Abstract
Macroinvertebrate community structure and assemblages associated with the planted, native submerged aquatic vegetation (SAV) species Heteranthera dubia (Jacq.) MacMillan and Potamogeton nodosus Poiret were examined in a series of constructed urban floodway wetlands, the Dallas Floodway Extension Lower Chain of Wetlands, Dallas, TX, USA. Macroinvertebrate community metrics, including abundance, richness, diversity, and evenness associated with SAV and three different wetlands of varying construction completion dates, water sources (direct or wetland-channeled wastewater effluent), and ecosystem management stage (established/reference or developing) were compared and analyzed. Assemblages at sampling sites were also classified and related to vegetation and wetland physicochemical parameters. Plant species affected only macroinvertebrate abundance, with the less-dissected P. nodosus supporting higher counts than H. dubia. Wetland age and water-effluent type had the most substantial effect on macroinvertebrate communities. The older, longer-managed wetland and wetland-channeled effluent habitat consistently demonstrated higher quality metrics and biodiversity than newly constructed, direct effluent wetland habitat. Increased vegetation cover and wetland age, coupled with moderate water temperature, pH, and DO levels were characteristics of more rich and diverse macroinvertebrate communities, including pollutant-sensitive taxa, such as Ephemeroptera and Trichoptera.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Constructed wetlands are created for a variety of reasons, including environmental mitigation, recreation, floodwater management, wastewater effluent polishing, wildlife management, as well as agricultural and livestock uses (Kentula 1999). While constructed wetlands are commonplace, research regarding their floral and faunal community development and expansion is not. Assessing this structural and functional progress in created wetlands can provide valuable information to aquatic ecosystem managers and conservation biologists in terms of maximizing biodiversity, productivity, community development, and energy flow (Schad et al. 2016). Epiphytic macroinvertebrates are valuable candidates to evaluate this ecological development in new aquatic ecosystems because they are abundant, diverse, and form essential links in the transfer of organic material and energy between trophic levels (Gerking 1962; Krull 1970; Keast 1984; Engel 1988).
Studies have revealed the relationship between macroinvertebrate community structure and the abundance and morphology of submerged aquatic vegetation (SAV) (Downing and Cyr 1985; Brown 1988; Beckett and Aartila 1991; Beckett et al. 1992; Thorp et al. 1997; Collier et al. 1999). For example, SAV stands are known to support higher macroinvertebrate abundance and diversity than contiguous open habitat (Engel 1988; Thorp et al. 1997). In addition, macroinvertebrate colonization rates and densities are frequently higher on dissected-leaved versus non-dissected-leaved SAV, which has been attributed to higher vegetation surface areas, more substrate for attachment of reproductive forms, refuge from predation, and greater density of food resources (Brown 1988; Cheruvelil et al. 2002). Continuing to assess these relationships between SAV and macroinvertebrates in constructed wetlands provides valuable information for aquatic restoration and conservation efforts regarding the quality of faunal habitat provided by intentionally planted and volunteer SAV species. Researchers and restoration ecologists can use this information when planning, designing, and managing constructed wetlands and ascertaining ecologically important parameters needed for maximizing and sustaining their ecosystem services.
The intentions of this study were to investigate macroinvertebrate communities associated with two native SAV species intentionally planted in a newly constructed wetland floodway ecosystem. Heteranthera dubia (Jacq.) MacMillan (water stargrass) and Potamogeton nodosus Poiret (American pondweed) were chosen as plant species of interest to assess aquatic plant and animal relationships because of their different architecture, availability, applicability to aquatic vegetative community establishment efforts, and growth success in the study area. Both are perennial flowering SAV species native to the United States (Muenscher 1944). H. dubia is a grass-like herb with slender branching stems, which often roots at the nodes. Its yellow flowers rise above the water surface from leaf axils, and reproduces via seeds and fragmentation. H. dubia leaf morphology is linear or ribbon-like, thin, sessile, finely parallel-veined and without a distinct mid-vein (Stuzenbaker 1999). P. nodosus has submerged and floating elliptical leaves with an emergent fruiting spike, reproducing by seed, fragmentation, and production tubers. P. nodosus is less dissected than H. dubia, with fewer branches and larger, but fewer leaves and stems, resulting in lower surface area per plant in most cases. Both species are considered high in wildlife value for aquatic fauna, including ducks and wading birds (Stuzenbaker 1999). Previous studies assessing macroinvertebrate associations with H. dubia or P. nodosus include general colonization (Berg 1949), abundances (Beckett and Aartila 1991), taxa richness and Chironomidae spp. annual secondary productivity (Balci and Kennedy 2003a), macroinvertebrate herbivory on vegetation (Nachtrieb et al. 2007; Nachtrieb 2008), macroinvertebrate geographical variations (Harms et al. 2011), and secondary production and seasonal development of epiphytic Enallagma civile Hagen, 1861 (Schad et al. 2016).
Comparisons of macroinvertebrate community structure parameters (abundance, richness, diversity, and evenness) were made between the two SAV species in three wetlands, which were supplied by different wastewater effluent sources, constructed at different times, and thus represented different stages of native plant community development. Macroinvertebrate assemblages were also compared between sampling sites to assess variability based on environmental factors. It was expected that SAV species, wetland type, water source, sampling date, sampling site characteristics, and certain environmental parameters all would affect the macroinvertebrate community structure and assemblages. Specifically, it was predicted the more dissected (Beckett and Aartila 1991; Balci and Kennedy 2003a) SAV species, H. dubia, and the more mature vegetated wetlands would contain richer and more diverse macroinvertebrate communities. Denser population sizes of pollutant-sensitive taxa, such as Ephemeroptera and Trichoptera known to occur in similar systems, were expected at sites within the older constructed and vegetated wetland as well as those farther from a direct wastewater effluent source.
Materials and methods
Study area
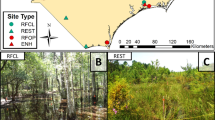
Epiphytic macroinvertebrate sampling was conducted in a series of constructed wetlands within The Dallas Floodway Extension Trinity River Project (DFE), Dallas, TX, constructed by the U.S. Army Corps of Engineers Fort Worth District (SWF) and the City of Dallas, TX (Fig. 1). Its main component, and this study’s location, consisted of a 6.5 km long chain of six wetlands located in south Dallas’ Trinity River Corridor termed the Lower Chain of Wetlands (LCOW). The LCOW's primary function was to provide overflow for floodwaters along the western side of the Trinity River in Dallas, TX. During low or normal Trinity River flow periods, the LCOW was hydraulically fed and water levels were maintained with discharge from the Dallas Central Wastewater Treatment Plant (CWTP) outflow to the Trinity River. In addition to flood conveyance, a major objective of the LCOW was to develop quality wetland habitat for waterfowl and other wildlife through ecosystem management. Beginning in 2005, this management was conducted by the U.S. Army Corps of Engineer's Lewisville Aquatic Ecosystem Research Facility (LAERF) and included native aquatic vegetation establishment (31 SAV, floating-leaved, and emergent species), nuisance or invasive plant management (biological, physical, and chemical control), seasonal water-level drawdowns to promote migratory waterfowl habitat, bird-nesting device installations, water quality monitoring, and biological community monitoring (Dick et al. 2015).
Wetlands, in this study, were distinguished as different types based upon their construction completion date and water source (direct wastewater effluent versus wetland-channeled effluent). One wetland type was Cell D, which received direct effluent from CWTP through a dedicated pipe, was completed in 2005 (5–6 years old at the initiation of this study), and had plant community management applied thereafter. Other wetland types included wetland cells E, E-West, F, F-North, and G, with construction completion occurring in 2009 (1–2 years old at the initiation of this study) and plant community management was applied similarly thereafter in all. Similarly to Cell D, wastewater effluent flowed directly into Cell E through a dedicated pipe, but then cascaded through weir gates to Cell F, and then Cell G (Fig. 1). Cell E-West was filled directly from the CWTP (much like Cell D and Cell E), while F-North was occasionally filled by overflow from Cell F. Wetland cells D (referred herein as the “reference” wetland), E (referred herein as the “developing direct effluent” wetland), and G (referred herein as the “developing channeled effluent” wetland) were chosen as wetlands of interest for this study because of the macroinvertebrate assemblage and community structure comparison possibilities between wetlands with different biological, physical, and chemical properties, such as age, vegetation community structure, fauna, and water quality.
Site design
Individual plants were transplanted in groups (by species), or founder colonies, into each wetland of interest and protected from vertebrate herbivores with exclosures to facilitate survival and establishment. The goal of founder colony establishment is SAV will eventually exceed a threshold of propagule production versus herbivorous and other environmental pressures, allowing for vegetative spread to unprotected areas (Smart et al. 1996, 1998; Smart and Dick 1999, 2005). Coupled with management of nuisance and desirable volunteer species, this approach was the general premise followed for ecosystem management throughout the LCOW.
Eighteen macroinvertebrate sampling locations were installed throughout the three wetland cells and were arranged in a two factor crossed design with factor 1 being SAV species (two levels—P. nodosus and H. dubia) and factor 2 being wetland type (three levels—reference, developing direct effluent, and developing channeled effluent) with three sites within each combination. Each site consisted of four 0.9 m tall, 0.9 m diameter, 3.81 cm × 3.81 cm mesh PVC-coated, welded-wire ring cages as protective exclosures for both species. Exclosures restricted herbivory to the extent that plant growth was adequate to support periodic destructive sampling needed to evaluate macroinvertebrate colonies. The exclosure mesh size was large enough to simulate a natural environment by allowing access to small predatory fauna while preventing entrance to large herbivores such as basking turtles. Each container-grown (10-cm diameter nursery pot) plant and associated exclosure was installed at a 0.45 m water depth. Vegetation was planted in spring and was fully established within exclosures for sampling by autumn (Fig. 1).
Data collection
Sampling of SAV and associated fauna from wetlands began in September 2010. Collections were made every 2 weeks for 1 year except during periods of plant senescence and flooding events (n = 14). Each of the 14 sampling dates from September 2010 to November 2011 yielded 18 samples per trip: SAV (n = 2), wetlands (n = 3), locations within wetlands (n = 3). Plants and associated faunal colonies were collected from individual exclosures by placing a polyester mesh bag (250 µm) sampling device carefully over the plant to minimize macroinvertebrate disruption. Sampling devices, purchased from Pentair Aquatic Eco-Systems (Apopka, FL), were sold as water or pond pre-filters and zooplankton collectors. After placement of the bag, a 20–50 cm plant segment was cut at its base. The bag opening was then “squeezed” shut and inverted while carefully removing the sample from the water, similar to the sampling protocol outlined in Balci and Kennedy (2003a). In addition to animal and plant collections, and to assess environmental relationships, general water quality parameters were recorded (dissolved oxygen (DO) mg/L, pH, temperature °C, and conductivity µs/cm) with a Hydrolab Quanta Multiparameter Sonde (Hach Hydromet Loveland, CO). Sites were further characterized based upon percent SAV coverage inside of exclosures, as well as their proximity to emergent wetland vegetation shoreline. The occurrence of any hydrologic changes (e.g., overbanking, flooding) was monitored (NOAA NWS, USGS, Fort Worth, TX and direct observations during sampling) throughout the study due to their known influence on floral and associated epiphytic macroinvertebrate colonization (Riis and Biggs 2003; Neckles et al. 2006; Owens et al. 2008).
Sample processing
In the laboratory, macroinvertebrates were rinsed from vegetation samples over a 250 µm sieve. Plants were inspected under a Nikon SMZ 1000 dissecting microscope (Nikon, Melville, NY) to remove macroinvertebrates not dislodged by the washing process. Macroinvertebrates were picked from debris retained by the sieve, sorted by taxon, and counted. Macroinvertebrates were identified to genus except those in early developmental stages or specimen damaged during the collection process (McCafferty 1983; Merritt et al 2008; Thorp and Covich 2010). After macroinvertebrates were removed, plants were dried at 105 °C for 24 h to obtain a plant dry weight for macroinvertebrate abundance estimations.
Macroinvertebrate abundance counts were expressed in relation to estimated surface areas of plants to more accurately reflect density and structure. This was accomplished using the relationships between plant surface area and dry biomass. Values reported by Balci and Kennedy (2003b) were used to estimate the surface area of the more finely dissected plant H. dubia, where 1 g of plant dry weight was equivalent to 678 cm2 of plant surface area. Surface area of P. nodosus was estimated by measuring leaves, stems, and petioles using techniques similar to Beckett and Aartila (1991). Leaf areas were calculated by considering them ellipses and then multiplying by two to account for both sides of the leaf. The formula for a cylinder was used to estimate both petiole and stem surface area for this species. All measurements were done with a digital caliper (Traceable®, Friendswood, TX). Leaf, stem, and petiole surface areas were summed to obtain a total surface area. Using the method, on average 1 g of dry plant biomass = 188 cm2 of plant surface area. Regression analysis and methods described by Sher-Kaul et al. (1995), Armstrong et al. (2003), and Feldmann and Noges (2009) were used to relate plant surface area to dry biomass measurements taken from P. nodosus.
Data analysis
Each macroinvertebrate community structure metric (abundance, richness, diversity, and evenness) was analyzed using a partly-nested design/repeated measures ANOVA because of the multiple sampling trips at the same locations (Quinn and Keough 2001; Morris 1996; McGoldrick and Mac Nally 1998). In this analysis, there were two between subjects factors, SAV (n = 2) and wetland type (n = 3), and one within plots/subjects factor, sampling date or time (n = 14). There were three sites for each of the six plant/wetland combinations. The Huynh–Feldt method for estimating the epsilon statistic (έ), used to estimate index of sphericity (ε) necessary for this linear model, was used to adjust the degrees of freedom (df) downwards for the within subjects tests to make them more conservative and decrease the risk of Type I or II statistical errors (Quinn and Keough 2001). Response variables included macroinvertebrate abundance per m2 surface area of vegetation (no./m2), taxa richness, Simpson's index of diversity and evenness (Simpson 1949; Hill 1973). Abundance data were natural-log transformed to meet statistical assumptions of normality and equal variance, although raw data are presented in figures. All statements of significance refer to an alpha level of 0.05. Data were analyzed using statistical software IBM SPSS Statistics 22.0 (IBM Armonk, NY, USA).
Classification and ordination of sampling sites were then completed to assess the relationships between biotic (taxa abundances) and environmental variables (temperature, pH, DO, conductivity, SAV coverage, shoreline distance (m), and wetland age in years). With abundance data, classification was done with cluster analysis using a Bray–Curtis similarity matrix (Jackson 1993; Clarke and Warwick 2001). Abundance data were squared root transformed and normalized (Jackson 1993; Heino 2000). Community groups were then delineated by ordination analysis using non-metric multidimensional scaling (NMDS) (Kruskal 1964; Kenkel and Orloci 1986; McRae et al. 1998; Shin and Fong 1999). Principal component analysis (PCA) was used to reduce environmental information between sampling sites into a few axes of habitat heterogeneity (Santos 1993; Braskerud et al. 2005). Environmental data were Log10 (x + 1) transformed because of the nature of PCA's eigenanalysis and correlation matrix of variables with various measures of magnitude as well as to approximate normal distributions (Heino 2000). Relationships between biotic and environmental data were then evaluated with a Pearson product moment correlation between NMDS dimensions and principal components with large eigenvalues or those responsible for the majority of variance (Clarke and Ainsworth 1993). Correlations were also done between principal components and taxa order groupings and PCA bi-plot overlaid to illustrate assemblage delineation between environmental parameters and sites. 2D NMDS ordination plots of wetlands/SAV were created with bubble plot overlays of pollutant-sensitive Ephemeroptera and Trichoptera abundances to elucidate ecological condition between sampling sites. In the results and discussion sections for sampling site classification and ordination, sites are coded as wetland—reference (Ref), developing direct effluent (DD), developing channeled effluent (DC), SAV—P. nodosus (Pnod), H. dubia (Het). All multivariate analyses were done using Primer 7 (Primer-E, Auckland, New Zealand).
Results
A principal component analysis (PCA) of environmental variables collected at the LCOW resulted in two principal components (PC1 and PC2) being responsible for a total cumulative variance of 81.7% with variables thought to be most important having loadings < -− 0.7 or > 0.7, similar to Braskerud et al. (2005) (Table 1). Principal component 1 (PC1) indicated pH, DO, and conductivity were negatively correlated with shoreline distance and vegetation cover. Temperature and wetland age were variables with the largest loads on PC2 and were also inversely correlated. Table 2 provides basic water quality results throughout the study. Epiphytic macroinvertebrate taxa collected from P. nodosus and H. dubia in three wetlands are given in Table 3. Thirty five unique arthropods, one annelid, and seven mollusk taxa were collected from September 2010 to November 2011 in a total of 252 samples. Included in the table are the different SAV and wetland types from which macroinvertebrates were observed, as well as taxa resolution used.
Mean taxa abundances per m2 surface area of vegetation (no./m2) of primary insect orders are given in Fig. 2. Statistical differences were observed in macroinvertebrate abundance between SAV species, with P. nodosus consistently supporting higher abundance than H. dubia, but no significant differences occurring among wetland types (Table 4, Fig. 3). Macroinvertebrate abundances varied by date throughout the sampling year, with lowest observed in the summer months, likely due to warm water temperatures. Significant differences in taxa richness, diversity, and evenness between wetland types and sampling dates were observed, but no significant differences occurred between SAV species. The reference and developing channeled effluent wetlands consistently represented higher richness, diversity, and evenness throughout the study (Fig. 3).
Mean insect taxa abundance (no./m2) (top), Diptera (bottom) between SAV and wetlands at The Dallas Floodway Extension Lower Chain of Wetlands (mean ± SD); (Pnod), Potamogeton nodosus; (Het), Heteranthera dubia; (Ref), reference wetland; (DD), developing direct effluent wetland; (DC), developing channeled effluent wetland
Macroinvertebrate abundance (no./m2) between SAV and sampling dates (top left), taxa richness between sampling dates and wetland types (top right); Simpson's index of diversity between sampling dates and wetland types (bottom left); evenness between sampling dates and wetland types (bottom left) at The Dallas Floodway Extension Lower Chain of Wetlands (mean + SD)
Cluster analysis and NMDS based on a Bray–Curtis similarity matrix of macroinvertebrate assemblages identified five (> 80%) similar location groups (A–E) of the eighteen sampling sites (Fig. 4). Total NMDS stress = 0.12, which was sufficiently low to give a quality representation of the high-dimensional data (Clarke and Gorley 2006). Breakdown of the total NMDS 2D stress into contributions from each of the 18 sample sites given on NMDS figure in parentheses. These groupings indicate differences between the developing direct effluent wetland (groups A and E) and the reference and developing channeled effluent wetlands (groups B, C, and D), specifically along the gradient of NMDS dimension 2 (Y-axis, Fig. 4). Distinguishing taxa associated with the reference and developing channeled effluent wetlands, included Collembola: Sminthuridae Sminthurides sp., Coleoptera: Haliplidae Peltodytes sp., Diptera: Stratiomyidae Stratiomys sp., Ephemeroptera: Baetidae Baetis sp. and Callibaetis sp., Hemiptera: Naucoridae Pelocoris sp., Odonata: Aeshnidae Anax sp., and Trichoptera: Hydropsychidae Hydropsyche sp. Additionally, similar assemblages were observed on each SAV species, as apparent from NMDS groups A, B, D, and E containing only one of the two plant species (Fig. 4).
Cluster analysis dendrogram (top) and plot of first two dimensions from non-metric multidimensional scaling (NMDS) (bottom) showing the delineation of five sampling site location groups (a–e) from classification results based on macroinvertebrate assemblage (Bray–Curtis) similarity. (Pnod), Potamogeton nodosus; (Het), Heteranthera dubia; (Ref), reference wetland; (DD), developing direct effluent wetland; (DC), developing channeled effluent wetland. Breakdown of the total NMDS 2D stress of 0.12 into contributions from each of the 18 sample sites given on NMDS bottom figure in parentheses
A Pearson product moment correlation between the first two NMDS dimensions and principal components 1 and 2 yielded a significant inverse relationship between NMDS dimension 2 and PC1 (r = − 0.536, p = 0.022) and PC2 (r = − 0.695, p = 0.001). NMDS dimension two separated the reference and developing channeled effluent wetland from the developing direct effluent wetland, PC1 is associated with pH, DO, conductivity, shoreline distance, and vegetation cover and PC2 temperature and wetland age. This, coupled with the community structure analysis, indicated the wetlands with more rich and diverse macroinvertebrate assemblages (reference and developing channeled effluent) were associated with increased age or maturity, vegetation cover, and moderate pH and DO levels (e.g., the biologically less diverse developing direct wetland had pH ranges > 10 and DO > 18 mg/l). In addition, the strong correlation between NMDS dimension 2 and PC2 demonstrated temperature and wetland age also influenced macroinvertebrate assemblages throughout the constructed wetland ecosystem. Results from the Pearson product moment correlations between PCs and individual taxa order groupings to illustrate assemblage delineation between environmental parameters and sites are given in Table 5 and overlaid on a PCA bi-plot (Fig. 5). Ephemeroptera had the strongest inverse correlation observed (PC2), illustrating its sensitivity to high water temperature, pH, and DO levels as well as its affinity for more mature and vegetated wetlands. Pollutant-sensitive taxa, such as Ephemeroptera and Trichoptera, were associated with increased wetland age, vegetation cover, and moderate temperature, pH and DO, while Dipterans dominated newer wetlands and those with higher temperature, DO, and pH (Figs. 5 and 6).
Bi-plot of the results from the first two principal components in the principal component analysis (PCA), which were responsible for 81.70% of total variance among environmental variable correlations. Taxa abundances, environmental parameters, and wetland type sampling site relationships shown. (Ref), reference wetland; (DD), developing direct effluent wetland; (DC), developing channeled effluent wetland
Non-metric multidimensional scaling ordination of sampling sites with bubble plot overlay showing mean Ephemeroptera (top) and Trichoptera (bottom) abundances (per m2 plant surface area) at each sampling location. (Pnod), Potamogeton nodosus; (Het), Heteranthera dubia; (Ref), reference wetland; (DD), developing direct effluent wetland; (DC), developing channeled effluent wetland
Discussion
Plant morphology generally contributes to epiphytic macroinvertebrate community structure dynamics (Downing and Cyr 1985; Beckett et al. 1992; Collier et al. 1999). In this study, only macroinvertebrate abundance (no./m2) was found to be significantly different between the two SAV species analyzed. Typically this difference has been attributed to plant surface area, with the more highly dissected leaves providing more surface area for macroinvertebrates to colonize resulting in increased population sizes (Brown 1988; Cheruvelil et al. 2002). In this study, however, P. nodosus (the less dissected plant) demonstrated higher abundance than H. dubia. These differences could be attributed to the greater and more rapid establishment of P. nodosus, resulting in denser and larger plant stands, which were colonized earlier than slower-growing, smaller stands of H. dubia. It should be noted that taxa richness, evenness, and diversity did not differ between the two plant species.
The most substantial source of variability in macroinvertebrate community structure was observed between wetland types and sampling dates. These differences were expected because of known seasonal influences on macroinvertebrate life cycles as well as vegetation and water quality differences between wetlands of varying temporal ecosystem management strategies (Balci and Kennedy 2003a). The reference (older, managed for 5–6 years, direct effluent) and developing channeled (newer, 1–2 years managed, channeled effluent) wetlands supported higher taxa richness, diversity, pollutant-sensitive taxa abundances such as Ephemeroptera and Trichoptera, and lower total Dipteran densities than the developing direct (newer, 1–2 years managed, direct effluent) wetland at most sampling dates. This suggests a more complex, developed trophic system in the older and channeled wetlands. Macroinvertebrate richness and diversity also increased depending on site characteristics such as increased age, vegetation coverage, proximity to the emergent vegetation shoreline, and moderate temperature, pH, and DO levels (Fig. 5, Table 2). These results support the concept that the most productive freshwater aquatic ecosystems have a diverse and robust SAV and emergent vegetation community (Carpenter and Lodge 1986; Engel 1988; James and Barko 1990, 1995; Newman 1991; Honnell et al. 1993; Rybicki and Landwehr 2007; Smart et al. 2009).
High faunal abundances, such as those of dipterans observed on P. nodosus in the developing direct effluent wetland, may not necessarily reflect high faunal biomass or general ecological condition. Functional studies, such as assessing individual species annual secondary production (biomass per area over time or mg/m2/year), standing stock biomass, and cohort and annual production/biomass (P/B) ratios, help quantify trophic dynamics and thus identify habitat quality (Benke 1976, 1979). As part of this study’s sampling effort, secondary production and seasonal development of the regionally ubiquitous, predatory damselfly Enallagma civile Hagen, 1861 (Odonata: Zygoptera) was determined and reported by Schad et al. (2016). The population at the LCOW represented the first documented record of multivoltine life cycles for E. civile, which may have been due to relatively high water temperatures maintained in this constructed ecosystem during much of the year. Secondary production values were in the upper range of known Odonata production values from similar systems and biomass growth rates were observed to be higher from populations on the better established macrophyte (P. nodosus) and in the longest established wetland (reference). The SAV stands contributed refugia and complex habitat structure to the aquatic ecosystem, which lead to high densities of macroinvertebrates and other fauna used as food resources by E. civile as well as its increased survivorship. Analogous to the community structure metric variability between wetlands and macrophytes documented in this paper, annual P/B, production to biomass (mg) ratio decreased from P. nodosus (11.11) > H. dubia (9.16) and from the reference (11.81) > developing channeled effluent (10.82) > developing direct effluent (8.84) wetland (Schad et al. 2016).
Water nutrient chemistry is known to influence both aquatic vegetation establishment and coverage (Smart et al. 1996) as well be an important driver of macroinvertebrates communities (Spieles and Mitsch 2000). In similar systems to the LCOW, it has been shown that macroinvertebrate community structure generally has significantly lower diversity closest to a wastewater source as compared to those closer to a riverine freshwater source (Spieles and Mitsch 2000). During our study, funding restraints prevented water quality analysis beyond basic parameters (water temperature, DO, pH, conductivity). However, this data still provided valuable information on the habitat suitability for aquatic flora and fauna during sampling (Table 2). All basic metrics were within limits for higher trophic levels such as centrarchids (Smart et al. 2009; Dick et al. 2015) and conducive to SAV community development (Smart et al. 1996). Two years after the macroinvertebrate sampling in this study, additional water nutrient chemistry (chlorophyll a, alkalinity, nitrogen, and total phosphorus) sampling occurred throughout the LCOW (Dick et al. 2015). Because nutrient data and macroinvertebrate data were not taken simultaneously, direct analysis here was not ideal, but it is worth discussing some general trends. In Dick et al. (2015), total phosphorous and chlorophyll a were highest near inflows of the reference and developing direct effluent wetlands, but reduced through the system as water reached the developing channeled wetland. Additionally, alkalinity increased through the system and was at its highest magnitude in the developing channeled wetland, indicating better conditions for vegetation growth and potential resultant macroinvertebrate colonization (Dick et al. 2015). These 2013 water nutrient chemistry dynamics were possibly similar to what occurred during the 2011 macroinvertebrate sampling and support the conclusion that macroinvertebrate communities responded to improving water quality as effluent passed through the system, corresponding to Spieles and Mitsch (2000).
In addition to wastewater effluent, the LCOW received irregular flood water pulses from the Trinity River. This occurred after heavy rains when the river reached approximately 9.14 m at the USGS monitoring station near downtown Dallas, TX. Two separate flooding events of the Trinity River occurred during macroinvertebrate sampling. The first occurred in September 2010 (2 weeks before sampling), crested at 12.62 m and lasted 3.5 days. The second occurred in May 2011 and crested at 9.75 m for the duration of 1 day (NOAA NWS, USGS, Fort Worth, TX). Macroinvertebrate community sampling had not commenced before the first overbanking event and the low intensity of the second prevented evaluation of community structure differences due to flood events. Seasonal flooding events are known to influence epiphytic macroinvertebrate community structure, however, populations generally recover in wetland habitats (Neckles et al. 2006). In this study, assemblages were not affected by the Trinity River’s overbanking events, likely due to the natural processes and short durations of the flooding. Dick et al. (2015) reported similar findings with fish assemblages and qualitative macroinvertebrate assessments before and after Trinity River overbanking events.
The use of constructed wetlands for polishing wastewater effluents, floodwater management, and environmental mitigation is expected to increase with human population growth and urban development (Kentula 1999). In order to maximize their values for recreation, wildlife management, and conservation, a better understanding of ecosystem functioning in these systems is needed. Current aquatic epiphytic macroinvertebrate research has clearly established the association of macroinvertebrate community structure (patterns) and morphology of SAV (Downing and Cyr 1985; Beckett et al. 1992; Collier et al. 1999). There are an increasing number of functional (process) studies such as species-specific annual secondary production estimations, but only a few have assessed functional parameters in correlation with SAV and surface area (Balci and Kennedy 2003a; Schad et al. 2016). A better understanding of the functional aspects of macroinvertebrate/SAV interactions in wetlands will lead to better designed and managed wetland ecosystems that maximize their value for trophic linkages thereby increasing their value for conservation and recreational purposes.
Although several environmental variables played a role in macroinvertebrate community structure and assemblages in our study, wetland age (amount of ecosystem management/vegetation establishment time) and proximity to a direct wastewater effluent source were most responsible for this variability. Assemblages within the developing direct effluent wetland (youngest and closest to effluent source) were classified as being statistically dissimilar to other sites. These results suggest that adequate time is needed to develop native vegetation communities necessary to create an ecologically productive constructed wetland system with a wastewater effluent source. A mechanism mimicking a wetland-channeled water source may also serve to enhance effluent water quality, vegetation establishment, and hasten the development of the ecosystem. The geometric design of wetland cells in constructed systems is also important. Wetland cell designs that provide gradually sloped planting shelves (0–1 m depths) and maximize the shoreline and riparian zones, promote the establishment of a native aquatic plant community that is diverse in both species and growth-forms. When planning, designing, constructing, and managing wetland floodways, these dynamics should be top priority in predicting trajectory of how quickly these systems will become ecologically functional or consist of sufficient habitat heterogeneity for the benefit of fish and other wildlife.
References
Armstrong N, Planas D, Prepas E (2003) Potential for estimating macrophyte surface area from biomass. Aquat Bot 75:173–179
Balci P, Kennedy JH (2003a) Comparison of chironomids and other macroinvertebrates associated with Myriophyllum spicatum and Heteranthera dubia. J Freshw Ecol 18:187–197
Balci P, Kennedy JH (2003b) Measures of plant surface-areas for Eurasian watermilfoil and water stargrass. J Aquat Plant Manage 41:119–122
Beckett DC, Aartila TP (1991) Invertebrate abundance on Potamogeton nodosus: effects of plant surface area and condition. Can J Zool 70:300–306
Beckett DC, Aartila TP, Miller A (1992) Contrasts in density of benthic invertebrates between macrophyte beds and open littoral patches in Eau Galle Lake, Wisconsin. Am Midl Nat 127:77–90
Benke AC (1976) Dragonfly production and prey turnover. Ecology 57:915–927
Benke AC (1979) A modification of the Hynes method to estimating secondary production with particular significance for multivoltine populations. Limnol Oceanogr 24:168–171
Berg CO (1949) Limnological relations of insects to plants of the genus Potamogeton. T Am Microsc Soc 68–4:279–291
Braskerud BC, Tonderski KS, Wedding B, Bakke R, Blankerberg B, Ulen B, Koskiaho J (2005) Can constructed wetlands reduce the diffuse phosphorus loads to eutrophic water in cold temperate regions? J Environ Qual 34:2145–2155
Brown C (1988) Relationships of phytomacrofauna to surface area in naturally occurring macrophyte stands. J N Am Benthol Soc 7:129–139
Carpenter SR, Lodge DM (1986) Effects of submersed macrophytes on ecosystem processes. Aquat Bot 26:341–370
Cheruvelil K, Soranno P, Madsen J, Roberson M (2002) Plant architecture and epiphytic macroinvertebrate communities: the role of an exotic dissected macrophyte. J N Am Benthol Soc 21:261–277
Clarke KR, Ainsworth M (1993) A method of linking multivariate community structure to environmental variables. Mar Ecol Prog Ser 92:205–218
Clarke KR, Warwick RM (2001) Change in marine communities: an approach to statistical analysis and interpretation, 2nd edn. Primer-E, Plymouth
Clarke KR, Gorley RN (2006) Primer v6: user manual/tutorial. Primer-E, Plymouth
Collier K, Champion P, Croker G (1999) Patch- and reach-scale dynamics of a macrophyte-invertebrate system in a New Zealand Lowland Stream. Hydrobiologia 392:88–97
Dick GO, Dodd LL, Schad AN, Smith DH, Owens CS (2015) Dallas floodway extension lower chain of wetlands and grasslands ecological management and monitoring. Status report to the USACE SWF. https://www.swf.usace.army.mil/Portals/47/docs/PAO/DFE/PDF/DFE_March_2015_Status_Report.pdf. Accessed Nov 2019
Downing JA, Cyr H (1985) Quantitative estimation of epiphytic invertebrate populations. Can J Fish Aquat Sci 42:1570–1579
Engel S (1988) The role and interactions of submersed macrophytes in a shallow Wisconsin Lake. J Freshwater Ecol 4:329–341
Feldmann T, Noges P (2009) Seasonal and vertical changes in the surface area/biomass ratio of Potamogeton lucens L. in a clear and turbid shallow lake. J Aquat Plant Manage 47:116–121
Gerking SH (1962) Production and food utilization in a population of bluegill sunfish. Ecol Monogr 32:31–78
Harms NE, Grodowitz M, Kennedy JH (2011) Insect Herbivores of Water Stargrass (Heteranthera dubia) in the U.S. J Freshw Ecol 16:185–194
Heino J (2000) Lentic macroinvertebrate assemblage structure along gradients in spatial heterogeneity, habitat size and water chemistry. Hydrobiologia 418:229–242
Hill MO (1973) Diversity and evenness: a unifying notation and its consequences. Ecology 54:427–432
Honnell DR, Madsen JD, Smart RM (1993) Effects of selected exotic and native aquatic plant communities on water temperature and dissolved oxygen. Info Exchange Bull A-93-2. U.S Army Engineer WES, Vicksburg
Jackson D (1993) Multivariate analysis of benthic invertebrate communities: the implication of choosing particular data standardizations, measures of association, and ordination methods. Hydrobiologia 268:9–26
James WF, Barko JW (1990) Macrophyte influences on the zonation of sediment accretion and composition in a north-temperate reservoir. Arch Hydrobiol 120:129–142
James WF, Barko JW (1995) Effects of submersed macrophytes on sediment resuspension in March Lake, Minnesota. In: Proceedings, 29th annual meeting, APCRP, 168–175. Miscellaneous Paper A-95-3, U.S. Army Engineer WES, Vicksburg, MS.
Keast A (1984) The introduced aquatic macrophyte, Myriophyllum spicatum, as habitatfor fish and their invertebrate prey. Can J Zool 62:1289–1303
Kenkel NC, Orloci L (1986) Applying metric and nonmetric multidimensional scaling to ecological studies: some new results. Ecology 67:919–928
Kentula ME (1999) Wetland restoration and creation National water summary on wetland resources. USGS Water-Supply Paper 2425:87–92
Krull JN (1970) Aquatic plant-macroinvertebrate associations and waterfowl. J Wildlife Manage 34:707–718
Kruskal JB (1964) Multidimensional scaling by optimizing goodness of fit to a nonmetric hypothesis. Psychometrika 29:1–27
Merritt RW, Cummins KW, Berg MB (2008) Aquatic insects of North America, 4th edn. Kendall Hunt Publishing, Dubuque
McCafferty WP (1983) Aquatic entomology: the fishermen's and ecologists' illustrated guide to insects and their relatives. Jones & Bartlett Learning, Burlington
McGoldrick JM, Nally M (1998) Impact of flowering on bird community dynamics in some central Victorian eucalypt forests. Ecol Res 13:125–139
McRae G, Camp DK, Lyons WG, Dix TL (1998) Relating benthic infaunal community structure to environmental variables in estuaries using nonmetric multidimensional scaling and similarity analysis. Environ Monit Assess 51:233–246
Morris DW (1996) Coexistence of specialist and generalist rodents via habitat selection. Ecology 77:2352–2352
Muenscher WC (1944) Aquatic plants of the United States. Comstock Publishing Company Inc., Cornell Univeristy, Binghamton
Nachtrieb JG (2008) The impact of invertebrates to four aquatic macrophytes: Potamogeton nodosus, P. illinoensis, Vallisneria americana, and Nymphaea Mexicana. Thesis, University of North Texas
Nachtrieb JG, Grodowitz MJ, Smart RM (2007) Impact of invertebrate herbivory on native aquatic macrophytes. Technical Report ERDC/TN APCRP-BC-9, U.S. Army Engineer WES, Vicksburg, MS
Neckles HA, Murkin HR, Cooper JA (2006) Influences of seasonal flooding on macroinvertebrate abundance in wetland habitats. Freshw Biol 23:311–322
Newman RM (1991) Herbivory and detritivory on freshwater macrophytes by invertebrates: a review. J North Am Benthol Soc 10:89–114
Owens CS, Smart RM, Dick GO (2008) Effects of water level fluctuation on Vallisneria americana Michx growth. J Aquat Plant Manage 46:117–119
Quinn GP, Keough MJ (2001) Experimental design and data analysis for biologists. Cambridge University Press, Cambridge
Riis T, Biggs BJF (2003) Hydrologic and hydraulic control of macrophyte establishment and performance in streams. Limnol Oceanogr 48:1488–1497
Rybicki NB, Landwehr JM (2007) Long-term changes in abundance and diversity of macrophyte and waterfowl populations in an estuary with exotic macrophytes and improving water quality. Limnol Oceanogr 52:1195–1207
Santos R (1993) A multivariate study of biotic and abiotic relationships in a subtidal algal stand. Mar Ecol Prog Ser 94:181–190
Schad AN, Kennedy JH, Dick GO (2016) Secondary production and seasonal development of epiphytic Enallagma civile Hagen, 1861 (Odonata: Coenagrionidae) in a newly constructed urban wetland floodway ecosystem. Aquat Insect 73:159–173
Sher-Kaul S, Oertlie B, Castella E, Lachavanne JB (1995) Relationship between biomass and surface area of six submerged aquatic plant species. Aquat Bot 51:147–154
Shin PKS, Fong KYS (1999) Multiple discriminant analysis of marine sediment data. Mar Pollut Bull 39:285–294
Simpson EH (1949) Measurement of diversity. Nature 163:688
Smart RM, Dick GO (1999) Propagation and establishment of aquatic plants: a handbook for ecosystem restoration projects. Technical Report A-99-4, U.S. Army Engineer WES, Vicksburg, MS
Smart RM, Dick GO (2005) Update to the propagation and establishment of aquatic plants handbook. APCRP Miscellaneous Paper TR-05-4, U.S. Army ERDC, Vicksburg, MS
Smart RM, Doyle RD, Madsen JD, Dick GO (1996) Establishing native submersed aquatic plant communities in southern reservoirs. Technical Report A-96-2, U.S. Army Engineer WES, Vicksburg, MS
Smart RM, Dick GO, Doyle RD (1998) Techniques for establishing native aquatic plants. J Aquat Plant Manage 36:44–49
Smart RM, Dick GO, Snow JR, Honnell DR, Smith DH, Smith JK (2009) Ecological effects of exotic and native aquatic vegetation, ERDC/EL TR-09-10. U.S. Army ERDC, Vicksburg
Spieles DJ, Mitsch WJ (2000) Macroinvertebrate community structure in high- and low-nutrient constructed wetlands. Wetlands 20:716–729
Stuzenbaker CD (1999) Aquatic and wetland plants of the western gulf coast. TPWD Press, Austin
Thorp AG, Jones RC, Kelso DP (1997) A comparison of water-column macroinvertebrate communities in beds of differing submersed aquatic vegetation in the tidal freshwater Potomac River. Estuaries 20:86–95
Thorp JH, Covich AP (2010) Ecology and classification of North American freshwater invertebrates. Academic Press, San Diego
Acknowledgements
This project was funded by the US Army Corps of Engineers Fort Worth District for biological monitoring of the Dallas Floodway Extension Lower Chain of Wetlands. We are indebted to Julie Nachtrieb, Nathan Harms, Dian Smith, Meredith Evans, Todd Sliger, David Holbrook, and Emily Yeoman for their help in the field and lab.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Schad, A.N., Kennedy, J.H., Dick, G.O. et al. Aquatic macroinvertebrate richness and diversity associated with native submerged aquatic vegetation plantings increases in longer-managed and wetland-channeled effluent constructed urban wetlands. Wetlands Ecol Manage 28, 461–477 (2020). https://doi.org/10.1007/s11273-020-09724-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11273-020-09724-1