Abstract
Purpose
Polychlorinated biphenyls (PCBs) represent a large group of recalcitrant environmental pollutants, differing in the number of chlorine atoms bound to biphenyl ring. Due to their excellent technological properties, PCBs were used as heat-transfer media, for filling transformers and condensers, as paint additives, etc. With increasing knowledge of their toxicity, transfer to food chains and accumulation in living organisms, their production ended in most countries in the 1970s and in 1984 in the former Czechoslovakia. But even a quarter of century after the PCB production ceased, from contaminated areas, the volatile PCBs evaporate and contaminate much larger areas even at very distant parts of the world. For this reason, PCBs still represent a global problem. The main method of PCB removal from contaminated environment is at present the expensive incineration at high temperatures. With the aim of finding effective alternative approaches, we are studying biological methods for PCB removal from the environment. In this paper, we summarise 10 years of studies using long-term PCB-contaminated soil from a dumpsite in South Bohemia, targeted for the use of plants (phytoremediation) and their cooperation with microorganisms in the root zone (rhizoremediation).
Materials and methods
Long-term contaminated soil from Lhenice dumpsite, more than hundred kilograms of homogenised material, was used in microcosms (pots and buckets), and field plots were established at the site. Tested plants include among others tobacco, black nightshade, horseradish, alfalfa and willow. Aseptic plant cell and tissue cultures were from the collection of the IOCB. Microorganisms were our own isolates. The paper summarises experiments done between 1998 and 2008 with real contaminated soil, both vegetated and non-vegetated. PCB analysis was performed by GC-ECD, metabolic products identified mostly using 2D-GC/MS-MS and synthetic standards, whereas molecular methods included quantitative PCR and sequencing.
Results
The soil was used both for preparation of field plots at the site and for greenhouse and laboratory tests in microcosms. The results include analyses of changes in PCB content in untreated and vegetated soil, PCB uptake and distribution in different parts of various plant species, analysis of products formed, identification and characterisation of cultivable and non-cultivable bacteria both in rhizosphere and in bulk soil. Different treatments and amendments were also tested. Experiments in real contaminated soil were accompanied by in vitro experiments using aseptic cultures of plant biomass, genetically modified (GM) plants and bacteria, to allow identification of players responsible for PCB metabolisation in soil. The time-span of the experiments allows extrapolating some of the results and drawing conclusions concerning the effectivity of exploitation of various plant species and treatments to remove PCBs from soils.
Discussion
The approach using plants proved to represent a viable alternative to costly incineration of PCB-contaminated soils. The recent studies using molecular methods show that plants are responsible for the composition of consortia of microorganisms present in their root zone, including those with ability to degrade the chlorinated aromatic compounds.
Conclusions
In addition to uptake, accumulation and partial metabolisation of PCBs by plants, compounds produced by plants allow survival of microorganisms even in poor soils, serve as carbon and energy source, and can even induce the degradation pathways of different xenobiotics. Thus, the choice of proper plant species is crucial for effective cleaning of different polluted sites. Our study shows how the efficiency of PCB removal is dependent on the plant used.
Recommendations and perspectives
The use of plants in biological remediation of different organic xenobiotics proved to be a useful approach. Further improvement can be expected by application of specifically tailored GM plants and use of selective conditions ensuring high remediation potential based on optimal composition of the soil microbial consortia designed for the needs of given site.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Polychlorinated biphenyls (PCBs) represent a large group of recalcitrant environmental pollutants, differing in the number of chlorine atoms bound to biphenyl ring. Due to their excellent technological properties, PCBs were used as heat-transfer media, for filling transformers and condensers, as paint additives, etc. With increasing knowledge of their toxicity, transfer to food chains and accumulation in living organisms, their production ended in most countries in the 1970s and in 1984 in the former Czechoslovakia. But even a quarter of century after the PCB production ceased from contaminated areas, the volatile PCBs evaporate and contaminate much larger areas even at very distant parts of the world. For this reason, PCBs still represent a global problem (Safe 1994; Holoubek et al. 2001; Anonymous 2006). The main method of PCB removal from contaminated environment is at present the expensive incineration at high temperatures. With the aim of finding effective alternative approaches, we are studying biological methods for PCB removal from the environment. In this paper, we summarise 10 years of studies using long-term PCB-contaminated soil from a dumpsite in South Bohemia, targeted at the use of plants (phytoremediation) and their cooperation with microorganisms in the root zone (rhizoremediation).
Bioremediation uses biological systems including bacteria, fungi and plants for the removal of toxic compounds from the environment (Cunningham and Berti 1993; Kas et al. 1997). In natural conditions, these organisms often show mutual co-operation and support each other’s abilities. Plants exude sugars, amino acids, phenols or terpenes which can be used as carbon sources by microorganisms or as co-substrates or inducers of bacterial degradative enzymes (Fletcher et al. 1995; Macek et al. 2000). Phytoremediation and rhizoremediation, as shown in books collating reviews on these processes, principles and field applications (McCutcheon and Schnoor 2003; Mackova et al. 2006; Dowling and Doty 2009) are thus the most promising means of PCB removal in long-term perspective.
2 Materials and methods
2.1 The soil and the biological material
Practical use of bioremediation for PCB removal from contaminated soil using plants must inevitably combine the activity of plants themselves, and their bacterial counterparts. To get an insight into processes responsible for PCB removal under real conditions in the contaminated environment, we studied the abilities of microorganisms (the cultivable ones) isolated from contaminated environments and the abilities of plants themselves to take up, accumulate and metabolise PCB (including aseptic plants cultivated in vitro). The development of molecular methods allowed us to achieve a qualitatively new level in gained information, with the aim to manipulate the activity of soil bacterial consortia by choice of proper plants. In our study, we grew plants of different species in an area contaminated with PCBs. Since 1999, each year after a period of plant growth, the PCB content and differences in microbial growth in vegetated and non-vegetated soil, rhizoplane and rhizosphere of plants from the experimental field was tested.
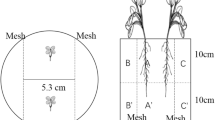
Long-term contaminated soil from Lhenice dumpsite (560 m above sea level, South Bohemia, Middle European climatic zone), about 200 kg of homogenised material each year, was used in microcosms (pots and buckets), and field plots were established at the site. In some years, more than 600 kg of soil were homogenised for experiments. The soil at this dumpsite originates from a tarmacadam-producing plant at Milevsko in South Bohemia, where main contamination comes from the heat-transfer medium Delotherm (produced in former Czechoslovakia by Chemko Strazske Company till about 1984). This commercial product of polychlorinated biphenyls contained mostly Delor 103 and Delor 106 mixtures of PCB (consisting of congeners with similar level of chlorination as Aroclor 1242 and 1260, respectively). The sandy soil contaminated in between 1960 and 1990 was removed from the tarmacadam plant and deposited at Lhenice dumpsite in 1996 (R. Kaplanek, personal communication). Detailed characteristics of the soil including analysis of heavy metal contamination can be found in papers describing natural plant species growing in this particular contaminated soil (Pavlikova et al. 2007). Uncontaminated soil of the same composition was not available. The whole dumpsite was mapped for PCB content, the area with highest content (between 400 and 500 ug PCB per gram of dry soil (ppm) in 1999) was used for preparation of experiments. Each year, new soil was dug, homogenised and analysed before using for experiments. Independent experiments were performed every year to study PCB removal and presence of microorganisms in the soil vegetated with selected plants and the same non-vegetated soil. At the end of each experiment, soil samples collected from non-vegetated area and from the rhizosphere of tested plant species were analysed for PCB content and compared with the initial concentration of tested soil in the given year.
Microorganisms used for amendments were our own isolates from Lhenice and other different contaminated soils.
After preliminary testing for degradation ability of over 40 plant cell and tissue cultures in vitro (Mackova et. al. 1997a, b; Kucerova et al. 1999) and evaluation of spontaneously occurring vegetation at the dumpsite (Pavlikova et al. 2007), some plant species were selected for testing in contaminated soil, Aseptic plant cell and tissue cultures were from the plant cell culture collection of the Institute of Organic Chemistry and Biochemistry (IOCB). From each species, more cultures were used for testing (Kucerova et al. 1999; Chroma et al. 2002a). The following selected plants were used for both field and pot experiments: Nicotiana tabacum (tobacco), Solanum nigrum (black nightshade), Salix sp. (willow), Medicago sativa (alfalfa) and Silybum marianum (thistle), while Armoracia rusticana (horseradish), Morus rubra (mulberry) and Zea mays (maize) were used in mesocosms only.
Experiments with real contaminated soil started in 1998, using both vegetated and non-vegetated soil (weeds removed manually). Tobacco, nightshade, mulberry, alfalfa and horseradish were pregrown to the size of 10 cm in non-contaminated soil, after washing to remove the starting soil the plantlets were transferred into the contaminated soil. Willows were from cuttings, while thistle and maize were grown directly from seeds.
PCB analysis was performed by gas chromatography (GC)-ECD (Burkhard et al. 1997; Ryslava et al. 2003); metabolic products were identified mostly using 2D-GC/MS-MS (Rezek et al. 2008) and synthetic standards; the total number of colony forming units, pseudomonads and related non-fermenting bacteria were estimated in the rhizosphere and rhizoplane areas of cultivated plants. Since 2002, in parallel to the above analyses, soil DNA extraction was also performed to analyse community profiling. The molecular methods included analysis of bphA gene presence (Erb and Wagner-Dobler 1993; Ryslava et al. 2003), terminal-restriction fragment length polymorphisms (T-RFLP) (Liu et al. 1997), and stable isotope probing analysis (Tillmann et al. 2005; Leigh et al. 2007; Uhlik et al. 2009a, b).
2.2 Cultivation of bacteria and plant tissue cultures
Bulk non-vegetated soil (weeds regularly removed manually, root zone soil (defined as the bulk soil beneath the plant) and rhizosphere soil samples were collected using an ethanol-disinfected shovel. For rhizosphere samples, fine roots (diameter, approximately 1 mm) were carefully separated, loose soil was removed by shaking, and then the tightly bound rhizosphere soil was collected. Samples were homogenised in separate mounds, and then approximately 50 g soil was placed in plastic bags for storage prior to microbial analyses, and approximately 20 g was stored in aluminium foil for determination of the PCB concentration. Extracted bacteria were cultivated on PCA medium to determine the total number of colony forming units, biphenyl (BP)-utilising bacteria were cultivated by spreading organisms suspended from 10 g soil onto basal mineral agar prepared with Difco Noble agar (Becton, Dickinson and Co., Sparks, MD, USA) with BP vapour provided as the sole carbon source (Leigh et al. 2006). Colonies were subjected to a BP-clearing zone assay and/or dibenzofuran (an aromatic compound with structure similar to biphenyl, Becher et al. 2000) using methods described previously (Sylvestre 1980; Leigh et al. 2006), except that the clearing zone assay (Sylvestre 1980) was performed with BP rather than 4-bromo-BP (Leigh et al. 2006), and colonies producing clearing zones were enumerated. Different numbers of BP-utilising colonies were randomly selected from plates of soil at time zero for isolation and further screening. Colonies were isolated, screened and further verified by growth in liquid culture with BP crystals as sole carbon source in basal mineral medium. Bacteria that were capable of growing in the liquid medium on biphenyl were further identified and screened for the presence of genes encoding for biphenyl dioxygenase subunit bphA1 and PCB degradation abilities.
Analysis of PCB degradation by bacterial isolates at laboratory conditions was performed in 250 ml Erlenmeyer flasks in 50 ml of mineral medium with biphenyl as co-substrate and 50 µg/ml of PCB mixture Delor 103 for 14 days at 28 C on a rotary shaker. The commercial PCB mixture, Delor 103, contains 59 individual congeners substituted with 3–5 chlorines per biphenyl molecule. After 14 days cultivation, the flasks were heated (95 C for 10 min) and residual amount of PCB was estimated.
Plant cells of tested species from the collection of IOCB cultivated in vitro were incubated with 50 µg/ml of Delor 103 similarly to bacteria, in Murashige-Skoog medium at 26 C, for 14 days (Burkhard et al. 1997; Mackova et al. 1997a, b).
In first years (1999–2003), when working only with cultivable microorganisms, the total number of colony forming units (CFU), pseudomonads and related non-fermenting species were estimated in the samples. From Pseudomonas agar, individual colonies were isolated and particular bacterial species were identified using Nefermtest (LACHEMA Brno, a.s.) containing 24 biochemical tests for differentiating non-fermentative bacteria. Later with adoption of molecular methods, total soil DNA has also been extracted from each sample and 16SrDNA (Zhou et al. 1996; Ryslava et al. 2003; Demnerova et al. 2005; Ionescu et al. 2009) was analysed by T-RFLP. The ability of each isolated bacteria to degrade PCBs was analysed (Demnerova et al. 2005).
In a typical experiment, unless otherwise stated, field plots were planted each year in late May or early June. The samples of plant tissue, the non-vegetated soil, rhizoplane and rhizospheric soil vegetated with tested plants were collected for microbial analysis and estimation of residual PCB content in November after 6 months from the beginning of the experiment. No fertilizers or watering were used. The control samples consisted of the same soil, non-vegetated, with weeds removed manually.
2.3 Pot experiments
Indoor and outdoor systems were used in pot experiments, 20 l metal buckets filled with the PCB-contaminated soil from the Lhenice dumpsite were kept at open-air conditions of middle European climatic zone, while some microcosm experiments were performed in plastic vessels containing 500 ml of soil, separated by aluminium foil from plastic surface to minimise losses caused by adsorption. The tested plants were grown in phytotron at 28 C, 16/8 hour day/night conditions in contaminated soil and the same non-vegetated soil was used as the control. Plants were grown for 6 months and then the residual amount of PCBs was analysed. Parallel with DNA extraction, also the total number of microorganisms, pseudomonads and related non-fermenting bacteria were estimated in the rhizosphere and rhizoplane areas of cultivated plants.
2.4 Analysis of polychlorinated biphenyls
The analytical approach used for bacterial and plant cells to estimate their ability to degrade PCB was very similar. For the use of plant biomass, it was slightly modified (Burkhard et al. 1997). After homogenisation (of plant cultures only) and subsequent sonication, the contents of the flasks were extracted by 10 ml of hexane at 20 C on a rotary shaker for 2 h. Following phase separation, the upper, hexane layer was subjected to GC analysis. Samples were analysed using a Hewlett-Packard 5890 gas chromatograph with an electron capture detector and a fused silica capillary column (30 m, 0.20 mm inner diameter) coated with 0.25-µm immobilised phase SE-54 with nitrogen as the carrier gas (flow rate 1 ml/min). Using GC analysis with EC detector, 22 of the Delor 103 congeners were assigned to peaks with areas larger than 0.5% of the total area of all 59 individual chromatographic peaks. For the calculation of the residual amount of PCBs, the above mentioned 22 chromatographic peaks were used. These 22 congeners represent 80–90% of the total sample amount. Controls containing heat-killed cells were included to establish that observed changes in the content of congeners were dependent exclusively on the activity of living cells. Using standard conditions for the preparation of samples for GC analysis, the accuracy of the results obtained was within 15%. Results were calculated from the residual amounts of each congener peak of the sample, comparing with the respective peaks of the controls, or using the value recommended by US Environmental Protection Agency (EPA) for expressing the total content of PCBs as a sum of recommended ”indicator” congeners. For Delor 103, the following congeners (IUPAC nomenclature) were recommended: PCB no. 28 (GC peak no.7), PCB no. 52 (peak no.10) and PCB no. 101 (peak no.21; EPA: US/EPA Methods 8089/8081).
2.4.1 Polychlorinated biphenyl analysis—field and pot experiments
Tested soil was dried overnight and sieved through a mesh with 1 mm pore size. Then 1 g of the soil was Soxhlet extracted with hexane for 4 h. The extract was concentrated to approximately 1 ml in volume by nitrogen flow, purified on a Florisil column, diluted with hexane to exactly 10 ml volume, then diluted 1/100 for analysis by GC. Triplicate soil samples from each sampling date and treatment were air-dried for 24 h at room temperature, then homogenised with mortar and pestle and sieved through a 1-mm mesh. Aliquots of soil (1 g) were extracted in hexane for 4 h using a micro-scale modification of the EPA method for Soxhlet extraction (EPA Method 3540C; Ryslava et al. 2003). Extracts were concentrated under a nitrogen stream and subjected to florisil cleanup (EPA Method 3620B) before analysis. Each sample was analysed in duplicate using a Hewlett-Packard 5890 gas chromatograph with an electron capture as described above. The total PCB concentration was estimated based on indicator congeners using US EPA methods 8089/8081 for expressing the total content of PCBs in soil as a sum of 6 recommended indicator congeners (PCB no. 28—2,4,4′-trichlorobiphenyl, PCB no. 52—2,2′,5,5′-tetrachlorobiphenyl, PCB no. 101—2,2′,4,5,5′-pentachlorobiphenyl, PCB no. 138—2,2′,3,4,4′,5-hexachlorobiphenyl, PCB no. 153—2,2′,4,4′,5,5′-hexachlorobiphenyl, PCB no. 180—2,2′,3,4,4′,5,5′-heptachlorobiphenyl). The effect of the different treatments on PCB concentration was assessed by analysis of variance employing general linear modelling using the SPSS 14.0 software (SPSS, Inc., Chicago, Ill, USA).
2.5 Identification and characterisation of microorganisms
Ten-gram samples of non-vegetated soil, rhizospheric soil and roots with attached soil layer (representing rhizoplane bacteria) were extracted by shaking with 90 ml of peptone water or 1% w/v sodium pyrophosphate solution (from 2000; Leigh et al. 2006) and glass beads for 1 h at room temperature. After allowing the extract to settle for 30 min, serial dilutions were made in basal mineral medium and were spread onto Plate Count Agar (OXOID; total counts of bacteria), Pseudomonas agar (Oxoid) for detection of non-fermenting bacteria and related species and also basal mineral agar plates prepared with Noble agar. Biphenyl was provided as the sole carbon source by placing crystals in the Petri dish lid to preferably cultivate bacteria-degrading biphenyl. For liquid cultivation, a medium with 0.5% peptone was used for enrichment of bacteria preferring biphenyl as carbon source. Enrichment cycle was not repeated, but the first one prolonged to 5 days. Individual colonies isolated from Pseudomonas agar were chosen for identification by Nefermtest (Lachema Brno, Czech Republic). Individual strains of non-fermenting bacteria were tested for their ability to degrade PCBs.
Since 2003, the cultivable bacteria were characterised by 16S rDNA using method described by Borneman et al. (1996). Starting in 2005, some non-cultivable bacteria in the contaminated soil were also identified using T-RFLP of genes encoding 16S rRNA, following the method of Liu et al. (1997). Recently also stable isotope probing was introduced to widen the knowledge of bacteria involved in PCB degradation in contaminated soil (Uhlik et al. 2009a, b).
Total DNA extraction
Total DNA was extracted using a PowerMax™ Soil DNA Isolation Kit (MoBio Company) following standard protocol with the exception that, after final elution, DNA was concentrated by adding 0.2 ml of 5 M NaCl and 10.4 ml of ethanol, incubated overnight at 20°C, and transferred gradually into 2-ml microtubes with 20 µg of glycogen, each time centrifuged. Finally, the pellet was dissolved in 20 µl of water. DNA concentration was evaluated by absorbance measurements at 260 and 280 nm.
Polymerase chain reaction amplification of bphA1 gene
The product of bphA, biphenyl-2,3-dioxygenase is a multicomponent enzyme, containing four subunits. This enzyme determines the substrate specificity of PCB-degrading bacteria. The small subunit of dioxygenase, encoded by bphA1, is responsible for incorporation of two molecules of oxygen to one molecule of PCB and is highly homologous among different species. Bacterial colonies were scraped off agar plates and dissolved in 50 μl of TE buffer. The samples were heated (95°C for 10 min) and centrifuged. BphA1 genes were amplified by polymerase chain reaction (PCR) using forward and reverse primers F352 and R674 or R1178 (Ryslava et al. 2003). PCR reaction mixture consisted of 5 μl of template DNA, 1 μl of each primer (10 mM), 1 μl of deoxynucleoside triphosphate mixture (10 mM), 5 μl of 10xPCR buffer, 3 μl of MgCl2, 0.2 μl of Taq-polymerase and sterile distilled water up to 50 μl of each reaction volume. PCR amplifications were performed in an automated thermal cycler Techne (Progene) with an initial denaturing (94°C for 5 min), followed by 30 cycles of denaturation (94°C, 30s), annealing (40°C, 30 s), and extension (72°C, 1 min) and concluded by a single final extension (72°C, 10 min). The presence of amplified part of bphA1 gene was confirmed by gel electrophoresis on a 2% agarose gel with TAE buffer.
16S rDNA amplification and sequencing
Bacterial colonies were scraped off agar plates and suspended in 50 μl of TE buffer, followed by heating (95°C for 10 min) and centrifugation. 16S rDNA genes were amplified by PCR using a forward primer hybridising at position eight to 27 and reverse primer hybridizing at the complement of positions 1507 to 1492 (Escherichia coli numbering). Each 50 μl sample of PCR reaction mixture consisted of 5 μl of template DNA, 1 μl of each primer (10 mM), 2,5 μl of deoxynucleoside triphosphate mixture (2,5 mM), 5 μl of 10 × PCR buffer, 5 μl of MgCl2 and sterile distilled water up to 50 μl of each reaction volume. PCR was carried out under following conditions: an initial denaturing (94°C for 6 min), followed by 35 cycles of denaturation (94°C, 1 min), annealing (50°C, 2 min), and extension (72°C, 2 min) and single final extension (72°C, 6 min). After PCR, the amplified DNA was purified with PCR purifying kit (Sigma) and checked by gel electrophoresis on a 1% agarose gel. The sequence of 16S rDNA was determined by CEQ 2000 XL DNA sequencer (Beckman Coulter, USA) using the protocols recommended by the manufacturer for “CEQ 2000 Dye Terminator Cycle Sequencing” with Quick Start kit using the fluorescent dye-labelled dideoxynucleotides and standard 16S rDNA sequencing primers or the samples were submitted to Genomac Co., Czech Republic, for commercial sequencing service. Homology searching of the resulting sequence data was carried out in comparison with 16S rDNA sequences in the NCBI nucleotide database by using BLAST database.
3 Results
This study aimed to describe the effect of different plants and find conditions to improve the performance of phyto- and rhizoremediation in PCB removal from soil. The Lhenice soil was used both for preparation of field plots at the site, for greenhouse and laboratory tests in microcosms and for application of molecular methods. The results include analyses of changes in PCB content in untreated and vegetated soil, PCB uptake and distribution in different parts of various plant species, analysis of products formed, identification and characterisation of cultivable and non-cultivable bacteria both in rhizosphere and in bulk soil. The results of phytoremediation at field conditions and pot experiments were compared with the results obtained in laboratory experiments performed with aseptic plant tissue cultures of the same species cultivated in vitro. The experimental designs were modified in time according to the changing knowledge and availability of new methods. The main differences in the experiments were the complexity of the analysed factors, starting with changes in the PCB content in soil and plants, through analysis of cultivable degraders, exploitation of e.g. terminal restriction fragment length polymorphism, to metagenomic approaches and stable isotope probing. With the soil from Lhenice dumpsite a large amount of information has been gained and this paper can give just a short overview of the main findings.
Separately published results include those obtained in vitro (Mackova et al. 1997a, b; Kucerova et al. 1999; Chroma et al. 2002a, b) and analyses of PCB distribution in plants grown in this soil (Ryslava et al. 2003), comparison of PCB content in vegetation that appeared on the dumpsite naturally (Pavlikova et al. 2007), analysis of products formed from PCBs by plants (Kucerova et al. 2000; Rezek et al. 2008, 2009) and their toxicity (Gichner et al. 2007). Different treatments and amendments were also tested as summarised under 2.7, but these studies were not fully evaluated yet. Lhenice soil served also for testing of genetically modified (GM) plants and GM bacteria (Villacieros et al. 2005). Plants have been prepared bearing bacterial bphC gene or genes for toluenedioxygenase (Novakova et al. 2009; Sylvestre et al. 2009) and tested within contained use in laboratory using the Lhenice soil. The results obtained within the time span of the experiments with the long-time contaminated soil allow extrapolating some of the results and drawing conclusions concerning the effectivity of exploitation of various plant species and treatments to remove PCBs from soils.
3.1 Field experiments
Since the beginning of experiments with real contaminated soil, the total counts of bacteria pseudomonads and related non-fermenting species present in field samples were estimated, as illustrated by results of the year 1999 in Table 1. It can be seen that thistle performed better than alfalfa in supporting both total number of bacteria and the number of pseudomonads, at that time considered to be main candidates for high degradation ability in soil. Species isolated from rhizosphere and rhizoplane area of thistle were identified as: Pseudomonas sp., P. stutzeri, P. fluorescens, from rhizosphere and rhizoplane area of alfalfa: P. putida, P. fluorescens, P. pseudoalcaligenes. Individual strains isolated from contaminated soil were tested in laboratory conditions for their ability to grow with biphenyl as the sole carbon source, with quite poor (not exceeding 104 CFU/ml of medium) growth obtained in comparison with the control strain Burkholderia xenovorans LB400, positively degrading PCBs (Gibson et al. 1993).
3.2 Polychlorinated biphenyl content in soil
The results of the repeated experiments performed using soil of the same site confirmed that natural attenuation at this dumpsite is responsible for some decrease of the PCB content. The initial soil prepared for experiments in particular years shows a substantial decrease in PCB content—from 470 µg/g in 1999, the PCB concentration dropped to average 330 µg/g in 2003 and to 200 µg/g in 2005. The dumpsite is far from homogeneous in its PCB content, but in the area selected for our experiments, the measurements in 1999 showed differences within 10%, and this soil was further homogenised before use. The effects of tested plant species are illustrated by Tables 2 and 3. Rhizosphere and rhizoplane of alfalfa, tobacco and nightshade grown at field plots in 1999 served as source of bacterial consortia tested for PCB degradation ability under laboratory conditions in medium with commercial PCB mixture Delor 103, initial concentration 50 µg PCB/ml and 500 µg biphenyl/ml as co-substrate. The content of PCB remaining after 10 days incubation of different bacterial consortia, expressed as percentage of PCB concentration in dead cell controls, is shown in Table 4. After the year 1999, thistle (Silybum marianum) was no longer used, despite its good growth, as PCB removal results in soil vegetated by thistle did not differ from those of control soil and also tests in vitro showed poor performance of all five of its tested culture strains—Table 5 shows the results of one only thistle strain, OP35, for illustration.
3.3 Pot and laboratory experiments
The pot experiments also showed the beneficial effect of plants on the number of microorganisms found in the rhizosphere and especially in the rhizoplane area (see Table 2). In this case, bacterial strains were not taxonomically identified, only microscopically (data not shown).
The data in Table 3 show results revealing that the final PCB concentration of soil vegetated with tobacco and nightshade decreased to 76% and 78%, respectively, of that in non-vegetated soil. Soil cultivated with alfalfa in pots showed comparable (18.5% versus 15%) decrease of PCB concentration to that seen in the field experiment.
The ability to degrade PCBs was analysed using the enriched microbial consortia isolated from rhizosphere in pots (not individual bacterial strains), incubated with biphenyl as co-substrate and Delor 103 under laboratory conditions. It can be seen that bacterial consortia enriched from non-vegetated soil and from samples of vegetated soil exhibited different ability to degrade PCBs. The average PCBs concentration remaining in media after incubation with these bacterial consortia is summarised in Table 4. The highest degradation efficiency was shown by bacteria isolated from the non-vegetated soil. This fact does not exclude the possibility that in the rhizosphere or rhizoplane area, different bacterial populations were selected which can prefer other co-substrate of plant origin instead of biphenyl. Data show a substantial decrease in PCB concentration of peaks representing congener 7 (Cl in position 2,4,4′), 10 (Cl in 2,2′,5,5′) and 21 (Cl in 2,2′,4,5,5′).
Aseptic in vitro plant cultures of the same species as used in pot experiments were followed for their ability to metabolise PCBs. Plant tissue cultures were cultivated as submerged cultures for 14 days with 50 ppm of Delor 103. Results are shown in Table 5.
The best results during in vitro experiments were obtained with the hairy root clone SNC-9O of S. nigrum. In the pot experiments, after the cultivation period, the soil vegetated with this plant also contained by 14–23% less PCBs than initial soil and 5–15% less than non-vegetated control soil. On the other hand, the results obtained with in vitro plant cultures cannot be simply generalised for whole plant of the same species, because cultures in vitro exhibit much spontaneous variability (Macek et al. 1998). This can be illustrated by our results if e.g. we examined more than ten different in vitro cultures of S. nigrum hairy roots for their ability to metabolise PCBs, the results obtained for particular strains were ranging between 0 for some strains and 70% conversion by the best ones under the same conditions (Mackova et al. 1997a, b, Kucerova et al. 1999).
In between 1999 and 2003, the best results were obtained in soil vegetated with N. tabacum (see Table 5). Results obtained in 2004 pointed to another possible alternative—horseradish (A. rusticana). Horseradish in comparison with other tested plants supported better the growth of rhizosphere microflora, and revealed the highest decrease of PCB concentration in soil. This plant species is more suitable for the climate in Central Europe than tobacco (N. tabacum); however, we repeated the experiment again with both plant species in 2005.
Further experiments were focused on isolation and characterization of PCB degraders. From the contaminated soil vegetated with three different plant species (see Table 3) and non-vegetated soil, bacterial strains growing on minimal medium with biphenyl as a sole source of carbon and energy, were isolated and tested also for the presence of bphA gene (coding biphenyldioxygenase) and ability to degrade PCBs. The results proved the presence of bphA in six strains and their ability to degrade PCBs (Table 6). Their taxonomical characterization was performed by biochemical tests and also based on sequencing of 16SrDNA. Following experiments were focused on abilities and degrading efficiency of isolated degraders in real contaminated soil. First, they were added to contaminated soil without any plant species. Soil was amended with bacterial suspensions to get 107 CFU/g soil at the beginning of the experiment. Total content of PCBs was 130 mg/kg of soil. The results obtained after 5 months of incubation of pots with contaminated soil at 22 C are summarised in Table 7. The lowest residual content of PCBs was detected in pots with added strain KH6—Ochrobactrum anthropi which was originally isolated from control, non-vegetated PCB-contaminated soil in the year 2004. Pots with added strains originally isolated from rhizosphere did not show such significant decrease of PCBs. Augmentation of indigenous population with other tested bacterial species did not bring any improved results. Better results were obtained in experiments with indigenous microbial population supported by nutrients or compounds promoting PCB degradation either by addition of individual chemicals or presence of plants supplying their products.
Other experiments not shown here, carried out during several years included genetically modified bacteria, genetically modified plants, consortia of plants and bacterial degraders, effect of AM fungi, these experiments were done often in close cooperation with different partner laboratories. Due to space limitation, here are presented results of only part of performed experiments, illustrating the work done. In other approaches, we applied surfactants increasing bioavailability of xenobiotics and conditions affecting stress behaviour and living conditions of organisms. Use of genetically modified plants revealed their higher resistance to increased concentrations of PCBs, but only recently, more efficient degradation ability also (Macek et al. 2008; Novakova et al. 2009). In case of genetically modified bacteria used in the field conditions, a slight increase of PCB degradation was observed. However, modified bacteria did not survive, in concurrence of indigenous microbes (de Carcer et al. 2007). Surfactants increased bioavailability of xenobiotics but their toxicity negatively affected indigenous bacteria including degraders. The best results were obtained when consortia of plants and artificially added bacterial degraders were applied or in case when soil was vegetated with different plants allowing establishment of specific indigenous rhizosphere community.
4 Discussion
Resistant plants naturally occurring at the Lhenice dumpsite were identified (Pavlikova et al. 2007), and their accumulation of PCB analysed. The approach using plants proved to represent a viable alternative to costly incineration of contaminated soils (McCutcheon and Schnoor 2003). In terms of parameters like plant survival, support of degrading microorganisms, PCB removal, some of the selected dicotyledonous plants seem to be suitable test organisms to assess effectivity of phyto/rhizoremediation processes in soil or contaminated sediments. Recent studies using molecular methods have discussed, that plants are responsible for the composition of microbial consortia present in their root zone, including those with ability to degrade the chlorinated aromatic compounds (Ryslava et al. 2003; Chekol et al. 2004; Singer 2006; Leigh et al. 2006; Ionescu et al. 2009; Rezek et al. 2009; McGuiness and Dowling 2009). The experiments documented that not all seemingly beneficial conditions and combinations brought desirable effect. Successful bioremediation requires an understanding of site-specific factors that limit biotransformation and specificity and stereoselectivity of enzymes involved (Jurcek et al. 2008; Zarevucka and Wimmer 2008) and also bioremediation engineers and scientists need to satisfy the physiological and nutritional requirements of specific degraders and ensure that a competitive advantage is provided to desirable (but not undesirable) biotransformation pathways (Singer et al. 2003; Mackova et al. 2007). Achieving this can be a very complex task. One challenge in investigating phyto/rhizoremediation in real contaminated soil is to obtain good growth of the plants despite the possible toxicity of polluting compounds. Contaminated sites sometimes include low-quality soil. As generally known in agricultural practice, optimal water supply and nutrient conditions should be chosen to produce optimal growth of the plants. In case of phytoremediation, this does not necessarily mean highest growth, but optimal for the required performance. This is the reason why crop plants would be suitable candidates, because their agrotechnical requirements are well known. On the other hand, for food and feed security reasons such plants are not always the best choice. Better candidates seem to be technology or energy crops (Griga et al. 2003; Tlustos et al. 2006), and especially GM trees (Doty et al. 2007; van Aken 2008).
Hairy root cultures of different plant species proved to be a useful tool in basic research for phytoremediation purposes (Doran 1997; Soudek et al. 1998). Actually, ours were the first experiments using transformed hairy root cultures for phytoremediation studies of organics (Mackova et al. 1997a, b) and also heavy metals (Macek et al. 1994). For comparing different plant species under in vitro conditions, trends obtained from analysis of more individual strains of each species must be followed. Evaluation based on one single strain of each species (Wilken et al. 1995) cannot be considered representative (Macek et al. 2000). If an in vitro culture does not transform some organic compound, one should not make conclusions about the ability of the species, while in case the culture does perform some biotransformation (including detoxication), it can be concluded the plant has proper enzymatic systems to perform the required reaction even in vivo (Macek et al. 2000). Plant metabolites of polychlorinated biphenyls in hairy root culture of black nightshade and tobacco described Rezek et al. (2007, 2008). The exploitation of in vitro cultures for bioremediation studies is discussed in detail in a recent study by Doran (2009).
The experiments in contaminated soil confirmed the in vitro findings and showed that selected plants helped to remediate soil highly contaminated with PCBs. This outcome may be ascribed to the stimulation and support of indigenous PCB-degrading bacterial consortia in the rhizosphere of specific plants, as well as to the degradative ability of the plants themselves. This shows that phytoremediation and rhizoremediation are interrelated processes, based on mutual co-operation of different organisms, where the responsibility of microorganisms is in:
-
modification of rhizosphere by production of extracellular enzymes and plant growth factors
-
formation of mutualistic relationships with plants allowing to grow in nutrient-deficient soil
-
enhance uptake of certain mineral nutrients
while action of plants includes:
-
supply of compounds present in exudates that can be utilised by microorganisms as carbon and energy sources
-
regulation of deposits of soil water
-
release of plant compounds that solubilize contaminants whereby they are more available to both plant and microbial degradative enzymes
A number of bacterial strains have been identified in a wide variety of contaminated environments in association of various plant species and with enzymes capable of degrading different toxic organic compounds. Great potential of rhizosphere but also endophytic bacteria was recently discussed (McGuiness and Dowling 2009; Weyens et al. 2009). These processes can occur as natural processes, but also engineered systems can be used. When thinking about commercial use and before commercially viable systems are available, a lot of laboratory and pilot scale work remains to be done using plant-associated rhizospheric bacteria to degrade a wide range of toxic organic compounds of concern in soil. High importance of molecular studies giving information about complexity and structural changes in microbial diversity was shown (Narasimhan et al. 2003; Cebron et al. 2007; Uhlik et al. 2009b).
5 Conclusions
The soil from Lhenice dumpsite is serving to allow identification of players responsible for PCB metabolisation in soil. In accordance with the relatively slow performance of plants in biological remediation, long time is needed for experimental evaluation of different factors affecting the process. Results shown in this paper illustrate part of our approach to study and improve phyto/rhizoremediation of PCB in contaminated soil.
When performing tests with living organisms, one has to deal with natural variability due to genetic differences and to differences caused by complex and complicated analytical approaches. In our studies, despite the variability within some years, the long duration of repeated experiments allows us to draw some conclusions. Neither the used soil (each year newly acquired from the same locality and homogenised for the experiments) nor the used test design influenced this result drastically. The practice of taking uniform plantlets of the same size and age, in some cases even micropropagated in vitro under aseptic conditions and transferred to the contaminated soil is therefore strongly recommended. Given the restrictions applied by a feasible number of replicates, the increase in replicate number to more than five in laboratory studies and three in mesocosm studies using five plants per pot is in our conditions probably not realistic due to limited spatial and financial capacity and considering the number of tested plants, sets of experimental conditions and necessity to perform other research tasks. Despite this fact, the results obtained in different scales, for different plants and setups, show high agreement and important is the same trend found in different years.
From the data presented, it can be concluded that the experimental conditions in the various test systems were suitable to study bioremediation process in PCB-contaminated soil. There is a variation in performance of different plant species, changing from year to year as effect of different weather conditions, but obtained PCB conversions were comparable between laboratory and field investigations and in vegetated soil better than in control non-vegetated soil of same composition, contamination and conditions.
The comparison of the different test designs in this study demonstrated that differences in PCB removal effectivity were rather linked to the presence of different plant species in the soil than to the different degree of complexity of the test systems. In most years, the field observation data were in agreement with pot experiments and in vitro studies (if all performed). The advantage of in vitro degradation tests with cultivated plant tissues is that they can be performed at highly controllable and reproducible conditions and low costs, whereas pot studies reflect more environmental realistic conditions, but are time consuming and cost intensive. In this study, we demonstrated that both the simple and complex experimental set-ups resulted in reliable results and were in agreement with tests performed in field plots at the dumpsite itself.
In addition to uptake, accumulation and partial metabolisation of PCBs by plants, compounds produced by plants allow survival of microorganisms even in poor soils, serve as carbon and energy source, and can even induce the degradation pathways of different xenobiotics, e.g. the effect of terpene-utilising isolates and their relevance to enhanced biotransformation of polychlorinated biphenyls in soil has been described by Hernandez et al. (1997). Very important is the fact, that plants influence the composition of consortia of microorganisms in soil surrounding the roots, within rhizosphere. Thus, the choice of proper plant species is crucial for effective cleaning of different polluted sites. Our study shows how much the efficiency of PCB removal is dependent on the plant used. Even the untreated soil exhibits some decrease of the presence of contaminants and their metabolic products, but to achieve a substantial degradation, the soil conditions have to be manipulated artificially to ensure optimal composition of microbial consortia by selecting right plants and nutrients for both. In the tested soil, the best performance was obtained with tobacco and horseradish.
6 Recommendations and perspectives
The use of plants in biological remediation of different organic xenobiotics proved to be a useful approach also in the case of PCB. Further improvement can be expected by application of specifically tailored GM plants (Macek et al. 2008; Novakova et al. 2009; Sylvestre et al. 2009), and use of selective conditions ensuring high remediation potential based on optimal composition of the soil microbial consortia designed for the needs of a given site. The removal of recalcitrant xenobiotics like PCB is much faster in vegetated soil in comparison to non-vegetated bulk soil. Changes in the composition of rhizospheric microorganisms, especially the increased presence of degraders, could be ascribed to the beneficial effect of selected plant species. Stable isotope probing allows obtaining yet unavailable insight also into the situation at real contaminated sites (Uhlik et al. 2009a, b) and together with metagenomic approaches allows to optimise the performance even of the until now non-cultivable microorganisms. The early results already show how large potential is hidden in microorganisms which we are at present unable to cultivate in the laboratory and thus their identification and manipulation of performance of rhizospheric consortia seems to be crucial for increase of effectivity of bioremediation processes.
References
Anonymous (2006) Agency for Toxic Substances and Disease Registry. 2006. CERCLA priority list of hazardous compounds.Washington, DC. http://www.atsdr.cdc.gov/cercla/05list.html
Becher D, Specht M, Hammer E, Francke W, Schauer F (2000) Cometabolic degradation of dibenzofuran by biphenyl-cultivated Ralstonia sp. strain SBUG 290. Appl Environ Microbiol 66:4528–4531
Borneman JP, Skroch PW, O’Sullivan PJA, Rumjanek NG, Jansen JL, Nienhuis J, Triplett EW (1996) Molecular microbial diversity of an agricultural soil in Wisconsin. Appl Environ Microbiol 62:1935–1943
Burkhard J, Mackova M, Macek T, Kucerova P, Demnerova K (1997) Analytical procedure for the estimation of PCB transformation by plants. Anal Commun Roy Soc 34:287–290
Cebron AL, Bodrossy N, Stralis-Pavese AC, Singer IP, Thompson JI, Prosser IJ, Murrell JC (2007) Nutrient amendments in soil DNA stable isotope probing experiments reduce the observed methanotroph diversity. Appl Environ Microbiol 73:798–807
Chekol T, Vough LR, Chaney RL (2004) Phytoremediation of polychlorinated biphenyl-contaminated soils: the rhizosphere effect. Environ Int 30:799–804
Chroma L, Mackova M, Kucerova P, in der Wiesche C, Burkhard J, Macek T, Chroma L, Mackova M, Kucerova P, in der Wiesche C, Burkhard J, Macek T (2002a) Enzymes in plant metabolism of PCBs and PAHs. Acta Biotechnologica 22:35–41
Chroma L, Mackova M, Demnerova K, Macek T (2002b) Decolorization of RBBR by plant cells and correlation with the transformation of PCBs. Chemosphere 49:739–748
Cunningham SD, Berti WR (1993) Remediation of contaminated soils with green plants: an overview. In Vitro Cell Dev Biol 29:207–212
de Carcer DA, Martin M, Mackova M, Macek T, Karlson U, Rivilla R (2007) The introduction of genetically modified microorganisms designed for rhizoremediation induces changes on native bacteria in the rhizosphere but not in the surrounding soil. ISME J 1:215–223
Demnerova K, Mackova M, Spevakova V, Beranova K, Kochankova L, Lovecka P, Ryslava E, Macek T (2005) Two approaches to biological decontamination of ground-water and soil polluted by aromatics-characterization of microbial populations. Int Microbiol 8:205–211
Doran PM (ed) (1997) Hairy roots: culture and application. Harwood Academic Publishers, London, pp 133–138
Doran PM (2009) Application of plant tissue cultures in phytoremediation research: incentives and limitations. Biotechnol Bioeng 103:60–76
Doty SL, James CA, Moore AL, Vajzovic A, Singleton GL, Ma C, Khan Z, Xin G, Kang JW, Park AY et al (2007) Enhanced phytoremediation of volatile environmental pollutants with transgenic trees. Proc Natl Acad Sci USA 104:16816–16821
Dowling DN, Doty SL (2009) Improving phytoremediation through biotechnology. Curr Opin Biotechnol 20:1–3
Erb RW, Wagner-Dobler I (1993) Detection of polychlorinated biphenyl degradation genes in polluted sediments by direct DNA extraction and polymerase chain reaction. Appl Environ Microbiol 59:4065–4073
Fletcher JS, Donnelly PK, Hegde RS (1995) Biostimulation of PCB-degrading bacteria by compounds released from plant roots. In: Hinchee RE, Anderson DB, Hoeppel RE (eds) Bioremediation of recalcitrant organics. Battelle Press, Columbus, pp 131–136
Gibson DT, Cruden DL, Haddock JD, Zylstra GJ, Brand JM (1993) Oxidation of polychlorinated biphenyls by Pseudomonas sp. LB400 and Pseudomonas pseudoalcaligenes KF707. J Bacteriol 175:4561–4564
Gichner T, Lovecka P, Kochankova L, Mackova M, Demnerova K (2007) Monitoring toxicity, DNA damage, and somatic mutations in tobacco plants growing in soil heavily polluted with polychlorinated biphenyls. Mutat Res 629:1–6
Griga M, Bjelkova M, Tejklova E (2003) Phytoextraction of heavy metals by fibre crops: Linum usitatissimum L. case study. In: Kalogerakis N, Psillakis E (eds) Proceedings of the 2nd European Bioremediation Conference, Chania, Crete, TU Crete, pp 353–356
Hernandez BS, Koh SC, Chial M, Focht DD (1997) Terpene-utilizing isolates and their relevance to enhanced biotransformation of polychlorinated biphenyls in soil. Biodegradation 8:153–158
Holoubek I, Kocan A, Holoubkova I, Kohoutek J, Falandysz J, Roots O, Staffova K (2001) Polychlorinated biphenyls contaminated sites worldwide. The case of the Central and Eastern European countries. In: Robertson LW, Hansen LG (eds) PCBs. Recent advances in environmental, toxicology and health effects. The University Press of Kentucky, Lexington
Ionescu M, Beranova K, Dudkova V, Kochankova L, Demnerova K, Macek T, Mackova M (2009) Isolation and characterization of different plant associated bacteria and their potential to degrade polychlorinated biphenyls. Int Biodeterior Biodegrad 63:667–672
Jurcek O, Wimmerova M, Wimmer Z (2008) Selected chiral alcohols: enzymic resolution and reduction of convenient substrates. Coord Chem Rev 252:767–781
Kas J, Burkhard J, Demnerova K, Kostal J, Macek T, Mackova M, Pazlarova J (1997) Perspectives in biodegradation of alkanes and PCBs. Pure Appl Chem 69:2357–2369
Kucerova P, Mackova M, Polachova L, Burkhard J, Demnerova K, Pazlarova J, Macek T (1999) Correlation of PCB transformation by plant tissue cultures with their morphology and peroxidase activity changes. Coll Czech Chem Commun 64:1497–1509
Kucerova P, Mackova M, Chroma L, Burkhard J, Triska J, Demnerova K, Macek T (2000) Metabolism of polychlorinated biphenyls by Solanum nigrum hairy root clone SNC-9O and analysis of transformation products. Plant Soil 225:109–115
Leigh MB, Prouzova P, Mackova M, Macek T, Nagle DP, Fletcher JS (2006) Polychlorinated biphenyl (PCB)-degrading bacteria associated with trees in a PCB-contaminated site. Appl Environ Microbiol 72:2331–2342
Leigh MB, Pellizari VH, Uhlik O, Sutka R, Rodrigues J, Ostrom NE, Zhou J, Tiedje JM (2007) Biphenyl-utilizing bacteria and their functional genes in a pine root zone contaminated with polychlorinated biphenyls (PCBs). ISME J 1:134–148
Liu W, Marsh T, Cheng H, Forney L (1997) Characterization of microbial diversity by determining terminal restriction fragment length polymorphisms of genes encoding 16S rRNA. Appl Environ Microbiol 63:4516–4522
Macek T, Kotrba P, Suchova M, Skacel F, Demnerova K, Ruml T (1994) Accumulation of cadmium by hairy root cultures. Biotechnol Lett 16:621–624
Macek T, Mackova M, Burkhard J, Demnerova K (1998) Introduction of green plants for the control of metals and organics in environmental remediation. In: Holm FW (ed) Effluents from alternative demilitarization technologies. NATO PS Series 1, vol. 12. Kluwer Academic Publishers, Dordrecht, Boston, pp 71–85
Macek T, Mackova M, Kas J (2000) Exploitation of plants for the removal of organics in environmental remediation. Biotechnol Advance 18:23–35
Macek T, Kotrba P, Svatos A, Novakova M, Demnerova K, Mackova M (2008) Novel roles for genetically modified plants in environmental protection. Trends Biotechnol 26:146–152
Mackova M, Macek T, Burkhard J, Ocenaskova J, Demnerova K, Pazlarova J (1997a) Biodegradation of polychlorinated biphenyls by plant cells. Int Biodeterior Biodegrad 39:317–325
Mackova M, Macek T, Kucerova P, Burkhard J, Pazlarova J, Demnerova K (1997b) Degradation of polychlorinated biphenyls by hairy root culture of Solanum nigrum. Biotechnol Let 9:787–790
Mackova M, Dowling D, Macek T (eds) (2006) Phytoremediation and rhizoremediation. Theoretical background. FOCUS on Biotechnology series, vol. 9A. Springer, Dordrecht, p 300
Mackova M, Vrchotova B, Francova K, Sylvestre M, Tomaniova M, Lovecka P, Demnerova K, Macek T (2007) Biotransformation of PCBs by plants and bacteria, consequences of plant–microbe interactions. Eur J Soil Biol 43:233–241
McCutcheon SC, Schnoor JL (eds) (2003) Phytoremediation: transformation and control of contaminants. Wiley, New York
McGuiness M, Dowling D (2009) Plant-associated bacterial degradation of toxic organic compounds in soil. Int J Environ Res Public Health. doi:10.3390/ijerph60x000x
Narasimhan KC, Basheer VB, Bajic B, Swarup S (2003) Enhancement of plant-microbe interactions using a rhizosphere metabolomics-driven approach and its application in the removal of polychlorinated biphenyls. Plant Physiol 132:146–153
Novakova M, Mackova M, Chrastilova Z, Szekeres M, Demnerova K, Macek T (2009) Cloning the bacterial bphC gene into Nicotiana tabacum to improve the efficiency of PCB phytoremediation. Biotechnol Bioeng 102:29–37
Pavlikova D, Macek T, Mackova M, Pavlik M (2007) Monitoring native vegetation on a dumpsite of PCB-contaminated soil. Int J Phytoremed 9:71–78
Rezek J, Macek T, Mackova M, Triska J (2007) Plant metabolites of polychlorinated biphenyls in hairy root culture of black nightshade Solanum nigrum SNC-9O. Chemosphere 69:1221–1227
Rezek J, Macek T, Mackova M, Triska J, Ruzickova K (2008) Hydroxy-PCBs, methoxy-PCBs and hydroxy-methoxy-PCBs: Metabolites of polychlorinated biphenyls formed in vitro by tobacco cells. Environ Sci Technol 42:5746–5751
Rezek J, in der Wiesche C, Mackova M, Zadrazil F, Macek T (2009) Biodegradation of PAHs in long-term contaminated soil cultivated with European white birch (Betula pendula) and red mulberry (Morus rubra) tree. Int J Phytorem 11:1–16
Ryslava E, Krejcik Z, Macek T, Novakova H, Demnerova K, Mackova M (2003) Study of PCB degradation in real contaminated soil. Fresen Environ Bull 12:296–301
Safe SH (1994) Polychlorinated biphenyls (PCBs): environmental impact, biochemical and toxic responses, and implications for risk assessment. Crit Rev Toxicol 24:87–149
Singer AC (2006) The chemical ecology of pollutant biodegradation. Bioremediation and phytoremediation from mechanistic and ecological perspectives. In: Mackova M, Dowling D, Macek T (eds) Phytoremediation and rhizoremediation. Theoretical background. Focus on biotechnology, 9A. Springer, Dordrecht, pp 5–21
Singer AC, Crowley DE, Thompson IP (2003) Secondary plant metabolites in phytoremediation and biotransformation. Trends Biotechnol 21:123–130
Soudek P, Podlipna R, Vanek T (1998) Phytoremediation of heavy metals by hairy root culture of Armoracia rusticana. Int J Biodeterior Biodegrad 42:235–236
Sylvestre M (1980) Isolation method for bacterial isolates capable of growth on p-chlorobiphenyl. Appl Environ Microbiol 39:1223–1224
Sylvestre M, Macek T, Mackova M (2009) Transgenic plants to improve rhizoremediation of polychlorinated biphenyls (PCBs). Curr Opin Biotechnol 20:242–247
Tillmann S, Strompl C, Timmis KN, Abraham WR (2005) Stable isotope probing reveals the dominant role of Burkholderia species in aerobic degradation of PCBs. FEMS Microbiol Ecol 52:207–217
Tlustos P, Pavlikova D, Szakova J, Fischerova Z, Balik J (2006) Exploitation of fast growing trees in metal remediation. In: Mackova M, Dowling D, Macek T (eds) Phytoremediation and rhizoremediation. Theoretical background. Focus on biotechnology series, vol. 9A. Springer, Dordrecht
Uhlik O, Jecna K, Leigh MB, Mackova M, Macek T (2009a) DNA-based stable isotope probing: a link between community structure and function. Sci Total Environ 407:3611–3619
Uhlik O, Jecna K, Mackova M, Vlcek C, Hroudova M, Demnerova K, Paces V, Macek T (2009b) Biphenyl-metabolizing bacteria in the rhizosphere of horseradish and bulk soil contaminated by polychlorinated biphenyls as revealed by stable isotope probing. Appl Environ Microbiol. doi:10.1128/AEM.00466-09
Van Aken B (2008) Transgenic plants for phytoremediation: helping nature to clean up environmental pollution. Trends Biotechnol 26:225–227
Villacieros M, Whelan CM, Mackova M, Molgaard J, Sanchez-Contreras M, Lloret J, Aguirre de Carcer D, Bolanos L, Oruezabal RI, Macek T, Karlson U, Dowling DN, Martin M, Rivilla R (2005) PCB rhizoremediation by Pseudomonas fluorescens F113 derivatives using a Sinorhizobium meliloti nod system to drive bph gene expression. Appl Environ Microbiol 71:2687–2694
Weyens N, van der Lelie D, Taghavi S, Vangronsveld J (2009) Phytoremediation: plant–endophyte partnerships take the challenge. Curr Opin Biotechnol 20:248–254
Wilken A, Bock C, Bokern M, Harms H (1995) Metabolism of different PCB congeners in plant cell cultures. Environ Chem Toxicol 14:2017–2022
Zarevucka M, Wimmer Z (2008) Plant products for pharmacology: application of enzymes in their transformations. Int J Mol Sci 9:2447–2473
Zhou J, Bruns MA, Tiedje JM (1996) DNA recovery from soils of diverse composition. Appl Environ Microbiol 62:316–322
Acknowledgements
The authors thank the support of projects COST 859, the Grant Agency of the Czech Republic 525/09/1058, and projects funded by Czech Ministry of Education 2B06156, 6046137305, Z40550506. The help of many colleagues and students is highly appreciated. Especially, Mr. Rudolf Kaplanek deserves special thanks for his enthusiasm, patience and permission to access to site.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editors: Peter Schröder and Jean-Paul Schwitzguébel
Rights and permissions
About this article
Cite this article
Mackova, M., Prouzova, P., Stursa, P. et al. Phyto/rhizoremediation studies using long-term PCB-contaminated soil. Environ Sci Pollut Res 16, 817–829 (2009). https://doi.org/10.1007/s11356-009-0240-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-009-0240-3