Abstract
This work sought to ascertain survival and possible changes in levels of glycogen, triglycerides, total lipids, cholesterol, protein, and lipid peroxidation in gills, liver, and muscle of bullfrog tadpoles (Lithobates catesbeianus) exposed to low concentrations of atrazine (2.5 μg L−1), glyphosate (18 μg L−1), and quinclorac (0.025 μg L−1) at laboratorial conditions. Tadpoles showed a reduction of glycogen and triglyceride in all organs and an increase in lipid peroxidation (LPO) compared with control animals. Total lipid in gills and muscle increased in exposure to atrazine, and gills alone in exposure to glyphosate, but decreased in gills, liver, and muscle after quinclorac. Cholesterol increased in gills and liver after atrazine, in gills and muscle after glyphosate, and decreased in liver after quinclorac. Total protein in gills decreased after exposure to all herbicides, increased in muscle after atrazine, and in liver and muscle after quinclorac. These findings show that at concentrations of these herbicides tested can lead to an increase in energy expenditure to maintain homeostasis and survival of these animals despite the increase in lipid peroxidation levels in all organs analyzed. Responses observed can be one of the factors responsible for the decline in the number of amphibians around the world.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The way humans use the world’s natural resources has led to significant impacts on other species that inhabit the planet. One example of this phenomenon is the decline in the number of amphibians over the last few decades, an extinction that has no precedent in any animal class in this period and which may be the result of an isolated action or interaction of different factors, such as habitat loss, ultraviolet radiation, global warming, diseases, over-harvesting, and/or the introduction of agrochemicals, especially pesticides—even at low levels—into the environment (Allran and Karasov 2000; Blaustein et al. 2003; Boone et al. 2005; David and Kartheek 2015; Davidson 2004; Gascon et al. 2005; Relyea 2003; Sayim 2008).
Approximately 1 % of agrochemicals used in the field reach their specific targets. The remaining 99 % can move through the different environmental compartments and may have an indirect effect on non-target organisms exposed to contaminants (Belluck et al. 1991).
Amphibians are among the animals that may be indirectly exposed to these agrochemicals, and the exposure may account for the great amphibian mortality that has been observed in recent years. Tadpoles appear to be more sensitive than adults, which is consistent with the greater fragility of these animals in the larval stage (Blaustein et al. 2003; David and Kartheek 2015; Johansson et al. 2006; Murphy et al. 2000; Sayim 2008; Wang et al. 2001).
The environmental changes induced by the use of agricultural chemicals can interfere with physiological and biochemical parameters in non-target aquatic organisms, affecting functions such as growth, development, and reproduction (Upasani and Balaraman 2001; Venkataramana et al. 2006). Nwani et al. (2010) and Roy and Hänninen (1993) reported that the indiscriminate use of herbicides may have harmful effects on aquatic organisms, possibly leading to sublethal effects, such as biochemical and metabolic changes in the tissues of exposed animals.
According to Oba et al. (2009), some animals’ responses on exposure to chemicals, such as pesticides, can include a variety of metabolic changes: mobilization of energy substrates, as expressed by depleted glycogen stores; lipolysis; inhibition of protein synthesis; increased muscle protein catabolism; and changes in the levels of fatty acids and cholesterol.
Dornelles and Oliveira (2014), working with bullfrog tadpoles exposed to the same pesticides as in this study, reported changes in the biochemical composition (glycogen, proteins, lipids, and triglycerides) of the animals. This response seems to be related to a depletion of energy reserves and increase in energy expenditure, possibly to detoxify and allow the animals to survive environmental change. A similar response pattern was observed by other authors in different species (El-Banna et al. 2009; Ganeshwade 2012; Moyes and Schulte 2010; Salbego et al. 2010; Sounderraj et al. 2011; Vutukuru 2005).
Different animals when exposed to pesticides have had their levels of lipid peroxidation (LPO) increased in different organs, which may lead to a chemical damage and possible cell death if the antioxidant and defense system were not enough to counteract this damage (Al-Othman et al. 2011; Champe and Harvey 2006; Dornelles and Oliveira 2014; El-Banna et al. 2009; Patil et al. 2009; Uchendu et al. 2012). The LPO is among the best predictors of systemic level of damage induced by reactive oxygen species (ROS) (Sayeed et al. 2003) and is one of the molecular mechanisms involved in the toxicity of pesticides to bodies (Kehrer 1993; Kavitha and Venkateswara Rao 2007).
It is known that the gill epithelium is responsible for breathing, elimination of nitrogen excreted, osmoregulation, and other functions directly related to animal survival; furthermore, the gills are in direct contact with the environment which can act as a site of absorption, magnifying the action of these herbicides when compared to other organs (Bernabò et al. 2008), making this an interesting organ for analysis after the exposure of the animals to these herbicides.
Of the organs widely used for toxicological studies, the liver stands out mainly for having a central role in major functions of the organism, being responsible for the biotransformation and the detoxification of exogenous substances, with its capacity to convert hydrophobic substances into water-soluble products that can be secreted readily from the body. Furthermore, the liver is a metabolically active organ responsible for many vital functions, such as bile production and excretion; excretion of bilirubin, cholesterol, hormones, and drugs; metabolism of fats, proteins, and carbohydrates; storage of glycogen, vitamins, and minerals; synthesis of plasma proteins, such as albumin, and clotting factors; and uptake of amino acids, lipids, carbohydrates, and vitamins and their subsequent storage, metabolic conversion, and release into the blood and bile (Guillouzo 1998; Vickers 1994). These important activities and the exposure to different substances make the liver one of the more susceptible organs to injury.
The muscle tissue is composed mainly of proteins and is responsible for providing the contractile force to movement (Hill et al. 2012). Moyes and Schulte (2010) state that in muscle, ATP is produced by three major mechanisms: by high-energy transfer from creatine phosphate to ADP, by glycolysis, and by oxidative phosphorylation, characterized as a low metabolic rate process.
Since our experimental model has generalist eating habits and voracious predatory behavior, analyses of the liver and the muscle provides information about not only the storage of nutrients but also the action of these herbicides in a long period (7 days of exposure) in an organ with a high (liver) and another with a low (muscle) metabolic rate.
According to Massoud et al. (2011), there is a paucity of information on the toxicity of pesticides at sublethal concentrations and sensitive biomarkers related to the metabolism of these animals. Therefore, research on the impact of these contaminants in exposed communities of aquatic animals—even at low concentrations—is justified.
Taking this lack of information into account, and the results obtained in the laboratory with tadpoles exposed to concentrations within the range permitted by law (Dornelles and Oliveira 2014), the present work sought to assess potential changes in biochemical parameters, levels of lipoperoxidation, and survival in bullfrog tadpoles exposed to concentrations below legal limits of atrazine, glyphosate, and quinclorac. Our hypothesis states that these herbicides, even at realistic concentrations found in the natural environment, are able to introduce changes in the energy reserves and oxidative status in different organs in prometamorphic tadpoles.
Material and methods
Chemicals
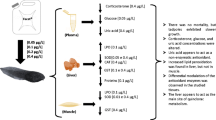
All chemical reagents used were obtained from Merck and Sigma-Aldrich. For toxicity testing, three herbicides in concentrations below legal limits—atrazine (3 μg L−1) (US Environmental Protection Agency 1985), glyphosate (65 μg L−1) (Brasil and Meio Ambiente 2005), and quinclorac—were used. For the latter, we used a concentration below that found in natural bodies of water (Marchezan et al. 2007; Silva et al. 2009), considering that no specific legislation for maximum allowable concentrations in natural water bodies is available for quinclorac (Marchezan et al. 2007). All pesticides were used as commercial formulations: atrazine (A): Primóleo® 400 g L−1 (Syngenta); glyphosate: Roundup Original® 306 g L−1 (Monsanto); and quinclorac: Facet® 500 g kg−1 (Basf). The use of these herbicides is justified by their wide use in different types of crops, not only in Brazil but in several countries (Dornelles and Oliveira, 2014).
The concentrations chosen for the present study were 2.5 μg L−1 for atrazine, 18 μg L−1 for glyphosate, and 0.025 μg L−1 for quinclorac. The pesticides were applied only once, at nominal concentrations according to Dornelles and Oliveira (2014) in respect to the time of permanency in water of these chemicals: between 34.8 and 742 days at pH between 2.9 and 6.0 for atrazine, between 7 and 10 weeks for glyphosate (Montgomery et al. 2008), and between 21 and 30 days for quinclorac (Barceló and Hennion, 2003; Rodrigues and Almeida 1998).
Experimental model
For this study, 76 bullfrog (Lithobates catesbeianus) tadpoles were procured from a frog farm in the municipality of Imbé, state of Rio Grande do Sul, Brazil. All the tadpoles were 3 months old and had no visible limbs (Landis and Yu 2003), and they were in the larval stage 25, according to Gosner (1960).
The animals were transported in air-filled plastic bags to the Conservation Physiology Laboratory at Pontifícia Universidade Católica do Rio Grande do Sul (PUCRS), where they were individually measured, weighed, and randomly divided into groups.
Experimental design
The tadpoles were distributed to a control group (Co 7 = only water, without herbicides) and three exposure groups (A 2.5 = atrazine 2.5 μg L−1; G 18 = glyphosate 18 μg L−1; Q 0.025 = quinclorac 0.025 μg L−1). Each experiment was performed using 8 or 10 tadpoles for treatment, and in duplicate, being 16 the final number of animals in the control group and 20 in the herbicide groups. All aquariums contained 12 L of water each, with constant aeration, a water temperature of around 22 ± 2 °C, pH 6.2 ± 0.3, and a 12-h light/dark cycle. The animals were fed once daily (with approximately 5 % of the early biomass in each aquarium) with the same fish feed used at the frog farm, with a crude protein content of 38 %.
A static exposure system was used in accordance with a Standard Guide for Conducting the Frog Embryo Teratogenesis Assay—Xenopus (FETAX, ASTM 1998). The levels of ammonia in the water were monitored daily by a commercial kit (LabCom) and showed no toxic values (variation between 0 until 0.5). The levels of pH and dissolved oxygen were monitored too (Sanxin SX721 Portable pH/ORP Meter) and remained within the established by FETAX guide (ASTM 1998), with the treatment pH between 6.5 and 9. The conductivity and dissolved oxygen do not have values established by the FETAX guide. Our values of dissolved oxygen remained between 3.5 and below 5 mg L−1 (ASTM 1998).
The total duration of the experiment was 14 days: 7 days for acclimation and 7 days of exposure to herbicides. Herbicides were introduced in the aquariums on the eighth day after the beginning of the experiment—the commercial formulations, both liquid (atrazine and glyphosate) and powder (quinclorac), were diluted in distilled water and added only once to the aquariums at a particular concentration. The time of exposures for 7 days was based on the half-life of atrazine, glyphosate, and quinclorac in water (Solomon et al. 1996; Giesy et al. 2000; Rodrigues and Almeida 1998, respectively). In the control group, the animals remained for seven more days under the same acclimation conditions. At the end of the experiment, all animals were euthanized by the freezing method, and the left and right gills, liver, and muscle were removed by dissection.
All research protocols used in this work were authorized by the Pontifícia Universidade Católica do Rio Grande do Sul Animal Research Ethics Committee with registration number CEUA 11/00250, as set forth in approval letter number 157/11-CEUA, December 2011.
Biochemical analysis
All biochemical analyses of tissue specimens were performed in quadruplicate, by spectrophotometric methods, and the respective results were expressed in milligrams per gram of tissue. Glycogen was extracted by Van Handel’s method (1965) and quantified as glucose after acid hydrolysis (HCl) and neutralization (Na2CO3) (Geary et al. 1981), using a commercial Glucose Oxidase Kit (Labtest). The total protein concentration was determined by the colorimetric biuret method: the sample is added to the reagent kit, vortexed, placed in a bath, cooled, and read in a spectrophotometer. A commercial kit was used (Total Protein Kit, Labtest); lipid, triglyceride, and cholesterol extraction was performed using the chloroform/methanol method (2:1) (Folch et al. 1957). Lipid content was determined by the sulfo-phospho-vanillin reaction (Frings and Dunn, 1970), and triglycerides, by the lipoprotein lipase method, using the commercial Triglycerides GPO-ANA Kit (Bio-Diagnostic). Cholesterol was determined with the Liquiform Kit (Labtest). Measurement of lipid peroxidation was carried out by the TBARS (TBA-reactive substances) method: 150 μL of 10 % trichloroacetic acid (TCA), 50 μL of tissue homogenate, 100 μL of 0.67 % thiobarbituric acid (TBA), and 50 μL of distilled water are added to a test tube (total volume 350 μL). The tube is shaken, incubated at 100 °C for 15 min, and cooled for 10 min. Then, 300 μL of n-butyl alcohol is added to the sample for extraction of the colored product from aqueous solution. Tubes are shaken for 45 s and centrifuged for 10 min at 3000 rpm. The supernatant was added to the spectrophotometer cuvette and read at 535 nm. Results were expressed as nanomoles per milligram of protein.
Statistical analysis
Comparisons between the experimental and control group were made using the Statistical Package for the Social Sciences 12.0 (SPSS) environment, using the Kolmogorov-Smirnov test for normality. The data was compared with control group for Student’s T test to the independent sample. The results were expressed as mean ± standard deviation. The level of significance was set at p < 0.05.
Results
Gills
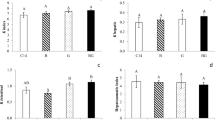
There were significant reductions in glycogen levels in the gills of animals when exposed to atrazine (x̅ = 0.044 mg glycogen/g wet weight), glyphosate (x̅ = 0.027 mg glycogen/g wet weight), and quinclorac (x̅ = 0.032 mg glycogen/g wet weight) as compared with animals in the control group (x̅ = 0.197 mg glycogen/g wet weight) (Fig. 1(A1)). In comparison to the control group (x̅ = 0.166 mg cholesterol/g wet weight), total lipid levels were increased in animals who were exposed to atrazine (x̅ = 0.539 mg total lipids/g wet weight) and glyphosate (x̅ = 0.511 mg total lipids/g wet weight), but reduced in those who were exposed to quinclorac (x̅ = 0.071 mg total lipids/g wet weight) (Fig. 1(A2)). Triglyceride levels decreased in animals exposed to atrazine (x̅ = 0.059 mg triglycerides/g wet weight) and glyphosate (x̅ = 0.134 mg triglycerides/g wet weight) in relation to the control group (x̅ = 0.232 mg triglycerides/g wet weight) (Fig. 1(A3)). Cholesterol levels were increased in those exposed to atrazine (x̅ = 0.238 mg cholesterol/g wet weight) and glyphosate (x̅ = 0.439 mg cholesterol/g wet weight) as compared with control animals (x̅ = 0.095 mg cholesterol/g wet weight) (Fig. 1(A4)). Quinclorac exposure had no significant effect on triglyceride or cholesterol levels. Total protein level decreased in the animals who were exposed to glyphosate (x̅ = 0.093 mg protein/g wet weight) and quinclorac (x̅ = 0.091 mg protein/g wet weight) in relation to the control group (x̅ = 0.155 mg protein/g wet weight), but there was no significant difference in atrazine-exposed animals (Fig. 1(A5)). All herbicides increased LPO levels in relation to the control group (x̅ = 9.025 nmol TBARS/mg protein): levels were x̅ = 57.753 nmol TBARS/mg protein in animals exposed to atrazine, x̅ = 47.601 nmol TBARS/mg protein in those exposed to glyphosate, and x̅ = 57.849 nmol TBARS/mg protein in those exposed to quinclorac (Fig. 1(A6)).
Graphics A1, A2, A3, A4, A5, and A6: Levels of glycogen, total lipids, triglycerides, cholesterol, total proteins, and lipid peroxidation in the gills of Lithobates catesbeianus tadpoles exposed to the herbicides atrazine, glyphosate, and quinclorac (Co 7 control 7 days, A 2.5 atrazine 2.5 μg L−1; G 18 glyphosate 18 μg L−1, Q 0.025 quinclorac 0.025 μg L−1). The results are expressed as the mean ± standard deviation. The asterisk beside the bars indicates a significant difference compared to the control group 7 days, being p < 0.05
Liver
Statistically significant reductions in glycogen and triglyceride levels in the liver were noted in animals that were exposed to atrazine, glyphosate, and quinclorac. Glycogen levels declined in animals exposed to atrazine (x̅ = 0.047 mg glycogen/g wet weight), glyphosate (x̅ = 0.047 mg glycogen/g wet weight), and quinclorac (x̅ = 0.057 mg glycogen/g wet weight) in relation to the control group (x̅ = 0.113 mg glycogen/g wet weight) (Fig. 2(B1)). Total lipid level decreased after exposure to atrazine (x̅ = 0.429 mg total lipids/g wet weight) and quinclorac (x̅ = 0.171 mg total lipids/g wet weight) than to the control group (x̅ = 0.723 mg total lipids/g wet weight) (Fig. 2(B2)), but there was no significant difference in lipid level after glyphosate exposure. Triglyceride levels declined after exposure to atrazine (x̅ = 0.263 mg triglycerides/g wet weight), glyphosate (x̅ = 0.229 mg triglycerides/g wet weight), and quinclorac (x̅ = 0.180 mg triglycerides/g wet weight) in relation to the control group (x̅ = 0.345 mg triglycerides/g wet weight) (Fig. 2(B3)). Atrazine exposure led to an increase in cholesterol levels (x̅ = 0.262 mg cholesterol/g wet weight), but animals exposed to glyphosate (x̅ = 0.032 mg cholesterol/g wet weight) and quinclorac (x̅ = 0.053 mg cholesterol/g wet weight) showed a reduction in cholesterol levels as with controls (x̅ = 0.209 mg cholesterol/g wet weight) (Fig. 2(B4)). Total protein level decreased in the glyphosate group (x̅ = 1.013 mg protein/g wet weight) but increased in the atrazine (x̅ = 1.683 mg protein/g wet weight) and quinclorac (x̅ = 1.935 mg protein/g wet weight) groups in relation to the control group (x̅ = 1.454 mg protein/g wet weight) (Fig. 2(B5)). In the liver, as well as in the gills, all herbicides were associated with an increase in lipid peroxidation levels in relation to the control group (x̅ = 14.212 nmol TBARS/mg protein): x̅ = 22.656 nmol TBARS/mg protein in animals exposed to atrazine, x̅ = 22.419 nmol TBARS/mg protein in those exposed to glyphosate, and x̅ = 23.855 nmol TBARS/mg protein in those exposed to quinclorac (Fig. 2(B6)).
Graphics B1, B2, B3, B4, B5, and B6: Levels of glycogen, total lipids, triglycerides, cholesterol, total proteins, and lipid peroxidation in the liver of Lithobates catesbeianus tadpoles exposed to the herbicides atrazine, glyphosate, and quinclorac (Co 7 control 7 days, A 2.5 atrazine 2.5 μg L−1, G 18 glyphosate 18 μg L−1, Q 0.025 quinclorac 0.025 μg L−1). The results are expressed as the mean ± standard deviation. The asterisk beside the bars indicates a significant difference compared to the control group 7 days, being p < 0.05
Muscle
In muscle tissue, there were statistically significant decreases in glycogen levels on the exposure of glyphosate (x̅ = 0.007 mg glycogen/g wet weight) and quinclorac (x̅ = 0.007 mg glycogen/g wet weight), but there was no significant decrease for atrazine in relation to the control group (x̅ = 0.009 mg glycogen/g wet weight) (Fig. 3(C1)). Total lipid levels increased after exposure to atrazine (x̅ = 0.075 mg total lipids/g wet weight) but decreased in animals exposed to glyphosate (x̅ = 0.006 mg total lipids/g wet weight) and quinclorac (x̅ = 0.004 mg total lipids/g wet weight) in relation to the control group (x̅ = 0.008 mg total lipids/g wet weight) (Fig. 3(C2)). Triglyceride levels decreased after exposure to atrazine (x̅ = 0.021 mg triglycerides/g wet weight), glyphosate (x̅ = 0.039 mg triglycerides/g wet weight), and quinclorac (x̅ = 0.058 mg triglycerides/g wet weight) in relation to the control group (x̅ = 0.139 mg triglycerides/g wet weight) (Fig. 3(C3)). Regarding cholesterol levels, only atrazine (x̅ = 0.030 mg cholesterol/g wet weight) was associated with a significant increase in relation to the control group (x̅ = 0.022 mg cholesterol/g wet weight) (Fig. 3(C4)). All herbicides were associated with increases in total protein levels: x̅ = 0.424 mg protein/g wet weight after exposure to atrazine, x̅ = 0.391 mg protein/g wet weight after exposure to glyphosate, and x̅ = 0.525 mg protein/g wet weight after exposure to quinclorac, versus x̅ = 0.258 mg protein/g wet weight in the control group (Fig. 3(C5)). As in the gills and the liver, lipid peroxidation levels in muscle tissue increased after exposure to herbicides in relation to the control group (x̅ = 20.543 nmol TBARS/mg protein): levels were x̅ = 55.646 nmol TBARS/mg protein after atrazine exposure, x̅ = 69.186 nmol TBARS/mg protein after glyphosate exposure, and x̅ = 52.411 nmol TBARS/mg protein after quinclorac exposure (Fig. 3(C6)).
Graphics C1, C2, C3, C4, C5, and C6: Glycogen levels, total lipids, triglycerides, cholesterol, total proteins, and lipid peroxidation in the muscle of Lithobates catesbeianus tadpoles exposed to the herbicides atrazine, glyphosate, and quinclorac (Co 7 control 7 days, A 2.5 atrazine 2.5 μg L−1, G 18 glyphosate 18 μg L−1, Q 0.025 quinclorac 0.025 μg L−1). The results are expressed as the mean ± standard deviation. The asterisk beside the bars indicates a significant difference compared to the control group 7 days, being p < 0.05
Weight, size, and survival
All animals experienced increases in weight (Fig. 4(D1)) and size (Fig. 4(D2)) during the experiment. There was no mortality (Fig. 4(D3)), even among the animals exposed to herbicides.
Graphics D1, D2, and D3: Weight gain during the experiment compared to the initial weight of the groups (the value beside the bar indicates the percentage of weight gain), size at the beginning and end of the experiment (the value beside the bar indicates the percentage of increase in size during the experiment), and survival of Lithobates catesbeianus tadpoles until the end of the experiment (the value beside the bar indicates the percentage of survival) (Co 7 control 7 days, A 2.5 atrazine 2.5 μg L−1, G 18 glyphosate 18 μg L−1, Q 0.025 quinclorac 0.025 μg L−1, INIT initial, FIN final)
Discussion
The findings of our research were consistent with the hypothesis that there was no mortality, significant change in weight gain, and growth of these animals after exposure to herbicides in low concentrations. However, changes occurred in biochemical parameters in the animal tissues. According to Johansson et al. (2006), exposure to pesticides at levels as low as those found in nature usually does not cause mortality. A study was conducted in our laboratory with tadpoles exposed to higher concentrations of these pesticides, and the results also showed increased energy demands and no significant mortality in the animals (Dornelles and Oliveira 2014).
Pollutants such as herbicides are environmental stressors, which can induce an adaptive response in exposed animals due to changes in their metabolic balance, thus producing a physiological response in an attempt by the animal to reestablish the homeostasis (Roy and Hänninen 1993; Sasikala et al. 2011; Wendelaar Bonga 1997). The responses induced by this stress can occur as a result of metabolic changes, due to an increase in metabolic processes and a consequent increase in energy expenditure as a function of the stressor (Weissman 1990).
The decrease in glycogen concentrations in all tissues observed after exposure to the three tested herbicides is an expected response and similar to that found by other researchers, where this polymer acts as an immediately available energy reserve. This response may be due to the fact that pesticides usually affect energy metabolism, leading to an increase in energy expenditure, and glycogen can act as a rapid source of fuel—through the mobilization of stored glycogen—in times of stress exposure (Dornelles and Oliveira 2014; Dua et al. 2010; Oba et al. 2009; Tiwari and Singh 2003; Triebskorn et al. 1998; Venkataramana et al. 2006; Weissman 1990). So this polymer shows a protective function during periods of high energy demand, such as those caused by the presence of xenobiotics, acting as an immediately accessible energy reserve.
The effects of some pesticides on the lipid profile may be more pronounced in the skeletal muscles and liver and can cause a decrease in tissue lipid levels (Adamu and Kori-Siakpere 2011; Triebskorn et al. 1998). In our experiment, we observed a reduction in lipid levels in the liver after exposure to all herbicides and in the muscle after exposure to glyphosate and quinclorac, which may suggest mobilization of stored lipids, a common response in cases of exposure to chemical stressors (Adamu and Kori-Siakpere 2011). An increase of total lipids was observed in gills and muscle after exposure to atrazine, and it increased too in the gills after exposure to glyphosate. This response profile may be related to the sensitivity of each pesticide, the function, and the amount of metabolites in each organ. The liver has the highest values of total lipids and triglycerides followed by gills and muscle. Already, the glycogen content was higher in the gills followed by the liver and muscle, and total proteins were highest in the liver, followed by muscle and gills.
Little information is available about the effects of pesticides on lipid metabolism, but it is known that the liver has many functions such as synthesizing and processing of multiple substances, as lipoproteins, fatty acid, and glucose, to be transported to other areas of the body. Also, it is responsible for the maintenance of blood glucose, in addition to endo- and xenobiotic detoxification of substances that are chemically altered or excreted by the liver (Hill et al. 2012; Lasram et al. 2009; Moyes and Schulte 2010). This functional profile requires a high energy demand which can, together with the stress caused by the chemical, lead to the use of a larger amount of metabolites; this response also is related to the toxic potential of each herbicide to which the animal is exposed. This hypothesis is reinforced by the reduction in glycogen, lipid, and triglyceride reserves in the hepatic tissue of animals exposed to herbicides—and the reduction of protein levels in tadpoles exposed to glyphosate—in this study. After a stressful event, fat is used as the primary fuel source, in a process whereby triglycerides are metabolized into fatty acids and glycerol and then metabolized as fuel (Weissman 1990).
In this study, the herbicide atrazine led to an increase of cholesterol levels in all organs studied, the glyphosate determined a decrease of this metabolite in the liver and an increase in muscle, and quinclorac has an effect in the liver only, showing a decrease in cholesterol levels. The results showed that cholesterol levels were altered in different ways in the different types of tissues and showed a specific responsiveness and more sensitivity of the liver, which suggests that this organ acts in metabolizing these herbicides and that atrazine had a more powerful action on the cholesterol metabolism in relation to other herbicides.
Some authors, such as Weissman (1990), have shown that aquatic animals can alter their cholesterol levels on exposure to pesticides accordingly. Cholesterol levels may increase due to the stress induced by exposure to these agents, indicating an induced chemical disruption of lipid metabolism (Weissman 1990). The hypercholesterolemia observed in frogs exposed to pesticides can be the result of a decrease in the ratio of cholesterol conversion to bile acids or an impaired liver function. Conversely, pesticide exposure also can decrease cholesterol levels, leading to an increase in food intake (Adamu and Kori-Siakpere 2011; Sounderraj et al. 2011).
According Yeagle (1989) the cholesterol has been shown to be an ion pump regulator, and is some cases show an absolute dependence on cholesterol for activity. The author suggests that an essential role that cholesterol plays in mammalian cell biology is to enable crucial membrane enzymes to provide function necessary for cell survival. Studies of Na+K+ ATPase in human erythrocyte membranes showed inhibition of activity by high levels of membrane cholesterol. Similar results have been found in other membranes: in rabbit erythrocyte membranes, guinea pig erythrocyte membranes, rat liver membranes, and kidney basolateral membranes (Yeagle 1989). The variations found in the cholesterol levels may be a result of the disruption of the plasma membrane of cells after lipid peroxidation. We must also consider the possibility of using this molecule as a regulator of the electrogenic pump activity and indirectly in the ATP demand by the cells.
Proteins can also be involved in compensatory mechanisms in response to animal stress (Dornelles and Oliveira 2014). The reduction in protein levels in the gills is observed after the exposure to all herbicides, as well as the reduction in liver protein levels after exposure to glyphosate, which suggests that this decline in protein content can be due to a reduction in protein synthesis and/or an increase in proteolysis, and use of proteins in metabolic processes with respect to pesticide toxicity. These reductions in tissue protein content suggest that there are some pathways triggered after an attempt to restore depleted energy by breaking down protein to yield energetic fuel (Adamu and Kori-Siakpere 2011; Khan et al. 2002; Susan et al. 2010; Tiwari and Singh 2003; Venkataramana et al. 2006).
However, other responses may occur in the liver and muscle of aquatic animals exposed to pesticides, such as an increase in levels of protein. This may be due to a decrease in protein catabolism and/or to an increase in protein synthesis, as verified by Sahib et al. (1984) in the tissues of fish exposed to malathion. A similar response was observed in muscle tissue of tadpoles exposed to all herbicides (atrazine, glyphosate, and quinclorac) and in the liver of animals exposed to atrazine and quinclorac in the present study.
In the present work, the pattern of glycogen response to herbicide exposure was the same in all the tissues, which demonstrates the importance of polysaccharides to the energy homeostasis and survival of these animals. The other metabolites analyzed showed different responses according to the tissue and the herbicide. The intense depletion of energy reserves observed in this study may be associated with the high survival rate of tadpoles exposed to low concentrations of the tested herbicides (atrazine, glyphosate, and quinclorac) (Fig. 4(D3)).
Lipid peroxidation can provide important evidence of the toxicity of environmental pollutants. In this study, an increase in LPO levels after exposure to all herbicides and in all tissues analyzed was observed. These responses were more marked in gill tissue (5.2-fold to 6.3-fold increases) than in muscle (2.7-fold to 3.4-fold increases) and liver (1.6-fold increase), allowing us to suggest a higher antioxidant capacity in other organs, mainly the liver, compared to gills. Despite the increase in the lipid peroxidation levels, there was no mortality, which may be related to an antioxidant defense capacity both in enzymatic and non-enzymatic terms (Ballesteros et al. 2009). Farombi et al. (2008), Gultekin et al. (2000), and Brocardo et al. (2005) found similar results in their investigations.
Organisms respond to changes in the external environment by activating their mechanism of regulation of the homeostasis. In complex animals, homeostasis is maintained particularly in organs that serve as sites of exchange with the external environment, such as the gills (Hickman et al. 2001). As the gills are the first tissue to be exposed in amphibians, greater uptake of pesticides may occur and the response to exposure may be more intense (Streit 1992). The results of the present study reinforce this hypothesis, as glycogen, triglyceride, and total protein levels in gill tissue decreased and lipid peroxidation was markedly increased in response to all pesticides.
In animals, responses to stress are an adaptive mechanism that allows survival in the presence of stressors by maintenance of homeostasis. These responses can occur at the tissue level, which includes the mobilization of energy substrates. Depending on the intensity of the stressor, animals may be unable to tolerate the ensuing changes and exhibit reactions such as inhibition of growth, reproduction, or immune response (Lima et al. 2006).
Although there had been research on exposure of frogs to the herbicides such as atrazine, glyphosate, and quinclorac, our investigations have focused on behavioral effects, morphological deformities, changes in the developmental stages, and metabolic alterations; however, we were unable to find any similar research involving quinclorac.
Conclusion
The realistic concentrations of atrazine, glyphosate, and quinclorac used in this study had no effect on mortality, weight gain, or size in L. catesbeianus tadpoles exposed to the herbicides for 7 days. However, even using concentrations below the legal limits, the herbicides induced significant alterations in biochemical parameters, mainly reductions in the levels of glycogen, triglycerides, and proteins, and an increase in levels of lipid peroxidation in all organs studied, which are expected to have longer term effects. The intensity of the observed responses was dependent on the studied organ and herbicide to which the animals were exposed.
The use of energy reserves to maintain homeostasis and the survival of animals may influence other biological parameters, such as development and reproductive success. It is important to stress that the responses found in the present study and the aforementioned studies may differ depending on the species of animal, concentration of herbicide, and duration of exposure to the contaminant.
References
Adamu KM, Kori-Siakpere O (2011) Effects of sublethal concentrations of tobacco (Nicotiana tobaccum) leaf dust on some biochemical parameters of hybrid catfish (Clarias gariepinus and Heterobranchus Bidorsalis). Braz Arch Biol Technol 54:183–196
Al-Othman AM, Khaled SA, Gaber EE, Kareem Y, Zeid AA, Mourad AM, John PG (2011) Protection of α-tocopherol and selenium against acute effects of malathion on liver and kidney of rats. Afr J Pharm Pharmacol 5:1263–1271
Allran JW, Karasov WH (2000) Effects of atrazine and nitrate on northern leopard frog (Rana pipiens) larvae exposed in laboratory from posthatch through metamorphosis. Environ Toxicol Chem 19:2850–2855
ASTM – American Society for Testing and Materials. Active Standard (1998) Standard Guide for Conducting the Frog Embryo Teratogenesis Assay—Xenopus (FETAX) ASTM International, West Conshohocken, PA. E 1439:790-805
Ballesteros ML, Wunderlin DA, Bistoni MA (2009) Oxidative stress responses in different organs of Jenynsia multidentata exposed to endosulfan. Ecotoxicol Environ Saf 72:199–205
Barceló D, Hennion MC (2003) Pesticides and their degradation products: characteristics, usage and environmental behaviour. In: Trace determination of pesticides and their degradation products in water, techniques and instrumentation in analytical chemistry. Elsevier Science B 19, Amsterdam, pp.01-89
Belluck DA, Benjamin SL, Dawson T (1991) Groundwater contamination by atrazine and its metabolites: risk assessment, policy, and legal implications. In: Pesticide transformation products: fate and significance in the environment. ACS Symposium Series 459. American Chemical Society, Washington, DC. pp.254-273
Bernabò I, Brunelli E, Berg C et al (2008) Endosulfan acute toxicity in Bufo bufo gills: ultrastructural changes and nitric oxide synthase localization. Aquat Toxicol 86:447–456
Blaustein AR, Romansic JM, Kiesecker JM et al (2003) Ultraviolet radiation, toxic chemicals and amphibian population declines. Divers Distrib 9:123–140
Boone MD, Cowman D, Davidson C et al (2005) Evaluating the role of environmental contamination in amphibian population declines. In: Gascon C, Collins JP, Moore RD, Church DR, McKay JE, Mendelson JR III (eds) Amphibian conservation action plan. IUCN Species Survival Commission, Gland, Switzerland, pp 32–35
Brasil, Conselho Nacional do Meio Ambiente (2005) Resolução n.° 357, de 17/3/2005. Dispõe sobre a classificação dos corpos de água e diretrizes ambientais para o seu enquadramento, bem como estabelece condições e padrões de lançamento de efluentes, e dá outras providências
Brocardo PS, Pandolfo P, Takahashi RN et al (2005) Antioxidant defenses and lipid peroxidation in the cerebral cortex and hippocampus following acute exposure to malathion and/or zinc chloride. Toxicol 207:283–291
Champe C, Harvey RA (2006) Metabolismo dos lipídeos complexos e colesterol e metabolismo dos esteróides. In: Champe PC, Harvey RA (eds) Bioquímica Ilustrada, 2ath edn. Artes Médicas, Porto Alegre, pp 199–242
David M, Kartheek RM (2015) Malathion acute toxicity in tadpoles of Duttaphrynus melanostictus, morphological and behavioural study. J Basic Appl Zool 72:1–7
Davidson C (2004) Declining downwind: amphibian population declines in California and historical pesticide use. Ecol Appl 14:1892–1902
Dornelles MF, Oliveira GT (2014) Effect of atrazine, glyphosate and quinclorac on biochemical parameters, lipid peroxidation and survival in bullfrog tadpoles (Lithobates catesbeianus). Arch Environ Contam Toxicol 66:415–429
Dua R, Kumar V, Sunkaria A et al (2010) Altered glucose homeostasis in response to aluminium phosphide induced cellular oxygen deficit in rat. Indian J Exp Biol 722-730
El-Banna SG, Attia AM, Hafez AA, El-Kazaz SA (2009) Effect of garlic consumption on blood lipid and oxidant/antioxidant parameters in rat males exposed to chlorpyrifos. Slovak J Anim Sci 42:111–117
Farombi EO, Ajimoko YR, Adelowo OA (2008) Effect of butachlor on antioxidant enzyme status and lipid peroxidation in fresh water African catfish (Clarias gariepinus). Int J Environ Res Public Health 5:423–427
Folch J, Lees M, Sloane-Stanley GHA (1957) Simple method for isolation and purification of total lipids from animal tissues. Biol Chem 226:497–509
Frings CE, Dunn RA (1970) Colorimetric method for determination of total serum lipids based on the sulfophosphovanillin reaction. Am J Clin Pathol 53:89–91
Ganeshwade RM (2012) Biochemical changes induced by dimethoate (Rogor 30% EC) in the gills of fresh water fish Puntius ticto (Hamilton). J Ecol Nat Environ 4:181–185
Gascon C, Collins JP, Moore RD et al (2005) Amphibian conservation action plan. IUCN Species Survival Commission, Gland, Switzerland, 68p
Geary N, Langhans W, Scharrer E (1981) Metabolic concomitants of glucagon-induced suppression of feeding in the rat. Am J Physiol 241:330–335
Giesy JP, Dobson S, Solomon KR (2000) Ecotoxicological risk assessment for roundup herbicide. Rev Contam Toxicol 167:35–120
Gosner KL (1960) A simplified table for staging anuran embryos and larvae with notes on identification. Herpetologica 16:183–190
Guillouzo A (1998) Liver cell models in in vitro toxicology. Environ Health Perspect 106:511–532
Gultekin F, Ozturk M, Akdogan M (2000) The effect of organophosphate insecticide chlorpyrifos-ethyl on lipid peroxidation and antioxidant enzymes (in vitro). Arch Toxicol 74:533–538
Hickman JrCP, Roberts L, Larson A (2001) Homeostasis, osmotic regulation, excretion, and temperature regulation. In: Hickman JrCP, Roberts L, Larson A (eds) Integrated principles of zoology - 11th ed., pp.664-683
Hill RW, Wyse GA, Anderson M (2012) Fisiologia animal, 2nd edn. Artmed, Porto Alegre, 920 p
Johansson M, Piha H, Kylin H et al (2006) Toxicity of six pesticides to common frog (Rana temporaria) tadpoles. Environ Toxicol Chem 25:3164–3169
Kavitha P, Venkateswara Rao J (2007) Oxidative stress and locomotor behaviour response as biomarkers for assessing recovery status of mosquito fish, Gambusia affinis after lethal effect of an organophosphate pesticide, monocrotophos. Pestic Biochem Physiol 87:182–188
Kehrer JP (1993) Free radicals as mediators of tissue injury and disease. CRC Crit Rev Toxicol 23:21–48
Khan MZ, Farina F, Imtiaz A (2002) Effect of cypermethrin on protein contents in lizard Calotes versicolor in comparison to that in Frog Rana tigrina. Biol Sci 2:780–781
Landis WG, Yu MH (2003) Introduction to environmental toxicology: impacts of chemicals upon ecological systems, 3rd edn. Crc press, Boca Raton, Flórida, 509p
Lasram MM, Annabi AB, Elj NE et al (2009) Metabolic disorders of acute exposure to malathion in adult Wistar rats. Hazard Mater 163:1052–1055
Lima LC, Ribeiro LP, Leite RC et al (2006) Estresse em peixes. Rev Bras Reprod Anim 30:113–117
Marchezan E, Zanella R, Avila LA et al (2007) Rice herbicide monitoring in two Brazilian rivers during the rice growing season. Sci Agric 64:131–137
Massoud ASDAH, El-Fakhrany II, Allah MSS (2011) Toxicological effects of organosphorus insecticides and remediation technologies of its residues in aquatic system B. dimethoate. J Agric Res Kafer El-Sheikh Univ 37:516–533
Montgomery MP, Kamel F, Saldana TM et al (2008) Incident diabetes and pesticide exposure among licensed pesticide applicators: Agricultural Health Study 1993–2003. Am J Epidemiol 167:1235–1246
Moyes CD, Schulte PM (2010) Princípios De Fisiologia Animal. Porto Alegre, Artmed, pp 526–571
Murphy JE, Phillips CA, Beasley VR (2000) Aspects of amphibian ecology. In: Sparling DW, Linder G, Bishop CA (eds) Ecotoxicology of amphibians and reptiles, 1ath edn. Setac Press, Pensacola, FL, pp 141–178
Nwani CD, Lakra WS, Nagpure NS et al (2010) Toxicity of the herbicide atrazine: effects on lipid peroxidation and activities of antioxidant enzymes in the freshwater fish Channa punctatus (Bloch). Int J Environ Res Public Health 8:3298–3312
Oba ET, Mariano WS, Santos LRB (2009) Estresse em peixes cultivados: agravantes e atenuantes para o manejo sustentável. In: Tavares-Dias M (ed) Manejo e Sanidade de Peixes em Cultivo. Macapá, Embrapa Amapá, pp 389–424
Patil JA, Patil AJ, Sontakke AV, Govindwar SP (2009) Oxidative stress and antioxidants status of occupational pesticides exposed sprayers of grape gardens of western Maharashtra (India). J Environ Health Res 9:81–89
Relyea RA (2003) Predator cues and pesticides: a double dose of danger for amphibians. Ecol Appl 13:1515–1521
Rodrigues NR, Almeida FS (1998) Guia de herbicidas 4a edn. Editora dos Autores, Londrina, pp 137–142
Roy S, Hänninen O (1993) Biochemical monitoring of the aquatic environment: possibilities and limitations. In: Richardson M (ed.), Ecotoxicol Monit, VCH-Verlag, pp. 119-135
Sahib IK, Rao KR, Rao KV (1984) Effect of malathion on protein synthetic potentiality of the tissues of the teleost, Tilapia mossambica (Peters), as measured through incorporation of [14C] amino acids. Toxicol Lett 20:63–67
Salbego J, Pretto A, Gioda CR et al (2010) Herbicide formulation with glyphosate affects growth, acetylcholinesterase activity, and metabolic and hematological parameters in piava (Leporinus obtusidens). Arch Environ Contamin Toxicol 58:740–745
Sasikala G, Palanisamy P, Mallikaraj D et al (2011) Biochemical modulations induced by metasystox in fresh water snakeheaded fish Channa striata blood. Int J Pharm Biol Arch 2:772–774
Sayeed I, Parvez S, Pandey S et al (2003) Oxidative stress biomarkers of exposure to deltamethrin in freshwater fish, Channa punctatus Bloch. Ecotoxicol Environ Saf 56:295–301
Sayim F (2008) Acute toxic effects of malathion on the 21st stage larvae of the marsh frog. Turk J Zool 32:99–106
Silva DRO, Avila LA, Agostinetto D et al (2009) Monitoramento de agrotóxicos em águas superficiais de regiões orizícolas no sul do Brasil. Ci Rural 39:2383–2389
Solomon KR, Baker DB, Richards RP, Kenneth RD, Klaine SJ, Lapoint TW et al (1996) Ecological risk assessment of atrazine in North American surface waters. Environ Toxicol Chem 15:31–76
Sounderraj SFL, Sekhar P, Kumar PS et al (2011) Effect of systemic pesticide phosphamidon on haematological aspects of common frog Rana tigrina. I Int J Pharm Biol Arch 2:1776–1780
Streit B (1992) Bioaccumulation processes in ecosystems. Experientia 48:955–970
Susan TA, Sobha K, Veeraiah K et al (2010) Studies on biochemical changes in the tissues of Labeo rohita and Cirrhinus mrigala exposed to fenvalerate technical grade. J Toxicol Environ Health Sci 2:53–62
Tiwari S, Singh A (2003) Control of common freshwater predatory fish, Channa punctatus, through Nerium indicum leaf extracts. Chemosphere 53:865–875
Triebskorn R, Christensen K, Heim G (1998) Effects of orally and dermally applied metaldehyde on mucus cells of slugs (Deroceras reticulatum) depending on temperature and duration of exposure. J Mollus Stud 64:467–487
Uchendu C, Ambali SF, Ayo JO (2012) The organophosphate, chlorpyrifos, oxidative stress and the role of some antioxidants: a review. Afr J Agric Res 7:2720–2728
Upasani CD, Balaraman R (2001) Effect of vitamin E, vitamin C and spirulina on the levels of membrane bound enzymes and lipids in some organs of rats exposed to lead. Indian J Pharmacol 33:185–191
US Environmental Protection Agency (1985) EPA draft final list of recommendation for chemicals in the National Survey for Pesticides in Groundwater. Chem Regul Rep 9:1033
Van Handel E (1965) Estimation of glycogen in small amount soft tissue. Anal Biochem 11:256–265
Venkataramana GV, Sandhya Rani PN, Murthy PS (2006) Impact of malathion on the biochemical parameters of gobiid fish, Glossogobius giuris (Ham). J Environ Biol 27:119–122
Vickers AEM (1994) Use of human organ slices to evaluate the biotransformation and drug-induced side-effects of pharmaceuticals. Cell Biol Toxicol 10:407–414
Vutukuru SS (2005) Acute effects of hexavalent chromium on survival, oxygen consumption, hematological parameters and some biochemical profiles of the Indian major carp, Labeo rohita. Int J Environ Res Public Health 2:456–462
Wang X, Dong Y, Wang L et al (2001) Acute toxicity of substituted to Rana japonica tadpoles and mechanism-based quantitative structure-activity relationship (QSAR) study. Chemosphere 44:447–455
Weissman C (1990) The metabolic response to stress: an overview and update. Anesthesiol 73:308–327
Wendelaar Bonga SE (1997) The stress response in fish. Phys Rev 77:592–625
Yeagle PL (1989) Lipid regulation of cell membrane structure and function. FASEB J 3:1833–1842
Acknowledgments
We would like to thank Dr. Taran Grant, Dr. Nelson Ferreira Fontoura, the team at the PUCRS Conservation Physiology Laboratory, CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior) for supporting this study, and CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico) for granting a Productivity Fellowship (307303/2012) to the correspondent author.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Dornelles, M.F., Oliveira, G.T. Toxicity of atrazine, glyphosate, and quinclorac in bullfrog tadpoles exposed to concentrations below legal limits. Environ Sci Pollut Res 23, 1610–1620 (2016). https://doi.org/10.1007/s11356-015-5388-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-015-5388-4