Abstract
In Brazil’s littoral, many iron ore industries are located in areas of restinga, a type of coastal ecosystem; such industries represent stationary sources of iron and acid particulates. The industrial sector is under expansion, threatening the already fragile ecosystem. In the present study, the simulated impact of these emissions was studied on the early establishment stages of two native plant species, Sophora tomentosa and Schinus terebinthifolius. The results indicate that S. tomentosa display deficient germination and low root tolerance when exposed to iron dust and acidity. Toxic iron concentrations were found to accumulate only in the roots of S. tomentosa plants exposed to increasing doses of iron dust. In these plants, initial growth was reduced, leading to the conclusion that S. tomentosa was unable to regulate, and, thus, to overcome, the effects of the iron dust in the substrate. In contrast, the germination, root tolerance index, and initial growth of S. terebinthifolius were not affected by iron dust and acidity, revealing the species’ resistance to these pollutants. We propose that under similar field conditions, S. tomentosa would be adversely affected, whereas S. terebinthifolius would thrive. On a long-term basis, this imbalance is likely to interfere in the vegetational composition and dynamics of the affected ecosystem.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Brazil is the world’s largest producer of iron ore products, such as pellets, powder and sinter (UNCTAD 2006). The demand for metal goods is increasing worldwide; this consequently compels ore industries to expand their activities, which, in turn, can exacerbate pre-existent ecological problems caused by the sector. The industries constitute a source of geographic and environmental disturbance due to mining and the emission of iron dust and sulfur dioxide (SO2) (Wong and Tam 1977; Lopes et al. 2000; Paling et al. 2001). The iron dust, or iron ore particulate matter, represents the major pollutant released by these industries, during both the processing and storage of final products in open stock yards. These pollutants can be deposited either near the source or carried away, depending on the particle size, wind and landscape features. The SO2 is emitted by the furnaces when coal is burned during the ore pelletization process, thereby increasing the chances of acid particle formation (Lopes et al. 2000).
The effects of particulate matter on natural ecosystems are linked to its chemical constitution and the severity of the exposure to it. Excessive particle accumulation in the ecosystems can influence plant growth and also increases the plant susceptibility to other environmental stresses. This type of pollutant can directly affect photosynthesis, through abrasion, stomata blockage and smothering of the leaves, once the particles settle down on the organ surface (Hirano et al. 1995; Naidoo and Chirkoot 2004). Indirect effects may involve chemical and physical modification of the soil properties. Deposition of acid particles often alters soil pH and enhances the solubility of heavy metals (Farmer 1993). Germination and the early growth stages are the most vulnerable periods of a plant life cycle; thus, any environmental stress, combined with the sensitivity of the species, can interfere with a species’ establishment success (Fan and Wang 2000; Grantz et al. 2003). This condition is likely to affect the vegetation dynamic, causing further ecological problems (Narayan et al. 1994; Wen et al. 2006).
Iron hydroxides constitute the main ore exploited by the industries. Particulate matter derived from the crushing and beneficiation of iron ore is primarily inert and usually unavailable for plants as nutrient source. However, iron particulate accumulation in the soil due to heavy deposition or poor drainage, in combination with low pH of the substrate, may increase the availability of iron (Fe) to plants (Wong et al. 1978). Even though Fe is an essential micronutrient, high levels of this element in the soil can lead to toxicity or nutritional alterations, which can negatively affect plant metabolism (Connolly and Guerinot 2002).
Iron ore industries with full facilities, including factories, stock yards, and ship loading harbors, are present on the coastline of Espírito Santo state in Brazil. Those industries are often located in areas of natural ecosystem, termed restinga (Pereira 1990). Thus, the restinga vegetation is frequently at risk of chronic and acute exposures to iron dust and acid deposition since particles are constantly escaping during the handling process as well as during spills, which are unpredictable (Lopes et al. 2000). The situation is aggravated by the expansion policy of the industries.
However, information is scarce on how or whether the exposure to the related emissions affects the restinga vegetation. This study represents a first attempt to investigate the possible effects of iron dust and acidity on native species of the restinga, through testing germination and initial growth.
2 Materials and Methods
2.1 Area Description
The area exposed to the emissions is located on the littoral of Espírito Santo State, Brazil. The region, at 20°46′21.0″ S and 40°34′52.3″ W, hosts an iron ore industry, which is undergoing an expansion process, and fragments of restinga vegetation. The main ore exploited is itabirito, which is basically composed of hematite (Fe2O3) and quartz (SiO3); its handling and beneficiation generate iron ore particles, mainly coarse particulates, and sulfur dioxide (SO2). In the region, SO2 and total particles in suspension are usually within the standard limits (80 μg m−3 for both atmospheric pollutants), which were established by Brazil's office of environmental regulation. The industry plant includes a stockyard, two furnaces and a harbor for shipment of iron pellets and sinter, the final products. Incidents involving furnaces and spills of iron sinter and powder due to strong winds or troubleshooting of the loading equipment have been reported, although the actual amount of pollutants deposited in the nearby areas has not been disclosed.
According to Köppens classification, the climate in the region is tropical wet and dry (Aw), with a non-pronounced dry season during winter months. The annual precipitation is about 900 mm, the average air humidity is 70%, and the average temperature is 23.4°C.
2.2 Plant Material
Two native species, Schinus terebinthifolius Rardii (Anacardiaceae) and Sophora tomentosa L. (Leguminosae), were chosen to have their germination and initial growth tested under the influence of iron ore dust and different pH levels. These shrub species are components of the restinga after-dune plant community, and they help to stabilize the sand dunes (Gross 1993; Patiño et al. 2002; Lenzi and Orth 2004; Nogueira and Arruda 2006).
Ripened fruits of the two species were collected from plants growing on a restinga fragment, ‘Paulo César Vinha’ State Park, 30 km north of the iron ore industry and opposed to the prevalent wind direction. The seeds were removed, washed in distilled water and soaked first in a 5% sodium hypochlorite solution for 5 min and then in a 0.1% Captan solution for 1 min, to prevent microorganism infestation. After drying at room temperature, the seeds were selected for size uniformity and used within 15 days. Although carried out simultaneously, the experiments for each species were completely independent.
2.3 Iron Dust Material
The iron particulate matter used in the experiments was the manufactured powdered ore, the iron sinter, which is basically composed of metallic or elemental iron (Fe0) and which is often stockpiled in the open yard at the ore industry plant. The granulometric size of the powder ranges from coarse (2.5–10 μm ∅) to large (>50 μm ∅) particles, and its composition is described in Table 1.
2.4 Germination and Root Tolerance Index
The selected seeds were placed in sterile glass Petri dishes (9 cm ∅) containing germination paper and submitted to treatments with different combinations of pH and iron dust dose in a factorial 3 × 2 arrangement with four replicates, in a randomized block design. The pH values studied were 6.5 (distilled water), 5.0 and 3.0, which were achieved by adding diluted sulfuric acid solutions. The doses of iron dust were: 0.0 and 0.6 mg mm−2, based on the average amount daily deposited in the vicinity of an iron ore industry (Lopes et al. 2000). The pH 6.5 without iron dust treatment was used as the control. Each treatment used 100 seeds evenly distributed in four dishes/replicates. The Petri dishes were incubated in a germination chamber at a constant temperature of 27°C with a 16/8 h photoperiod. The solutions with different pH values and the iron dust in the dishes were renewed every 2 days to assure maximum exposure to the intended treatments. A seed was considered to be germinated when the protruded radicle was 2.0 mm long, which took approximately 24–48 h for S. terebinthifolius and 72 h for S. tomentosa. The germinated seeds were counted daily, and the mean cumulative results were expressed as percentages.
To assess the tolerance of pre-germinated seeds to iron dust and acidity, seeds with 2.0 mm protruded radicles were placed in sterile PVC trays (5 × 20 × 45 cm) containing germination paper with the corresponding treatments of iron dust and sulfuric acid solutions described above. For each treatment, 20 pre-germinated seeds were evenly distributed in five trays/replicates in a randomized block design. The trays were covered with polypropylene film and kept at a constant temperature of 27°C with a 16/8 h photoperiod. When necessary, the sulfuric acid solutions were replaced to assure desired pH levels. The primary root length was measured on the first, seventh and 15th days after the start of the experiment. Tolerance was determined by the root tolerance index (RTI) using the equation based on Rout et al. (2000):
2.5 Seedling Emergence and Growth
An additional experiment to assess tolerance to iron dust was set up under greenhouse conditions. In this experiment, 480 seeds of each of the two species were evenly sowed in 24 plastic pots (2.0 L, 9 cm ∅) filled with soil collected from the ‘Paulo César Vinha’ State Park. The soil composition and characteristics were determined (Table 2). The treatments consisted of different amounts of iron dust applied on the soil surface. The iron dust doses were: 0.0, 3.0, 6.0 and 12.0 g, based on Lopes et al. (2000). The experiment was designed in a randomized block arrangement with six replicates per treatment, with each replicate represented by one pot. Due to the sandy nature of the soil, the pots were watered with distilled water every 2 days. The emergence of seedlings, indicated by the expanded cotyledons, was recorded on the seventh, 14th, 21st and 28th days after the start of the experiment. The final number of emerged seedlings was expressed as mean percentage.
In order to evaluate the influence of iron dust on the initial growth, 35 days after the start of the experiment, the seedlings were thinned to two plants per pot, which comprised the new experimental unit. The number of leaves and height of the seedlings were recorded monthly. Standard Hoagland solution (100 mL) at 1/4 strength and pH 6.5 was applied every 15 days. At the end of 180th day, the plants were harvested.
The leaves, stem and roots were separated and washed in distilled water. The roots were then rinsed in a 1.0 mM ethylene diamine tetraacetic acid solution to minimize tissue contamination by external Fe. The total leaf area of each plant was measured using a plan meter (Delta MK2—Delta Devices Ltd, England). All plant parts were dried at 90°C and weighed. Later, a 0.5 g sample of the dried organ was subsequently wet-digested in a nitric-perchloric solution (3:1 v/v), and the total Fe content was determined by atomic absorption spectrophotometry (GBC Avanta—GBC Scientific Equipment Ltd, Australia) using a standard curve of iron solution for accuracy (MERCK Standard Solutions) (Kampfenkel et al. 1995).
2.6 Statistical Analysis
The experimental data were submitted to analysis of variance using a statistical program package (SAEG/UFV). The means of each parameter examined were further compared by Tukey’s test at P ≤ 0.05.
3 Results
3.1 Germination and Root Tolerance Index
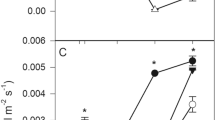
The germination of S. terebinthifolius was not affected by the iron dust and acidity. However, the germination of S. tomentosa was significantly (P ≤ 0.05) reduced by both pH and iron dust, but not by the interaction of them (Fig. 1). During the germination period, most of the S. tomentosa seeds under iron dust exposure developed a dark coloration on their coats, followed by spoiling.
Germination of Schinus terebinthifolius and Sophora tomentosa under different pH (6.5, 5.0 and 3.0) and iron dust (0.0 and 0.6 mg mm−2) exposure. Significant differences between pH levels and iron dust doses are indicated by upper- and lowercase letters, respectively, at P ≤ 0.05, according to Tukey’s test. Bars represent mean ± standard error (n = 4)
The RTI in S. terebinthifolius did not differ (P ≤ 0.05) from the control treatment, indicating that this species was tolerant to the imposed treatments. In S. tomentosa, however, RTI was negatively affected by acid pH and iron dust (Table 3). In the presence of iron dust, the radicles of S. tomentosa displayed stunted growth, and their surfaces were covered by a noticeable reddish-stained coat that probably originated from the deposition of iron oxides or plaques (Fig. 2).
3.2 Seedling Emergence, Growth and Iron Content
The seedling emergence of S. terebinthifolius was not affected (P ≤ 0.05) by the different doses of iron dust applied in this experiment (Table 4). In addition, the iron dust treatments did not interfere with the growth parameters of S. terebinthifolius. However, the emergence of S. tomentosa was consistently reduced by iron dust, even at the lowest dose applied (3.0 g). Stem height, leaf area and nodule dry weights of S. tomentosa were significantly reduced when the seedlings were exposed to iron dust. Roots of S. tomentosa were also stained with a reddish coat that could not be completely removed after the cleansing process.
The Fe content in the organs of S. terebinthifolius varied according to the treatment. The accumulation was pronounced in the roots, but the pattern was also observed in leaves and stems (Fig. 3). In S. tomentosa, Fe content increased in the stem and roots but decreased in the leaves (Fig. 3). Higher Fe content (P ≤ 0.05) was found in the leaves and stems of these two species at doses 6.0 and 12.0 g of iron dust.
4 Discussion
Of the two species assessed, only S. tomentosa germination was affected by the pH and iron dust exposure, despite adequate rehydration of the seeds in all treatments. The germination process in S. terebinthifolius was normal when compared to the control treatment.
Acidity may alter seed physiology, thereby affecting germination (Fan and Wang 2000). Low pH can impair metabolic processes, including glycolysis, by disrupting pH gradient balance within the cells. The reduced germination rate and low RTI of S. tomentosa observed at pH values of 5.0 and 3.0 might be related to respiratory alterations in the embryo, which apparently occurred even in the weaker acid condition (pH 5.0). A limited supply of energy can interfere in the radicle and hypocotyl growth, decreasing the germination capacity of sensitive species (McColl and Johson 1983), as S. tomentosa appears to be.
Color change in seed coat during germination is usually related to phenol oxidation (Rashid et al. 2005). Phenols are commonly present in many parts of the seeds, including the coat and embryo, and they are primarily related to the regulation of seed germination as well as to defense against herbivory and pathogen infestation (Muscolo et al. 2004; Rashid et al. 2005). Although S. terebinthifolus is rich in polyphenols (Lima et al. 2006), no distinct color alteration was observed on the seeds’ coats when iron dust was present. The fast germination of the species, which usually occurred within the first 48 h, could have altered the seeds’ metabolism and contributed to phenols degradation by enzymatic activity, as has been reported for sunflower seeds (Hansel et al. 2001; De Leonardis et al. 2006).
In contrast, the browning of the S. tomentosa seed coat in the treatments with iron dust was probably due to phenol oxidation by the metal. The presence of elemental Fe0, as in iron dust, can cause oxidation of phenol to quinines (Rush et al. 1995). Hydroquinone, a reduced quinone, has a cytotoxic potential that can diminish cell division rate and growth (El-Bargathi and Asoyri 2007). In addition, increased Fe supply can reduce respiratory capacity due to enzyme inactivation by interaction with sulphydryl groups, as was observed in Phragmites australis (Fürtig et al. 1999). Hence, it is thought that the oxidizing characteristic of the iron dust might have accelerated the oxidation of seed coat phenols and/or contributed to the accumulation of internal Fe in the seeds of S. tomentosa, which might have affected the germination process.
After the beneficiation of iron ore, metallic or elemental Fe0 is generated, which is a form of iron that is unavailable to plants. In the presence of water and dissolved oxygen, the elemental Fe0 reverts to its ferrous state Fe2+, which is readily absorbed by roots (Azevedo and Chasin 2003; Schmidt 2003; Pereira and Freire 2005). Nevertheless, Fe2+ in the surrounding rizosphere may react with oxygen and then precipitate, forming mineral plaques of iron oxides (Fe2O3), as were observed on the roots of some wetland plant species (Tanaka et al. 1966); alternatively, Fe2+ may be oxidized by enzymatic activity. The presence of a reddish stain on the root surfaces of S. tomentosa may indicate the formation of mineral precipitates. On the roots of S. terebinthifolius, no such stains were observed, indicating that iron plaques had not been formed on the organ surface. One of the reasons for the lack of iron plaques on the S. terebinthifolius roots could be the chelation of the iron by organic acids or polyphenols released by the organ. This phenomenon could prevent both iron precipitation and iron toxicity, as has been reported for other plant species (Briat 2007).
Depending on size extension of the plaques formed on the roots, they can either diminish the Fe2+ uptake, preventing toxic levels, or interfere in the absorption of other minerals by acting as a physical barrier (St. Cyr. and Campbell 1996). In the first case, the plaque could act as a protective barrier against metal accumulation and the development of further oxidative stress (Sinha et al. 1997). However, in the second case, the growth and development of the roots can be negatively affected due to inadequate nutrition (Yamauchi 1989; Ye et al. 1998). The low RTI of S. tomentosa might in part be linked to the plaque formation phenomenon and the cytotoxic effect of phenol oxidation.
Vegetation contamination by metals, including Fe, has been observed in areas containing iron ore tailings (Wong and Tam 1977; Alvarez et al. 2003). However, in our experiment that tested the effects of iron dust in the initial growth, the mean Fe content in the leaves and stems of S. terebinthifolius or S. tomentosa did not reach the toxic values reported for most plant species, as cited by Pugh et al. (2002). However, in the roots of both species, the levels exceeded the toxic values of 500 ppm (Pugh et al. 2002). Despite this, S. terebinthifolius exhibited no growth reduction or injury symptoms as a result of Fe excess, which suggests that the species is resistant to iron dust in the soil.
Regardless the fact that the Fe content in the leaves of S. tomentosa decreased as the amount of applied iron dust increased, the content in the roots increased and affected the seedlings' growth. The reduced emergence and limited growth of S. tomentosa indicate that, during the initial stages of seedling establishment, this species was affected by the iron dust in the soil. High levels of iron oxides in the soil can lead to iron overlays on the roots which, like the root plaques, affect the rizosphere interactions (Hansel et al. 2001). The reddish coats on the roots of S. tomentosa that were exposed to iron dust treatments indicate the presence of iron oxide on the organ surface and may explain the poor nodulation and growth observed in this species. Indeed, high levels of Fe applied to leguminous species can interfere in the growth of aerial organs by obstructing both nodulation and survival of the symbiotic mycorrhiza, Rhizobium trifolli (Lie and Brotonegoro 1969; Whelan and Alexander 1985). Since the distinction between iron overlays and plaques could not be made, the iron precipitates observed on the roots of S. tomentosa, and the remarkably high Fe content of the organ can be partially attributed to the ineffectiveness of the cleansing process, which could have masked the true values.
5 Conclusion
Our results indicate that the two native species assessed in this study might be differentially affected by emissions of the iron ore industry under field conditions. While S. terebinthifolius shows resilience by not having its germination or growth affected, S. tomentosa might be at a disadvantage upon acute exposure to particulate matter, iron or acid once its initial establishment is affected. Despite the fact that the physiological implications of this abiotic stress upon vegetation are still poorly understood, the resulting competitive imbalance, as a long-term consequence, can affect the restinga flora composition.
References
Alvarez, E., Fernández Marcos, M. L., Vaamonde, C., & Fernández-Sanjurjo, M. J. (2003). Heavy metals in the dump of an abandoned mine in Galicia (NW Spain) and in the spontaneously occurring vegetation. The Science of the Total Environment, 313, 185–197. doi:10.1016/S0048-9697(03)00261-4.
Azevedo, F. A., & Chasin, A. A. M. (2003). Metais: gerenciamento da toxicidade. São Paulo: Atheneu.
Briat, J. F. (2007). Iron dynamics in plants. Advances in Botanical Research, 46, 137–180. doi:10.1016/S0065-2296(07)46004-9.
Connolly, E. L., & Guerinot, M. L. (2002). Iron stress in plants. Genome Biology, 3, 10241–10245. doi:10.1186/gb-2002-3-8-reviews1024.
De Leonardis, A., Albanese, T., & Macciola, V. (2006). Biodegradation in vivo and in vitro of chlorogenic acid by a sunflower-seedling (Helianthus annuus) like-polyphenoloxidase enzyme. European Food Research and Technology, 223, 295–301. doi:10.1007/s00217-005-0209-5.
El- Bargathi, M., & Asoyri, H. (2007). Effect of phenol and naphtol and giberellic acid on seed germination of Allium cepa L. (Onion). Journal of Science and its Application, 1, 6–13.
Fan, H. B., & Wang, Y. H. (2000). Effects of simulated acid rain on germination, foliar damage, chlorophyll contents and seedling growth of five hardwood species growing in China. Forest Ecology and Management, 126, 321–329.
Farmer, A. (1993). The effects of dust on vegetation: a review. Environmental Pollution, 79, 63–75.
Fürtig, K., Pavelic, D. M., Brunold, C., & Brändle, R. (1999). Copper and Iron-induced injuries in roots and rhizomes of reed (Phragmites australis). Limnologica, 29, 60–63.
Grantz, D. A., Garner, J. H. B., & Johnson, D. W. (2003). Ecological effects of particulate matter. Environment International, 29, 213–239.
Gross, C. L. (1993). The reproductive ecology of Canavalia rosea (Fabaceae) on Anak Krakatau, Indonesia. Australian Journal of Botany, 41, 591–599.
Hansel, C. M., Fendorf, S., Sutton, S., & Newville, M. (2001). Characterization of Fe plaque and associated metals on the roots of mine-waste impact aquatic plants. Environmental Science and Technology, 35, 3863–3868.
Hirano, T., Kiyota, M., & Aiga, I. (1995). Physical effects of dust on leaf physiology of cucumber and kidney bean plants. Environmental Pollution, 89, 255–261.
Kampfenkel, K., Van Montagu, M., & Inze, D. (1995). Effects of iron excess on Nicotiana plumbaginifolia plants. Plant Physiology, 107, 725–735.
Lenzi, M., & Orth, A. I. (2004). Caracterização funcional do sistema reprodutivo de aroeira vermelha (Schinus terebinthifolius Raddi), em Florianópolis-SC, Brasil. Revista Brasileira de Fruticultura, 26, 198–201.
Lie, T. A., & Brotonegoro, S. (1969). Inhibition of root–nodule formation in leguminous plants by EDTA. Plant and Soil, 30, 339–342.
Lima, M. R. F., Luna, J. S., Santos, A. F., et al. (2006). Anti-bacterial activity of some Brazilian medicinal plants. Journal of Ethnopharmacology, 105, 137–147.
Lopes, S. A., Oliva, M. A., & Martinez, C. A. (2000). Impacto das imissões de dióxido de enxofre e deposição de material particulado de ferro em espécies vegetais de restinga: avaliação ecofisiológica. In E. Espíndola, C. Paschoal, O. Rocha, M. Bohrer, & A. Oliveira Neto (Eds.), Ecotoxicologia (pp. 53–71). São Paulo: RiMa.
McColl, J. G., & Johnson, R. (1983). Effects of simulated acid rain on germination and early growth of Douglas-fir and Ponderosa pine. Plant and Soil, 74, 125–129.
Muscolo, A., Panuccio, M. R., & Sidari, M. (2004). The effect of phenols on respiration enzymes in seed germination. Plant Growth Regulation, 35, 31–35.
Naidoo, G., & Chirkoot, D. (2004). The effects of coal dust on photosynthetic performance of the mangrove, Avicennia marina in Richards Bay, South Africa. Environmental Pollution, 127, 359–366.
Narayan, D., Agrawal, M., Pandey, J., & Singh, J. (1994). Changes in vegetation characteristics downwind of an aluminum factory in India. Annals of Botany, 73, 557–565.
Nogueira, E. M. L., & Arruda, V. L. V. (2006). Fenologia reprodutiva, polinização e sistema reprodutivo de Sophora tomentosa L. (Leguminosae–Papilionoideae) em restinga da praia da Joaquina, Florianópolis, sul do Brasil. Biotemas, 19, 29–36.
Paling, E. I., Humphries, G., McCardle, I., & Thomson, G. (2001). The effects of iron ore dust on mangroves in Western Australia: lack evidence for stomatal damage. Wetland Ecology Management, 9, 363–370.
Patiño, S., Jeffree, C., & Grace, J. (2002). The ecological role of orientation in tropical convolvulaceous flowers. Oecologia, 130, 373–379.
Pereira, O. J. (1990). Caracterizações fitofisionômica da restinga de Setiba, Guarapari–ES. Simpósio sobre ecossistemas da costa sul e sudeste brasileira, III. Annals pp. 207–219. São Paulo: ACIESP.
Pereira, W. S., & Freire, R. S. (2005). Ferro zero: uma nova abordagem para o tratamento de águas contaminadas com compostos orgânicos poluentes. Química Nova, 28, 130–136.
Pugh, R. E., Dick, D. G., & Fredeen, A. L. (2002). Heavy metal (Pb, Zn, Cd, Fe, and Cu) contents of plant foliage near the Anvil Range lead/zinc mine, Faro, Yukon Territory. Ecotoxicology and Environmental Safety, 52, 273–279.
Rashid, A., Furness, N. H., Eliis, B. E., & Upadhyaya, M. K. (2005). Inhibition of seed germination and seedling growth by hounds-tongue (Cynoglosum officinale L.). Weed biology and Management, 5, 143–149.
Rout, G. R., Samantaray, S., & Das, P. (2000). Effects of chromium and nickel on germination and growth in tolerant and non-tolerant populations of Echinochloa colona (L.) Link. Chemosphere, 40, 855–859.
Rush, J. D., Cyr, J. E., Zhao, Z., & Bielski, B. H. (1995). The oxidation of phenol by ferrate (VI) and ferrate (V). A pulse radiolysis and stopped flow study. Free radical Research, 22, 349–360.
Schmidt, W. (2003). Iron solutions: acquisition strategies and signaling pathways in plants. Trends in Plant Science, 8, 188–193.
Sinha, S., Gupta, M., & Chandra, P. (1997). Stress induced by iron in Hydrilla verticillata, (I. F.) Royele: response of antioxidants. Ecotoxicology and Environmental Safety, 38, 286–291.
St. Cyr, L., & Campbell, P. G. C. (1996). Metals (Fe, Mn, Zn) in the root plaque of submerged aquatic plants collected in situ: relations with metal concentrations in the adjacent sediments and in the root tissue. Biogeochemistry, 33, 45–76.
Tanaka, A., Loe, R., & Navasero, S. A. (1966). Some mechamism involved in the development of iron toxicity symptoms in rice plant. Plant and Soil, 12, 158–164.
UNCTAD (2006). The iron market, 2005–2007. United Nations Conference on Trade and Development, UNCTAD Trust Fund Project. Accessed November/2007. www. unctad.org.
Wen, D., Kuang, Y., Liu, S., Zhang, D., Lu, Y., & Li, J. (2006). Evidences and implications of vegetation damage from ceramic industrial emission on a rural site in the Pearl River delta of China. Journal of Forestry Research, 17, 7–12.
Whelan, A. M., & Alexander, M. (1985). Effects of low pH and high Al, Mn and Fe levels on the survival of Rhizobium trifolii and the nodulation of subterranean clover. Plant and Soil, 92, 363–371.
Wong, M. H., Chan, K. C., & Choy, C. K. (1978). The effect of the iron ore tailing on the costal environment of Tolo harbor, Hong Kong. Environmental Research, 15, 342–356.
Wong, M. H., & Tam, F. Y. (1977). Soil and vegetation contamination by iron ore tailing. Environmental Pollution, 14, 242–254.
Yamauchi, M. (1989). Rice bronzing in Nigeria caused by nutrient imbalances and its control by potassium sulfate application. Plant and Soil, 117, 275–286.
Ye, Z., Baker, A. J. M., Wong, M. H., & Willis, A. J. (1998). Zinc, lead and cadmium accumulation and tolerance in Typha latifolia as affected by iron plaque on the root. Aquatic Botany, 61, 55–67.
Acknowledgments
This research was made possible by Universidade Federal de Viçosa, Conselho Nacional de Desenvolvimento Cientifico e Tecnologia, Fundação de Amparo à Pesquisa do Estado de Minas Gerais and SAMARCO Mineração S.A.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kuki, K.N., Oliva, M.A. & Costa, A.C. The Simulated Effects of Iron Dust and Acidity During the Early Stages of Establishment of Two Coastal Plant Species. Water Air Soil Pollut 196, 287–295 (2009). https://doi.org/10.1007/s11270-008-9776-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11270-008-9776-y