Abstract
This field study investigated the phytoremediation potential of two arsenic (As) hyperaccumulating fern species, Pityrogramma calomelanos var. austroamericana and Pteris vittata over 27-month duration at a disused As-contaminated cattle-dip site located at Wollongbar, NSW, Australia. Ferns planted in January 2009 were harvested following 10, 22 and 27 months of growth. A detailed soil sampling was undertaken in June 2009 (initial, n = 42 per plot) and limited sampling in April 2011 (after 27 months, n = 15 per plot) to measure total and phosphate-extractable As concentrations in soil at 0 − 20-, 20 − 40- and 40 − 60-cm depths. The choice of the limited number of samples was considered sufficient to estimate the changes in soil As concentration following phytoremediation based on a geostatistical model. The average frond dry biomass, As concentration and As uptake were significantly (P < 0.001 − 0.05) greater in P. calomelanos var. austroamericana than P. vittata, at all three harvests (1.6 − 4.3, 1.3 − 1.5 and 2.2 − 5.7 times, respectively). After 27-months of growth, P. calomelanos var. austroamericana removed 8,053 mg As (i.e. cumulative over three harvests) in plot B (25.4 kg As ha−1) that was 2.65 times higher than that depleted by P. vittata (3,042 mg As in plot A (9.7 kg As ha−1)). The cumulative frond As uptake data of the two fern species revealed that P. calomelanos var. austroamericana extracted 1.7 − 3.9 % and P. vittata removed 0.53 − 1.5 % of total As from soil at three depths. However, for the surface (0 − 20 cm) and subsurface (40 − 60 cm) layers, the (post-experiment) soil As data indicated that total As concentration in soil was reduced by 49 and 63 % (P < 0.05), respectively, using P. calomelanos var. austroamericana; and 17 and 15 % (P > 0.05), respectively, by P. vittata. Our results show that phytoremediation time based on observed changes in soil As based on limited sampling is not reliable; hence, it is recommended that the frond As uptake should be considered in order to evaluate the phytoremediation efficiency of the two fern species at the experimental site. Using As uptake of the two fern species, we estimate that with P. calomelanos var. austroamericana it would take 55 − 125 years to decrease mean total As content below the ecological investigation level (20 mg kg−1) in the surface and subsurface soils, whereas with P. vittata 143 − 412 years would be required to achieve this target.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pesticides containing arsenic (As) were frequently used to control ticks in cattle from the early 1900s to 1955 at cattle-dip sites, now disused across large expanses of New South Wales (NSW), Australia (Smith et al. 1998, 2006). The repeated application of arsenical pesticides (as sodium arsenite) at these sites has inadvertently led to the contamination of surrounding soils with elevated (1,000 to 3,000 mg kg−1) and highly variable concentrations of As (Smith et al. 1998; Kimber et al. 2002; Niazi et al. 2011a). Considering that As is highly toxic (Adriano 2001; Mandal and Suzuki 2002), and present in readily and/or potentially available forms in soils adjoining the cattle-dip sites (Smith et al. 2006; Niazi et al. 2011b), immediate attention is required for the restoration of these sites using a suitable remediation strategy.
Several reports suggest that phytoremediation of As-contaminated sites using As-hyperaccumulating ferns is a viable and efficient in situ remediation technology (Gonzaga et al. 2006; Kertulis-Tartar et al. 2006; Vangronsveld et al. 2009; Niazi et al. 2011c). Many ferns in the Pteris genus, e.g. Pteris vittata, Pteris longifolia, Pteris cretica and Pteris umbrosa, have been reported as As-hyperaccumulators (Ma et al. 2001; Zhao et al. 2002; Wei et al. 2007), few non-Pteris fern species have also been identified as As-hyperaccumulators including, e.g. Pityrogramma calomelanos (silver fern) and P. calomelanos var. austroamericana (gold dust fern) (Francesconi et al. 2002; Kachenko et al. 2007). P. vittata (Chinese brake fern) is a well-known As-hyperaccumulator, which can accumulate >3,500 mg kg−1 As in fronds on a dry weight (DW) basis (Ma et al. 2001; Tu and Ma 2002; Kertulis-Tartar et al. 2006). This fern species has been extensively used in many pot studies to investigate its potential for the phytoextraction of As from As-contaminated or As-spiked soils (Cao et al. 2003; Caille et al. 2004; Fayiga and Ma 2006; Gonzaga et al. 2007, 2008, 2009; Shelmerdine et al. 2009). However, a few field studies have evaluated the phytoextraction efficiency of these fern species for the remediation of As-contaminated sites. For example, in a field study at a copper–chromium–arsenate (CCA)-contaminated site, Kertulis-Tartar et al. (2006) reported that P. vittata would take 8 years to reduce the mean total As content in the surface (from 190 to 140 mg kg−1, 0–15 cm) soil below 3.7 mg kg−1, the limit set by Florida Department of Environmental Protection for the remediation of As-contaminated sites. The remediation estimate was based on the reduction in the mean total soil As calculation after a 24-months phytoremediation period during which repeated frond harvests were performed. Similarly, in another field study by Salido et al. (2003), P. vittata was reported to accumulate As up to 2,740 mg As kg−1 DW and the authors estimated that 8 years would be required to reduce the acid-extractable As concentration in soil from a mean value of 82 to 40 mg As kg−1, the limit set by EPA. Conversely, in a field study Reichmann et al. (2004) indicated that the performance of P. vittata to accumulate As from an As-contaminated soil decreased after subsequent frond harvests and the fern species did not regrow well in the field. They attributed this to the inefficiency of P. vittata to compete with weeds and suggested that this fern species might not have practical applications for the phytoremediation of As in Australian conditions.
The fern, P. calomelanos var. austroamericana—a lesser-known As-hyperaccumulating fern species—has been recently identified in Australia (Kachenko et al. 2007). This species accumulated up to 16,413 mg kg−1 DW As in fronds when exposed to 500 mg kg−1 As concentration under glasshouse conditions (Kachenko et al. 2007). The present study is an extension of our earlier work (Niazi et al. 2011c) where we compared the phytoextraction potential of P. vittata and P. calomelanos var. austroamericana after a short period (10 months), at a highly variable As-contaminated cattle-dip site in NSW, Australia. Prior to this study, no field experiment has been performed to investigate the phytoextraction efficiency of P. calomelanos var. austroamericana. The short-term experimental data from the earlier study revealed that P. calomelanos var. austroamericana produced 1.6 times greater frond dry biomass and accumulated twofolds more As than that of P. vittata (Niazi et al. 2011c). From the plant As uptake data of the two fern species, it was predicted that P. calomelanos var. austroamericana would take approximately (100 years) half the time required by P. vittata (≥200 years) to reduce the total soil As content (at 0–60-cm depth) below 20 mg kg−1, the ecological investigation level (EIL) limit of As in soil in Australia (NEPM 1999). It was suggested that under field conditions at the experimental site P. calomelanos var. austroamericana performs better than P. vittata in extracting soil As. However, long-term data, multiple frond harvests, are required to evaluate the phytoextraction efficiency of these fern species and to estimate the time required to remediate the As-contaminated site below the Australian EIL value.
This long-term field study compared the phytoremediation potential of P. calomelanos var. austroamericana and P. vittata after three harvests during a 27-month period. Soil samples were analysed for total As concentration after three frond harvests to assess the change in soil As, at the disused dip site. The specific objectives were (1) to evaluate the phytoremediation efficiency of P. calomelanos var. austroamericana against the well-known As-hyperaccumulator, P. vittata after three harvests of fronds; (2) to determine the remediation time for the two fern species from As uptake data after multiple harvests; and (3) to compare the As uptake remediation time with the estimated time based on limited (post-experiment) soil sampling data.
Materials and methodology
Fern species and experimental site
Two As-hyperaccumulating fern species, P. vittata and P. calomelanos var. austroamericana were used in this study. P. vittata was obtained from the Randwick City Council Nursery (Sydney, Australia) and P. calomelanos var. austroamericana was propagated from spores in the glasshouse.
The experimental site is a disused cattle-dip site at Wollongbar Agricultural Institute in northern NSW, Australia (28 °49′12.0″ S, 153 °23′49.2″ E) where As-based pesticides were first used in 1953 to treat ticks in the livestock. The soil properties at the site have been described in the earlier study by Niazi et al. (2011c).
Ferns transplantation and experimental set up
In January 2009, 42 uniform (5–6 fronds stage) plants of P. vittata and P. calomelanos var. austroamericana were transplanted in the field into hand-dug holes (~10 cm deep × 10 cm wide) in two separate equal sized (3.15 m2) plots (referred to as Plot A and Plot B, respectively). The ferns were planted in a systematic design on the nodes of a 30 × 30 cm grid, leaving a buffer strip of 1 m between the two plots (details for the experimental design lay-out are available in Niazi et al. (2011c)).
A drip irrigation system was installed to irrigate ferns for 5 min, twice a day at 12-h intervals using a timer. A black nylon weed mat was spread within the experimental area to reduce the weed growth, and a shade cloth was erected over the experimental area to protect the ferns from excessive sunlight. A slow-release fertiliser (4–5 g plant−1, Osmocote® plus), containing N/P/K of 16:4.4:10 w/w% was used to supply the ferns with basic plant nutrients (Niazi et al. 2011c).
Frond harvests
During the 27-month experimental period, fronds of both fern species were harvested in December 2009, November 2010 and April 2011after 10, 22 and 27 months of transplanting, respectively. At each harvest, young fronds and fiddleheads were left to facilitate re-growth for successive frond harvests. The harvested fronds were thoroughly washed using a three-step washing procedure (tap water-0.1 M HCl-deionised water), and dried in a fan-forced oven at 70°C for 72 h (Niazi et al. 2011c). The dry matter yield was recorded and the samples were ground (<1 mm) and digested in a mixture (1:1) of concentrated HNO3 and HClO4 acids (Miller 1998) for As analysis.
Soil sampling
Soil sampling was undertaken at two stages, a detailed sampling in June 2009 (initial, after 5 months of fern transplantation) and limited sampling in April 2011(after 27 months of fern growth). Soil samples were taken using a hand-driven stainless steel soil corer to a depth of 60 cm and divided into three sections to obtain soil samples for 0–20-, 20–40- and 40–60-cm depths. In June 2009, soil samples (n = 42 per plot) were extracted around each fern in both Plot A and Plot B, and 18 samples were also collected from the buffer strip between the two plots in a systematic design, as described elsewhere (Niazi et al. 2011c). In April 2011 after 27 months of experimental period, soil samples (n = 15 per plot) were taken around 15 randomly selected ferns in each of the experiments plots, the choice of the number of samples was considered sufficient to assess the changes in soil As content following phytoremediation based on a linear mixed (geostatistical) model described elsewhere (Niazi et al. 2011a).
Soil samples were air-dried and sieved (<2 mm). Sub-samples (<200 μm) were oven-dried at 105°C overnight and digested in a mixture of acids (HF, HCl, HNO3, H2SO4 and HClO4) to determine the total As concentration using the method described by Huang and Fujii (1996). The samples were extracted using 0.5 M (KH2PO4) phosphate solution to measure the plant available (bioavailable) As in soils (Niazi et al. 2011c).
The soil and plant digests were analysed for As using an inductively couple-plasma atomic emission spectrometer (ICP-AES, Varian® Vista AX CCD). Reagent blanks (in triplicate) and National Institute of Standards and Technology reference plant material (pine needles no. 1575) and soils (Montana 2710 and San Janquin 2709) were used for quality assurance to check the efficiency of the digestion procedures and ICP-AES analysis. Recovery of As was within ±10 % of the recommended values. The relative standard deviation (RSD) was <2 % for ICP-AES analyses. Three reagent blanks were included with phosphate extraction of the soil samples and As in the extracts was analysed using a Varian® Spectra 220Z hydride-generation atomic absorption spectrometer (HG-AAS). The RSD was <2 % for the HG-AAS analysis.
Statistical analysis
Geostatistical analyses, including spatial maps of total As in soil, spatial analysis of variance (ANOVA) and analysis of covariance (ANCOVA) were performed using R version 2.10.1 (R Development Core Team 2008) as described in Niazi et al. (2011c). Spatial ANOVA was used to differentiate the soil (total and phosphate-extractable) As concentrations between Plot A and Plot B. ANCOVA was used to compare the performance of P. vittata and P. calomelanos var. austroamericana in terms of their dry matter yield, frond As concentration and As uptake. The bioconcentration factors (BFs; concentration of As in fern fronds to concentration of As in soil) were also calculated based on the total (BFT) and phosphate-extractable (BFP) soil As concentrations. The significant differences between the mean BFs of the two fern species were evaluated using t test.
To test significance difference between the mean soil As concentrations (total and phosphate-extractable at three depths) determined in June 2009 (initial) and April 2011 (after 27 months), a 95 % confidence interval was calculated around the mean total As values using a geostatistical model, as discussed in detail by Niazi et al. (2011a).
Results and discussion
Both P. calomelanos var. austroamericana and P. vittata established successfully in the experimental plots at the studied field site, and showed no obvious plant toxicity symptoms during the 27-month experimental period. After each frond harvest, P. calomelanos var. austroamericana ferns were observed to re-establish more rapidly and successfully than P. vittata. More fiddleheads and juvenile fronds were observed in P. calomelanos var. austroamericana than in P. vittata (data not shown here). During the 27-month experimental period, we observed that P. vittata was less efficient in competing with weeds as it has a less dense canopy as compared to P. calomelanos var. austroamericana.
Frond harvests
Frond dry matter yield
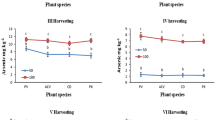
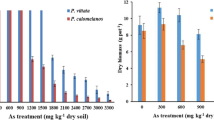
At all three harvests, the dry biomass yield of P. calomelanos var. austroamericana was higher than that of P. vittata (ANCOVA; P < 0.001 − 0.05; Table 1). The average frond dry biomass was 130, 151 and 68 g plant−1 for P. calomelanos var. austroamericana as compared to 81, 39 and 16 g plant−1 for P. vittata at the 10-, 22- and 27-month harvest, respectively (Table 1). The higher frond dry biomass produced at the second (22-month) harvest in P. calomelanos var. austroamericana was ascribed to slightly longer growing time (12 months) at this harvest as compared to the first and third harvests. Conversely, P. vittata showed a decreasing trend in the frond dry matter yield from the first to third harvest, which is in agreement with our field observations, suggesting that P. vittata was less suited to the field conditions at the site. The 27-month harvest yielded the least frond biomass in both fern species, due to the shortest growth period (5-month) for this harvest. Over the experimental (phytoremediation) period of 27 months, P. calomelanos var. austroamericana yielded 12,147 g total frond dry matter in Plot B (38.6 ton ha−1) which is substantially greater than 5,151 g of frond dry biomass observed for P. vittata in Plot A (16.4 ton ha−1).
Arsenic concentration in ferns
For all three harvests, the mean frond As concentration was significantly (ANCOVA; P < 0.001 − 0.05) higher in P. calomelanos var. austroamericana (887, 423 and 581 mg kg−1 DW, respectively) than the corresponding concentration in P. vittata (674, 292 and 401 mg kg−1 DW; Table 1). Arsenic concentration in fronds of both fern species was highly variable at three harvests (Table 1), which was attributed to the varying As concentration in the soil at the site as described earlier (Niazi et al. 2011c) and differences in re-growth of different plant species after each harvest.
Fern As uptake
The average frond As uptake at all three harvests was significantly (ANCOVA; P < 0.001 − 0.05) higher in P. calomelanos var. austroamericana (124, 64 and 40 mg plant−1, respectively) than in P. vittata (27, 14 and 7 mg plant−1; Table 1). P. calomelanos var. austroamericana fronds removed 8,053 mg As over three harvests from soil in Plot B (approximately 25.4 kg As ha−1) after 27 months of growth, which is 2.65 times greater than the total As extracted by P. vittata in Plot A (3,042 mg As; approximately 9.7 kg As ha−1).
Fern bioconcentration factors
For all three harvests, the average BFs (based on the total, BFT and phosphate-extractable, BFP soil As) were significantly (P < 0.001; t test) higher in P. calomelanos var. austroamericana (BFT = 0.82 − 1.30 and BFP = 13 − 21) than in P. vittata (BFT = 0.45 − 0.80 and BFP = 8 − 14). In earlier pot studies, P. vittata has been reported to possess BF generally >10 (Ma et al. 2001; Tu et al. 2002; Zhao et al. 2002; Xie et al. 2009), where soluble form of As was added to experimental soils and equilibrated (aged) for a relatively short period of time. Wei et al. (2006) reported that ageing of As in soil at an Au-mineralization site reduced its plant (fern) availability and decreased the mean BF value for P. vittata to 0.9. In the present study, the decrease in the BF value for both fern species over three harvests was attributed to the reduced bioavailability of As at the site. The bioavailable content of As was relatively small due to the high As sorption capacity of the soil at the studied site with the presence of high free Fe concentration (16 %) and low soil pH (4.80). It has been suggested in earlier studies (Wei and Chen 2006; Wei et al. 2006; Niazi et al. 2011c) that BF values based on the extractable or bioavailable soil As should be considered under field conditions, and the results from this long-term study support this observation.
The higher total frond dry biomass after three successive harvests observed for P. calomelanos var. austroamericana is in agreement with our previous results from the short-term study where 1.61 times greater frond biomass was reported for P. calomelanos var. austroamericana than that of P. vittata after 10 months of growth (Niazi et al. 2011c). However, the difference between the total frond dry matter yield of the two fern species became more pronounced over the three harvests, with significantly (2.36 times) larger cumulative frond biomass observed for P. calomelanos var. austroamericana as compared to P. vittata. The long-term (27 months) data support our earlier suggestion that the subtropical field conditions at this site are better suited for P. calomelanos var. austroamericana than P. vittata. A decreasing trend in the frond biomass yield of P. vittata was observed which could be partly contributed to competition of weeds. This concurs with Reichmann et al. (2004), who reported that the phytoextraction efficiency of P. vittata might decline due to unfavourable growing conditions, such as drought and weeds under field conditions.
Shelmerdine et al. (2009) evaluated the phytoextraction efficiency of P. vittata grown on a large number of As-contaminated soils over a period of 9 months under glasshouse conditions. They reported that the frond dry matter yield of P. vittata was reduced in the second and third frond harvests; each harvest was performed at 3-month interval. This was more noticeable in ferns grown on highly contaminated soils (300 to 2,330 mg As kg−1 soil), from mine-spoil waste and smelting processes. The authors suggested that P. vittata was not suitable for remediation of these soils (constituting primary As minerals, e.g. arsenopyrite) with high total soil As content and less labile soil As pool. In the current study, the poor performance of P. vittata in the field may be due to low bioavailability of As in soil despite of being high total soil As concentration at the site (Table 2), whereby As was strongly sorbed onto Fe oxide minerals. This could have caused a reduction in frond biomass in P. vittata over the experimental period at each successive harvest (see Table 1). Conversely, P. calomelanos var. austroamericana showed a consistent growth pattern and produced relatively high frond biomass over three harvests, suggesting that this fern species was better suited for remediation of the studied site.
Our data show that the concentration of As in fronds of P. calomelanos var. austroamericana and P. vittata at all three harvests was lower than the values reported in previous field studies (Salido et al. 2003; Kertulis-Tartar et al. 2006). Kertulis-Tartar et al. (2006) reported a mean As concentration of 3,186 mg kg−1 DW in fronds of P. vittata growing on a CCA-contaminated site in soil with alkaline pH (7.4) and sandy texture (88 % sand). The lower frond As concentration in the two fern species observed in the present study could be attributed to the high As sorption capacity of the soil due to its high free Fe content and low pH, and long ageing time (over 40 years) for As at this site (Niazi et al. 2011c). Arsenic sorption on Fe oxyhydroxides increases at low soil pH and in the presence of high free Fe, thus reducing the bioavailability of As in soil (Smith et al. 1998; de Mello et al. 2007; Pan et al. 2010; Niazi et al. 2011b). This was evident from the lower phosphate-extractable soil As concentration (≤13.4 % of total soil As) in the plots at the studied site. In addition, the low BFT of P. calomelanos var. austroamericana (≤1.3) and P. vittata (≤0.80) also affirm our earlier explanation that phytoavailability of As was low at the site mainly because it was bound with the Fe oxides.
P. calomelanos var. austroamericana showed a stronger trend in As uptake over the subsequent frond harvests and depleted twice as much As from the soil than that of P. vittata, suggesting that this fern species was more suitable to extract As from soil at the site.
In the current study, the difference in phytoextraction performance of P. calomelanos var. austroamericana and P. vittata could also be attributed to disparities in physiological mechanisms involved in the detoxification of As in frond tissue. For P. vittata, the production of low-molecular-weight thiols has been reported to detoxify As in the ferns while they were exposed to As-rich environments or contaminated soils (Cai et al. 2004). Whereas in P. calomelanos var. austroamericana, the formation of arsenide–sulfide (AsIII–S2−) compounds may be responsible for the biochemical reduction of AsV to AsIII during As transportation from root to shoot, which could play a pivotal role in the detoxification of As in this species (Kachenko et al. 2010). Further studies are warranted to elucidate if these physiological functions are responsible for the different uptake patterns observed in our study.
Total and phosphate-extractable soil As concentrations for three depths
Total As concentration (initial and after 27 months) in soil at the two experimental plots was spatially heterogeneous (Fig. 1). Spatial variability pattern in soil As at this site has been previously described in a geostatistical context (Niazi et al. 2011a); soil As concentration significantly (P < 0.05) increased toward the cattle-dip, conferring a spatial trend over As content in soil adjacent to the studied cattle-dip. This variability in soil As concentration was reported to be associated with the cattle dipping process, disposal of the As-contaminated dip sediment in the vicinity of the cattle dip and pumping-out of the dipping fluid from the dip bath (Kimber et al. 2002; Niazi et al. 2011a).
The spatial variability maps of As showing remediation trend in the total soil As concentration for three depths in plot A (P. vittata) and plot B (P. calomelanos var. austroamericana) of the experimental area; a, c, e initial total As concentration in soil determined in June 2009 after 5 months of fern transplanting; b, d, f final As concentration in soil measured in April 2011, after 27 months of fern growth. The two experimental plots were separated by a buffer strip (dotted lines) as shown in the figure above. The data presented here correspond to the total soil As concentration in Table 2
The mean total soil As concentration in the topsoil after 27 months of fern growth was observed to be reduced by 17 % in Plot A (P. vittata plot; P > 0.05 (909 − 754 mg kg−1)) and 49 % in Plot B (P. calomelanos var. austroamericana plot; P < 0.05 (753 − 385 mg kg−1); Table 2; Fig. 1a, b). For the second layer (20 − 40 cm), there was no decline in average total soil As concentration in Plot A, while 11 % (P > 0.05; 396 − 352 mg kg−1) decrease was depicted in Plot B (Table 2; Fig. 1c, d). In Plot A and Plot B, the average total As content in the soil at the lowest (40 − 60 cm) depth was reduced by 15 % (P > 0.05; 323 − 276 mg kg−1) and 63 % (P < 0.05; 324 − 121 mg kg−1), respectively, after 27 months of remediation period (Table 2; Fig. 1e, f). These results indicate that greater amount of soil As was reduced by P. calomelanos var. austroamericana than using P. vittata at the site. The decrease in total soil As concentration after the 27-month period for the two experimental plots is also evident in Fig. 1, particularly for the surface and the deepest layers in Plot B. The variability in the total soil As concentration appeared to be slightly reduced in both plots at all three depths (Fig. 1).
After 27 months, the mean phosphate-extractable As concentration in soil showed a significant reduction of 28 % (P < 0.05) in Plot B for the lowest depth (40 − 60 cm); however, negligible (P > 0.05) change was observed in the top two depths (see Table 2). Conversely, the average phosphate-extractable As concentration in Plot A significantly (P < 0.05) increased for the first two depths, and reduced by 8 % (P > 0.05) for the lowest (40 − 60 cm) layer.
The results demonstrate that both fern species decreased the concentration of total As in soil at the studied site from the surface (0 − 20 cm) and the lowest (40 − 60 cm) layers, indicating that fern roots of both fern species extracted As from the surface and subsurface soils. Our findings are in agreement with those of Kertulis-Tarter et al. (2006), who reported 26 and 12 % decline in the total soil As concentrations after 24 months for 0 − 15 and 30 − 60 cm depths, respectively, using P. vittata. They reported a maximum decrease (43 %) in the total soil As concentration at 15 − 30-cm depth. In the current study, no reduction was observed for the total soil As content at the second (20 − 40 cm) depth in Plot A (P. vittata); and only 11 % As was reduced in the soil in Plot B by P. calomelanos var. austroamericana at this depth. Immediately, it is unclear what factors or processes caused minimal or no depletion of As in soil with both fern species at the second depth. Further detailed examinations are required to elucidate the root distribution, density and morphology of these fern species grown at the field site.
In the As-hyperaccumulating fern species, excretion of root exudates, such as low-molecular-weight organic acids have been reported to be associated with the solubilization of As in the rhizosphere soil, thereby increasing the available As content in soil for fern uptake (Gonzaga et al. 2006, 2009). In the current study, both P. calomelanos var. austroamericana and P. vittata roots can express a similar mechanism to enhance the available As concentration in soil for plant (fern) uptake. The differences in As accumulation observed between the two fern species, with P. calomelanos var. austroamericana showing greater As accumulation, could be ascribed to the differences in root density and mass between the two species and variability in As solubilization mechanisms for the two species in the rhizosphere soil cannot be discounted. Future studies are warranted to elucidate the rhizosphere characteristics of these fern species.
Time estimation for remediation of the experimental site
The remediation time for the study site was estimated using the (cumulative) As uptake data of the two fern species obtained after three successive frond harvests over 27 months of the experimental period (see “Fern As uptake” section), as described in our earlier study (Niazi et al. 2011c). It was assumed that the ferns would remove equal amount of As from each soil depth. Over the 27 months of phytoremediation period, P. calomelanos var. austroamericana extracted 1.7 − 3.9 % of total As which was 2.6 − 3.3 times more than the As extracted by P. vittata (0.53 − 1.5 %). Using the As uptake data of both fern species, we predict that with P. calomelanos var. austroamericana, it would take 55 − 125 years to remediate soil (at three depths) to bring the total As level below the EIL value of 20 mg kg−1 (40 kg As ha−1), whereas P. vittata would require 143 − 412 years (for surface and the deepest layers) to achieve the same target. These remediation estimates are in agreement with those made in our short-term study (Niazi et al. 2011c) where we used (10 months) As uptake data of the two fern species to assess remediation time. From the short-term data, it was predicted that P. calomelanos var. austroamericana would take half the time (~100 years) than P. vittata (>200 years) to bring the total As content (at 0 − 60 cm depth) below the EIL value of 20 mg kg−1.
The remediation time was also estimated for the site based on the decrease in the mean total soil As concentration in the two experimental plots after 27 months of phytoremediation (i.e. Mean reduction in total soil As after 27 months (mg kg−1) = Initial As − Final As concentration after 27 months). We estimated that P. calomelanos var. austroamericana (Plot B) would take 5.5 years for the top soil (0 − 20 cm) and 4 years for the lowest subsoil (40 − 60 cm) to reduce the total As content below the EIL limit of 20 mg kg−1 for total As in Australia (NEPM 1999). Conversely, using P. vittata it would take 13 years for the top soil (0 − 20 cm) and 15 years for the soil at the lowest depth (40 − 60 cm) to decline the total As content below 20 mg kg−1. At the 20 − 40 cm depth, remediation time-estimation was only possible for P. calomelanos var. austroamericana plot (Plot B), as no decrease in soil As concentration occurred in Plot A using P. vittata (see Table 2). Our calculations suggest that 20 years would be required to decrease total soil As at 20 − 40-cm layer using P. calomelanos var. austroamericana.
The remediation time estimated by measuring change in post-experiment total soil As concentration and using the fern As uptake data showed considerable differences. These disparities in remediation time were attributed to the large differences between the soil As decrease and the fern As removal over 27 months of the experimental duration. The mass balance of total As was calculated by using the cumulative fern As uptake data from three frond harvests and the total reduction in soil As content observed for three depths following 27 months of phytoremediation period (Kertulis-Tartar 2005). The fern As uptake data showed that 9.7 kg As ha−1 was extracted from the soil by P. vittata (Plot A) and 25.4 kg As ha−1 was removed using P. calomelanos var. austroamericana (Plot B). In contrast, a total of 404 kg As ha−1 was calculated to have been depleted from the soil in Plot A (P. vittata; at 0 − 20- and 40 − 60-cm depths) and 1,230 kg As ha−1 in Plot B (P. calomelanos var. austroamericana; for three depths) after 27 months of experiment. The mass balance calculations of As show that the measured reduction in soil As content in Plot A (404 kg ha−1) and Plot B (1,230 kg ha−1) was 42 and 48 times higher than the amount of As extracted by P. vittata (9.7 kg As) and P. calomelanos var. austroamericana (25.4 kg As) in their respective plots. Our mass balance calculations of soil–plant As concur with Kertulis-Tartar (2005), who reported that the reduction in soil As (1,444 g As, for 0 − 15, 15 − 30 and 30 − 60-cm depths) was 55 times greater than the total As (26.4 g) removed by P. vittata over 24 months of the experimental period at a CCA-contaminated site. The author ascribed this discrepancy in mass balance of As to the highly heterogeneous soil As concentration at the studied site, suggesting that slight variations in sampling places might have resulted in large differences in soil As concentration. In the current study, the concentration of total As in soil was extremely variable (Fig. 1) at the dip site, with As concentrations varying on a sub-meter scale (Niazi et al. 2011a). Additionally, limited number of soil samples taken after 27 months of phytoremediation may not be representative enough to reflect the actual change in soil As concentration. It is most likely that minor variations in (post-experiment) soil sample locations have caused large differences (errors) in the soil As concentration measurements and subsequently in As removal data.
It is evident from our calculations that estimation of the As removal rate and/or remediation time based on the observed changes in the post-experiment soil As concentrations is not a reliable option. Therefore, the As uptake data of the two fern species should be used for calculating the removal rate of As from the soil in order to predict the remediation time; the post-experiment soil As concentration measurements could only be useful to indicate As-removal trend of both fern species to extract As from the soil at various depths, as it was observed in the current study. However, soil analysis for As from the three depths provided useful information about relative changes in soil As in the two experimental plots and this information could be used in conjunction with plant uptake data to further refine the estimates for phytoremediation time required at the site. Remediation calculations on plant uptake data are more reliable than the soil As based data because at this site there is probably little or no loss of As from leaching as As is strongly adsorbed onto Fe oxides and any changes in soil As concentration are likely to be due to plant uptake of As.
The remediation estimates are based on the assumption that both fern species would show the same As-accumulation trend to deplete As from soil over time, as it was observed during this study for the 27-month period after three frond harvests. Our remediation assessments are based on the mean total soil As concentration and they may result in the underestimation or overestimation of the remediation time at the site. Since As concentrations in soil at the studied site are highly variable (Fig. 1); therefore, it is expected that some areas in the two experimental plots would take shorter period of time before As concentration fall below the EIL limit of 20 mg kg−1. Conversely, some of the area under both fern species would need longer than the estimated period for remediation. This phytoremediation experiment is continuing; further frond harvests and (post-harvest) soil sampling would be undertaken to determine As concentration in fern fronds and in soils. Such data would be useful to confirm the remediation trend of the two fern species evaluated in this study. The results from our field experiment demonstrate that phytoremediation of this As-contaminated site using the two fern species will take a long time (>50 to 412 years); and it was apparent that P. calomelanos var. austroamericana would take 2.6–3.3-fold less time than P. vittata to remediate As in soil below the EIL value, at the studied site.
Conclusions
The results from this long-term field study revealed that P. calomelanos var. austroamericana accumulated much higher (2.65 times) As than P. vittata over the 27-month experimental period. Our remediation time calculations based on soil As concentrations demonstrated that both fern species would take 6–15 years to remediate As in soil below the EIL value (20 mg kg−1); whereas remediation time estimate on the basis of fern As uptake data showed that a long time (>50–412 years) would be required by the two fern species in order to achieve the same target. The remediation time calculations suggest that frond As uptake data of the two fern species should be considered to evaluate the As extraction efficiency and viability of these fern species for the phytoremediation of As contaminated soils. P. calomelanos var. austroamericana was better suited than P. vittata to the existing environmental conditions at the experimental site. However, future field studies are warranted to evaluate the phytoremediation potential of these As-hyperaccumulating fern species on other As-contaminated sites with varying soil properties (e.g. pH, free Fe, clay contents) and As concentrations.
References
Adriano DC (2001) Trace elements in terrestrial environments. Biogeochemistry, bioavailability and risks of metals. 2nd edn. Springer-Verlag, NY, 219–261.
Cai Y, Su J, Ma LQ (2004) Low molecular weight thiols in arsenic hyperaccumulator Pteris vittata upon exposure to arsenic and other trace elements. Environ Pollut 129(1):69–78
Caille N, Swanwick S, Zhao FJ, McGrath SP (2004) Arsenic hyperaccumulation by Pteris vittata from arsenic contaminated soils and the effect of liming and phosphate fertilisation. Environ Pollut 132(1):113–120
Cao XD, Ma LQ, Shiralipour A (2003) Effects of compost and phosphate amendments on arsenic mobility in soils and arsenic uptake by the hyperaccumulator, Pteris vittata L. Environ Pollut 126(2):157–167
de Mello J, Talbott J, Scott J, Roy W, Stucki J (2007) Arsenic speciation in arsenic-rich Brazilian soils from gold mining sites under anaerobic incubation. Environ Sci Pollut Res 14(6):388–396
R Development Core Team (2008) R: a language and environment for statistical computing. R foundation for statistical computing, Vienna, Austria, ISBN 3-900051-07-0, URL http://www.R-project.org/
Fayiga AO, Ma LQ (2006) Using phosphate rock to immobilize metals in soil and increase arsenic uptake by hyperaccumulator Pteris vittata. Sci Total Environ 359(1–3):17–25
Francesconi K, Visoottiviseth P, Sridokchan W, Goessler W (2002) Arsenic species in an arsenic hyperaccumulating fern, Pityrogramma calomelanos: a potential phytoremediator of arsenic-contaminated soils. Sci Total Environ 284(1–3):27–35
Gonzaga MIS, Santos JAG, Ma LQ (2006) Arsenic phytoextraction and hyperaccumulation by fern species. Sci Agric 63(1):90–101
Gonzaga MIS, Ma LQ, Santos JAG (2007) Effects of plant age on arsenic hyperaccumulation by Pteris vittata L. Water Air Soil Pollut 186(1–4):289–295
Gonzaga MIS, Santos JAG, Ma LQ (2008) Phytoextraction by arsenic hyperaccumulator Pteris vittata L. from six arsenic-contaminated soils: repeated harvests and arsenic redistribution. Environ Pollut 154(2):212–218
Gonzaga MIS, Ma LQ, Santos JAG, Matias MIS (2009) Rhizosphere characteristics of two arsenic hyperaccumulating Pteris ferns. Sci Total Environ 407(16):4711–4716
Huang PM, Fujii R (1996) Selenium and arsenic. In: Sparks DL (ed) Methods of soil analysis. Part 3. Chemical methods. Soil Science Society of America Inc, Wisconsin, pp 793-831.
Kachenko AG, Bhatia NP, Singh B, Siegele R (2007) Arsenic hyperaccumulation and localization in the pinnule and stipe tissues of the gold-dust fern (Pityrogramma calomelanos (L.) Link var. austroamericana (Domin) Farw. using quantitative micro-PIXE spectroscopy. Plant Soil 300(1–2):207–219
Kachenko AG, Grafe M, Singh B, Heald SM (2010) Arsenic speciation in tissues of the hyperaccumulator P. calomelanos var. austroamericana using X-ray absorption spectroscopy. Environ Sci Technol 44(12):4735–4740
Kertulis-Tartar GM (2005) Arsenic hyperaccumulation by Pteris vittata L. and its potential for phytoremediation of arsenic contaminated soils. PhD thesis, University of Florida, Florida, USA.
Kertulis-Tartar GM, Ma LQ, Tu C, Chirenje T (2006) Phytoremediation of an arsenic-contaminated site using Pteris vittata L.: a two-year study. Int J Phytorem 8 (4):311-322.
Kimber SWL, Sizemore DJ, Slavich PEG (2002) Is there evidence of arsenic movement at cattle tick dip sites? Aust J Soil Res 40(7):1103–1114
Ma LQ, Komar KM, Tu C, Zhang WH, Cai Y, Kennelley ED (2001) A fern that hyperaccumulates arsenic—a hardy, versatile, fast-growing plant helps to remove arsenic from contaminated soils. Nature 409:579
Mandal BK, Suzuki KT (2002) Arsenic round the world: a review. Talanta 58(1):201–235
Miller RO (1998) Nitric-perchloric acid wet digestion in an open vessel. In: Kalra YP (ed) Handbook of reference methods for plant analysis. CRC Press, Boca Raton, pp 57–61
National Environment Protection (Assessment of Soil Contamination) Measure (NEPM) (1999) Investigation levels for soil and groundwater. National Environmental Protection Council Service Corporation, Adelaide
Niazi NK, Bishop TFA, Singh B (2011a) Evaluation of spatial variability of soil arsenic adjacent to a disused cattle-dip site, using model-based geostatistics. Environ Sci Technol 45(24):10463–10470
Niazi NK, Singh B, Shah P (2011b) Arsenic speciation and phytoavailability in contaminated soils using a sequential extraction procedure and XANES spectroscopy. Environ Sci Technol 45(17):7135–7142
Niazi NK, Singh B, Van Zwieten L, Kachenko AG (2011c) Phytoremediation potential of Pityrogramma calomelanos var. austroamericana and Pteris vittata L. grown at a highly variable arsenic contaminated site. Int J Phytorem 13(9):912–932
Pan YF, Chiou C, Lin TF (2010) Adsorption of arsenic(V) by iron-oxide-coated diatomite (IOCD). Environ Sci Pollut Res 17(8):1401–1410
Reichmann KG, Gravel MR, Burren BG, Mayer DG, Wright CL (2004) Bioremediation of soil arsenic at a contaminated dip site using Pteris vittata. Paper presented at the Proceedings of the 25th Biennial Conference of the Australian Society of Animal Production, University of Melbourne, Collingwood, 25:305
Salido A, Hasty KL, Lim J, Butcher DJ (2003) Phytoremediation of arsenic and lead in contaminated soil using Chinese brake fern (Pteris vittata) and Indian mustard (Brassica juncea). Int J Phytorem 5:89–103
Shelmerdine PA, Black CR, McGrath SP, Young SD (2009) Modelling phytoremediation by the hyperaccumulating fern, Pteris vittata, of soils historically contaminated with arsenic. Environ Pollut 157(5):1589–1705
Smith E, Naidu R, Alston AM (1998) Arsenic in the soil environment: a review. Adv Agron 64:149–195
Smith E, Owens G, Naidu R (2006) Arsenic in the Australian environment. In: Naidu R, Smith E, Owens G, Bhattacharya P, Nadebaum P (eds) Managing arsenic in the environment. From soil to human health, CSIRO, Collingwood, Victoria, pp 627–638
Tu C, Ma LQ (2002) Effects of arsenic concentrations and forms on arsenic uptake by the hyperaccumulator ladder brake. J Environ Qual 31(2):641–647
Tu C, Ma LQ, Bondada B (2002) Arsenic accumulation in the hyperaccumultaor Chinese brake and its utilisation potential for phytoremediation. J Environ Qual 31:1671–1675
Vangronsveld J, Herzig R, Weyens N, Boulet J, Adriaensen K, Ruttens A, Thewys T, Vassilev A, Meers E, Nehnevajova E, van der Lelie D, Mench M (2009) Phytoremediation of contaminated soils and groundwater: lessons from the field. Environ Sci Pollut Res 16(7):765–794
Wei CY, Chen TB (2006) Arsenic accumulation by two brake ferns growing on an arsenic mine and their potential in phytoremediation. Chemosphere 63(6):1048–1053
Wei CY, Sun X, Wang C, Wang WY (2006) Factors influencing arsenic accumulation by Pteris vittata: a comparative field study at two sites. Environ Pollut 141(3):488–493
Wei CY, Wang C, Sun X, Wang WY (2007) Arsenic accumulation by ferns: a field survey in southern China. Environ Geochem Health 29(3):169–177
Xie QE, Yan XL, Liao XY, Li X (2009) The arsenic hyperaccumulator fern Pteris vittata L. Environ Sci Technol 43(22):8488–8495
Zhao FJ, Dunham SJ, McGrath SP (2002) Arsenic hyperaccumulation by different fern species. New Phytol 156(1):27–31
Acknowledgments
Nabeel gratefully acknowledges the Higher Education Commission of Pakistan for the award of a PhD scholarship. The project has been assisted by the NSW Government through its Environmental Trust. The authors are thankful to Dr. Thomas F. A. Bishop and Michael Nelson (The University of Sydney) for their help in geostatistical analysis. We are also thankful to Stephen Kimber, George Nastase, Joshua Rust, Scott Petty, Victor Warren and Desmond Cook at Wollongbar Agricultural Research Institute for field assistance. Finally, we are thankful to Dr. Elena Maestri (Editor) and two anonymous referees for their constructive comments on the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible editor: Elena Maestri
Rights and permissions
About this article
Cite this article
Niazi, N.K., Singh, B., Van Zwieten, L. et al. Phytoremediation of an arsenic-contaminated site using Pteris vittata L. and Pityrogramma calomelanos var. austroamericana: a long-term study. Environ Sci Pollut Res 19, 3506–3515 (2012). https://doi.org/10.1007/s11356-012-0910-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-012-0910-4