Abstract
Conversion of native forest to pastures is a common practice worldwide; it has complex effects on river biota that are related to activity type and intensity. This work was conducted in order to evaluate the effects of cattle grazing on environmental features and to select the most appropriate measures based on the macroinvertebrate community as indicators of ecological changes. Physicochemical features, riparian ecosystem quality, habitat condition, and benthic macroinvertebrates were investigated in streams draining pastures and were compared to reference streams located in nonimpacted native forested catchments. Strong evidence of sediment deposition was observed at pasture streams, which had higher percentage of sand in the streambed but also increased levels of suspended solids. Pasture sites had significantly higher water temperature and conductivity as well as lower dissolved oxygen concentrations than did forested sites. Both riparian quality and in-stream habitat condition were degraded at pasture sites. Among metrics, total richness, Ephemeroptera, Plecoptera, and Trichoptera (EPT) richness, Plecoptera richness, Shannon diversity, percentage of dominant taxon, percentage of Chironomidae species, shredder richness, and percentage of predator resulted to be the most consistent measures by displaying stronger responses to impairment. Our results suggest that forest conversion to pasture for livestock practices is diminishing macroinvertebrate biodiversity and potentially changing functioning and dynamics of Patagonian mountain streams as well. Damage prevention on riverine landscapes by restoring riparian ecosystems, replanting missing vegetation in buffer areas, and limiting livestock access to the riverbanks could contribute to enhance the ecological integrity at converted areas. A more extended and comprehensive use of macroinvertebrate metrics could contribute to better understand and evaluate impact effects of these extended land use activities and to promote conservation strategies.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Aquatic environments are influenced by land use activities in many ways. The conversion of native forests into pastures for cattle grazing is a generalized practice worldwide, with negative impacts on riparian and in-stream conditions. Changes in water temperature, primary productivity, sediment inputs, and nutrient enrichment as well as alterations in stream hydrology and habitat structure are examples of the effects that pasture conversion may exert on stream environments (Allan 2004; Riley et al. 2003).
In riverine landscapes, riparian vegetation is one of the most sensitive components to grazing activities. Removal or clearing of riparian forest alters both shading and the balance between allochthonous matter input and primary productivity (Davies and Nelson 1994; Rutherford et al. 1997, 2004; Arnaiz et al. 2010). While shade reduces water temperature and buffers its daily range, the balance of organic matter fractions is vital to detritivorous species.
Additionally, natural riparian cover also reduces the progressive erosion of the catchment and, at a local scale, of the stream bank during floods (Zimmerman et al. 2003; Lester and Boulton 2008; Magner et al. 2008). Erosion is extremely relevant for the biota; it is linked to the entrainment and deposition of inert fine particles (Wood and Armitage 1997; Larsen et al. 2011), increases turbidity, reducing primary production and available light for visual predators (Davies-Colley and Smith 2001), and changes the substratum structure and benthic habitat by filling interstices (Larsen et al. 2011). This phenomenon has been strongly associated with the decrease in richness and density of several sensitive taxa (Buendia et al. 2013)
Transformation of forest into pasture for livestock extensive grazing has been a generalized practice in Patagonian mountains since the 1960s (Carabelli and Scoz 2008). While nonpoint sources of pollution and physical alteration of stream ecosystems appears to be a major threat to aquatic biota in the region, the relationship between nonintensive pastoral activities and in-stream integrity is still difficult to assess and measure. Traditional chemical evaluations of water quality have been largely inadequate because pollution from nonpoint sources may be transient and often unpredictable, and interpreting the impact on biota may be confounded by the co-occurrence of physical habitat disturbance (Barbour et al. 1996).
Biological monitoring is one of the most appropriate means to detecting effects of nonpoint source pollution and catchment alterations on the river biota (Bonada et al. 2006). In this sense, benthic macroinvertebrates are natural monitors of environmental quality and can potentially reveal the effects of episodic as well as cumulative pollution and habitat alteration (Plafkin et al. 1989; Barbour et al. 1996). Regarding Patagonian environments, these metrics were successfully implemented in the recent years, for both agricultural (Miserendino and Masi 2010; Miserendino et al. 2011) and urban environments (Miserendino et al. 2008).
A multimetric approach allows evaluating the effect of various stressors on different features of the benthic communities; each metric is a characteristic of the biota that changes in a predictable manner as environmental stress increases in intensity (Barbour et al. 1996); most metrics are based on relative abundances or number of taxa of a specific taxonomic or functional group. In this sense, richness and diversity metrics increase when habitat and food sources are adequate to support development of many species. Similarly, the number of sensitive taxa (Ephemeroptera, Plecoptera and Trichoptera aggregated, known as EPT, or each one independently) is related to the good ecological status; therefore, a loss of taxa in this group suggests quality impoverishment, as well as loss of key features of the environment (i.e., riparian coverage, flow diversity, shade, and litter input) (Plafkin et al. 1989). The Shannon diversity index combines both richness and evenness; it is an expression of the community composition. Although it is expected to decrease when facing perturbation, it does not always show a monotonic response and can increase in cases of small disturbances. The same response is sometimes observed with functional metrics and with general density (Barbour et al. 1996).
Tolerance metrics, such as the relative composition of Oligochaeta and some Diptera families, and the percentage of the dominant taxon, shows a positive response facing different levels of environmental perturbation.
Specific changes in functional feeding group evidence deviations in stream ecological processes such as production and food source availability. In this sense, shredders will become affected if source of litter input is affected by removal of riparian vegetation or if palatability decreases, while other specialized groups such as scrapers will suffer from differences in solar energy flux and sedimentation processes (Riley et al. 2003). An increase in contribution or richness of other less specialized groups such as collector feeders is often related to an impoverishment of the stream health or an increase of the resources.
In this paper, we (1) assess the effects of cattle grazing on environmental features of patagonian streams by contrasting pasture-converted versus reference-forested sites and (2) evaluate the sensitivity of different metrics based on macroinvertebrate community attributes on a seasonal basis, in order to select the most appropriate measures to analyze the impact of pasture activities on freshwater courses.
2 Materials and Methods
2.1 Study Area and Site Selection
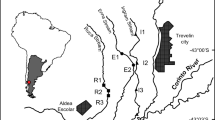
The study region is a transitional area located in the mountains and piedmont in northwestern Chubut province, Argentina (Fig. 1). It is located in the ecotone between two phytogeographical provinces, the Subantarctic forest and the Patagonian steppe. The Subantarctic forest is constituted by perennial (Austrocedrus chilensis, Nothofagus dombeyi, and Maytenus boaria) and deciduous tree species (N. pumilio and N. antarctica), whereas the shrub and herbaceous strata are composed mainly of Chusquea culeou, Berberis buxifolia, Fuchsia magellanica, Aristotelia chilensis, Oenothera odorata, Fragaria chiloensis, and Geranium sp. The valleys are dominated by herbaceous shrub-like steppe vegetation, where the dominant species are Mulinum spinosum, Stipa speciosa, S. humilis, Corynabutilon bicolor, Verbena tridens, Nassauvia glomerulosa, and Berberis heterophylla (Tell et al. 1997). The studied basins are similar in size, gradient, and geology and belong to the Andean-humid and sub-Andean sub-humid regions (Del Valle et al. 1998; Paruelo et al. 1998)
Sampling sites were located in catchments subjected to grazing pressure (Pas: pasture) and in catchments with nonmanaged native forests (NF: native forest) as reference. Within each category, we selected three sampling sites (codes for pasture sites are as follows: MG, manguera; LÑ, los Ñires, NYF: Nant y Fall; and codes for reference sites: CHI, chiquito; ALO, loro; COM, comisario).
All sites selected to assess pasture impact had been cleared from the native Nothofagus spp. forest (∼60–70 years ago) by logging and fires and had been intensively grazed, in which the livestock was sustained by the herbaceous stratum. Selected sites present a mix of cattle types mostly consisting of sheep and cows, and occasionally horses. Throughout the year, the ranching practices involve cattle rotation between lowlands (autumn, winter, and spring) and upland highlands (summer grazing only).
We selected streams by taking into account the similitude among streams within a geographical range and by considering the accessibility to the study reach. Additionally, we reviewed existing data sources in different governmental administration offices in order to select appropriate sites for each land use type (Dirección Provincial de Bosques y Parques (DPByP), Instituto Nacional de Tecnología Agropecuaria (INTA)). The study was conducted in May (autumn), September (winter), and December (spring) 2005, as well as in February (summer) 2006, under normal hydrological conditions (avoiding rainstorms or extremely high discharge events).
2.2 Site Characterization
At each site, percentages of boulder, cobble, gravel, pebble, and sand in the reach were estimated using a 1-m2 grid (Gordon et al. 2004). Average depth was assessed from five measurements with a calibrated stick along one transverse profile across the channel. Wet and dry widths (from bank to bank) of the channel were also determined. Current speed was measured in mid channel (average of three trials) by timing a float as it moved over a distance of 10 m (Gordon et al. 2004). Discharge data was calculated by combining depth, wet width, and current velocity as in Gordon et al. (2004). Air and water temperature were obtained with a mercury thermometer.
At each occasion, specific conductance, pH, turbidity, and dissolved oxygen were measured in the mid channel section with a Horiba U2 probe. For nutrient quantification, water samples were collected below the water surface, kept at 4 °C, and transported to the laboratory for analysis. Total nitrogen (TN) and total phosphorus (TP) were determined on unfiltered samples digested with persulfate, whereas nitrate plus nitrite nitrogen (NO3+ NO2), ammonia (NH4), and soluble reactive phosphate (SRP) were analyzed using standard methods (APHA 1994). Total suspended solids (TSS) were estimated by filtering a known volume of water with pre-weighted fiberglass filters, dried (110 °C, 4 h), and re-weighted.
Attributes of the riparian vegetation were examined at each site using an adaptation of the Riparian corridor index (QBR index by Munné et al. 2003) for Patagonian streams: the QBRp (Kutschker et al. 2009). This index combines information from total cover, structure, complexity, and naturalness of vegetation and the degree of channel alteration (e.g., bank modifications, dredging, etc.). The total QBRp score ranges from 0 (extreme degradation) to 100 points (excellent quality, natural riparian forest). QBRp was applied in summer, given that vegetation cover is at its peak and species identification is more effective.
Habitat condition index (HCI) was applied to evaluate habitat quality using the assessment procedure for high gradient streams of Barbour et al. (1999). This method ranks 10 river channel features (e.g., epifaunal substrate availability, frequency of riffles, etc.) from 0 to 20. A score of 200 points indicates that the river is natural and pristine and in its best possible condition (range: 150–200). This index evaluates the ability of the stream’s physical habitat to support a given fauna then measures the spatial heterogeneity of the stream (Castela et al. 2008).
2.3 Biological Sampling
Quantitative macroinvertebrate samples were taken with a Surber sampler (0.09 m2; 250 μm in pore size). On each reach, three samples from riffles and three samples from pools (n = 6) were taken. Samples were fixed in situ with 4 % formaldehyde and sorted in the laboratory under at least ×5 magnification. Macroinvertebrate species were identified to the lowest possible taxonomic level using regional keys (Domínguez and Fernández 2009), and counted.
We calculated a set of macroinvertebrate community descriptors for each site and sampling date, including richness measures: taxa richness (SR) and Ephemeroptera, Plecoptera and Trichoptera (EPT) richness, Plecoptera richness, Trichoptera richness, Ephemeroptera richness, and Orthocladiinae richness; enumeration measures: total density (TD), Plecoptera density, Trichoptera density, Ephemeroptera density (Barbour et al. 1999); and composition measures: Shannon-Weaver diversity index (H’), percentage of dominant taxon, Biotic Monitoring Patagonian Streams index (BMPS, a biotic index previously adapted for use in the Patagonian region; Miserendino and Pizzolón 1999), percentage of EPT taxa, percentage of Chironomidae, percentage of Orthocladiinae, percentage of Hydropsychidae, percentage of EPT to Chironomidae, and percentage of Orthocladiinae to Chironomidae; and trophic measurements: richness and percentage of each functional feeding group.
The BMPS is an adaptation of the Biological Monitoring Working Party index (BMWP; Armitage et al. 1983) and is obtained from a table of 95 families with different degrees of pollution sensitivity (scores 1–10) present in Patagonia. The total BMPS score ranges from 0 to >150.
Following Barbour et al. (1996), metrics were tested for sensitivity by comparing grazed and ungrazed sites. The sensitivity of each metric was judged according to the degree of interquartile overlap in box-and-whisker plots. Metrics were judged to have one of four sensitivity values: a sensitivity of 3 (strong) if no overlap existed in the interquartile range; a sensitivity of 2 (strong) if there was some overlap that did not extend to the medians; a sensitivity of 1 (weak) if there was a moderate overlap of interquartile ranges but at least 1 median was outside the range; and a sensitivity of 0 if the interquartile overlap was considerable, with no discrimination between reference and impaired sites (Barbour et al. 1996).
2.4 Statistical Analysis
To evaluate differences in physicochemical parameters between pasture and reference sites, Kruskall-Wallis analysis was performed (Sokal and Rohlf 1995). The relative important environmental factors in explaining the variability in macroinvertebrate metrics and the performance of metrics on a seasonal basis was evaluated by using a redundancy analysis (RDA) using CANOCO (ter Braak and Smilauer 1999). RDA was chosen because previous inspection of the data revealed a linear mode rather than a unimodal response in the biotic variables (ter Braak and Smilauer 1998). All environmental variables included in Table 1 were used, initially, to evaluate the response of community descriptors and sites to environmental gradients. Variables (except pH) and community attributes (except H’) were transformed (log(x) + 1) prior to analysis. Variables that were strongly correlated with others (those with an inflation factor >10) in the initial analysis were removed, and a further analysis was carried out with the remaining environmental variables. A Monte Carlo permutation test (9999 permutations) was used to verify the significance of the models (ter Braak and Smilauer 1998).
3 Results
3.1 Physicochemical and Environmental Conditions
Substrate size ranged between fine sediments and boulder; however, overall smaller substrate fractions and higher deposition of sand/gravel at river bottom were detected for pasture sites (Table 1). Mean water temperature values were comprised between 1.7 (ALO) and 13.6 °C (NYF). Pastures had significantly higher water temperature than did reference sites (Kruskall-Wallis p < 0.05) as well as higher conductivity values and lower dissolved oxygen contents than did reference ones (Kruskall-Wallis p < 0.05) (Table 1). Sedimentation impacts were associated with pasture sites as evidenced in total suspended solids’ values (pastures > reference sites, Mann-Whitney p < 0.04). However, nutrient values were within the expected range for patagonian environments, with neither extreme values nor differences between sites recorded.
According to the QBRp index, the riparian ecosystem displayed good (COM and CHI) to very good condition (ALO) at reference sites, while moderate (NyF and LÑ) to strong (MG) riparian ecosystem alteration was detected at pasture sites (Fig. 2a). At the latter sites, a strong riparian modification with a loss of up to 80 % of the riparian forest was observed, MG being the most degraded site, with scarce vegetation cover and composed mainly of exotic herbaceous species (Plantago lanceolata, Rumex acetosella, Artemisia absinthium, and Trifolium repens).
Habitat quality according to the HA score was optimal for the reference sites, which had riverbanks and margins in good condition. Pasture sites, NyF and MG, presented suboptimal conditions, while the worst situation was observed for LÑ, which had marginal quality (Fig. 2b). The main alterations detected in the riverbed were margin erosion due to vegetation removal, and embeddedness owed to an increase in sediment. Deposition was evident in both depositional and transport sections, mainly at LÑ stream, where margins were highly unstable and prone to collapse.
3.2 Macroinvertebrate Community Analyses
A total of 127 taxa were identified and counted in the study (Appendix I). Minimum density was recorded at ALO site while the highest value was found at the pasture site NyF (1322 and 7702 ind.m−2, respectively) (Appendix I). At pasture sites, the macroinvertebrate community was dominated by sensitive species, but with certain plasticity features (univoltine, collector-filtererers, and herbivorous detritivores), such as the hydropsychid Smicridea annulicornis at EÑ, the gripopterigids Antarctoperla michaelseni and Notoperlopsis femina, at MG, and the sericostomatid Parasericostoma ovale at NyF. In all cases, the mean density of these species almost duplicates the density of the second most abundant species.
Annelida species were present at all impacted sites; this group, which is highly tolerant to pollution, was represented by L. variegatus as the most abundant species, followed by Limnodrilus spp. (Appendix 1). Contrary to this, Plecoptera and Trichoptera orders showed the largest number of intolerant species (i.e., species that were present only at reference sites). Several species were completely absent from impacted sites: these were the stoneflies Alfonsoperla sp., Araucanioperla sp., Pictetoperla gayi and Austronemoura quadrangularis, and the caddisflies Metrichia patagonica, M. neotropicalis, Austrocosmoecus hirsutus, Monocosmoecus sp., Austrocentrus valgiformis, and Parasericostoma cristatum.
3.3 Metrics
Selected metrics (Table 2) were able to discriminate different degrees of biotic conditions. Metrics of richness (total richness and EPT, Plecoptera, and Orthocladiinae richness) were all lower at pasture than reference sites, displaying the expected response; sensitivity was strong (Type 3) for all cases with no overlap to some overlap of interquartile ranges. On the other hand, density measures were less sensitive to impairment, with total density, Ephemeroptera density, and Trichoptera density being higher at impaired sites than at reference ones (Fig. 3a).
Distribution of values of selected metrics at pasture (black bars) and reference sites (white bars) during this study (Patagonia, Argentina). Range bars show nonoutlier range, boxes represent interquartile ranges (25–75 %); small squares represent median values; the sensitivity type for each metric is indicated in each figure, and designated as in Barbour et al. (1996). a Richness and density metrics. b Composition metrics. c Trophic metrics
Composition measures, such as Shannon diversity, percentage of dominant taxon, and the biotic index (BMPS) showed a strong sensitivity to impairment. Other metrics that had the same tendency were percentage of Chironomids, percentage of Orthocladiinae, and the ratio percentage of Orthocladiinae to Chironomids, but with a lower degree of sensitivity (types 1 and 2). As expected, percentages of Hydropsychidae and EPT to Chironomids decreased at reference sites. In an unexpected trend, the percentage of EPT taxa increased at impaired sites (Fig. 3b).
Among trophic metrics, shredder richness and percentage of predators displayed a strong (type 3) and predicted response with the lowest values recorded at impaired sites. Values obtained for richness of gatherer, predator, and collector-filterer and the percentage of collector gatherers were also lowest at pasture sites; on the other hand, collector-filterers decreased at reference sites, and all these metrics were poorly sensitive (Fig. 3c).
3.4 Multivariate Analysis
Results of the RDA analysis (first two axes) are summarized in Table 3 and shown in Fig. 4a, b. Species environment correlations were 0.926 and 0.622 for the first and second axes, respectively. Monte Carlo test was significant for the first and for all canonical axes (Table 3). The strongest explanatory factors were physical and chemical variables, which explained 46.3 and 82.7 % of the variance in species data and in the species-environmental relationship, respectively. A first environmental gradient of water temperature and conductivity was highly correlated with RDA1, in which both variables changed seasonally and are in relation to the riparian corridor. A second gradient was highlighted by RDA2 and was defined by pH, soluble reactive phosphorus, total suspended solids, and percentage of sand; all variables strongly related to stream impairment (Fig 4a,b).
The metric percentage of dominant taxon, percentage of filterers, percentage EPT/C, percentage of EPT, total density, Ephemeroptera density, and percentages of Hydropsychidae and predator taxa were grouped on the left upper quadrant (negative end of RDA 1 and positive end of RDA2), which are strongly correlated with high values of conductivity, water temperature, percentage of sand, soluble reactive phosphorus, and total suspended solids. A second group of metrics, on the other hand, was identified on the lower right quadrant. This group included measures that showed a negative response to impairment, such as shredder richness, EPT richness, BMPS, Shannon diversity, Plecoptera richness, collector-gatherer richness, percentage of Chironomiidae, percentage of Othocladiinae, percentage of predators, percentage of gatherers, total richness, and Orthocladiinae richness. All were negatively related to conductivity, water temperature, percentage of sand, soluble reactive phosphorus, and total suspended solids (Fig 4a).
Ordination of all sampling sites (Fig 4b) highlights a good consistency among reference sites; all reference sites had low conductivity, lower sand contribution in substrates, lower values of soluble relative phosphorus, and cooler water. According to the ordination, the most impaired site corresponded to EÑ with most dates (EÑ1, EÑ4, EÑ2) showing high water temperature and conductivity being located on the left upper quadrant. In addition, EÑ2 displaying high values of total dissolved solids, soluble reactive phosphorus and sand predominance on substrates was located on the positive end of RDA2.
Regarding seasonal variances, the RDA analysis highlighted that the impact was more evident for the low water periods (February and December), for most of the cases.
4 Discussion
This research highlights a series of detrimental effects that forest conversion into pasture and grazing practices are producing on aquatic ecosystems. Even when no evidence of nutrient enrichment was detected, changes on suspended solids, conductivity, and water temperature were significant at managed sites (Vondracek et al. 2000). The disturbances derived from forest clearing, in addition to the stocking and trampling actions at grazed sites, were well reflected by a group of taxonomic, compositional, and functional metrics of the benthic community.
Local extinction of sensitive taxa and biodiversity loss have been reported in streams subjected to pastoral development and land use practices that strongly affect the riparian ecosystem (Roque et al. 2003; Binckley and Resetarits 2009; Magoba, and Samways 2010; Parkyn and Smith 2011). Effects seem to be more dramatic at low order streams, such as those studied here (Richardson and Danehy 2007). Our results strongly support the fact that certain benthic species have been eliminated from reaches at pasture streams and that a strong decrease in richness (total taxa, Plecoptera, EPT, Orthocladiinae) and diversity (H') was documented when comparing pasture with reference sites. Sensitive, nontolerant taxa were the most affected; in this respect, a 20 % decrease in EPT richness and a 43 % loss in the Plecoptera assemblage were observed at pasture sites, which is similar to the findings from other geographic regions (Quinn and Hickey 1990; Niyogi et al. 2007; Collier 2008).
Our analysis suggests that, although all pasture streams selected for this study showed differences among them, dissimilarities within stream type were lower than between types (reference vs pastures). In this sense, when contrasting forested versus pasture sites, the main environmental changes observed were related with physicochemical features such as substrate, water temperature, conductivity, total suspended solids, and soluble reactive phosphorous. Consequently, those metrics that consistently responded to impairment were those known as sensitive to fine sediment increases, modification in thermal regime, and changes in food quality/quantity. In our work, these variables were also represented among others by percentage of dominant taxon, EPT richness, BMPS index, Shredder richness, and Shannon diversity.
Some of the analyzed metrics related with organism abundance displayed a weak response to impairment (e.g., total density, Ephemeroptera density, and Trichoptera density). Barbour et al. (1996) mentioned that since density is an element of production, its response can vary according to disturbance type and its magnitude. As shown by several studies, it may increase due to a subsidy in nutrient loading that enhances the overall production of the system (Miserendino et al. 2008; Álvarez-Cabria et al. 2010; Buendia et al. 2013), while it may decline due to impoverished global condition of the habitat, or even due to extreme pollution.
A likely environmental driver in separation among benthic assemblages of our streams was a reduction of the available in-stream habitat, coupled with decreased bed stability (Maloney et al. 2005; Maloney and Feminella 2006). This situation was marked at EÑ, which had the lowest in-stream habitat quality (HA score: marginal), and was also the poorest in terms of community composition. Physical integrity at this reach was highly compromised, with characteristics such as unstable margins, increased fine sediments, and alterations in the riffle-pool sequence due to frequent channel modification. This type of human intervention is highly recurrent in agricultural landscapes across Patagonian mountains (Brand and Miserendino 2011; Miserendino et al. 2011).
Riparian forest is an important feature of riverine landscapes, and as such it fulfills a key role, providing both habitat complexity as well as large volumes of palatable food and organic debris (Read and Barmuta 1999; Richardson and Danehy 2007; Arnaiz et al. 2010). At our pasture sites, the alteration of the riparian forest varied from moderate to strong (EÑ and MG), and, as a result, the community resulted impoverished, with lower values of Shredder richness, diversity, EPT richness, and BMPS index than those reported at reference sites.
Riparian condition is usually related to medium scale management practices, such as the fencing out of livestock from riparian zones and land clearing history (Danger and Robson 2004; Arnaiz et al. 2010). Generally, the amount of remaining forest appears to be a determinant of the macroinvertebrate community condition, as proposed by Scarsbrook and Halliday (1999), and Richardson (2008). In line with these observations, a strong reduction in shredders’ taxa at pasture streams can probably be linked to the decrease in allochthonous input, as a consequence to the absence or simplification of riparian forest (Richardson 2008). Instead, both richness and relative abundance of collector-gatherers showed a tendency to decrease at similar sites. There is some indication that in streams where riparian vegetation has been removed, the number and richness of gatherer species increases. Mesa (2014) found that density, richness, and diversity of collector-gatherers increased significantly in sites with degraded riparian quality in subtropical mountain streams, showing the sensitivity of this functional feeding group to anthropogenic changes, which is consistent with other authors’ findings (Danger and Robson 2004; Thompson and Townsend 2004; Compin and Céréghino 2007; Miserendino and Masi 2010).
Riparian forest removal usually promotes an increase in higher peak temperatures (both hot and cold) and greater average annual temperatures due to the incidence of direct solar radiation (Richardson and Danehy 2007). This in turn may increase primary producer density (Niyogi et al. 2007) and, at the same time, may have deleterious effects on cold stenothermic species such as some Plecoptera and Trichoptera (Rutherford et al. 1997). In the present work, the observed changes of stonefly assemblages and the dramatic diminution in richness (type 3) of this endemic group clearly reveals the deleterious effects of pasture conversion. Another probable detrimental effect was the loss of appropriate sites for imaginal emergence for certain species; thus, the lack of refuges for aerial phases could avoid species recolonization of species with stream-dwelling larvae at a reach scale (Miserendino 2004).
Off-stream watering facilities for livestock are uncommon in Patagonia. Therefore, stock trampling has severe consequences because livestock is concentrated on stream banks and on the channel for water consumption. It generally affects adult stages of many macroinvertebrate species by degrading riparian habitat, at the same time removing substrate and releasing fine sediment to the current, increasing in-stream embeddedness. A decrease in taxa richness and macroinvertebrate abundance has been associated with reductions of available bank and littoral habitats due to livestock activity (Harrison and Harris 2002; Arnaiz et al. 2010). Another consequence of trampling is the modification in the trophic network, because trampling reduces primary production and periphyton palatability through deposition of fine sediment on the stream bottom. A strong decreasing density or absence of species in the grazer/scraper group (Meridialaris laminata, M. diguilina, Penaphlebia chilensis, and Luchoelmis cekalovici) was in coincidence with sedimentation peaks (14.9 mg l−1), especially at the most degraded site which is EÑ. Several works (e.g., Quinn 2000) pointed that 10 mg l−1 of TSS is a threshold value to prevent from deleterious effects on living aquatic biota, especially on aquatic insects.
Low water season showed high temperature and conductivity, while high discharge periods were characterized by increased turbidity levels. In line with this, Becker and Robson (2009) found that macroinvertebrate community composition differed between some site categories during autumn, but not during spring (Arnaiz et al. 2010), indicating that low discharge periods may provide a better evidence of impoverished conditions resulting from agricultural management.
A useful ecological indicator of disturbance should have the following characteristics: easily measured, sensitive, anticipatory, and integrative across key environmental gradients (Cairns et al. 1993; Dale and Beyeler 2001; Maloney and Feminella 2006). In this study, we selected four categories of metrics related to species richness, species composition, density, and function. Our results demonstrate the effectiveness of the set of metrics we used to indicate the initial stages of habitat degradation. The categories that better evidenced the effect of impairment were richness (mostly total, EPT, and Plecoptera richness), and composition metrics, while trophic and density indexes showed a weak response to land use-derived changes.
Metrics here assessed are valuable tools that could be used in monitoring different stages of impairment at areas converted into pasture. Moreover, it will help to better understand the functioning of streams subject to changes in grazing modalities. Until the present, extensive livestock grazing has been the more widespread practice in Patagonia, but there have been recent increases in feedlot modalities as an alternative practice. Recommendation to prevent damage on riverine landscapes would be to restore riparian ecosystems by replanting missing vegetation in buffer areas and to exclude livestock from river shores by limiting their access only to specific places. This could contribute to enhance the ecological integrity of stream draining pasture lands.
References
Allan, J. D. (2004). Influence of land use and landscape setting on the ecological status of rivers. Limnetica, 23(3-4), 187–198.
Álvarez-Cabria, M., Barquín, J., & Antonio Juanes, J. (2010). Spatial and seasonal variability of macroinvertebrate metrics: do macroinvertebrate communities track river health? Ecological Indicators, 10(2), 370–379.
Armitage, P. D., Moss, D., Wright, J. F., & Furse, M. T. (1983). The performance of a new biological water quality score system based on macroinvertebrates over a range of unpolluted running water sites. Water Research, 17, 333–347.
Arnaiz, O. L., Wilson, A. L., Watts, R. J., & Stevens, M. M. (2010). Influence of riparian condition on aquatic macroinvertebrate communities in an agricultural catchment in south-eastern Australia. Ecological Research, 26(1), 123–131.
Barbour, M. T., Gerritsen, J., Griffith, G. E., Frydenborg, R., McCarron, E., White, J. S., & Bastian, M. L. (1996). A framework for biological criteria for Florida streams using benthic macroinvertebrates. Journal of the North American Benthological Society, 15(2), 185–211.
Barbour, M.T., Gerritsen, J., Snyder, B.D., & Stribling, J.B. (1999). Rapid bioassessment protocols for use in streams and wadeable rivers: periphyton, benthic macroinvertebrates and fish, 2nd ed. U.S. Environmental Protection Agency, Washington, DC. EPA. 841-b-99-002.
Becker, A., & Robson, B. (2009). Riverine macroinvertebrate assemblages up to 8 years after riparian restoration in a semirural catchment in Victoria, Australia. Marine and Freshwater Research, 60, 1309–1316.
Binckley, C., & Resetarits, W. J. (2009). Spatial and temporal dynamics of habitat selection across canopy gradients generates patterns of species richness and composition in aquatic beetles. Ecological Entomology, 34(4), 457–465.
Bonada, N., Prat, N., Resh, V. H., & Statzner, B. (2006). Developments in aquatic insect biomonitoring: a comparative analysis of recent approaches. Annual Review of Entomology, 51, 495–523.
Brand, C., & Miserendino, M. L. (2011). Characterizing Trichoptera trophic structure in rivers under contrasting land use in Patagonia, Argentina. In K. Majecka, J. Majecki, & J. Morse (Eds.), Proceedings of the 13th international symposium on trichoptera (pp. pp. 29–40). Bialowieza: Magnolia Press.
Buendia, C., Gibbins, C. N., Vericat, D., Batalla, R. J., & Douglas, A. (2013). Detecting the structural and functional impacts of fine sediment on stream invertebrates. Ecological Indicators, 25, 184–196.
Cairns, J., Jr., McCormic, P., & Niederlehner, B. (1993). A proposed framework for developing indicators of ecosystem health. Hydrobiologia, 263, 1–44.
Carabelli, F., & Scoz, R. (2008). Human-Induced Alterations in Native Forests of Patagonia, Argentina. In: R. Lafortezza, J. Chen, R. Sanesi, & T. Crow, T. (Eds.), Patterns and Processes in Forest Landscapes (pp. 89-105). Springer Netherlands
Castela, J., Ferreira, V., & Graça, M. A. S. (2008). Evaluation of stream ecological integrity using litter decomposition and benthic invertebrates. Environmental Pollution, 153(2), 440–449.
Collier, K. J. (2008). Temporal patterns in the stability, persistence and condition of stream macroinvertebrate communities: relationships with catchment land-use and regional climate. Freshwater Biology, 53(3), 603–616.
Compin, A., & Céréghino, R. (2007). Spatial patterns of macroinvertebrate functional feeding groups in streams in relation to physical and land-cover in southwestern France. Landscape Ecology, 22, 1215–1225.
Dale, V. H., & Beyeler, S. C. (2001). Challenges in the development and use of ecological indicators. Ecological Indicators, 1, 3–10.
Danger, A., & Robson, B. (2004). The effects of land use on leaf-litter processing by macroinvertebrates in an Australian temperate coastal stream. Aquatic Sciences, 66(3), 1–9.
Davies, P. E., & Nelson, M. (1994). Relationships between riparian buffer widths and the effects of logging on stream habitat, invertebrate community composition and fish abundance. Australian Journal of Marine and Freshwater Research, 45, 1289–1305.
Davies-Colley, R., & Smith, D. (2001). Turbidity, suspended sediment and water clarity: a review. Journal of American Water Resources Association, 37, 1085–1101.
del Valle, H. F., Elissalde, N. O., Gagliardini, D. A., & Milovich, J. (1998). Status of desertification in the patagonian region: assessment and mapping from satellite imagery. Arid Soil Research and Rehabilitation, 12, 1–27.
Domínguez, E., & Fernández, H. R. (2009). Macroinvertebrados bentónicos sudamericanos. Sistemática y biología. Tucumán, Argentina: Fundación Miguel Lillo.
Gordon, N. D., McMahon, T. A., Finlayson, B. L., Gippel, C. J., & Nathan, R. J. (2004). Stream hydrology. An introduction for ecologists. Sussex, England: John Wiley & Sons, Ltd.
Harrison, S., & Harris, I. (2002). The effects of bankside management on chalk stream invertebrate communities. Freshwater Biology, 47, 2233–2245.
Kutschker, A., Brand, C., & Miserendino, M. L. (2009). Evaluación de la calidad de los bosques de ribera en ríos del NO del Chubut sometidos a distintos usos de la tierra. Ecología Austral, 19, 19–34.
Larsen, S., Pace, G., & Ormerod, S. J. (2011). Experimental effects of sediment deposition on the structure and function of macroinvertebrate assemblages in temperate streams. River Research and Applications, 267, 257–267.
Lester, R. E., & Boulton, A. J. (2008). Rehabilitating agricultural streams in Australia with wood: a review. Environmental Management, 42(2), 310–326.
Magner, J., Vondracek, B., & Brooks, K. N. (2008). Grazed riparian management and stream channel response in southeastern Minnesota (USA) streams. Environmental Management, 42(3), 377–390.
Magoba, R., & Samways, M. (2010). Recovery of benthic macroinvertebrate and adult dragonfly assemblages in response to large scale removal of riparian invasive alien trees. Journal of Insect Conservation, 14(6), 627–636.
Maloney, K. O., & Feminella, J. W. (2006). Evaluation of single- and multi-metric benthic macroinvertebrate indicators of catchment disturbance over time at the fort benning military installation, Georgia, USA. Ecological Indicators, 6(3), 469–484.
Maloney, K. O., Mulholland, P. J., & Feminella, J. W. (2005). Influence of catchment-scale military land use on physical and organic matter conditions in small southeastern plains streams (USA). Environmental Management, 35, 667–691.
Mesa, L. M. (2014). Influence of riparian quality on macroinvertebrate assemblages in subtropical mountain streams. Journal of Natural History, 48(19-20), 1153–1167.
Miserendino, M. L. (2004). Effects of landscape and desertification on the macroinvertebrate assemblages of rivers in Andean Patagonia. Archive fur Hydrobiology, 159, 185–209.
Miserendino, M. L., & Masi, C. I. (2010). The effects of land use on environmental features and functional organization of macroinvertebrate communities in Patagonian low order streams. Ecological Indicators, 10(2), 311–319.
Miserendino, M. L., & Pizzolón, L. (1999). Rapid assessment of river water quality using macroinvertebrates: a family level biotic index for the Patagonic Andean zone. Acta Limnologica Brasieliensia, 11, 137–148.
Miserendino, M. L., Brand, C., & Di Prinzio, C. Y. (2008). Assessing urban impacts on water quality, benthic communities and fish in streams of the Andes Mountains, Patagonia (Argentina). Water, Air, and Soil Pollution, 194(1-4), 91–110.
Miserendino, M. L., Casaux, R. J., Archangelsky, M., Di Prinzio, C. Y., Brand, C., & Kutschker, A. M. (2011). Assessing land-use effects on water quality, in-stream habitat, riparian ecosystems and biodiversity in Patagonian northwest streams. Science of the Total Environment, 409(3), 612–624.
Munné, A., Prat, N., Solá, C., Bonada, N., & Rieradevall, M. (2003). A simple field method for assessing the ecological quality of riparian habitat in rivers and streams: QBR index. Aquatic Conservation: Marine and Freshwater Ecosystems, 13(2), 147–163.
Niyogi, D., Koren, M., Arbuckle, C. J., & Townsend, C. R. (2007). Stream communities along a catchment land use gradient: subsidy-stress responses to pastoral development. Environmental Management, 39, 213–225.
Parkyn, S., & Smith, B. (2011). Dispersal constraints for stream invertebrates: setting realistic timescales for biodiversity restoration. Environmental Management, 48(3), 602–614.
Paruelo, J. M., Beltrán, A., Jobbágy, E., Sala, O. E., & Golluscio, R. A. (1998). The climate of Patagonia: general patterns and controls on biotic processes. Ecologia Austral, 8, 85–101.
Plafkin, J., Barbour, M.T., Porter, K.D., Gross, S.K., & Hughes, R.M. (1989). Rapid bioassessment protocols for use in streams and rivers: Benthic macroinvertebrate and fish. Office of Water, US Environmental Protection Agency, Washington, DC, EPA/440/4.
Quinn, J. M. (2000). Effects of pastoral development. In K. J. Collier & M. J. Winterbourn (Eds.), New Zealand stream invertebrates: ecology and implications for management (pp. 208–229). Christchurch: New Zealand Limnological Society.
Quinn, J. M., & Hickey, C. W. (1990). Characterization and classification of benthic invertebrate communities in 88 New Zealand rivers in relation to environmental factors. New Zealand Journal of Marine and Freshwater Research, 24, 387–409.
Read, D., & Barmuta, L. (1999). Comparisons of benthic communities adjacent to riparian native eucalypt and introduced willow vegetation. Freshwater Biology, 42, 359–374.
Richardson, J. S. (2008). Aquatic arthropods and forestry: effects of large- scale land use on aquatic systems in Nearctic temperate regions. Canadian Entomologist, 140, 495–509.
Richardson, J. S., & Danehy, R. J. (2007). Zones in temperate forests. Forest Science, 53(2), 131–147.
Riley, R. H., Townsend, C. R., Niyogi, D. K., Arbuckle, C. A., & Peacock, K. A. (2003). Headwater stream response to grassland agricultural development in New Zealand. New Zealand Journal of Marine and Freshwater Research, 37, 389–403.
Roque, F., Strixino, G., Agostinho, R. C., & Fogo, J. C. (2003). Benthic macroinvertebrates in streams of the Jaragua State Park (Southeast of Brazil) considering multiple spatial scales, (Ward 1989). Journal of Insect Conservation, 7, 63–72.
Rutherford, J., Blackett, S., Blackett, C., Saito, L., & Davies-Colley, R. J. (1997). Predicting the effects of shade on water temperature in small streams. New Zealand Journal of Marine and Freshwater Research, 31, 707–721.
Rutherford, J. C., Marsh, N. A., Davies, P. M., & Bunn, S. E. (2004). Effects of patchy shade on stream water temperature: how quickly do small streams heat and cool? Marine and Freshwater Research, 55, 737–748.
Scarsbrook, M. R., & Halliday, J. (1999). Transition from pasture to native forest land-use along stream continua: effects on stream ecosystems and implications for restoration. New Zealand Journal of Marine and Freshwater Research, 33, 293–310.
Sokal, R. R., & Rohlf, F. J. (1995). Bometry (3rd ed.). New York: W.H. Freeman and Company. 887 pp.
Tell, G., Izaguirre, I., & Quintana, R. (1997). Flora y fauna patagónicas. Bariloche: Ediciones Caleuche.
ter Braak, C.J.F., & Smilauer, P. (1998). CANOCO reference manual and user´s guide to Canoco for Windows. Software for Canonical Community Ordination (Version 4). Microcomputer power. Ithaca, N.Y.
ter Braak, C. J. F., & Smilauer, P. (1999). CANOCO for Windows (Version 4.02). A FORTRAN program for canonical community ordination. Wageningen: Centre for Biometry Wageningen.
Thompson, R. M., & Townsend, C. R. (2004). Land-use influences on New Zealand stream communities: effects on species composition, functional organization, and food-web structure. New Zealand Journal of Marine and Freshwater Research, 38(4), 595–608.
Vondracek, B., Frost, J. A., & Mumford, K. G. (2000). Impacts of rotational grazing and riparian buffers on physicochemical and biological characteristics of Southeastern Minnesota USA, Streams. Environmental Management, 26(6), 629–641.
Wood, P. J., & Armitage, P. D. (1997). Biological effects of fine sediment in the lotic environment. Environmental Management, 21(2), 203–217.
Zimmerman, J. K. H., Vondracek, B., & Westra, J. V. (2003). Agricultural land use effects on sediment loading and fish assemblages in two Minnesota (USA) watersheds. Environmental Management, 32, 93–105.
Aknowledgments
This study was supported by CONICET (PIP 5733) and Project A.W.A.R.E. Foundation (P-001036) and PADI Foundation (19/2006). The authors wish to thank Dr. C.Y. Di Prinzio, Dr. R. Casaux, and Lic. C. Masi for fieldtrip assistance, Dr. M. Archangelsky and Dr. P. Pessacq for helping in species identification, Dr. G. Martin for comments on the manuscript, and the two anonymous reviewers for valuable comments that greatly improved the manuscript. This is a Scientific Contribution no. 118 from LIESA.
Author information
Authors and Affiliations
Corresponding author
Appendix 1
Appendix 1
Rights and permissions
About this article
Cite this article
Brand, C., Miserendino, M.L. Testing the Performance of Macroinvertebrate Metrics as Indicators of Changes in Biodiversity After Pasture Conversion in Patagonian Mountain Streams. Water Air Soil Pollut 226, 370 (2015). https://doi.org/10.1007/s11270-015-2633-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-015-2633-x