Abstract
A total of 191 yeasts were isolated from 197 samples collected from eight estuarine mangrove forests along four different coastlines of Thailand (Andaman Sea and the East, North and West coasts of the Gulf of Thailand). Of these, 178 isolates were identified as 32 species in 16 genera of Ascomycota, 12 species in nine genera of Basidiomycota, and 13 isolates as potential new species, respectively. Mangroves located along the Andaman Sea coastline had a higher yeast diversity at the species and genera levels than those along the Gulf of Thailand. Kluyveromyces siamensis was the most frequently isolated species, whilst Candida tropicalis was the only species isolated at all eight sites. Screening isolated yeast strains belonging to genera previously reported as oleaginous yeast plus the 13 potential new species, revealed two oleaginous strains, Rhodotorula sphaerocarpa 11-14.4 and Saitozyma podzolica 11-11.3.1. Both of these strains were isolated from the same mangrove forest on the Andaman Sea coastline. They could accumulate lipid when suspended in glucose solution without any supplementation, while the fatty acid composition and oil profile of Rh. sphaerocarpa 11-14.4 and Sait. podzolica 11-11.3.1 were similar to vegetable oil and cocoa butter, respectively.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Mangroves, an ecotone ecosystems, that are transitional between terrestrial and marine habitats (Alongi et al. 2001) in estuaries of tropical and sub-tropical climate regions (Kathiresan and Bingham 2001). Mangroves are unusually rich in microbial species including yeasts. The yeasts are involved in detrital food chain in this ecosystem that is rich in degrading plant materials (Meyers et al. 1971) and a food source for some marine invertebrates and zooplanktons (Araujo et al. 1995; Kutty and Phili 2008; Fell et al. 2011).
In particular, some yeasts can accumulate intracellular lipids at high levels in the form of triacylglycerols as oil droplets (yeast oil) when they are grown under an excess carbon but limited nitrogen condition (Thanh 2006). The lower nitrogen condition causes an increased adenosine monophosphate (AMP) deaminase activity, which leads to decreased AMP and isocitrate dehydrogenase activity, and an accumulation of citrate in the TCA cycle. The citrate subsequently enters the fatty acid synthesis pathway upon conversion to acetyl CoA. Yeasts that accumulate intracellular lipid above 20% (w/w) by dry cell weight (DCW) are defined as oleaginous (Ratledge 1989).
Yeast oils have already been developed as an alternative source of biofuels and high-valued oils for oleochemical industries (Papanikolaou et al. 2001; Sitepu et al. 2014; Bandhu et al. 2018). The major fatty acids in most yeast oils are myristic (C14:0), palmitic (C16:0), palmitoleic (C16:1), stearic (C18:0), oleic (C18:1), linoleic (C18:2) and linolenic (C18:3) acids, which is similar to those of plant oils that are used as feedstock for biodiesel production (Beopoulos and Nicaud 2012). Oils of several oleaginous yeast strains, such as Yarrowia lipolytica, Rhodosporidium toruloides and Cryptococcus curvatus, are mainly composed of steric, oleic and palmitic acids, which is similar to cocoa butter, a high value natural fat extracted from cocoa beans (Hassan et al. 1994, 1995; Papanikolaou et al. 2001, 2003; Wu et al. 2011). Oils of Rhodotorula mucilaginosa IIPL32 have a fatty acid composition, physicochemical and tribophysical properties suitable as a renewable base oil for biolubricant production (Bandhu et al. 2018). Oils of Pichia segobiensis SSOH12 had a considerable amount (16%) of palmitoleic acid, an omega 7, and may be suitable for medical applications (Schulze et al. 2014).
The usage of yeast for oil production provides better benefits over plants or other microorganisms, as yeasts grow fast without effects from changing climatic or seasonal conditions, not requiring land space for plantation compared with plant cultivation and easier for production expansion versus single cell algae, ability to use diverse carbon sources (Sitepu et al. 2014). The component of oil produced from yeasts will be different dependent on the carbon sources used (Ageitos et al. 2011). Therefore, yeast oil can be used in a variety of industries.
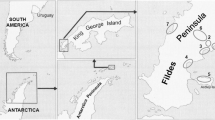
There are around 2400 km2 of mangrove forests in Thailand (Pumijumnong 2014), which are situated across four distinctively separate coastal regions along the Andaman Sea and the Gulf of Thailand (GOT) (Vibulsresth et al. 1975) namely: (i) the Andaman Sea coastline on the west of Thailand, which covers the five provinces of Ranong, Phuket, Krabi, Trang and Satul; (ii) the northern GOT coastline, which covers the four provinces of Samut Prakarn, Samut Sakorn, Samut Songkram and Bangkok (iii) the eastern GOT coastline, which covers the five provinces of Trat, Chantaburi, Rayong, Cholburi and Chachoengsao; and (iv) the western GOT coastline, which covers the eight provinces of Petchaburi, Prachuap Khirikhan, Chumporn, Surathani, Nakorn Sri Thamarat, Songkhla, Pattani and Naratiwat (Fig. 1).
Oleaginous yeasts have been found in various habitats, such as soil and the surface of flowers, fruits or plant leaves (Schulze et al. 2014; Jiru et al. 2016; Maina et al. 2017). Mangrove forests are known to have a high yeast diversity, and as such there were several reports on new yeast species isolated from different mangrove forests and coastlines in Thailand (Limtong et al. 2004, 2007; Limtong and Yongmanitchai 2010; Am-In et al. 2008, 2011). Lipid accumulation by yeasts is somewhat strain dependent, and not species or genus dependent (Polburee et al. 2015). There was a high success rate in obtaining oleaginous yeast strain by screening from those capable to grow in nitrogen-limited medium (Kraisintu et al. 2010; Sitepu et al. 2013). The high yeast diversity in mangrove forests increases the chance to isolate oleaginous yeast strain including new species of oleaginous yeast. Moreover, as the fatty acid composition of yeast oil is species dependent, the new species of oleaginous yeast isolated increases chance to obtain new high value yeast oil.
The objective of this study was to isolate diverse yeasts using nitrogen-limited medium from eight mangrove forests located in six provinces across the four different coastlines of Thailand, identifying the diversity of isolated yeasts, screening them for oleaginous yeast strains and characterizing their lipid production.
Materials and methods
Sample collection and yeast isolation
A total of 197 samples, comprised of soil, water and decayed biological matter, were collected from eight mangrove forests at four different coastlines (Tables 1 and 2). The sampled sites were (i) Ranong Biosphere Reserve (RBR; Ranong province) and (ii) Mangrove Resource Development station (MRDS) no. 11 (Ranong province) on the Andaman Sea coastline; (iii) MRDS no. 7 (Samut Songkhram province) on the northern GOT coastline; (iv) MRDS no. 2 (Chanthaburi province) (v) MRDS no. 4 (Trat province) and (vi) MRDS no. 45 (Trat province) on the eastern GOT coastline; and (vii) Pranburi National Forest Park (PNFP; Prachuap Khiri Khan province) and (viii) MRDS no. 6 (Phetchaburi province) on the western GOT coastline (Fig. 1). The latitude and longitude of these sampled mangrove forests are shown in Table 1.
Each sample (1 g or 1 ml) was inoculated into 10 ml of nitrogen-depleted medium (NDM; 20 g/l glucose, 0.85 g/l KH2PO4, 0.15 g/l K2HPO4·3H2O, 0.5 g/l MgSO4·7H2O, 0.1 g/l NaCl, 0.1 g/l CaCl2·6H2O, 0.5 mg/l H3BO3, 0.04 mg/l CuSO4·5H2O, 0.1 mg/l KI, 0.2 mg/l FeCl3·3H2O, 0.4 mg/l MnSO4·H2O, 0.2 mg/l Na2MOO4·2H2O, 0.4 mg/l ZnSO4·7H2O, pH 5.5) and supplemented with 100 mg/l chloramphenicol and incubated at 30 ºC, 200 rpm for 2 d (Thanh 2006). The resultant culture was streaked on NDM-agar and incubated at 30 ºC for 5 d. Purification to clonality of the culture was performed by the streak plate method on YM agar (3 g/l yeast extract, 3 g/l malt extract, 5 g/l peptone, 10 g/l glucose, 20 g/l agar, pH 5.5). Each clonal culture was kept on a YM agar slant at 4 ºC for further study.
Molecular identification of yeast isolates
Genomic DNA extraction
Yeast cells were lysed in 200 µl lysis solution (60% (w/v) Yatalase, 6% (v/v) Yatalase buffer, 6% (w/v) RNase, 4.65% (w/v) NaCl, 10 mM K2HPO4, 100 mM EDTA, pH 8) at 37 °C for 1–2 h, vortex mixed with glass beads (diameter 0.8 mm; Sigma-Aldrich Co., LLC., U.S.A.) in 8% (w/v) sodium dodecyl sulphate (67 µl) for 1.5 min and incubated at 60 °C for 10 min. The cell lysate was further mixed with 87 µl of 3 M sodium acetate (pH 5) on ice, centrifuged at 20,600×g, 4 °C for 5 min, and the supernatant was transferred into Acroprep 96 multi-well filter plate (PALL®, Pall corporation, U.S.A.) containing 110 µl isopropanol in each well. The DNA pellet obtained after centrifugation (1580×g, 16 °C for 5 min) was washed twice with 200 µl of 70% (v/v) ethanol, dissolved in 60 µl TE buffer and kept at −20 °C until used.
(GTG) 5 fingerprinting pattern
The extracted genomic DNA (1–20 ng/µl) was PCR amplified in a 10 µl reaction volume comprised of 1 µl DNA template, 0.2 µl of 10 mM (GTG)5 primer (5′-GTGGTGGTGGTGGTG-3′) (Meyer et al. 1993), 5 µl 2 × Go Tag green and 3.8 µl distilled water. The thermal cycling was performed at 95 °C for 5 min followed by 40 cycles of 95 °C for 45 s, 50 °C for 1 min and 72 °C for 1 min, and then a final 72 °C for 6 min. The PCR products were resolved by 1.5% (w/v) agarose gel electrophoresis and visualized after ethidium bromide staining. The resolved pattern was defined as the (GTG)5 fingerprint pattern. The (GTG)5 fingerprint patterns were grouped according to their respective relevant similarity.
Sequencing of the 26S rRNA gene (D1/D2 domain)
The 26S rRNA gene at the D1/D2 domain (LSU D1/D2 domain) was PCR amplified using the NL1 (5′-GCATATCAATAAGCGGAGGAAAAG-3′) and NL4 (5′-GGTCCGTGTTTCA AGACGG-3′) primers (O’Donnell and Gray 1995). The PCR was performed in a 20 µl reaction volume containing 2 µl DNA template, 0.4 µl of each primer (10 pmol/µl), 10 µl 2 × Go Tag green and 7.2 µl distilled water, and was thermocycled at 94 °C for 3 min, followed by 36 cycles of 94 °C, 52 °C and 72 °C, each for 30 s, and then a final 72 °C for 5 min. The PCR product was purified by washing with sterile deionized water, filtered through a MinElute® multi-well filter plate (QIAGEN sciences, U.S.A.) and then used as the DNA template for the thermal cycle sequencing reaction. Primers used were NL1, NL4, NL3A (5′-GAGACCGATAGCGAACAAG-3′) and NL2A (5′-CTTGTTCG CTATCGGTCTC) (Kurtzman and Robnett 1998). Cycle sequencing was performed in a 10 µl reaction volume using a BigDye® Terminator V3.1 Cycle Sequencing kit (Applied Biosystems, U.S.A.). Each reaction contained 1–2 µl DNA template and 1.5 µl of the respective primer (1 pmol/μl). Samples were thermocycled at 96 °C for 10 min, followed by 25 cycles of 96 °C for 10 s, 50 °C for 5 s and 60 °C for 4 min, followed by a final 60 °C for 10 min. The PCR products were purified and sequence analyzed using an Auto-sequencer ABI Prism 3130xl Genetic analyzer (Applied Biosystems, USA) following the manufacturer’s instructions.
The DNA sequence was manually edited using the BioEdit program version 7.2.5 (Hall 1999) and compared to those in the online database available in the NCBI GenBank using the BLASTn program.
Biodiversity analysis
Yeast diversity in each sampling sites was analyzed using Shannon–Wiener index (H′), \(H^{\prime} = - \sum\nolimits_{{i = 0}}^{s} {Pi\left( {\ln Pi} \right)}\) where Pi is the proportion of the number of yeast strains in each species to the total number of yeast strains in that sampling site (Shannon 1948; Spellerberg and Fedor 2003). Yeast species evenness in sampling sites was determined using equitability (EH), \(E_{H} = \frac{{H^{\prime}}}{{\ln S}},\) where S is total number of yeast species in each sampling site (Pielou 1975). Evenness indices range from 0 to 1, where a value close to 1 means there is complete evenness among all species in that area (Heip et al. 1998).
Nile red staining
Yeast isolates that were designated as likely to belong to genera known to contain oleaginous yeasts, plus the potential new species, were grown on modified YM agar (20 × diluted nitrogen source) at 30 °C for 5 days. The resultant cells were suspended in Nile red solution (50 µg/ml in acetone diluted 100 × with 25% (v/v) dimethyl sulfoxide) and incubated at 30 °C for 10 min. The intracellular lipid accumulation was preliminary examined under a fluorescence microscope (Olympus BX51, USA) using a U-WNB2 filter with excitation and emission wavelengths of 470–490 and 520 nm, respectively (Greenspan et al. 1985). The known oleaginous yeast, Lipomyces starkeyi JCM 5995, was used as a positive control.
Analysis of the intracellular lipid content
Nile red stained yeasts that showed an oil droplet bigger than 2/3 of the cell size were selected for analysis of their intracellular lipid content. One loopful of yeast grown on YM agar was transferred into YM broth (50 ml) in a 250-ml flask and incubated at 30 °C, 200 rpm for 24 h. A 15-ml aliquot was then inoculated into YM broth (150 ml) in a 500-ml flask and incubated at 30 °C, 200 rpm for 48 h. The resultant culture was collected by centrifugation (9803×g, 4 °C, 15 min) and the cell pellet was washed with lipid production medium (LPM; modified from Galafassi et al. (2012) to contain 50 g/l glucose, 1 g/l yeast extract, 0.05 g/l MgSO4·7H2O, 1 g/l KH2PO4, 1 g/l (NH4)2SO4, 0.01 g/l NaCl, 0.01 g/l CaCl2.·2H2O, pH 5.5). The washed cells were inoculated into 150 ml LPM in a 500-ml flask and incubated at 30 °C, 200 rpm for 6 days. Cells were then harvested by centrifugation (as above), washed with distilled water and lyophilized. The intracellular lipid level of the lyophilized cells was determined as described previously (Folch et al. 1957). The lyophilized cells (1 g DCW) were suspended in 20 ml of 2:1 (v/v) chloroform: methanol, sonicated at 37 kHz, room temperature for 30 min (Elmasonic, E60H model, Germany) and centrifuged at 9803×g, 4 °C for 40 min. The supernatant was harvested, dried by evaporation at room temperature and the weight of the lipid residue was measured.
Analysis of the fatty acid composition
Lipid was extracted and converted to fatty acid methyl esters (FAMEs) as reported (Anamnart et al. 1998). In brief, 1 g of wet cells was saponified with 0.8 ml of 10% (w/v) potassium hydroxide in methanol at 80 °C for 2 h. After cooling down to room temperature, 1 ml of petroleum ether was added to remove the unsaponified materials in the reaction mixture. The obtained aqueous phase was acidified by 0.3 ml of 6N hydrochloric acid and extracted with diethyl ether to recover the fatty acids. The fatty acid fraction was evaporated to dryness under nitrogen gas and derivatized to FAMEs using BF3/MeOH, and then the FAMEs were extracted in hexane and further analyzed on a flame ionization detector system using gas chromatography (GC; Agilent Technologies 6890N, USA) equipped with an INNOWAX capillary column (30 m × 0.3 mm, 0.2 μm film thickness) as reported (Limsuwatthanathamrong et al. 2012). Helium was used as the carrier gas at a flow rate of 2.3 ml/min. The temperature program started at 150 °C and was then increased to 180 °C, 200 °C and 205 °C at a rate of 10 °C/min, 5 °C/min and 0.5 °C/min, respectively. The temperature was then held at 205 °C for 2 min before increasing to 250 °C at 5 °C/min and maintained for 5 min. The FAMEs were identified by comparison with reference standards.
Phenotypic characterization of the oleaginous yeast isolates
To confirm the molecular identification results of the isolated oleaginous yeasts, morphological and physiological characteristics of the isolated oleaginous yeasts were determined and then compared to the description of type strains in Kurtzman et al. (2011). Cell morphology was observed under light microscopy, while the colony morphology, including the texture, color, surface, elevation and margin were recorded visually. Carbon assimilation tests were conducted using an ID 32 C kit (BioMerieu, France) following the manufacturer's instructions. Formation of pseudomycelium and true mycelium were examined by the Dalmau slide culture method at 25 °C for up to 14 days (Kurtzman et al. 2011).
Determination of lipid accumulation in glucose solution
Capability of the isolated oleaginous yeasts to accumulate oil in the absence of a nitrogen source was determined (Lin et al. 2011). One loopful of oleaginous yeasts grown on YM agar was transferred into YM broth (50 ml) in a 250-ml flask and incubated at 30 °C, 200 rpm for 24 h. A 15-ml aliquot was then inoculated into YM broth (150 ml) in a 500-ml flask and incubated at 30 °C, 200 rpm for 48 h. The cells were then collected by centrifugation (4 °C, 9803×g, 10 min), washed with sterile distilled water twice, and suspended in 150 ml glucose solution (40 g/l) in a 500-ml flask and incubated at 30 °C with agitation at 200 rpm for 60 h. Every 12 h, cells (three independent culture flasks) were harvested by centrifugation, washed with sterile distilled water and lyophilized. The DCW, lipid content (% (w/w, DCW)) and lipid yield were determined.
Results
Isolation and molecular identification of yeast isolates
A total of 191 yeasts were isolated and categorized by PCR (GTG)5 fingerprinting. The obtained (GTG)5 fingerprint patterns were manually grouped into 94 distinct patterns. One representative strain from each of the 94 patterns was then selected for molecular operational taxonomic unit (MOTU) identification and species inference by comparing their LSU D1/D2 domain sequence (500–600 bp) with those deposited in the NCBI GenBank database using the BLASTn program. Yeast strains with six or more nucleotide substitutions (> 1%) were likely to be different species, while strains with 0–3 nucleotide substitutions were assigned as conspecific or sister species (Kurtzman and Robnett 1998). In this study, yeast strains with less than 1% nucleotide substitution were assigned as the same species, and the isolates with the same (GTG)5 fingerprint pattern were assumed to be the same. The term ‘aff.’ (species affinis) was used to indicate a potentially new species that affiliated to but not identical to its closest known species. As a result, the 191 isolated yeast strains were classified to ascomycetous yeasts (156), basidiomycetous yeasts (34) and yeast-like algae (1). From the 156 yeast strains belonging to the Ascomycota, 147 strains were classified to 32 species in 16 genera. Another nine strains were assigned as five new species within the genera Wickerhamomyces sp. (two strains), Yamadazyma sp. (one strain), Clavispora sp. (one strain), Pichia sp. (three strains) and Candida sp. (two strains). Thirty one out of the 34 strains belonging to the Basidiomycota were classified to 12 species in nine genera, while the three remaining strains were assigned as potential new species in the genera Sakaguchia, Heterocephalacria and Goffeauzamy. One other strain was classified as a potential new species in the genus Prototheca, a yeast-like non-photosynthetic algae with yeast-like cells and colonies. The number of each species of yeasts isolated, including the accession number of the LSU D1/D2 domain sequence of each representative yeast species submitted to the GenBank database are shown in Table 2.
Yeast diversity and community
Andaman Sea coastline
At RBR, a total of 24 yeast strains were isolated from 20 collected samples. Nineteen and four strains belonged to Ascomycota (thirteen species in eight genera) and Basidiomycota (four species in three genera), respectively. The remaining basidiomycetous yeast was ascribed as new species closely related to Heterophalacria arrabidensis with 2.44% nucleotide substitutions. The most frequently isolated species were Candida aaseri and C. thaimuengensis with a fairly even distribution. The species isolated only at this site were Candida aaseri, C. intermedia, C. sorboxylosa, Pichia occidentalis, P. sporocuriosa, Rhodotorula sphaerocarpa, Suhomyces atakaporum and the new species (Tables 2 and 3). The Shannon diversity index (H') and community evenness (EH) of the isolated yeasts were 2.79 and 0.96, respectively (Table 3).
For MRDS no. 11, 34 yeast strains were isolated from 23 collected samples. Eighteen strains were ascomycetous yeasts (eight species in six genera) and 13 strains were basiodiomycetous yeasts (nine species in five genera). The remaining three strains were ascribed as new species closely related to Goffeauzyma gilvescens (one strain) and Candida silvanorum (two strains) with 8.15% and 12.79% nucleotide substitutions, respectively. The most frequently isolated species were Kluyveromyces siamensis and Schwanniomyces vanrijiae. The species isolated at this site only were Candida maltose, C. silvanorum, Hannaella phetchabunensis, Kwoniella dejecticola, Metahyphopichia laotica, Naganishia liquefaciens, Nag. albida, Rhodotorula toruloides, R. mucilaginosa, Schwanniomyces vanrijiae and the two new species (Tables 2 and 3). The H' and EH of the isolated yeasts were 2.83 and 0.96, respectively (Table 3).
Gulf of Thailand
At MRDS no. 7, 16 ascomycetous yeasts (six species in five genera) plus two new species closely related to Candida quercuum (two strains) and Candida ecuadorensis (one strain) with 5.13% and 6.78% nucleotide substitutions, respectively, were isolated from 25 collected samples. Candida tropicalis was the most prevalent species, while Ambrosiozyma monospora, Yamadazyma mexicana and the two new species were unique species to this site (Tables 2 and 3). The H' and EH of the isolated yeasts were 1.81 and 0.87, respectively (Table 3).
For MRDS no. 2, 12 yeast strains including four species in four genera of Ascomycota (11 strains) and one species of Basidiomycota (one strain) were isolated from 22 collected samples. Candida amphicis was only isolated from this site, while Kluyveromyces siamensis was by far the most common species isolated (Tables 2 and 3). The H' and EH of the yeast isolated were 1.47 and 0.91, respectively (Table 3).
For MRDS no. 4, 29 yeast strains were isolated from the 26 collected samples and contained nine species in seven genera of Ascomycota (18 strains) and three species in three genera of Basidiomycota (10 strains) plus one new species closely related to Candida californica with 2.23% nucleotide substitutions. Kluyveromyces siamensis and Rhodotorula paludigena were the most prevalent species. Candida nonsorbophila, C. orthopsilosis, Meyerozyma guilliermondii, Pseudozyma huberensis and Schwanniomyces polymorphus were unique species to this site (Tables 2 and 3). The H' and EH of the isolated yeasts were 2.27 and 0.89, respectively (Table 3).
Seventeen yeasts isolated from 25 samples collected at MRDS no. 45 were composed of 11 strains of ascomycetous yeasts (nine species in six genera), three strains of basidiomycetous yeasts (two species in two genera) plus two new species that were closely related to Sakaguchia lamellibrachiae (one strain) and Candida californica (two strains) with 2.44% and 2.22% nucleotide substitutions, respectively. The most frequently isolated species was Candida tropicalis. The new species (one strain) and Candida sithepensis were only isolated at this site (Tables 2 and 3). The H' and EH of the isolated yeasts were 2.48 and 0.97, respectively (Table 3).
At PNFP, 35 yeast strains were isolated from 34 collected samples. Thirty four strains were ascribed to five species in four genera of Ascomycota plus one new species closely related to Prototheca wickerhamii with 9.14% nucleotide substitutions. The most common species isolated was Kluyveromyces siamensis. In addition to the new species, Candida viswanathii and C. andamanensis were unique to this site (Tables 2 and 3). The H' and EH of the isolated yeasts were 1.18 and 0.66, respectively (Table 3).
Finally, at MRDS no. 6, 21 yeasts strains were isolated from 22 samples and were comprised of 20 strains of ascomycetous yeasts (seven species in five genera) plus one new species closely related to Yamadazyma mexicana with 2.64% nucleotide substitutions. The new species plus Wickerhamomyces anomalus, Kluyveromyces aestuarii and Pichia kudriavzevii were only isolated from this site, while the most common species was Kluyveromyces siamensis (Tables 2 and 3). The H' and EH of the isolated yeast were 1.91 and 0.92, respectively (Table 3). The relative abundance of the yeast species among the different sampling sites is shown in Fig. 2.
Nile red staining
Of the 191 isolated yeast strains, 30 were designated to belong to a genera that has been reported to contain oleaginous yeasts; Rh. paludigena, Rh. sphaerocarpa, Rh. toruloides, Rh. mucilaginosa, Sait. podzolica, Pap. flavescens, Pap. laurentii, Nag. liquefaciens, Nag. albida, Kwoniella dejecticola and Debaryomyces nepalensis (Li et al. 2008; Rossi et al. 2009; Ageitos et al. 2011; Kurtzman et al. 2011; Sitepu et al. 2012). These 30 yeast strains plus the 13 strains ascribed as potential new species were selected for preliminary determination of their intracellular lipid accumulation by Nile red staining. Of these 43 selected yeast strains, 11 (Rh. sphaerocarpa 11-14.4, Rh. paludigena BW7.3, Rh. paludigena 11-14.7, Rh. paludigena MTW11.1, Rh. paludigena BW1.3, Rh. paludigena BW8.1, Rh. paludigena WW1.2, Sait. podzolica 11-11.3.1, Pap. laurentii MTT4.1, Prototheca sp. NSP10-2 and Debaromyces nepalensis NSB6-1) showed an oil droplet bigger than 2/3 of the cell size and so were selected for further analysis of their lipid content. The oil droplet stained with Nile red of Rh. sphaerocarpa 11-14.4, Sait. podzolica 11-11.3.1 and Lipomyces starkeyi JCM 5995 (positive control) are showed in Fig. 3.
Nile red staining of a Rhodotorula sphaerocarpa 11-14.4, b Saitozyma podzolica 11-11-3.1 and c Lipomyces starkeyi JCM 5995 grown on modified YM agar (20 × diluted nitrogen source) at 30 °C for 5 days. Bright field (left) and fluorescent (right) images at × 100 magnification. Scale bar = 10 μm. Each image is a representative of at least three such fields of view per sample and two independent samples
Analysis of lipid accumulation
Analysis of the intracellular lipid content of the 11 selected yeast strains revealed that Rh. sphaerocarpa 11-14.4 and Sait. podzolica 11-11.3.1 had an intracellular lipid content in excess of 20% (w/w, DCW), at 27.3 ± 0.006 and 24.0 ± 0.010% (w/w, DCW), respectively, and so were designated as oleaginous strains (Fig. 4) and selected for further fatty acid composition analysis. Of interest, both these oleaginous strains (Rh. sphaerocarpa 11-14.4 and Sait. podzolica 11-11.3.1) were isolated from the same site (MRDS no. 11 (Ranong province) on the Andaman Sea coastline).
Intracellular lipid accumulation of the 11 yeast strains having an oil droplet bigger than 2/3 of their cell size. (1) Rh. sphaerocarpa 11-14.4 (2) Rh. paludigena BW7.3 (3) Rh. paludigena 11-14.7 (4) Rh. paludigena MTW11.1 (5) Rh. paludigena BW1.3 (6) Rh. paludigena BW8.1 (7) Rh. paludigena WW1.2 (8) Sait. podzolica 11-11.3.1 (9) Pap. laurentii MTT4.1 (10) Prototheca sp. NSP10-2 (11) Debaromyces nepalensis NSB6-1 and (control) Lipomyces starkeyi JCM 5995. Data are shown as the mean ± 1SD, derived from three independent trials
Fatty acid composition
The fatty acid composition of the two oleaginous strains is presented in Table 4. The major fatty acids were oleic, steric, palmitic and linoleic acids. The oil from Rh. sphaerocarpa 11-14.4 was comprised of oleic, palmitic and linoleic acids at 55.7, 18.9 and 12.4%, respectively, while myristic, linolenic and palmitoleic acids were present in lesser amounts. For Sait. podzolica 11-11.3.1, the three main fatty acids were steric (28.7%), oleic (28.7%) and palmitic (25.9%) acids, with linoleic and myristic acids as minor and trace amounts, respectively.
Phenotypic characteristics of the oleaginous yeast isolates
Morphological and physiological characteristics of the oleaginous strains, Rh. sphaerocarpa 11-14.4 and Sait. podzolica 11-11-3, were the same as their type strains, Rh. sphaerocarpum CBS5939T and Sait. podzolicus CBS 6819 T, respectively. The Rh. sphaerocarpa 11-14.4 and Sait. podzolica 11-11-3 were deposited in the Microbe Division/Japan Collection of Microorganisms, RIKEN BioResource Center, Tsukuba, Japan under numbers JCM 32,652 and JCM32653, respectively.
Lipid accumulation of the oleaginous yeasts in glucose solution
The Rh. sphaerocarpa 11-14.4 suspended in 40 g/l glucose solution had the highest lipid content at 32.1% (w/w, DCW) and lipid yield (3.04 g/l) at 48 h, while the cell biomass was highest (14.1 g/l) at 36 h (Fig. 5a). For Sait. podzolica 11-11.3.1 under the same conditions, the highest lipid content, lipid yield and cell biomass 25.8% (w/w DCW), 3.46 g/l and 13.40 g/l, respectively, were all found at 36 h (Fig. 5b).
Lipid accumulation in glucose solution of a Rh. sphaerocarpa 11-14.4 and b Sait. podzolica 11-11.3.1. Symbols used are: lipid content (filled circle), lipid yield (filled square) and dry cell weight (DCW) (filled triangle). The data are displayed as the mean ± SD, and are derived from triplicate experiments
Discussion
Based on the isolation condition performed in this study, ascomycetous yeasts were dominant and were found at all the sampling sites, whereas basidiomycetous yeasts were less prevalent and were not found at three sites (MRDS no.7, PNFP and MRDS no.6). Chi et al. (2012) used YPD as the isolation medium at 25 °C reported that ascomycetes yeasts were the most abundant yeasts in mangrove ecosystems in China, where they represented 95.2% of the 269 yeasts isolated from the sediment, water and various plant parts. Culture-independent studies on fungal diversity in mangrove forests of New Caledonia revealed that ascomycetes yeasts were dominant, and comprised 82% of the sequence reads (Arfi et al. 2012). The distribution and population of yeasts were depended on the type and concentration of organic materials (Kutty and Philip 2008).
Among the 44 known species isolated in this study, 33 species have been reported from a mangrove environment (Araujo et al. 1995; Fell et al. 2011; Chi et al. 2012; Kunthiphun et al. 2018). Many of these yeast species were likely to have been introduced from terrestrial environments and the marine water. Terrestrial and human-associated yeasts are introduced into mangrove forests by rain, rivers and human activity, while marine yeasts can influx into this convergence area from marine waters by tidal action, natural drainage and human activities (Kutty and Philip 2008; Fell 2012; Libkind et al. 2017; Hagler et al. 2017).
The most prevalent yeast species isolated in this study was Kluyveromyces siamensis (21.5%) followed by Candida thaimueangensis (12.0%) and Candida tropicalis (11.5%), while the remaining species were represented by only 1–6 isolated strains (0.5–3.1%). Kluyveromyces siamensis was the most frequently isolated species (5/8 sampling sites; MRDS no. 11, MRDS no. 2, MRDS no. 4, PNFP and MRDS no. 6). This species was first isolated from mangrove water in Thailand (Am-In et al. 2008) and then subsequently reported from mangrove forests in China (Chi et al. 2012) and Thailand (Kunthiphun et al. 2018). The high prevalence of occurrence of K. siamensis in this study may suggest that mangroves are its natural habitat.
Candida thaimueangensis was described by Limtong et al. (2007) from mangrove water in Thailand and then in mangroves in the USA (Fell et al. 2011), China (Chi et al. 2012) and Thailand (Kunthiphun et al. 2018). However, Bautista-Gallego et al. (2011) reported finding C. thaimueangensis in fermented olives in Spain. Nevertheless, the high prevalence of occurrence of C. thaimueangensis in this study of Thai mangroves could suggest that mangroves are its primary habitat.
Candida tropicalis, a pathogenic yeast of human and marine invertebrates (Moore and Strom 2003; Wang et al. 2007), was the most widely distributed species, being isolated from all the sampling sites in this survey. Moreover, C. tropicalis was reported to be the most typical yeast species in mangroves (Hagler et al. 2017) and was found in all mangrove ecosystems surveyed in China (Chi et al. 2012). However, this species is widely distributed, being isolated from clinical samples and considered as a human-associated species but is also common in a variety of natural sources, such as plants, fruits, flower, soil, water and food products (Kvasnikov et al. 1975; Lachance et al. 2011). Thus, the ecological association of this species is hard to ascertain. Other opportunistic pathogen yeast species found in this survey were C. viswanathii, C. orthopsilosis, C. aaseri, C. intermedia, Wickerhamomyces anomalus, Meyerozyma guilliermondii and Pichia kudriavzevii. They are often observed in feces of warm-blooded animals. However, there are few mammals in mangroves but lots of birds so a fecal origin from birds for such species is very likely.
Kluveromyces aestuarii is marine adapted species which typically found in mangroves habitat (Kutty and Philip 2008; Fell 2012; Hagler et al. 2017). K. aestuarii was first described by Fell (1961) from estuaries in Florida (USA) and subsequently in mangroves in Brazil (Araujo et al. 1995; Araujo and Hagler 2011) China (Chi et al. 2012) and Thailand (Kunthiphun et al. 2018). It was found in detritus feeding crabs and shipworms (mollusk) (Araujo et al. 1995). Kluveromyces aestuarii has been used as an indicator species for natural well-preserved mangrove ecosystems (Araujo and Hagler 2011). In this study, K. aestuarii was only isolated from one site, at MRDS no.6, located in Bang Khunsai, which contains the largest cockle breeding and cultivating grounds in Thailand (Pumijumnong 2014). Accordingly, the occurrence of K. aestuarii at MRDS no.6 may suggest that this site is situated in a good practice mangrove community, or it may simply reflect its proximity to the large cockle bed.
Rhodotorula paludigena and Rh. sphaerocarpa are frequently found in marine habitats but not exclusively. Rh. paludigena was found in plant nectars (Canto et al. 2017) and Rh. Sphaerocarpa was found in freshwater habitats (Brandão et al. 2017). The two remaining Rhodotorula species observed in this study, namely Rh. toruloides and Rh. mucilaginosa, are found in a wide range of natural habitats such as phyllophane (Khunnamwong et al. 2018) soil, decayed plants (Sampaio 2011) and marine habitats (Kurtzman et al. 2011).
Heavy rain with enhanced river flow and floods can wash out terrestrial and freshwater microbial populations and carry them to mangrove forests (Araujo et al. 1998). In this study, four yeast species that are commonly found in soil (Saitozyma podzolica, Candida sithepensis, Pichia terricola and P. chibodoensis) were isolated. And three strains of the Sait. podzolica isolated were from different kind of samples, one strain from water at RBR and the other two strains from soil and decaying moss at MRDS no. 11. Saitozyma podzolica has previously been reported as a typical soil-borne yeast, since it is frequently isolated from various kinds of soil, such as podzolic and sod-podzolic soils in the taiga zone (Babjeva and Rheshetova. 1975), peat soil (Golubev 1991), spruce forest soil (Yurkov et al. 2012a) and forest and grassland soils (Yurkov et al. 2012b). Moreover, Sait. podzolica has been reported to occur in other habitats that contained high organic matter and high moisture content, such as rotten wood in a beech forest (Middelhoven et al. 2006), decayed biomaterial in a mangrove forest (Kunthiphun et al. 2018), litters from the temperate forests (Mašínová et al. 2017) and from the spruce frorest (Štursová et al. 2012), Sphagnum moss (Kachalkin et al. 2008), and water from tropical lake (Brandão et al. 2017). The oil from the oleaginous Sait. podzolica CPOH4, isolated from peat bog soil in the black forest of Germany, had a fatty acid profile suitable for biodiesel production (Schulze et al. 2014).
The highest diversity of yeasts was found at MRDS no. 11 (11 genera and 17 species) followed by RBR (10 genera and 16 species), which are both located in Ranong province on the Andaman Sea coastline. Shannon diversity index (H') of MRDS no. 11 and RBR were 2.83 and 2.79, respectively. Mangrove forests in the Ranong province were the least invaded by human activities compared to the other provinces in this study (Department of Marine and Coastal Resources 2014). The RBR has been declared a UNESCO Biosphere Reserve area (Pumijumnong 2014). The MRDS no. 11 and RBR are mature mangrove forests (Pumijumnong 2014) which have Rhizophora apiculata as their dominant plant species (Mangrove Conservation Office 2018). The R. apiculata is the highest biomass-mangrove plant species (Meepol 2010). Carbon sequestration in the MRDS no. 11 and RBR was higher than the other surveyed mangrove forests (Mangrove Conservation Office 2018). So they are potentially richer in nutrients for yeasts. Additionally, Candida tropicalis was the only human-associated species isolated at MRDS no. 11. This low level of human-associated yeast species indicates the lower level of human influence (Hagler et al. 2017; Libkind et al. 2017).
The MRDS2 and PNFP sites, located in Chanthaburi and Prachuap Khiri Khan provinces, respectively, have the lowest yeast diversity. More than 50% of the mangrove area in these provinces had been invaded for agriculture and aquaculture (Department of Marine and Coastal Resources 2014). The distribution of yeasts is influenced by the type and concentration of available nutrients (Kutty and Philip 2008), plant vegetation (Yurkov et al. 2012b; Hagler et al. 2017) and anthropogenic activities (Yurkov et al. 2012b; Yurkov 2017).
Among the 30 yeast strains designated to belong to a genera that has been reported to contain oleaginous yeast species plus the 13 strains ascribed as potential new species, only two strains (6.67%; Rh. sphaerocarpa 11-14.4 and Sait. podzolica 11-11-3.1) were found to be oleaginous yeasts. This result confirmed that the oleaginicity was species and genus independent. Interestingly, both of them were isolated from the same site (MRDS no. 11, Andaman Sea coastline), where there was the highest yeast diversity and so the potential chance to isolate such. Screening of oleaginous yeast from yeasts inherent in genus reported to contain oleaginous yeast provided the chance of finding more oleaginous yeast (Sitepu et al. 2013).
The Rh. sphaerocarpa 11-14.4 had an intracellular lipid content of 27% (w/w, DCW). Only 5% of oleaginous yeasts have been reported to accumulate intracellular lipid more than 25% (w/w, DCW) (Agetios et al. 2011). L. starkeyi (Angerbauer et al. 2008), Rhodotorula glutinis (Beopoulos et al. 2009) and Rhodotorula toruloides (Zhao et al. 2008) were reported to accumulate oil up to 70% (w/w, DCW). The lipid content of yeasts is markedly influenced by the growth conditions, such as the carbon source, C/N ratio, other nutrients, oxygen level, pH and temperature (Sitepu et al. 2013). Lipid content of Rhodotorula sphaerocarpa UCDFST 68–43 was increased from 25.95 to 36.6% (w/w, DCW) when nitrogen was removed from medium (Sitepu et al. 2013). Saitozyma podzolica CPOH4 grown in modified YM medium containing 50 g/l glucose at 25 °C, 130 rpm for 120 h had an intracellular lipid content of 34.6% (w/w, DCW) (Schulze et al. 2014). The intracellular lipid content of Sait. podzolica DMKU-CPC19 (1) grown in nitrogen-limited medium at 28 °C, 150 rpm for 120 h was 30.1% (w/w, DCW) (Polburee et al. 2015). This makes it impossible to compare our results to others (Ageitos et al. 2011).
The major fatty acids of Rh. sphaerocarpa 11-14.4 oil were oleic and palmitic acids, which is similar to vegetable oils, such as rapeseed (Hoekman et al. 2012), that are generally used as a raw material for biodiesel production. The oil from Rh. sphaerocarpa 11-14.4 could, therefore, serve as a raw material for biodiesel production. The major fatty acids of Sait. podzolica 11-11-3.1 oil were steric, oleic and palmitic acids, similar to those of cocoa butter, a high value natural fat extracted from cocoa beans (Zarringhalami et al. 2012). Cocoa butter has several applications in the food, medicine and cosmetic industries, such as providing the texture and structure of chocolate (Wang and Maleky 2018), increasing the HDL cholesterol and improvement of UV-induced erythema resistance (Marsu et al. 2004; Heinrich et al. 2006) and inhibition of lipid peroxidation (Marsu et al. 2004)). Cocoa butter like-lipids (CBLs) have been reported to be accumulated in several oleaginous yeasts, such as Cutaneotrichosporon (Cu.) curvatus ATCC 2059 (Hassan et al. 1994), Yarrowia lipolytica LGAM S(7)1 (Papanikolaou et al. 2001) and Yarrowia lipolytica ACA-DC 50109 (Papanikolaou et al. 2003) and Cu. oleaginosus DSM11815 (Wei et al. 2017), while Rh. toluroids Y4 produced CBLs under sulfate-limited conditions (Wu et al. 2011). The fatty acid composition and profile of the yeast oil depends on the culture medium and condition (Sitepu et al. 2014). The oil of Sait. podzplica CPOH4 grown in medium containing glucose as the sole carbon source had oleic (59.4%) and palmitic (18.4%) acids as the major fatty acids (Schulze et al. 2014), which is different from that for Sait. podzolica 11-11-3.1 in this study.
When grown in LPM medium, Rh. sphaerocarpa 11-14.4 and Sait. podzolica 11-11.3.1 had a lipid content of 27.3 and 24% (w/w, DCW), respectively, after 6 days. But when suspended in glucose solution, the lipid content of Rh. sphaerocarpa 11-14.4 and Sait. podzolica 11-11.3.1 increased to 32.1 and 25.8% (w/w, DCW), respectively, after 48 h and 36 h, respectively. This indicated that the cell growth and lipid accumulation of Rh. sphaerocarpa 11-14.4 and Sait. podzolica 11-11.3.1 could occur separately. This characteristic makes the time to reach the maximum lipid content level shorter. Likewise, the cell propagation and lipid accumulation of Lipomyces starkeyi AS 2.1560 has also been reported to be temporally separated (Lin et al. 2011).
Conclusions
Ascomycetous yeasts (thirty-two species in sixteen genera), Basidomycetous yeasts (twelve species in nine genera), and 13 potential new species were distributed in the 8 mangrove forests across 4 different coastlines of Thailand. Occurrence frequency of oleaginous yeast was 3.82% (two out of one hundred ninety one yeast isolates). Oil of the Rhodotorula sphaerocarpa 11-14.4 had oleic acid and palmitic acid as major fatty acids and so it was suitable to be raw material for biodiesel and biolubricant productions. Oil of the Saitozyma podzolica 11-11.3.1 was similar to cocoa butter used in food industry in that it contained high saturated fatty acids.
References
Ageitos JM, Vallejo JA, Veiga-Crespo P, Villa TG (2011) Oily yeasts as oleaginous cell factories. Appl Microbiol Biotechnol 90:1219–1227. https://doi.org/10.1007/s00253-011-3200-z
Alongi DM, Wattayakorn G, Pfitzner J, Tirendi F, Zagorskis I, Brunskill GL, Davidson A, Clough BF (2001) Organic carbon accumulation and metabolic pathways in sediments of mangrove forest in Southern Thailand. Marine Geo 179:85–103. https://doi.org/10.1016/S0025-3227(01)00195-5
Am-In S, Yongmanitchai W, Limtong S (2008) Kluyveromyces siamensis sp. nov., an ascomycetous yeast isolated from water in a mangrove forest in Ranong Province. Thailand. FEMS Yeast Res 8:823–828. https://doi.org/10.1111/j.1567-1364.2008.00396.x
Am-In S, Limtong S, Yongmanitchai W, Jindamorakot S (2011) Candida andamanensis sp. nov., Candida laemsonensis sp. nov. and Candida ranongensis sp. nov., anamorphic yeast species isolated from estuarine waters in a Thai mangrove forest. J Syst Evol Microbiol 61:454–461. https://doi.org/10.1099/ijs.0.022038-0
Anamnart S, Tolstorukov I, Kaneko Y, Harashima S (1998) Fatty acid desaturation in methylotrophic yeast Hansenula polymorpha strain CBS 1976 and unsaturated fatty acid auxotrophic mutants. J Biosci Bioeng 85:476–482. https://doi.org/10.1016/S0922-338X(98)80065-2
Angerbauer C, Siebenhofer M, Mittelbach M, Guebitz G (2008) Conversion of sewage sludge into lipids by Lipomyces starkeyi for biodiesel production. Bioresour Technol 99:3051–3056. https://doi.org/10.1016/j.biortech.2007.06.045
Araujo FV, Hagler AN (2011) Kluyveromyces aestuarii, a potential environmental quality indicator yeast for mangroves in the State of Rio de Janeiro, Brazil. Braz J Microbiol 42:954–958. https://doi.org/10.1590/S1517-838220110003000014
Araujo FV, Soares CAG, Hagler AN, MendonÇa-Hagler LC (1995) Ascomycetous yeast communities of marine invertebrates in a southeast Brazilian mangrove ecosystem. Antonie Van Leeuwenhoek 68:91–99
Araujo FV, Medeiros RJ, Mendonça-Hagler LC, Hagler AN (1998) A preliminary note on yeast communities of bromeliad-tank water waters of Rio de Janeiro, Brazil. Rev Microbiol 29:118–121. https://doi.org/10.2307/3758110
Arfi Y, Buée M, Marchand C, Devasseur A, Record E (2012) Multiple markers pyrosequencing reveals highly diverse and host specific fungal communities on the mangrove tree Avicenia marina and Rhizophora stylosa. FEMS Microbiol Ecol 79:433–444. https://doi.org/10.1111/j.1574-6941.2011.01236.x
Babjeva IP, Rheshetova IS (1975) A new yeast species Candida podzolica sp. nov. isolated from the soil. Microbiology 44:333–338
Bandhu S, Khot MB, Sharma T, Sharma OP, Dasgupta D, Mohapatra S, Hazra S, Khatri OP, Ghosh D (2018) Single cell oil from oleaginous yeast grown on sugarcane bagasse-derived xylose: An approach toward novel biolubricant for low friction and wear. ACS Sustainable Chem Eng 6:275–283. https://doi.org/10.1021/acssuschemeng.7b02425
Bautista-Gallego J, Rodríguez-Gómez F, Barrio E, Querol A, Garrido-Fernández A, Arroyo-López FN (2011) Exploring the yeast biodiversity of green table olive industrial fermentations for technological applications. Int J Food Microbiol 147:89–96. https://doi.org/10.1016/j.ijfoodmicro.2011.03.013
Beopoulos A, Nicaud JM (2012) Yeast: A new oil producer? OCL 19(1):22–28. https://doi.org/10.1051/ocl.2012.0426
Beopoulos A, Cescut J, Haddouche R, Uribelarrea J-L, Molina-Jouve C, Nicaud J-M (2009) Yarrowia lipolytica as a model for bio-oil production. Prog Lipid Res 48:375–387. https://doi.org/10.1016//j.plipres.2009.08.005
Brandão LR, Vaz ABM, Espírito Santo LC, Pimenta RS, Morais PB, Libkind D, Rosa LH, Rosa CA (2017) Diversity and biogeographical patterns of yeast communities in Antarctic, Patagonian and tropical lakes. Fungal Ecol 28:33–43. https://doi.org/10.1016/j.funeco.2017.04.003
Canto A, Herrera CM, Rodriguez R (2017) Nectar-living yeasts of a tropical host plant community: diversity and effects on community-wide floral nectar traits. PeerJ 5:23517. https://doi.org/10.7717/peerj.3517
Chi ZM, Liu TT, Chi Z, Liu GL, Wang ZP (2012) Occurrence and diversity of yeasts in the mangrove ecosystems in Fujian, Guangdong and Hainan provinces of China. Ind J Microbiol 52:346–353. https://doi.org/10.1007/s12088-012-0251-5
Department of Marine and Coastal Resources (2017) Situation of marine and coastal resources and coastal erosion of Thailand 2017. Department of Marine and Coastal Resources, Ministry of Natural Resources and Environment. Bangkok, Thailand. https://www.dmcr.go.th/detailLib/40 50. Accessed 25 Nov 2018
Fell JW (1961) A new species of Saccharomyces isolated from a subtropical estuary. Antonie Van Leeuweenhoek 27:27–30
Fell JW (2012) Yeast in marine environment. In: Jones EBG, Pang KL (eds) Marine fungi and fungal-like organisms. Walter de Gruyter, Berlin, pp 91–102
Fell JW, Statzell-Tallman A, Scorzett G, Gutierrez MH (2011) Five new species of yeasts from fresh water and marine habitats in the Florida Everglades. Antonie Van Leeuwenhoek 99:533–549. https://doi.org/10.1007/s10482-010-9521-6
Folch J, Lees M, Sloane-Stanley G (1957) A simple method for the isolation and purification of total lipid from animal tissues. J Biol Chem 226:497–509
Galafassi S, Cucchetti D, Pizza F, Franzosi G (2012) Lipid production for second generation biodiesel by the oleaginous yeast Rhodotorula graminis. Bioresour Technol 111:398–403. https://doi.org/10.1016/j.biortech.2012.02.004
Golubev WI (1991) Taxonomic evaluation of mycocins produced by basidiomycetous yeast Cryptococcus podzolicus. Mikrobiologiia 60:115–121
Greenspan P, Mayer EP, Fowler SD (1985) Nile red: a selective fluorescent stain for intracellular lipid droplets. J Cell Biol 100:965–973
Hagler AN, Mendonça-Hagler LC, Pagnocca FC (2017) Chapter 2 Yeasts in aquatic ecotone habitats. In: Buzzini P, Lachance MA, Yurkov A (eds) Yeasts in natural ecosystems: Diversity. Springer International Publishing AG, Cham, pp 63–85
Hall TA (1999) Bioedit: a user-friendly biological sequence alignment editor and analysis program for windows 95/98/nt. Nucl Acids 41:95–98. https://doi.org/10.14601/Phytopathol_Mediterr-14998u1.29
Hassan M, Blanc PJ, Pareilleux A, Goma G (1994) Selection of fatty acid auxotrophs from the oleaginous yeast Cryptococcus curvatus and production of cocoa butter equivalents in batch culture. Biotechnol Lett 16:819–824. https://doi.org/10.1007/BF00133960
Hassan M, Blanc PJ, Pareilleux A, Goma G (1995) Production of cocoa butter equivalents from prickly-pear juice fermentation by an unsaturated fatty acid auxotroph of Cryptococcus curvatus grown in batch culture. Process Biochem 30(7):629–634. https://doi.org/10.1016/0032-9592(94)00061-1
Heip CHR, Herman PMJ, Soetaert K (1998) Indices of diversity and evenness. Oceanis 24:61–87
Hoekman SK, Broch A, Robbins C, Ceniceros E, Natarajan M (2012) Review of biodiesel composition, properties and specifications. Renew Sustain Energy Rev 16:143–169. https://doi.org/10.1016/j.rser.2011.07.143
Jiru TM, Abate D, Kiggunda N, Pohl C, Groenewald M (2016) Oleaginous yeasts from Ethiopia. AMB Express 6:78
Kachalkin AV, Glushakova AM, Yurkov AM, Chernov IY (2008) Characterization of yeast groupings in the Phyllosphere of Sphagnum mosses. Microbiology 77:474–481
Kathiresan K, Bingham BL (2001) Biology of mangrove and mangrove ecosystems. Adv Mar Biol 40:81–251. https://doi.org/10.1016/S0065-2881(01)40003-4
Khunnamwong P, Jindamorakot S, Limtong S (2018) Endophytic yeast diversity in leaf tissue of rice, corn and sugarcane cultivated in Thailand assessed by a culture-dependent approach. Fungal Biology 122:785–799. https://doi.org/10.1016/j.funbio.2018.04.006
Kraisintu P, Yongmanitchai W, Limtong S (2010) Selection and optimization for lipid production of a newly isolated oleaginous yeast, Rhodosporidium toruloides DMKU3-TK16. Kasetsart J (Nat Sci) 44:436–445
Kunthiphun S, Chokreansukchai P, Hoondee P, Tanasupawat S, Savarajara A (2018) Diversity and characterization of cultivable oleaginous yeasts isolated from mangrove forests. World J Microbiol Biotechnol 34:125. https://doi.org/10.1007/s11274-018-2507-7
Kurtzman CP, Robnett CJ (1998) Identification and phylogeny of ascomycetous yeasts from analysis of nuclear large subunit (26S) ribosomal DNA partial sequences. Antonie Van Leeuwenhoek 73:331–371
Kurtzman CP, Fell JW, Boekhout T (2011) The Yeasts: a taxonomic study, 5th edn. Elsevier, Amsterdam
Kutty SN, Philip R (2008) Marine yeast-a review. Yeast 25:465–483. https://doi.org/10.1002/yea.1599
Kvasnikov EI, Nagornaia SS, Shchelokova IF (1975) Yeast flora of plant rhizosphere and phyllosphere. Mikrobiologiia 44:339–346
Lachance MA, Boekhout T, Scorzetti G, Fell JW, Kurtzman CP (2011) Candida Berkhout (1923). In: Kurtzman CP, Fell JW, Boekhout T (eds) The yeasts: a taxonomic study, 5th edn. Elsevier, Amsterdam, pp 987–1062
Li Q, Du W, Liu D (2008) Perspectives of microbial oils for biodiesel production. Appl Microbiol Biotechnol 80:749–756. https://doi.org/10.1007/s00253-008-1625-9
Libkind D, Buzzini P, Turchetti B, Rosa CA (2017) Chapter 1 Yeasts in Continental and Seawater. In: Buzzini P, Lachance MA, Yurkov A (eds) Yeasts in natural ecosystems: Diversity. Springer International Publishing AG, Cham, pp 1–51
Limsuwatthanathamrong M, Sooksai S, Chunhabundit S, Noitung S, Ngamrojanavanich N, Petsom A (2012) Fatty acid profile and lipid composition of farm-raised and wild-caught sandworms, Perinereis nuntia, the diet for marine shrimp broodstock. Asian J Anim Sci 6:65–75. https://doi.org/10.3923/ajas.2012.65.75
Limtong S, Yongmanitchai W (2010) Candida chanthaburiensis sp. nov., Candida kungkrabaensis sp. nov. and Candida suratensis sp. nov., three novel yeast species from decaying plant materials submerged in water of mangrove forest. Antonie Van Leeuwenhoek 98:379–388. https://doi.org/10.1007/s10482-010-9451-3
Limtong S, Srisuk N, Yongmanitchai W, Kawasaki H (2004) Three new thermotolerant methylotrophic yeasts, Candida krabiensis sp. nov., Candida sithepensis sp. nov., and Pichia siamensis sp. nov., isolated in Thailand. J Gen Appl Microbiol 50:119–127. https://doi.org/10.2323/jgam.50.119
Limtong S, Yongmanitchai W, Kawasaki H, Seki T (2007) Candida thaimueangensis sp. nov., an anamorphic yeast species from estuarine water in a mangrove forest in Thailand. Int J Syst Evol Microbiol 57(3):650–653. https://doi.org/10.1099/ijs.0.64698-0
Lin J, Shen H, Tan H, Zhao X (2011) Lipid production by Lipomyces starkeyi cells in glucose solution without auxiliary nutrients. J Biotech 152:184–188. https://doi.org/10.1016/j.jbiotec.2011.02.010
Liu XZ, Wang QM, Göker M, Groenewald M, Kachalkin AV, Lumbsch HT, Millanes AM, Wedin M, Yurkov AM, Boekhout T, Bai FY (2015) Towards an integrated phylogenetic classification of the Tremellomycetes. Stud Mycol 81:85–147. https://doi.org/10.1016/j.simyco.2015.12.001
Maina S, Pateraki C, Kopsahelis N, Paramithiotis S, Drosinos EH, Papanikolaou S, Koutinas A (2017) Microbial oil production from various carbon sources by newly isolated oleaginous yeasts. Eng Life Sci 17:333–344. https://doi.org/10.1002/elsc.201500153
Mangrove Conservation Office (2018) Status of mangrove in Ranong. Department of marine and coastal resources. https://km.dmcr.go.th/th/c_1/s_19141. Accessed 25 Dec 2018
Marsu J, Voutilainen S, Nurmi T, Rissanen TH, Virtanen JK, Kaikkinen J, Nen KN, Salonen JT (2004) Dark chocolate consumption increase HDL cholesterol concentration and chocolate fatty acid may inhibit lipid peroxidation in healthy humans. Free Radical Biol Med 37:1351–1359. https://doi.org/10.1016/j.freeradbiomed.2004.06.002
Mašínová T, Bahnmann BD, Větrovský T, Tomšovský, Merunková K, Baldrian P (2017) Drivers of yeast community composition in the litter and soil of temperate forest. FEMS Microbiol Ecol 93. https://doi.org/10.1093/femsec/fiw223
Meepol W (2010) Carbon sequestration of mangrove forest at ranong biosphere reserve. J For Manag 4:29–44
Meyer W, Mitchell TG, Freedman EZ, Vilgalys R (1993) Hybridization probes for conventional DNA fingerprinting used as single primers in the polymerase chain reaction to distinguish strains of Cryptococcus neoformans. J Clin Microbiol 31:2274–2280
Meyers SP, Ahearn DG, Miles P (1971) Characterization of yeasts in Barataria Bay. La St Univ Coastal Stud Bull 6:7–15
Middelhoven WJ (2006) Polysaccharides and phenolic compounds as substrates for yeasts isolated from rotten wood and description of Cryptococcus fagi sp. nov. Antonie Van Leeuwenhoek 90:57–67. https://doi.org/10.1007/s10482-006-9060-3
Moore MM, Strom MS (2003) Infection and mortality by the yeast Metschnikowia bicuspidate var. bicuspidate in chinook salmon fed live adult brine shrimp (Artemia franciscana). Aquaculture 200:43–57. https://doi.org/10.1016/S0044-8486(02)00271-5
O’Donnell K, Gray LE (1995) Phylogenetic relationships of the soybean sudden death syndrome pathogen Fusarium solanif sp. phaseoli inferred from rDNA sequence data and PCR primers for its identification. Mol Plant Microbe Interact 8:709–716
Papanikolaou S, Chevalot I, Komaitis M, Aggelis G, Marc I (2001) Kinetic profile of the cellular lipid composition in an oleaginous Yarrowia lipolytica capable of producing a cocoa-butter substitute from industrial fats. Antonie Van Leeuwenhoek 80:215–224. https://doi.org/10.1023/A:1013083211405
Papanikolaou S, Muniglia L, Chevalot I, Aggelis G, Marc I (2003) Accumulation of a cocoa-butter-like lipid by Yarrowia lipolytica cultivated on agro-industrial residues. Curr Microbiol 46:124–130. https://doi.org/10.1007/s00284-002-3833-3
Pielou EC (1975) Ecological diversity. Willey & Sons, New York
Polburee P, Yongmanitchai W, Lertwattanasakul N, Ohashi T (2015) Characterization of oleaginous yeasts accumulating high levels of lipid when cultivated in glycerol and their potential for lipid production from biodiesel-derived crude glycerol. Trans Br Mycol Soc 119:1194–1204. https://doi.org/10.1016/j.funbio.2015.09.002
Pumijumnong N (2014) Mangrove Forests in Thailand. In: Faridah-Hanum I, Latiff A, Hakeem KR, Ozturk M (eds) Mangrove Ecosystems of Asia: Status, Challenges and Management Strategies. Springer, New York Heidelberg Dordrecht London, New York, pp 61–79
Ratledge C (1989) Biotechnology of oils and fats. In: Ratledge C, Wilkinson SG (eds) Microbial Lipids. Academic Press, London, pp 567–668
Rossi M, Buzzini P, Cordisco L, Amaretti A (2009) Growth, lipid accumulation, and fatty acid composition in obligate psychrophilic, facultative psychrophilic, and mesophilic yeasts. FEMS Microbiol Lett 69:363–372. https://doi.org/10.1111/j.1574-6941.2009.00727
Sampaio JP (2011) Rhodotorula Harrison (1928). In: Kurtzman CP, Fell JW, Boekhout T (eds) The Yeasts: a taxonomic study, 5th edn. Elsevier, Amsterdam, pp 1912–1915
Schulze I, Hansen S, Grobhans S, Rudszuck T (2014) Characterization of newly isolated oleaginous yeasts Saitozyma podzolicus, Trichosporon porosum and Pichia segobiensis. AMB Express 4:24–34. https://doi.org/10.1186/s13568-014-0024-0
Shannon CE (1948) A mathematical theory of communication. Bell Syst Tech J 27:379–423. https://doi.org/10.1002/j.1538-7305.1948.tb01338.x
Sitepu I, Ignatia L, Franz A, Wong D (2012) An improved high-through put Nile red fluorescence assay for estimating intracellular lipids in a variety of yeast species. J Microbiol Methods 91:321–328. https://doi.org/10.1016/j.mimet.2012.09.001
Sitepu I, Sestric R, Ignatia L, Levin D (2013) Manipulation of culture conditions alters lipid content and fatty acid profiles of a wide variety of known and new oleaginous yeast species. Bioresour Technol 144:360–369. https://doi.org/10.1016/j.biortech.2013.06.047
Sitepu I, Garay LA, Sestric R, Levin D (2014) Oleaginous yeasts for biodiesel: Current and future trends in biology and production. Biotechnol Adv 32:1336–1360. https://doi.org/10.1016/j.biotechadv.2014.08.003
Spellerberg IF, Fedor PJ (2003) A tribute to Claude Shannon (1916–2001) and a plea for more rigorous use of species richness, species diversity and the ‘Shannone–Wiener’ Index. Global Ecol Biogeogr 12:177–179. https://doi.org/10.1046/j.1466-822X.2003.00015.x
Štursová M, Žifčákova L, Leigh MB, Burgess R, Baldrian P (2012) Cellulose utilization in forest litter and soil: identification of bacterial and fungal decomposers. FEMS Microbiol Ecol 80:735–746. https://doi.org/10.1111/j.1574-6941.2012.01343.x
Thanh VN (2006) Lipomyces orientalissp.nov., a yeast species isolated from oil in Vietnam. Int J Syst Evol Microbiol 56:2009–2013. https://doi.org/10.1099/ijs.0.64241-0
Vibulsresth S, Ketruangrots C, Sriplung N (1975) Distribution of mangrove forest as revealed by the Earth Resources Technology Satellite (ERTS-1) Imagery. Technical Report No. 751003 Natl Res Council Appl Sci Res Corp Thailand. 75
Wang H, Maleky F (2018) Effects of cocoa butter triacylglycerides and minor compounds on oil migration. Food Res Int 106:213–224. https://doi.org/10.1016/j.foodres.2017.12.057
Wang X, Chi Z, Yue L, Li J (2007) A marine killer yeast against the pathogenic yeast strain in crab (Portunus trituberculatus) and an optimization of the toxin production. Microbiol Res 162:77–85. https://doi.org/10.1016/j.micres.2006.09.002
Wei Y, Siewers V, Nielsen J (2017) Cocoa butter-like lipid production ability of non-oleaginous and oleaginous yeasts under nitrogen-limited culture conditions. Appl Microbiol Biotechnol 101:3577–3585. https://doi.org/10.1007/s00253-017-8126-7
Wu S, Zhao X, Shen H, Wang Q, Zhao ZK (2011) Microbial lipid production by Rhodosporidium toruloides under sulfate-limited conditions. Bioresour Technol 102:1803–1807
Yurkov AM (2017) Yeast of the soil—obsure but precious. Yeast 35:369–378. https://doi.org/10.1002/yea.3310
Yurkov A, Wehde T, Kahl T, Begerow D (2012a) Aboveground deadwood deposition supports development of soil yeasts. Diversity 4:453–474. https://doi.org/10.3390/d4040453
Yurkov AM, Kemler M, Begerow D (2012a) Assessment of yeast diversity in soils under different management regimes. Fungal Ecol 5:24–35. https://doi.org/10.1016/j.funeco.2011.07.004
Zarringhalami S, Sahari MA, Barzegar M, Hamidi-Esfehani Z (2012) Production of cocoa butter replacer by dry fractionation, partial hydrogenation, chemical and enzymatic inter-esterification of tea seed oil. Food Nutrition Sci 3:184–189. https://doi.org/10.4236/fns.2012.32027P2.32027
Zhao X, Kong X, Hua Y, Feng B, Zhao ZK (2008) Medium optimization for lipid production through co-fermentation of glucose and xylose by the oleaginous yeast Lipomyces starkeyi. Eur J Lipid Sci Technol 110:405–412. https://doi.org/10.1002/ejlt.200700224
Acknowledgements
This research was funded by the National Research University Project, Office of Higher Education Commission (WCU-58-016-EN) and the Thailand Research Fund through the Royal Golden Jubilee Ph.D. Program (Grant No. PHD/0059/2558). The authors would like to thank Dr. Robert D. J. Butcher, Faculty of Science, Chulalongkorn University for his assistance in English proofreading and precious comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations
Rights and permissions
About this article
Cite this article
Hoondee, P., Wattanagonniyom, T., Weeraphan, T. et al. Occurrence of oleaginous yeast from mangrove forest in Thailand. World J Microbiol Biotechnol 35, 108 (2019). https://doi.org/10.1007/s11274-019-2680-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-019-2680-3