, limonene, and α-zingiberene, present in the EOs evoke responses in Aedes aegypti antennae. Sesquiterpenes α-curcumene, β-sesquiphellandrene, zingiberene, and β-bisabolene from Z. officinale EO (Campbell 2009), as well as trans-caryophyllene and Ocimum forskolei (Dekker et al. 2011), also induced antennal responses by the antennae of A. aegypti females.
Larvicidal and oviposition deterrent potential
Essential oils from plants can represent an alternative source of eco-friendly and biodegradable mosquito ovicides (Benelli 2015b), larvicides (Pavela 2015) and adult repellents (Barnard 1999). A growing number of researches concentrated on the effectiveness of plant EOs against mosquito young instars, with special reference to larvae (Sukumar et al. 1991; Benelli 2015b; Pavela 2015). According to Tawatsin et al. (2001), the bioactivity of EOs depends on various factors, including the plant species and cultivar, the growing conditions, the harvesting time, the storage conditions, and the extraction method (see also Pavela and Benelli 2016).
In our assays, the EO extracted from the rhizome of Z. cernuum showed high toxicity against third instar larvae of Anopheles stephensi (LC50 = 41.34 μg/ml), Aedes aegypti (LC50 = 44.88 μg/ml), Culex quinquefasciatus (LC50 = 48.44 μg/ml), Anopheles subpictus (LC50 = 51.42 μg/ml), Aedes albopictus (LC50 = 55.84 μg/ml), and Culex tritaeniorhynchus (LC50 = 60.20 μg/ml). Concerning the bioactivity of other EOs and extracts from the Zingiberaceae family, Rahuman et al. (2008) evaluated the larvicidal activity of 4-gingerol from Z. officinalis, against A. aegypti (4.25 ppm) and C. quinquefasciatus (5.52 ppm). Sutthanont et al. (2010) reported that Z. zerumbet and Kaempferia galanga EOs are effective on A. aegypti, with LC50 of 48.88 and 53.64 ppm, respectively. Tewtrakul et al. (1998) showed the toxicity of Z. zerumbet ethanol extract on anopheline larvae, with LD50 of 18.9 μg/ml. The Z. cassumunar EO is effective against A. aegypti larvae (LT50 = 1.4 min) (Jantan et al. 2003). Pitasawata et al. (2007) and Champakaew et al. (2007) noted that the C. zedoaria EO showed larvicidal activity on A. aegypti, with LC50 of 33.45 ppm. Z . zerumbet EO also showed larvicidal toxicity on A. aegypti and A. nuneztovari, with LC50 of 89.8 and 62.8 μg/ml, respectively (Tewtrakul et al. 1998). Other studies reported that Z. officinale EO tested at 20 mg/ml and 700 μl/ml effectively repelled stored product pests, such as Sitophilus zeamais and Prostephanus truncatus adults (Ogbonna et al. 2014).
The Z. cernuum EO tested in this study was mainly composed of δ-3-carene, trans-caryophyllene, and α-humulene. Recently, several effective mosquitocidal molecules have been identified in the EOs of other Indian plants. For example, Govindarajan and Benelli (2016a) investigated the toxicity of α-humulene and β-elemene from Syzygium zeylanicum EO on A. albopictus (LC50 = 6.86 and 11.15 μg/ml), C. tritaeniorhynchus (LC50 = 7.39 and 12.05 μg/ml), and A. subpictus (LC50 values were 6.19 and 10.26 μg/ml). Further research focusing on potential synergic larvicidal effects occurring among the abovementioned molecules is ongoing.
Concerning the oviposition deterrent potential, we observed that the range of OAI achieved by the Z. cernuum EO tested at 200 and 250 μg/ml compared with controls ranged from −0.7 to −0.8. Recently, a growing number of studies focused on the oviposition deterrent activity of plant extracts and EOs against mosquito vectors of economic importance (Elango et al. 2009). However, few of them investigated the oviposition deterrent potential of Zingiber species. Coria et al. (2008) reported 100% oviposition deterrent effect obtained with Melia azedarach leaf extract tested at 1 g/l concentration against Aedes aegypti. Prajapati et al. (2005) noted that the bark EO of Cinnamomum zeylanicum reduced the oviposition rates of A. aegypti to 50% when tested at 33.5 ppm. Autran et al. (2009) recorded the oviposition deterrent effect of EO obtained from leaves, inflorescences, and stems of Piper marginatum; the EOs from leaves and stems of P. marginatum exhibited oviposition deterrent effect on A. aegypti females at 50 and 100 ppm concentrations and the number of eggs laid was significantly lower (<50%), if compared to control.
Biotoxicity on mosquito predators
It is worthy to note that the toxicity of Z. cernuum EO on the mosquito predators A. bouvieri, D. indicus, G. affinis, and P. reticulata was very low, with LC50 values always higher than 3000 μg/ml. EOs have been recently recognized as novel and reliable biopesticides, which do not induce resistance and have few toxic effects on human health and non-target species. For example, scarce toxicity of P. kesiya EO on A. bouvieri, D. indicus, and G. affinis was noted, with LC50 from 4135 to 8390 mg/ml, and in agreement with the present results, G. affinis has been found less susceptible to EO, if compared to A. bouvieri and D. indicus (Govindarajan et al. 2016a). S. zeylanicum EO tested on G. affinis showed LC50 = 20,374.26 μg/ml (Govindarajan and Benelli 2016a). Moreover, Heracleum sprengelianum EO, lavandulyl acetate and bicyclogermacrene, tested on A. bouvieri, D. indicus, and G. affinis, led to LC50 ranging from 414 to 4219 μg/ml (Govindarajan and Benelli 2016b). Taken together, the data reported above underline the environmental-friendly nature of botanicals from selected Asian plant species, which can be further considered for as larvicides and oviposition deterrents in urban and peri-urban areas.
Conclusions
Overall, the present research sheds light on the chemical composition of the EO of Z. cernuum, as well as on its larvicidal and oviposition deterrent activity on six important mosquito species. Notably, really limited non-target effects of Z. cernuum EO were found on four important mosquito predators. Therefore, the results from this study supported our hypothesis to consider the Z. cernuum EO for the development of effective and eco-friendly larvicides and oviposition deterrents effective against a broad range of mosquito vector species.
References
Adams RP (2007) 4th Ed. Carol Stream, Illinois: Allured Publishing Corporation; Identification of essential oil components by gas chromatography/mass spectroscopy
Almeida RN, Fátima Agra M, Negromonte Souto Major F, Sousa DP (2011) Essential oils and their constituents: anticonvulsant activity. Molecules 16:2726–2742
Ansari MA, Mittal PK, Razdan RK, Sreehari U (2005) Larvicidal and mosquito repellent activities of pine (Pinus longifolia, family: Pinaceae) oil. J Vect Borne Dis 42:95–99
Ansari MA, Razdan RK, Tandon M, Vasudevan P (2000b) Larvicidal and repellent actions of Dalbergia sisoo Roxb. (F. Leguminosae) oil against mosquitoes. Bioresour Technol 73:207–211
Ansari MA, Vasudevan P, Tandon M, Razdan RK (2000a) Larvicidal and mosquito repellent action of peppermint (Mentha piperita) oil. Bioresour Technol 71:267–271
Araujo ECC, Silveira ER, Lima MAS, Neto MA, Andrade I, Lima MAA (2003) Insecticidal activity and chemical composition of volatile oils from Hyptis martiusii Benth. J Agric Food Chem 51:3760–3762
Astani A, Reichling J, Schnitzler P (2010) Comparative study on the antiviral activity of selected monoterpenes derived from essential oils. Phytother Res 24:673–679
Autran ES, Neves IA, Da Silva CS, Santos GK, Da Câmara CA, Navarro DM (2009) Chemical composition, oviposition deterrent and larvicidal activities against Aedes aegypti of essential oils from Piper marginatum Jacq. (Piperaceae). Bioresour Technol 100:2284–2288
Barnard DR (1999) Repellency of essential oils to mosquitoes (Diptera: Culicidae). J Med Entomol 36:625–629
Bendjeddou D, Lalaoui K, Satta D (2003) Immunostimulating activity of the hot water-soluble poly-saccharide extracts of Anacyclus pyrethrum, Alpinia galanga and Citrullus colocythis. J Ethnophamacol 88:155–160
Benelli G (2015a) Research in mosquito control: current challenges for a brighter future. Parasitol Res 114:2801–2805
Benelli G (2015) Plant-borne ovicides in the fight against mosquito vectors of medical and veterinary importance: a systematic review. Parasitol Res 114(9):3201–3212
Benelli G, Mehlhorn H (2016) Declining malaria, rising dengue and Zika virus: insights for mosquito vector control. Parasitol Res 115:1747–1754
Bhuiyan MNI, Chowdhury JU, Begum J (2009) Chemical investigation of the leaf and rhizome essential oils of Zingiber zerumbet (L.) Smith. Bangladesh. Aust J Pharm 4:9–12
Burkill IH (1966) A dictionary of the economic products of the Malay Peninsula. Vol I: A-H, Vol II: I-Z; Art Printing Works: Kuala Lumpur, 2402 p.
Campbell C, Gries R, Gries G (2011) Forty-two compounds in eleven essential oils elicit antennal responses from Aedes aegypti. Entomol Exp Appl 138:21–32
Campbell CJ (2009) Analyses of essential and edible oils, and constituents therein, as candidate repellents for the yellow fever mosquito Aedes aegypti L. (Diptera: Culicidae). Simon Fraser University
Carvalho AFU, Melo VMM, Craveiro AA, Machado MIL, Bantim MB, Rabelo EF (2003) Larvicidal activity of the essential oil from Lippia sidoides Cham. against Aedes aegepti L. Mem Inst Oswaldo Cruz 98:569–571
Cavalcanti ESBC, Morais SM, Lima MAA, Santana EWP (2004) Larvicidal activity of the essential oil from Brazilian plants against Aedes aegypti. Mem Inst Oswaldo Cruz 99:541–544
Champakaew D, Choochote W, Pongpaibul Y, Chaithong U, Jitpakdi A, Tuetun B, Pitasawat B (2007) Larvicidal efficacy and biological stability of a botanical natural product, zedoary oil-impregnated sand granules, against Aedes aegypti (Diptera: Culicidae). Parasitol Res 100:729–737
Cheng SS, Chang HT, Chang ST, Tsai KH, Chen WJ (2003) Bioactivity of selected plant essential oils against the yellow fever mosquito Aedes aegypti larvae. Bioresour Technol 89:99–102
Cheng SS, Chang HT, Lin CY, Chen PS, Huang CG, Chen WJ, Chang ST (2009c) Insecticidal activities of leaf and twig essential oils from Clausena excavata against Aedes aegypti and Aedes albopictus larvae. Pest Manag Sci 65:339–343
Cheng SS, Chua MT, Chang EH, Huang CG, Chen WJ, Chang ST (2009a) Variations in insecticidal activity and chemical composition of leaf essential oils from Cryptomeria japonica at different ages. Bioresour Technol 100:465–470
Cheng SS, Huang CG, Chen WJ, Kuo YH, Chang ST (2008) Larvicidal activity of tectoquinone isolation from red heartwood-type Cryptomeria japonica against two mosquito species. Bioresour Technol 99:3617–3622
Cheng SS, Liu JY, Huang CG, Hsui YR, Chen WJ, Chang ST (2009b) Insecticidal activities of leaf essential oils from Cinnamomum osmophloeum against three mosquito species. Bioresour Technol 100:457–464
Cheng SS, Liu JY, Tsai KH, Chen WJ, Chang ST (2004) Chemical composition and mosquito larvicidal activity of essential oils from leaves of different Cinnamomum osmophloeum provenances. J Agric Food Chem 52:4395–4400
Chericoni S, Flamini G, Campeol E, Cioni PL, Morelli I (2004) GC-MS analysis of the essential oil from the aerial parts of Artemisia verlotiorum: variability during the year. Biochem Sys Ecol 32:423–429
Coria C, Almiron W, Valladares G, Carpinella C, Ludueña F, Defago M (2008) Larvicide and oviposition deterrent effects of fruit and leaf extracts from Melia azedarach L. on Aedes aegypti (L.) (Diptera: Culicidae). Bioresour Technol 99:3066–3070
Dargahi L, Razavi-Azarkhiavi K, Ramezani M, Reza Abaee M, Behravan J (2014) Insecticidal activity of the essential oil of Thymus transcaspicus against Anopheles stephensi. Asian Pac J Trop Biomed 4:589–491
Dekker T, Ignell R, Ghebru M, Glinwood R, Hopkins R (2011) Identification of mosquito repellent odours from Ocimum forskolei. Parasit Vectors 4:1–7
Deo PG, Hasan SB, Majumdar SK (1988) Toxicity and suitability of some insecticides for household use. Int Pest Control 30:118–129
Dharmagadda VSS, Naik SN, Mittal PK, Vasudevan P (2005) Larvicidal activity of Tagetus patula essential oil against three mosquito species. Bioresour Technol 96:1235–1240
Do Nascimento JC, David JM, Barbosa LCA, De Paula VF, Demuner AJ, David JP, Conserva LM, Ferreira JRJC, Guimaraes EF (2013) Larvicidal activities and chemical composition of essential oils from Piper klotzschianum (Kunth) C. DC. (Piperaceae). Pest Manag Sci 69:1267–1271
El-Akhal F, El Ouali LA, Ez Zoubi Y, Greche H, Guemmouh R (2014) Chemical composition and larvicidal activity of essential oil of Origanum majorana (Lamiaceae) cultivated in Morocco against Culex pipiens (Diptera: Culicidae). Asian Pac J Trop Biomed 4:746–750
El-Akhal F, Greche H, Ouazzani CF, Guemmouh R, El Ouali LA (2015) Chemical composition and larvicidal activity of Culex pipiens essential oil of Thymus vulgaris grown in Morocco. J Mater Environ Sci 1:214–219
Elango G, Bagavan A, Kamaraj C, Abduz Zahir A, Abdul Rahuman AC (2009) Oviposition-deterrent, ovicidal, and repellent activities of indigenous plant extracts against Anopheles subpictus Grassi (Diptera: Culicidae). Parasitol Res 105:1567–1576
Finney DJ (1971) Probit analysis. Cambridge University Press, London, pp 68–72
Foko GA, Tamesse JL, Fekam F (2011) Adulticidal effects of essential oils extracts from Capsicum annum (Solanaceae), Piper nigrum (Piperaceae) and Zingiber officinales (Zingiberaceae) on Anopheles gambiae (Diptere-Culicidade), vector of malaria. J Entomol 8:152–163
Govindarajan M, Benelli G (2016a) α-Humulene and β-elemene from Syzygium zeylanicum (Myrtaceae) essential oil: highly effective and eco-friendly larvicides against Anopheles subpictus, Aedes albopictus and Culex tritaeniorhynchus (Diptera: Culicidae). Parasitol Res 115:2771–2778
Govindarajan M, Benelli G (2016b) Eco-friendly larvicides from Indian plants: effectiveness of lavandulyl acetate and bicyclogermacrene on malaria, dengue and Japanese encephalitis mosquito vectors. Ecotoxicol Environ Saf 133:395–402
Govindarajan M, Rajeswary M, Arivoli S, Samuel T, Benelli G (2016b) Larvicidal and repellent potential of Zingiber nimmonii (J. Graham) Dalzell (Zingiberaceae) essential oil: an eco-friendly tool against malaria, dengue and lymphatic filariasis mosquito vectors? Parasitol Res 115:1807–1816
Govindarajan M, Rajeswary M, Benelli G (2016a) Chemical composition, toxicity and effects on non-target organisms of Pinus kesiya essential oil: an eco-friendly larvicide against mosquito vectors. Ecotoxicol Environ Saf 129:85–90
Jain N, Srivastava SK, Aggarwal LKK, Ramesh S, Kumar S (2001) Essential oil composition of Zanthoxylum alatum seeds from northern India. Flavour Frag J 16:408–410
Jantan I, Ping WO, Visuvalingam SD, Ahmad NW (2003) Larvicidal activity of the essential oils and methanol extracts of Malaysian plants on Aedes aegypti. Pharm Biol 41:234–246
Jirovetz L, Buchbauer G, Pottachola M, Kalathil N (2003) Analysis of the essential oils of the leaves, stems, rhizomes and roots of the medicinal plant Alpinia galanga from southern India. Acta Pharma 53:73–81
Kamaraj C, Rahuman A, Mahapatra A, Bagavan A, Elango G (2010) Insecticidal and larvicidal activities of medicinal plant extracts against mosquitoes. Parasitol Res 107:1337–1349
Kamsuk K, Choochote W, Chaithong U, Jitpakdi A, Tippawangkosol P, Riyong D, Pitasawat B (2006) Effectiveness of Zanthoxylum piperitum-derived essential oil as an alternative repellent under laboratory and field applications. Parasitol Res 100:339–345
Kasarkar AR, Kulkarni DK (2011) Phytochemical investigation of the genus Zingiber from the family Zingiberaceae. Int J Pharm Res Allied Sci 2:648–649
Kiran SR, Bhavani K, Devi PS, Rao BRR, Reddy KJ (2006) Composition and larvicidal activity of leaves and stem essential oils of Chloroxylon swietenia DC against Aedes aegypti and Anopheles stephensi. Bioresour Technol 97:2481–2484
Koul O, Walia S, Dhaliwal GS (2008) Essential oils as green pesticides: potential and constraints. Biopest Int 4:63–84
Kramer WL, Mulla MS (1979) Oviposition attractants and repellents of mosquitoes: oviposition responses of Culex mosquitoes to organic infusions. Environ Entomol 8:1111–1114
Liu XC, Dong HW, Zhou L, Du SS, Liu ZL (2013) Essential oil composition and larvicidal activity of Toddalia asiatica roots against the mosquito Aedes albopictus (Diptera: Culicidae). Parasitol Res 112:1197–1203
Liu XC, Liu Q, Chen XB, Zhou L, Liu ZL (2015) Larvicidal activity of the essential oil from Tetradium glabrifolium fruits and its constituents against Aedes albopictus. Pest Manag Sci 71:1582–1586
Liu ZL, He Q, Chu SS, Wang CF, Du SS, Deng ZW (2012) Essential oil composition and larvicidal activity of Saussurea lappa roots against the mosquito Aedes albopictus (Diptera: Culicidae). Parasitol Res 110:2125–2130
Lucia A, Audino GA, Seccacini E, Licastro S, Zerba E, Masuh H (2007) Larvicidal effect of Eucalyptus grandis essential oil and turpentine and their major components on Aedes aegypti larvae. J Am Mosq Control Assoc 3:299–303
Menon AN (2007) Chemical composition of the essential oils from leaves and roots of ginger (Zingiber officinale) from Kerela. J Med Aromatic Pl Sci 29:78–80
Naqqash MN, Gökçe A, Bakhsh A, Salim M (2016) Insecticide resistance and its molecular basis in urban insect pests. Parasitol Res 115:1363–1373
Negi PS, Jayaprakasha GK, Jagan MRL, Sarariah KK (1999) Antibacterial activity of turmeric oil: a byproduct from Curcumin manufacture. J Agric Food Chem 47:4297–4300
Nguefack J, Leth V, Amvam HP, Mathur BS (2004) Evaluation of five essential oil from aromatic plant of Cameroon for controlling food spoilage and mycotoxin producing fungi. Int J Food Microbiol 94:329–994
Ogbonna CU, Eziah VY, Owusu EO (2014) Bioefficacy of Zingiber officinale against Prostephanus truncates Horn (Coleoptera: Bostrichidae) infesting maize. J Biopest 7:177–185
Padmakumari KP, Sreekumar MM, Sankarikutty B (2009) Composition of volatile oil of ginger (Zingiber officinale Roscoe) varieties from India. Indian Perfum 53:16–20
Park YU, Koo HN, Kim GH (2012) Chemical composition, larvicidal action, and adult repellency of Thymus magnus against Aedes albopictus. J Am Mosq Control Assoc 28:192–198
Pathak AK, Dixit VK (1988) Insecticidal and insect repellent activity of essential oils of Tridax procumbens and Cyathocline lyrata. Fitoterapia 59:211–214
Patricia FL, Mera EM, Daisy NS, Joao EC, Maricia OM, Angela AM (2003) Functional properties of spice extracts obtained via supercritical fluid extraction. J Agric Food Chem 51:2520–2525
Pavela R (2008a) Insecticidal properties of several essential oils on the house fly (Musca domestica L.) Phytother Res 22:274–278
Pavela R (2008b) Larvicidal effects of various Euro-Asiatic plants against Culex quinquefasciatus Say. larvae (Diptera: Culicidae). Parasitol Res 102:555–559
Pavela R (2015) Essential oils for the development of eco-friendly mosquito larvicides: a review. Ind Crop Prod 76:174–187
Pavela R, Benelli G (2016) Essential oils as ecofriendly biopesticides? Challenges and constraints. Tr Plant Sci 21:1000–1007
Pavela R, Govindarajan M (2017) The essential oil from Zanthoxylum monophyllum a potential mosquito larvicide with low toxicity to the non-target fish Gambusia affinis. J Pest Sci 90:369–378
Pitasawata B, Champakaewa D, Choochotea W, Jitpakdia A, Chaithonga U, Kanjanapothib D (2007) Aromatic plant-derived essential oil: an alternative larvicide for mosquito control. Fitoterapia 78:205–210
Prajapati V, Tripathi AK, Aggarwal KK, Khanuja SP (2005) Insecticidal, repellent and oviposition-deterrent activity of selected essential oils against Anopheles stephensi, Aedes aegypti and Culex quinquefasciatus. Bioresour Technol 96:1749–1757
Pushpanathan T, Jebanesan A, Govindarajan M (2008) The essential oil of Zingiber officinalis Linn (Zingiberaceae) as a mosquito larvicidal and repellent agent against the filarial vector Culex quinquefasciatus Say (Diptera: Culicidae). Parasitol Res 102:1289–1291
Rahuman AA, Gopalakrishnan G, Venkatesan P, Geetha K (2008) Isolation and identification of mosquito larvicidal compound from Abutilon indicum (Linn) Sweet. Parasitol Res 102:981–988
Rana VS, Verdeguer M, Blazquez MA (2008) A comparative study of rhizome essential oils of three Zingiber species from Manipur. Indian Perfum 52:17–21
Rattan RS (2010) Mechanism of action of insecticidal secondary metabolites of plant origin. Crop Prot 29:913–920
Sanjay J (2015) Phytochemical screening, antioxidant activity, multi-elemental and flavonoid analysis of rhizome extracts of Zingiber cernuum Dalzell. J Acad Indus Res 4:56–64
Santos RP, Nunes EP, Nascimento RF, Santiago GMP, Menezes GHA, Silveira ER, Pessoa ODL (2006) Chemical composition and larvicidal activity of the essential oils of Cordia leucomalloides and Cordia curassavica from the northeast of Brazil. J Braz Chem Soc 17:1027–1030
Scartezzini P, Speroni E (2000) Review on some plants of Indian traditional medicine with antioxidant activity. J Ethnopharmacol 171:23–43
Senthilkumar A, Kannathasan K, Venkatesalu V (2008) Chemical constituents and larvicidal property of the essential oil of Blumea mollis (D. Don) Merr. against Culex quinquefasciatus. Parasitol Res 103:959–962
Sharma RN, Gupta AS, Patwardhan SA, Hebbal-ker DS, Tare V, Bhonde SB (1992) Bioactivity of Lamiaceae plants against insects. Indian J Exp Biol 30:244–246
Sivagnaname N, Kalyanasundaram M (2004) Laboratory evaluation of methanolic extract of Atlantia monophylla (family: Rutaceae) against immature stages of mosquitoes and non-target organisms. Mem Inst Oswaldo Cruz 99:115–118
Somanabandhu A, Ruangrungsi N, Lange GL, Organ MG (1992) Constituents of the stembark of Zanthoxylum limonella. J Sci Soc Thailand 18:181–185
Srivastava AK, Srivastava SK, Shah NC (2000) Essential oil composition of Zingiber zerumbet (L.) Sm. from India. J Essent Oil Res 12:595–597
Sukumar K, Perich MJ, Boobar LR (1991) Botanical derivatives in mosquito control: a review. J Am Mosq Control Assoc 7:210–237
Sutthanont N, Choochote W, Tuetun B, Junkum A, Jitpakdi A, Chaithong U, Riyong D, Pitasawat B (2010) Chemical composition and larvicidal activity of edible plant-derived essential oils against the pyrethroid-susceptible and -resistant strains of Aedes aegypti (Diptera: Culicidae). J Vector Ecol 35:106–115
Tawastin A, Wratten SD, Scott RR, Thavara U, Techadamrongsin Y (2001) Repellency of volatile oils from plants against three mosquito vectors. J Vector Ecol 26:76–82
Tewtrakul S, Itchayapruk J, Chaitongruk P (1998) Mosquito larvicidal activity of Zingiber zerumbet Smith rhizomes. Songklanakarin J Sci Technol 20:183–187
Thomas TG, Rao S, Lal S (2004) Mosquito larvicidal properties of essential oil of an indigenous plant, Ipomoea cairica Linn. Jpn J Infect Dis 57:176–177
Tiwary M, Naik SN, Tewary DK, Mittal PK, Yadav S (2007) Chemical composition and larvicidal activities of the essential oil of Zanthoxylum armatum DC (Rutaceae) against three mosquito vectors. J Vect Borne Dis 44:198–204
Traboulsi AF, Taoubi K, El-Haj S, Bessiere JM, Rammal S (2002) Insecticidal properties of essential plant oils against the mosquito Culex pipiens molestus (Diptera: Culicidae). Pest Manag Sci 58:491–495
Urzúa A, Santander R, Echeverría J, Cabezas N, Palacios SM, Rossi Y (2010) Insecticide properties of the essential oils from Haplopappus foliosus and Bahia ambrosoides against the house fly, Musca domestica L. J Chil Chem Soc 55:392–395
Ward W, Benelli G (2017) Avian and simian malaria: do they have a cancer connection?. Parasitol Res 116(3):839–845
World Health Organization (2005) Guidelines for laboratory and field testing of mosquito larvicides. Communicable disease control, prevention and eradication, WHO pesticide evaluation scheme. WHO, Geneva, WHO/CDS/WHOPES/GCDPP/1.3
Xue RD, Barnard DR, Ali A (2001) Laboratory and field evaluation of insect repellents as oviposition deterrents against the mosquito Aedes albopictus. Med Vet Entomol 15:126–131
Youko S, Kiku K, Akio K (2000) Isolation of novel glucosides related to gingerdiol from ginger and their antioxidative activities. J Agric Food Chem 48:373–377
Acknowledgements
The authors extend their appreciation to the Deanship of Scientific Research at King Saud University for funding this work through research group No RG-1438-074.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Rajeswary, M., Govindarajan, M., Alharbi, N.S. et al. Zingiber cernuum (Zingiberaceae) essential oil as effective larvicide and oviposition deterrent on six mosquito vectors, with little non-target toxicity on four aquatic mosquito predators. Environ Sci Pollut Res 25, 10307–10316 (2018). https://doi.org/10.1007/s11356-017-9093-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-9093-3