Abstract
Metal-contaminated soil, from the El Arteal mining district (SE Spain), was remediated with organic (6 % compost) and inorganic amendments (8 % marble sludge) to reduce the mobility of metals and to modify its potential environmental impact. Different measures of metal bioavailability (chemical analysis; survival, growth, reproduction and bioaccumulation in the earthworm Eisenia andrei), were tested in order to evaluate the efficacy of organic and inorganic amendments as immobilizing agents in reducing metal (bio)availability in the contaminated soil. The inorganic amendment reduced water and CaCl2-extractable concentrations of Cd, Pb, and Zn, while the organic amendment increased these concentrations compared to the untreated soil. The inorganic treatment did not significantly reduce toxicity for the earthworm E. andrei after 28 days exposure. The organic amendment however, made the metal-contaminated soil more toxic to the earthworms, with all earthworms dying in undiluted soil and completely inhibiting reproduction at concentrations higher than 25 %. This may be due to increased available metal concentrations and higher electrical conductivity in the compost-amended soil. No effects of organic and inorganic treatments on metal bioaccumulation in the earthworms were found and metal concentrations in the earthworms increased with increasing total soil concentrations.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
One of the serious environmental problems associated with mining activities are the huge amounts of uncontrolled hazardous materials that are accumulated as tailings and waste rock deposits close to the mining area. Although mining itself affects relatively small areas, the tailings and solid waste deposits are important sources of metals. Metals, unlike organic contaminants, are generally immutable, not degradable and persistent in soil (Adriano et al. 2004).
Different techniques have been proposed to remediate metal-contaminated soils, some of which consist of very expensive or highly invasive treatments that can only be practiced ex situ and have a great impact on the ecosystem. Alternative stabilization techniques, based on the use of different soil amendments can be very effective in reducing metal bioavailability (Friels et al. 2003; Mench et al. 2006), improve soil physico-chemical and biological properties, do not generate by-products, are less expensive, and therefore more suitable for remediation of extensive areas of low-value land (Mench et al. 2003). In practice, such amendments often are waste products like marble sludge or compost (Melgar-Ramírez et al. 2012).
Criteria and standards for regulatory purposes for metals in soils usually are based on total concentrations as determined by acid digestion, but this is a conservative perspective. Total metal concentrations in the soil are not predictive of the potential risk of metal contamination in soil (Hooda and Alloway 1994; Morillo et al. 2004; Zhang et al. 2008). So, the main intent of applying amendments is not to change the total concentration but to reduce the bioavailable fraction, and by that to limit the risk to the environment.
The assessment of the overall effect of amendments and their efficacy for metal immobilization in contaminated soil may well combine physico-chemical and biological methods (Vangronsveld et al. 2000; González et al. 2011). Most contaminated sites have elevated concentrations of more than one element making the solution to the problem more complex, because remediation requires solutions effective for all target elements. In addition, it is more difficult to evaluate how these elements are affecting organisms. Several studies have demonstrated the adverse effects of metal contamination on plants, animals and bacteria (Escoto et al. 2007; Loureiro et al. 2005; Fountain and Hopkin 2004). For some organisms, the metal concentration in the soil solution may be predictive of metal availability. But other organisms, like soil invertebrates, are able to accumulate metals via a number of distinct uptake routes (Crommentuijn et al. 1994, 1997). Measures of bioavailability may either be direct or indirect, and chemical or biological (Lanno et al. 2004).
In this study, using the earthworm Eisenia andrei, different measures of metal bioavailability (chemical analysis, survival, growth, reproduction, and bioaccumulation), were tested in order to evaluate the efficacy of organic and inorganic amendments as immobilizing agents in reducing metal (bio)availability in a contaminated soil. Because the contaminated soil contained elevated concentrations of more than one element, a dilution approach was chosen to assess the effect of the amendments on the overall toxicity of the contaminated soil.
Material and methods
Soil and treatments
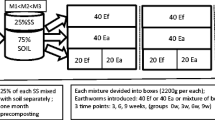
Contaminated soil was sampled in the El Arteal mining district (Almeria, SE Spain), near an old lead and silver smelting plant. Soil was sampled from the top 0–25 cm layer after removal of surface vegetation and litter. The soil was dried for 24 h at 60 °C and stored until use. The soil was mixed with two different amendments, inorganic sludge from marble industry (MS) and organic compost from greenhouses (CM). Characteristics of the soil and the amendments are given in Table 1. Treatments consisted of the addition of 8 % MS (w/w) or 6 % CM (w/w) to the contaminated soil. The compost (Table 1) was within the limits for heavy metal concentrations in compost and fertilizer products made from wastes, established by the Spanish legislation (Cd < 0.7, Cu < 70, Pb < 45 and Zn < 200 mg kg−1; BOE 2005). Amendments used were 2-mm sieved, and the amended soils were homogenized by hand for 15 min. Untreated contaminated soil (U-CS), contaminated soil treated with marble sludge (MS-CS), and contaminated soil treated with compost (CM-CS) were homogeneously mixed with the uncontaminated Lufa 2.2 reference soil to obtain concentrations of 100, 50.0, 25.0, 12.5, 6.25 and 3.13 % (w/w) of all soils.
Analytical methods
Cation-exchange capacity (CEC) was determined with 1 M Na acetate at pH 8.2 (Rhoades 1982). Total carbon content was analyzed by dry combustion in a LECO SC-144DR analyzer. Calcium carbonate equivalent content (CaCO3, %) was estimated manometrically (Williams 1948). Organic carbon (OC) content was determined by the difference between the amounts of total carbon and inorganic carbon from CaCO3. The particle-size distribution was determined by the pipette method (Loveland and Whalley 1991). pH and electrical conductivity were measured in a 1:2.5 soil/water suspension. The water holding capacity (WHC) was determined according to ISO (1999).
To determine total metal concentrations, very finely ground (<0.05 mm) soil samples were digested in concentrated acids (HNO3 + HF). Trace elements were measured by inductively coupled plasma-mass spectrometry (ICP-MS) using a Hewlett Packard 4500 STS spectrometer. The accuracy of the method was confirmed with analysis (six replicates) of the Standard Reference Material SRM2711. For all metals (Cd, Pb, and Zn) analyzed, average recoveries (n = 27) ranged between 94 and 101 % of the certified reference values. Water and CaCl2-extractable metal concentrations were obtained by shaking 20 g soil in 20 ml distilled water or 0.01 M CaCl2 solution, respectively, for 2 h at 200 rpm and subsequent filtration through a 0.45-μm cellulose membrane filter (Gelman Sciences, Michigan, USA). Trace elements were measured by ICP-MS.
Eisenia andrei bioassay
E. andrei (Lumbricidae) were obtained from a laboratory culture at the Department of Ecological Science, VU University, Amsterdam. Earthworms were cultured in horse manure free of any pharmaceuticals at 20 ± 1°C.
Before starting the test, untreated and treated soils, and their respective dilution series with Lufa 2.2 reference soil were dried at 50 °C and moistened at 60 % of their WHC. To check for the performance of the test organisms, in all tests controls of Lufa 2.2 soil were included. The test followed OECD guideline 222 (OECD 2004), with a 4-week exposure period of adult animals, followed by another 4-week incubation period of cocoons to allow for determination of the number of juveniles produced.
Before the start of the experiment, adult worms with well-developed clitellum were acclimatized in Lufa 2.2 soil for 24 h. For each test, four replicate test containers were used containing approx. 500 g soil (dry weight equivalent), with 10 earthworms being randomly assigned to each container. At the start of the test, each batch of 10 animals was weighed, and 56 animals were weighed individually to determine the range of starting weights (between 0.386 and 0.877 g; mean ± SD: 0.503 ± 0.09 g). Test containers were incubated at 20 °C and constant illumination. After introduction of the earthworms, 5 g of finely ground and moistened horse dung was placed in a small hole in the middle of the test soil as described by Van Gestel et al. (1989). During the experiments, test containers were opened at least once a week to aerate the test soils. Once a week, all test containers were weighed to correct for water loss and additional food was added if required.
After 4 weeks of incubation, test containers were emptied in a tray and surviving adults were collected by hand sorting, and weighed after washing in distilled water. Two earthworms of each replicate sample were placed on moist filter paper for 24 h to depurate their gut contents. The soil was returned into the test container and incubated for another 4 weeks to allow the cocoons to hatch. After this second 4-week period, containers were placed in a water bath at 60 °C to force the juveniles out of the soil. Juvenile worms were picked from the soil surface using a pair of tweezers and counted.
After depuration, earthworms were frozen, freeze-dried for 2 days and weighed. Dried earthworms were digested in 2 mL of a 4:1 mixture of nitric acid (65 % p.a.; Riedel-de-Haen) and hydrochloric acid (37 % p.a., Baker) in tightly closed Teflon bombs upon heating in a destruction oven at 140 °C for 7 h. After cooling, digests were diluted to 10 mL with demineralized water and analyzed on a Perkin-Elmer AAnalyst 100 flame atomic absorption spectrometer. Reference material Dolt 4 (animal tissue) was used to determine the accuracy of the analytical procedure. Average metal concentrations in the reference material varied between 104.2 and 106.2 % of the certified values.
Statistical analysis
Data from the bioassays in the different treatments, and the influence of treatments and total and extractable metal concentrations (water and CaCl2) on earthworm performance were analyzed by one-way ANOVA, and the significance of the differences between mean values was calculated by Tukey’s post hoc test at p < 0.05. EC50 values were estimated using a logistic model according to Haanstra et al. (1985). Toxic units for Cd, Pb, and Zn were calculated from earthworm internal concentrations divided by internal concentrations in control earthworms incubated for 28 days in the Lufa 2.2 soil, and effects were related to the sum of toxic units. All chemical and biological measures obtained in this study were selected for a factorial analysis (principal component analysis, PCA) in order to minimize the number of variables with high saturation for each factor and to detect structure in the relationship between variables. Missing values were removed pairwise, and the factor matrix was subjected to Varimax rotation, facilitating data interpretation. All analyses were run in the software package SPSS 15.0 for Windows.
Results
Soils and amendments properties
The contaminated soil was neutral (pH-H2O 7.1) and had a clay-loam texture (Table 1). The EC was low (1.41 dS m−1); the soil is considered saline when EC > 4 dS m−1 (U.S. Salinity Laboratory Staff 1954). The soil had low CEC (5.76 cmol+ kg−1) and low OC (1.26 g kg−1) compared to mean values for the Almería province (Sierra 2005), and high concentrations of Zn, Pb, and Cd compared to baseline proposed for South-Eastern Spain (Sierra et al 2007). The MS had a very high CaCO3 content (>99 %), alkaline pH (8.5), low EC value (2.0 dS m−1), and low trace element concentrations. The CM from greenhouse agriculture had a very high OC content (417 g kg−1), high EC value (7.2 dS m−1), and relatively low metal concentrations.
The inorganic treatment (MS-CS) significantly increased pH compared to untreated soil (U-CS) and soil amended with compost (CM-CS). Addition of compost significantly increased CEC, EC, OC content compared to U-CS and MS-CS (Table 2).
MS-CS treatment significantly (p < 0.01; Tukey’s post hoc test) decreased water (w) and CaCl2-extractable concentrations for all metals studied compared to untreated soil, with the exception of w-Pb, CaCl2-Pb and CaCl2-Cd (Fig. 1). After marble sludge application, w-Pb, w-Zn, and w-Cd concentrations were 10, 30, and 53 % lower, respectively, compared to untreated soil, while CaCl2-Pb and CaCl2-Zn concentrations were 51 and 27 % lower, respectively. No significant differences were found for CaCl2-Cd concentrations after inorganic treatment. Organic treatment (CM-CS) significantly (p < 0.01) increased water and CaCl2-extractable concentration of Pb, Zn, and Cd compared to untreated soil. After compost application, w-Pb, w-Zn, and w-Cd were 42, 52, and 30 % higher, respectively compared to untreated soil, while CaCl2-extractable Pb and CaCl2-Cd were 35 and 30 % higher, respectively. Dilution with Lufa 2.2 reference soil reduced total, water and CaCl2-extractable concentrations in the contaminated and amended soils, while it also reduced differences between amendments and non-amended soil (Fig. 1).
Water (left) and 0.01 M CaCl2 extractable (right) concentrations of Cd (top), Zn (middle) and Pb (bottom) in untreated contaminated soil (filled bars), contaminated soil treated with marble sludge (dashed bars), and contaminated soil treated with compost (open bars), for all concentrations of the studied soils diluted with Lufa 2.2 reference soil. Different letters mean significant differences (p < 0.05; Tukey’s post hoc test) between treatments in each concentration
Effect on Eisenia andrei survival, growth, reproduction, and metal bioaccumulation
No earthworm mortality was found in Lufa 2.2 reference soil. Survival was high and did not significantly differ between the untreated soil (U-CS) and the soil amended with marble sludge (MS-CS). However, in soil amended with compost (CM-CS), mortality was higher than 95 %. Survival was always close to 100 % when studied soils were diluted with Lufa 2.2 reference soil, and no differences were found between U-CS, MS-CS, and CM-CS.
After 4 weeks of exposure, weight of the adult earthworms in contaminated soil (U-CS) was increased by 7.6 %, while in MS-CS and CM-CS the worms lost 8.8 and 19.4 % weight, respectively. Earthworm weights tended to decrease at lower concentrations of contaminated soil (for all treatments: U-CS, MS-CS, CM-CS), although no significant differences were found.
Reproduction of E. andrei in the Lufa 2.2 control soil was high, with on average 40 juveniles per test container (Fig. 2), so exceeding the minimum number of 30 juveniles per vessel required by OECD Guideline 222 (OECD 2004). For all contaminated soils (U-CS, MS-CS, and CM-CS), the number of juveniles increased at higher dilution with Lufa 2.2 soil. EC50 values (based on total concentration of soil; with corresponding 95 % confidence intervals) for the effect on earthworm reproduction in MS-CS and U-CS were similar [51 (34.2–67.6) mg kg–1 and 46 (28.2–64.8) mg kg−1, respectively], while EC50 for CM-CS was lower [27 (21.6–32.1) mg kg–1], suggesting that application of compost increased toxicity. In treatments with compost, no juveniles were found at the two highest concentrations (100 and 50 % CM-CS, Fig. 2). For the lower concentrations (3.13, 6.25, 12.5, and 25 %), both amendments increased the number of juveniles compared to untreated soil, although significant differences were only found for CM-CS (Fig. 2). Reproduction was inversely related with total (Table 3), water and CaCl2-extractable concentrations of all metals studied (Table 4). For each dilution level (Fig. 2) differences between treatments were significant. Reproduction also showed a strong relationship with the sum of toxics unit estimated for Cd, Pb, and Zn (Fig. 3).
Reproduction of the earthworm Eisenia andrei after 28 days exposure to untreated contaminated soil (filled bars), contaminated soil treated with marble sludge (dashed bars), and contaminated soil treated with compost (open bars), for all concentrations of studied soil diluted with Lufa 2.2 reference soil and control (Lufa 2.2). Different letters means significant differences (p < 0.05; Tukey’s post hoc test) between treatments per each concentration. *>95 % mortality at this concentration
Eisenia andrei reproduction after 28 days exposure to metal-contaminated soil, untreated or treated with marble sludge or compost (in % of control) plotted against the summed Zn, Cd, and Pb concentrations in the earthworms, expressed as sum of Toxic Units (ΣTU). Toxic Units were obtained by scaling body concentrations in exposed earthworms against concentrations in earthworms incubated for 28 days in uncontaminated Lufa 2.2 reference soil
Metal concentrations in control earthworms incubated for 28 days in Lufa 2.2 reference soil were 0.049 mg Pb kg–1, 5.6 mg Cd kg–1, and 105 mg Zn kg–1 dry weight. The highest metal concentrations in the earthworms were found at the higher concentrations of untreated contaminated soil (U-CS): 91.4 ± 50.2 mg Pb kg–1 dry weight, 16.7 ± 5.2 mg Cd kg–1 dry weight, and 150 ± 27.1 mg Zn kg–1 dry weight. For all three elements studied, earthworm concentrations increased with increasing soil concentration. This increase was most pronounced for Cd and Pb (Fig. 4). Metal concentrations in the earthworms increased with higher total, water and CaCl2-extractable concentrations, for all metals in all soils (U-CS, MS-CS, CM-CS). No significant differences were found between treatments, although Pb and Cd concentrations in the earthworms were lower in undiluted soil treated with marble sludge compared to undiluted untreated soil (Fig. 3). Determination coefficients (r 2) were higher for the relationship between earthworm metal concentrations and total soil concentrations (Table 3) than for the relationships with water and CaCl2-extractable concentrations (Table 4).
Concentrations of Cd, Pb, and Zn in Eisenia andrei after 28 days exposure to untreated contaminated soil (filled bars), contaminated soil treated with marble sludge (dashed bars), and contaminated soil treated with compost (open bars), diluted with Lufa 2.2 reference soil and for control (Lufa 2.2). *>95 % mortality at this concentration
Indirect biological measures (survival, reproduction, and growth), direct biological measures (earthworm concentration), chemical measures of bioavailability (total, water extracted and CaCl2 extracted fractions), and soil properties (pH and EC) were selected for the PCA analysis. Three factors explained 81.5 % of the variability in the data (Table 5). The first factor, with 57.5 % of the variance explained, shows that reproduction (numbers of juveniles) of E. andrei was inversely related with water and CaCl2-extractable concentrations Cd, Pb, and Zn, pH, electrical conductivity, and total metal concentration in the soils. The second factor, with 18.0 % of the variance explained, showed that survival of E. andrei and growth rate were indirectly related with EC, w-Zn, and CaCl2-Pb. In the third factor, internal Cd, Pb, and Zn concentrations in the earthworms were directly related with pH and total concentrations of Cd, Pb, and Zn in the soils (Table 5).
Discussion
Total concentrations of Zn, Cd, and Pb in contaminated soil exceeded the background concentrations proposed (Sierra et al 2007) by between 5 and 19 times, respectively, indicating that Cd and Pb were the major contaminants in this soil. Although the amendments had no effect on total metal concentrations in soil, the water and 0.01 M CaCl2-extractable concentrations of all metals decreased significantly after the soils were amended with marble sludge (Fig. 1). CaCO3 must have played an important role in decreasing the solubility of Cd, Pb, and Zn, probably by precipitation as carbonates (Alloway et al. 1998; Garrido et al. 2005), by coprecipitation (Baes and Mesmer 1976) or adsorption onto iron and aluminum oxyhydroxides, which would precipitate on the carbonate particles forming coatings (Simón et al. 2002) in MS-CS. The effect of organic treatment (CM-CS), increasing water and CaCl2-extractable concentrations of all metals, could be due to the formation of complexes with soluble organic compounds (Hsu and Lo 2000). Other studies (Kiekens 1995; Saar and Weber 1980) have reported the fixation of trace elements by organic matter. These different behaviors may be associated with the use of different types of organic matter; in this way, complexes of trace elements with fulvic acids are water soluble, while metal complexation with high molecular weight of organic matter, such as humic acids, could reduce metal concentration in soil solution (Stevenson 1992). Survival of E. andrei in contaminated soil and soil amended with marble sludge was around 100 %, even when the soil was not diluted with Lufa 2.2 reference soil. But in soil treated with compost, survival was lower than 30 %. Because in all contaminated soils (U-CS, MS-CS, and CM-CS), total metal concentrations were lower than the LC50 values for E. andrei reported by Spurgeon et al. (1994) (300 mg Cd kg–1, 4,480 mg Pb kg–1, and 1,010 mg Zn kg–1), the mortality cannot be explained from total metal contents of the soils. Significantly higher EC values in CM-CS treated soils compared with U-CS and MS-CS (Table 2), and the inclusion of EC and survival for E. andrei in the PCA (Factor 2) with different sign (Table 5), suggest that survival was affected by EC. Other studies showed that the addition of 5.4 g NaCl L−1 (EC ≈ 11 dS m–1 estimated from equivalent conductivity of each ion) caused 50 % reduction in earthworm survival (Owojori et al. 2008). The inclusion of the growth rate in Factor 2 of the PCA suggests that, like survival, growth also was affected by EC. On the other hand, higher growth rates were found when the numbers of juveniles decreased, suggesting a trade off with higher reproduction implying lower growth rates (Van Gestel et al. 1992).
Earthworm reproduction was more sensitive than survival to changes in available metal concentrations. As shown by the EC50 values, application of marble sludge reduced toxicity of the contaminated soil for E. andrei, while application of compost increased toxicity. At each dilution level, total metal concentrations did not differ between treatments, but significant differences were found in reproduction (Fig. 2) in relation to water and CaCl2-extractable metal concentrations (Fig. 1). This suggests that reproduction was sensitive to changes in available metal concentrations (Becquer et al. 2005; Hobbelen et al. 2004; Spurgeon and Hopkin 1996). Considering all dilution levels, reproduction was inversely related with total (Table 3) and available metal concentrations (Table 4), suggesting that both concentrations affected reproduction of E. andrei. These results were confirmed by the PCA analysis, where reproduction (number of juveniles) and metal concentrations (total, water, and CaCl2-extractable) were grouped in the first component with opposite sign (Table 5).
The available metal concentrations were significantly decreased and reproduction significantly increased in the marble sludge amended soils compared to contaminated soil, although earthworm reproduction was around 80 % lower than in the control soil. These results suggest that other soil parameters like high pH (7.1–7.8) and fine soil texture (clay-loam) were not optimal for earthworm reproduction (Stein 1983; Van Gestel et al. 1992). The increase in reproduction when the earthworms were exposed to compost-amended soils at higher dilution with Lufa 2.2 (lower metal concentrations), could be due to the effects of the organic matter on soil structure (higher aeration and permeability). Reproduction was also affected by metal concentrations in the earthworms, which seems to agree with the findings of Van Gestel et al. (1993).
Metal concentrations in control earthworms were in agreement with values found by other authors in uncontaminated soils (Scaps et al. 1997; Van Gestel et al. 1993). The highest determination coefficients (r 2) were found for total soil concentrations (Table 3), suggesting that internal metal concentrations in the earthworms were mainly determined by total soil concentrations, probably due to ingestion of soil particles by the earthworms (Lanno et al. 2004). The PCA, including total metal concentration in soil and internal concentrations in the earthworms in the third Factor with the same sign, confirms this relationship (Table 5). Similar conclusions were drawn by other researchers (Veltman et al. 2007; Dai et al. 2004; Marinussen et al. 1997).
The metal concentrations in the earthworms decreased when decreasing total concentration in soil. The decrease was less steep for Zn than for Cd and Pb. Zn is an essential metal and its internal concentrations may be regulated to more constant levels by E. andrei (Peijnenburg et al. 1999; Van Gestel et al. 1993).
Although internal concentration is the most accurate measure of bioavailability (Lanno et al. 2004), our study suggests it cannot be used to estimate the effect of the amendments and subsequent dilution of the soils on the availability (water and CaCl2-extractable concentration) of metals. Other researchers have used earthworm concentrations to evaluate the metal-immobilizing efficiency of amendments in contaminated soils, and significant decreases in the concentrations before and after treatment were not found (Morgan et al. 2002; Grumiaux et al. 2010) Thus, to evaluate the effect of remediation on metal availability, other biological measures of bioavailability for earthworms (e.g., survival, growth, reproduction) or bioassays including organisms with different routes of exposure should be carried out.
In this study, we applied a dilution approach to assess the effect of a single amendment dose. The dilution approach was considered suitable as it did provide insight into the efficiency of the currently applied amendment doses for reducing metal bioavailability in contaminated soils. Alternatively, different amendment levels could have been applied to the contaminated soil. This would have provided insight in the amounts of the different amendments needed for decreasing metal solubility and bioavailability. In addition, it might have given a better insight into the possible effect of an amendment itself on the test organisms. Considering that the amendments at the recommended (maximum) dose did not show a clear reduction of metal bioavailability in the contaminated soil, testing different amendment doses probably would not have affected our conclusions. So, the dilution approach was suitable to answer our research question.
Conclusions
Organic and inorganic amendments applied had different effects on the survival and reproduction of the earthworm E. andrei in a metal-contaminated soil. Inorganic treatment with marble sludge caused metal (Cd, Pb, and Zn) immobilization, while the opposite effect was found for the amendment with organic compost, suggesting that care must be taken in the selection of organic amendments for soil remediation. No effects of organic and inorganic treatments on metal bioaccumulation in the earthworms were found, and earthworm internal concentrations were more related with total metal concentrations in soil than with available (water and 0.01 M CaCl2-extractable) concentrations. Other biological measures of bioavailability, in addition to bioaccumulation, must be used to estimate the effect of the amendments on the availability of metals in remediation tasks.
References
Adriano DC, Wenzel WW, Vangronsveld J, Bolan NS (2004) Role of assisted natural remediation in environmental cleanup. Geoderma 122:121–142
Alloway BJ, Thornton I, Smart GA, Sherlock JC, Quinn MJ (1998) Metal availability. Sci Total Environ 75:41–69
Baes CF Jr, Mesmer RE (1976) The hydrolysis of cations. Wiley, New York, New York
Becquer T, Dai J, Quantin C, Lavelle P (2005) Sources of bioavailable trace metals for earthworms from a Zn-, Pb-, and Cd-contaminated soil. Soil Biol Biochem 37:1564–1568
BOE (2005) Real decreto 824/2005, de 8 de julio, sobre productos fertilizantes. Bol Oficial Estado 171:25592–25669
Crommentuijn T, Doodeman CJAM, Doornekamp A, Van Der Pol JJC, Van Gestel CAM, Bedaux JJM (1994) Lethal body concentrations and accumulation patterns determine time-dependent toxicity of cadmium in soil arthropods. Environ Toxicol Chem 13:1781–1789
Crommentuijn T, Doornekamp A, Van Gestel CAM (1997) Bioavailability and ecological effects of cadmium on Folsomia candida (Willem) in an artificial soil substrate as influenced by pH and organic matter. Appl Soil Ecol 5:261–271
Dai J, Becquer T, Rouiller JH, Reversat G, Bernhard-Reversat F, Nahmani J, Lavelle P (2004) Heavy metal accumulation by two earthworm species and its relationship to total and DTPA-extractable metals in soils. Soil Biol Biochem 36:91–98
Escoto M, Fernandez J, Martín F (2007) Determination of phytotoxicity of soluble elements in soils, based on a bioassay with lettuce (Lactuca sativa L.). Sci Total Environ 378:63–66
Fountain MT, Hopkin SP (2004) A comparative study of the effects of metal contamination on Collembola in the field and in the laboratory. Ecotoxicology 13:573–587
Friels W, Lombi E, Horak O, Wenzel WW (2003) Immobilisation of heavy metals in soils using inorganic amendments in a greenhouse study. J Plant Nutr Soil Sci 166:191–196
Garrido F, Illera V, García-González MT (2005) Effect of the addition of gypsum- and lime-rich industrial by-products on Cd, Cu and Pb availability and leachability in metal-spiked acid soils. Appl Geochem 20:397–408
González V, Diez-Ortiz M, Simón M, Van Gestel CAM (2011) Application of bioassays with Enchytraeus crypticus and Folsomia candida to evaluate the toxicity of a metal-contaminated soil, before and after remediation. J Soils Sediment 11:1199–1208
Grumiaux F, Demuynck S, Schikorski D, Lemière S, Leprêtre A (2010) Assessing the effects of FBC ash treatments of metal-contaminated soils using life history traits and metal bioaccumulation analysis of the earthworm Eisenia andrei. Chemosphere 79:156–161
Haanstra L, Doelman P, Oude Vashaar JH (1985) The use of sigmoidal dose response curves in soil ecotoxicological research. Plant Soil 84:293–297
Hobbelen PHF, Koolhaas JE, van Gestel CAM (2004) Risk assessment of heavy metal pollution for detritivores in floodplain soils in the Biesbosch, The Netherlands, taking bioavailability into account. Environ Pollut 129:409–419
Hooda PS, Alloway BJ (1994) Changes in operational fractions of trace metals in two soils during two years of reaction time following sewage sludge treatment. Int J Environ Anal Chem 57:289–311
Hsu JH, Lo S-L (2000) Effect of dissolved organic carbon on leaching of copper and zinc from swine manure compost. Water Sci Technol 42:247–252
ISO (1999) Soil quality—inhibition of reproduction of Collembola (Folsomia candida) by soil pollutants. ISO 11267. International Standardization Organization, Geneva
Kiekens L (1995) Zinc. In: Alloway BJ (ed) Heavy metals in soils, 2nd edn. Blackie, Glasgow, UK, pp 284–305
Lanno R, Wells J, Conder J, Bradham K, Basta N (2004) The bioavailability of chemicals in soil for earthworms. Ecotoxicol Environ Saf 57:39–47
Loureiro S, Ferreira ALG, Soares MVM, Nogueira AJA (2005) Evaluation of the toxicity of two soils from Jales Mine (Portugal) using aquatic bioassays. Chemosphere 61:168–177
Loveland PJ, Whalley WR (1991) Particle size analysis. In: Smith KA, Mullis CE (eds) Soil analysis: physical methods. Marcel Dekker, New York, pp 271–328
Marinussen MPJC, van der Zee SEATM, de Haan FM (1997) Cu accumulation in Lumbricus rubellus under laboratory conditions compared with accumulation under field conditions. Ecotoxicol Environ Saf 36:17–26
Melgar-Ramírez R, González V, Sánchez JA, García I (2012) Effects of application of organic and inorganic wastes for restoration of sulphur-mine soil. Water Air Soil Pollut 223:6123–6131
Mench M, Bussiere S, Vangronsveld J, Manceau A (2003) Progress in remediation and revegetation of the barren Jales gold mine spoil after in-situ treatments. Plant Soil 249:187–202
Mench M, Renella G, Gelsomino A, Landi L, Nannipieri P (2006) Biochemical parameters and bacterial species richness in soils contaminated by sludge-borne metals and remediated with inorganic soil amendments. Environ Pollut 144:24–31
Morgan AJ, Evans M, Winters C, Gane M, Davies MS (2002) Assaying the effects of chemical ameliorants with earthworms and plants exposed to a heavily polluted metalliferous soil. Eur J Soil Biol 38:323–327
Morillo J, Usero J, Garcia I (2004) Heavy metal distribution in marine sediments from the southwest coast of Spain. Chemosphere 58:431–442
OECD (2004) Guideline for the testing of chemicals no. 222, earthworm reproduction test (Eisenia fetida/Eisenia andrei). Organization for Economic Cooperation and Development, Paris
Owojori OJ, Reinecke AJ, Rozanov AB (2008) Effects of salinity on partitioning, uptake and toxicity of zinc in the earthworm Eisenia fetida. Soil Biol Biochem 40:2385–2393
Peijnenburg WJGM, Baerselman R, de Groot AC, Jager T, Posthuma L, van Veen RPM (1999) Relating environmental availability to bioavailability: soil-type-dependent metal accumulation in the oligochaete Eisenia andrei. Ecotoxicol Environ Saf 44:294–310
Rhoades JD (1982) Cation exchange capacity. In: Page AL (ed) Methods of soil analysis, Part 2. American Society of Agronomy, Madison, WI, pp 149–157
Saar RA, Weber JH (1980) Lead (II)-fulvic acid complexes. Conditional stability, and implications for lead (II) mobility. Environ Sci Technol 14:877–880
Scaps P, Grelle C, Descamps M (1997) Cadmium and lead accumulation in the earthworm Eisenia fetida (Savigny) and its impact on cholinesterase and metabolic pathway enzyme activity. Comp Biochem Physiol 116:233–238
Sierra M (2005) Niveles de metales pesados y elementos asociados en suelos de la provincia de Almería. Parámetros que los afectan y riesgo de contaminación. Ph.D. Thesis, Univ. Granada, Spain
Sierra M, Martínez FJ, Aguilar J (2007) Baselines for trace elements and evaluation of environmental risk in soil of Almería (SE Spain). Geoderma 139:209–219
Simón M, Dorronsoro C, Ortiz I, Martín F, Aguilar J (2002) Pollution of carbonate with in a Mediterranean climate due to a tailing spill. Eur J Soil Sci 53:321–330
Spurgeon DJ, Hopkin SP (1996) Effects of variations in the organic matter content and pH of soils on the availability and toxicity of zinc to the earthworm Eisenia fetida. Pedobiologia 40:80–96
Spurgeon DJ, Hopkin SP, Jones DT (1994) Effects of cadmium, copper, lead and zinc on growth, reproduction and survival of the earthworm Eisenia fetida (savigny): assessing the environmental impact of point-source metal contamination in terrestrial ecosystems. Environ Pollut 84:123–130
Stein JK (1983) Earthworm activity: a source of potential disturbance of archaeological sediments. Am Antiq 48:277–289
Stevenson FJ (1992) Humus chemistry. Genesis, composition and reactions. Wiley, New York, 1992
U.S. Salinity Laboratory Staff (1954) Diagnosis and improvement of saline and alkali soils. Handbook. US Department of Agriculture, Washington DC
Van Gestel CAM, Van Dis WA, Van Breemen EM, Sparenburg PM (1989) Development of a standardized reproduction toxicity test with the earthworm species Eisenia andrei using copper, pentachlorophenol and 2,4-dichloroaniline. Ecotoxicol Environ Saf 18:305–312
Van Gestel CAM, Dirven-Van Breemen EM, Baerselman R (1992) Influence of environmental conditions on the growth and reproduction of the earthworm Eisenia andrei in an artificial soil substrate. Pedobiologia 36:109–120
Van Gestel CAM, Dirven-van Breemen EM, Baerselman R (1993) Accumulation and elimination of cadmium, chromium and zinc and effects on growth and reproduction in Eisenia andrei (Oligochaeta, Annelida). Sci Total Environ 134(Suppl 1):585–597
Vangronsveld J, Ruttens A, Colpaert J, van der Lelie D (2000) In situ fixation and phytostabilization of metals in polluted soils. In: Luo YM et al (eds) Proc. Int. Conf. Soil Remediation Soil-Rem. Chinese Acad. Sci., Hangzhou, China, pp 262–267
Veltman K, Huijbregts MAJ, Vijver MG, Peijnenburg WJGM, Hobbelen PHF, Koolhaas JE, Van Gestel CAM, Van Vliet PCJ, Hendriks AJ (2007) Metal accumulation in the earthworm Lumbricus rubellus. Model predictions compared to field data. Environ Pollut 146:428–436
Williams DE (1948) A rapid manometric method for determination of carbonate in soils. Soil Sci Soc Am Proc 13:127–129
Zhang JL, Liu JG, Li C, Nie YF, Jin YY (2008) Comparison of the fixation of heavy metals in raw material, clinker and mortar using a BCR sequential extraction procedure and NEN7341 test. Cem Concr Res 38:675–680
Acknowledgments
This study was funded by the projects: CTM2009-07921 (Science and Innovation Ministry of Spain and FEDER), and P07-RNM-03303 (Andalusian Government and FEDER). The first author expresses her gratitude to the Innovation and Science Ministry of Spain, for a FPI fellowship and supported exchange visits in VU University Amsterdam. The Department of Ecological Science at VU University is thanked for the opportunity to work there and hospitality during my stay.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
González, V., Díez-Ortiz, M., Simón, M. et al. Assessing the impact of organic and inorganic amendments on the toxicity and bioavailability of a metal-contaminated soil to the earthworm Eisenia andrei . Environ Sci Pollut Res 20, 8162–8171 (2013). https://doi.org/10.1007/s11356-013-1773-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-013-1773-z