Abstract
Changes in the position of altitudinal treelines and timberlines are considered useful indicators of climatic changes on tree growth and forest dynamics. We sought to determine if recent warming is driving contrasting growth responses of Himalayan birch, at moist treeline (Lete Lekh) and semi-arid timberline (Chimang Lekh) sites in the Trans-Himalayan zone of central Nepal. We used dendrochronological techniques to measure tree ring width (TRW) and basal area increment (BAI) of birch trees from climatically contrasting but nearby sites. The TRW series were correlated with climate records from nearby meteorological stations, and BAI was compared between populations to explore growth trends over recent decades. We found contrasting precipitation trends between nearby sites such that the wet site (Lete) is getting warmer and wetter, and the dry site (Chimang) is getting warmer and drier in recent decades. The radial growth of birch in both moist and semi-arid sites are positively correlated to spring (March–May) rainfall, and negatively correlated to mean and maximum temperature for the same period. The growth climate analysis indicated that moisture availability in early growing season is crucial for birch growth at these locations. The BAI of birch is declining more rapidly at the dry timberline than at the moist treelines in the recent decades, indicating that climatic warming might negatively impact birch radial growth where warming interacts with increasing spring drought in the region. Our work highlights contrasting growth response of birch to climate change at moist and semi-arid forests indicating that local climatic variation must be accounted for when assessing and forecasting regional patterns of tree growth in topographically complex regions like Trans-Himalaya, in order to make accurate predictions of vegetation responses to climate change.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Mountains provide habitats for a large portion of the world’s biodiversity (Beniston 2003). High mountain species are potentially most threatened under ongoing climate change because of their narrow distribution range, geographical isolation and unique ecological adaptations (Grabherr et al. 1994; La Sorte and Jetz 2010). Besides being susceptible to the impacts of a rapidly changing climate (Xu et al. 2009; Sharma and Tsering 2009; Aryal et al. 2012), mountains also provide valuable locations for the early detection of climatic change and its impacts on ecological and hydrological systems (Beniston 2003; Nogués-Bravo et al. 2007).
Temperature is usually the main limiting factor for tree growth in poleward and high-altitude treelines (Körner 2003; Holtmeier and Broll 2007; Harsch et al. 2009). Air and soil temperatures as well as topographic sheltering are important determinants of spatial patterns of tree seedling establishment at high altitude (Greenwood et al. 2014). Trees are, therefore, expected to increase growth and shift poleward and upward with warming temperatures and increased atmospheric CO2 concentration (Körner 2000; Borgaonkar et al. 2011; Gaire et al. 2014, 2016). However, this expectation only holds true if there are no other factors limiting growth at species distributional limits in these regions. Tree growth in high mountains could also be impaired by increases in temperature, mostly due to warming-induced water deficit (Cook et al. 2003; Sano et al. 2005; Lv and Zhang 2012; Yang et al. 2013; Dawadi et al. 2013; Liang et al. 2014; Qi et al. 2015; Tiwari et al. 2016; Gaire et al. 2016). Furthermore, in topographically complex regions such as the Trans-Himalayan zone of Nepal, rain shadow effects can lead to strong gradients of precipitation and drought stress over short distances, resulting in contrasting local limitations of tree growth and complicating prediction of regional changes (Schickhoff 2005; Schickhoff et al. 2015).
Tree ring width indices have proved to be useful proxies for growth climate interpretation, however, detrended ring width series retains less ecological and historical information such as forest disturbance, acute growth stress, and other low-frequency signals (Esper et al. 2002). Hence, unstandardized basal area increment (BAI) is often used as a more informative measure of tree growth trends in terms of total biomass production, than ring width measurements alone (Phipps and Whiton 1988; Peñuelas et al. 2011). Climatic warming, increased concentration of atmospheric CO2, and associated changes to water-use efficiency are predicted to enhance tree growth across many species and regions (Morison, 1993; Morgan et al. 2004; Norby et al. 2005). However, such anticipated positive growth trends have not been observed in many parts of the world (Peñuelas et al. 2011). Rather, tree growth has locally declined, with the potential for increasing levels of mortality due to warming-induced drought (Jump et al. 2006; Peñuelas et al. 2007; Allen et al. 2010). Hence, quantifying interannual responses of tree growth to local variation in climate is an important approach to understand tree growth patterns and forest productivity (Bunn et al. 2005; Yang et al. 2010) in climate sensitive high-elevation forests.
Himalayan birch (Betula utilis) typically forms the treeline in many parts of Himalaya (Miehe et al. 2015), but its growth across the various climatic zones and complex topography of central Nepal is poorly explored. The treeline is generally considered as the highest elevation at which a single upright tree with height greater than 2 m is found over the landscape (Hofgaard 1997; Körner 2003), and alpine timberlines represent the upper limit of closed mountain forest with tree density (trees >5 m tall) of at least 30% coverage (Wardle 1974; Holtmeier 2003). Trees typically show contrasting growth responses to warming temperatures at their upper and lower range edges (Peñuelas et al. 2008; Cavin and Jump 2016). However, differences in water availability might strongly modify expected responses to temperature. Consequently, we sought to determine the response of birch growth at its upper and lower range edge in high mountain forests which are subject to a contrasting moisture regime. Consequently, we set out to test expectations that the birch treeline should advance in areas with greater water availability (Schickhoff et al. 2015), and retreat in dry sites (Liang et al. 2014) despite general predictions of upward elevational shifts of mountain forest distributions (Harsch et al. 2009). Improving our understanding of local climate variability and its impact on tree growth and forest productivity is of high importance for our ability to predict spatial patterns of forest change and their consequences. Consequently, in this study, we hypothesized that elevated temperatures would have a greater negative impact on tree growth in dry versus wet high-altitude sites for B. utilis. We used tree ring records of B. utilis from contrasting climatic zones in the Trans-Himalayan region to (1) determine the limiting climatic factors for growth of B. utilis in moist and dry sites and (2) assess the growth trends of B. utilis at its upper and lower range edge with response to ongoing climate change.
Methods
Study area
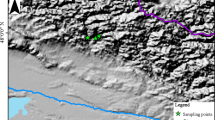
This work was conducted at Lete Lekh (28.6°N, 83.58°E, 3650–3900 m asl) and Chimang Lekh (28.75°N, 83.7°E, 3000–3300 m asl) of Mustang district in the Trans-Himalayan zone of central Nepal. Lete Lekh is a relatively moist area with alpine birch treeline in the north east facing slope, whereas Chimang Lekh is a semi-arid zone with birch timberline in the south–west facing slope. Lete treeline is the upper treeline and the Chimang timberline is the lower range edge of B. utilis in the southern part of Mustang District; however, at Chimang, the birch treeline reaches as high as 4000 m asl only in the sheltered north-facing slopes (personal observation). The sites are characterized by a contrasting precipitation regime within a short distance, and lie within the Annapurna Conservation Area (ACA). The Trans-Himalayan zone of central Nepal lies in the rain shadow formed by surrounding high mountains, where runoff water from snow cover is the main source of water (Aryal et al. 2012). The northern part of the Mustang lies in the Trans-Himalayan semi-arid dry zone, further north, the Tibetan type highland forms the driest zone of Nepal (Lomanthang: 200 mm annual rainfall) (Stainton 1972; Schickhoff 2005). The forested areas are thus confined to the southern part of the district covering only about 3.24% of land area (Government of Nepal 2010).
Species
Himalayan birch (Betula utilis D. Don) is a moderate-sized (<20 m tall) broadleaved pioneer tree species dominating an extensive area of subalpine forests up to 4500 m elevation, quite close to glaciers on northern slopes of the inner Himalayas (Stainton 1972; Zobel and Singh 1997; TISC 2002; Schickhoff 2005; Miehe et al. 2015). This species shows high variation in its local distribution possibly due to the influence of local-scale climate variability and topographic sheltering.
Field sampling
Field sampling was conducted during September–October of 2014 along transects on a topographically uniform area of the treeline at Lete and timberline at Chimang. Tree cores were collected from trees occurring in two treeline transects MT1 and MT2, each of which was 20 m × 120 m (hereafter MT for indicating moist treelines), and the third transect from a single timberline transect of 30 m × 50 m at Chimang (hereafter DT). The treeline sites included the treeline ecotone of B. utilis spanning the treeline and timberline.
We employed dendrochronological techniques for radial growth measurement and developing ring width chronology of trees. Single, two, or three cores were extracted from a tree using a 5.5 mm increment borer. All treeline trees were cored at the base of the tree (<30 cm), whereas DT trees were cored at breast height level. Tree cores were air-dried, mounted on sample holders, and sanded using progressively finer sandpaper according to standard methods (Fritts 1976). Ring widths were measured at a resolution of 0.01 mm with a LINTAB II measuring system (Rinntech Germany). Statistical analyses were performed using the software R (R Core Team 2015).
Meteorological data
Each study site has a meteorological station nearby (<4 km); however, the wet (Lete) site (28.63°N, 83.6°E, 2384 m asl) does not have temperature data for more than 15 years. The 15 years mean temperature of Lete was compared with temperature of Thakmarpha station (28.75°N, 83.7°E, 2566 m asl) for the same period and showed high correlation (r = 0.69, n = 15); hence, we used the Thakmarpha (1970–2013 ad) temperature records, and rainfall records of Lete and Chimang separately for growth climate analysis. The meteorological data showed mean annual rainfall of 1340 mm at Lete and 393 mm at Thakmarpha with mean annual temperature of 11.17 °C (Fig. 1).
Growth–climate response
Tree ring width (TRW) indices were employed to explore growth response to climate. We adopted visual inspection for cross-dating tree cores (Stokes and Smiley 1968), with statistical tests (sign-test and t test) using the software package TSAP-Win (Rinn 2003). Ring width measurements were detrended with a negative exponential using ARSTAN software (Cook 1985). We produced a standard tree ring width chronology of 107 years using 56 tree cores from 52 trees at MT1, 186 years using 27 tree cores from 23 trees at MT2, and of 122 years using 49 tree cores from 24 trees at DT (Table 1). To maintain a reasonable sample depth, we limit the tree ring width chronology to the period representing a minimum of ten cores for each site. Growth–climate relationships were determined by correlating site standard chronology with monthly climatic variables (total rainfall, mean air temperature) from June of the previous growth year until October of the current growth year (Fritts 1976). Given the previously identified importance of spring season (March–May) moisture sensitivity of birch for high-elevation birch populations in the central Himalaya (Dawadi et al. 2013; Liang et al. 2014; Gaire et al. 2016), we also correlated the site chronology with mean climate for the March–May period.
Basal area index (BAI) chronology
BAI is commonly used to assessing tree and stand growth since it allows accurate quantification of tree productivity (Rubino and McCarthy 2000; Peñuelas et al. 2011). The BAI sigmoidal growth model is an appropriate means for detecting changes in tree growth avoiding detrending and standardizing employed in calculation of RWI (Phipps and Whiton 1988; Esper et al. 2002; Salzer et al. 2009). Here we used individual tree BAI to produce mean unstandardized BAI series across all trees at each site for each year.
Ring width was converted into tree BAI according to the following standard formula:
where ‘R’ is the radius of the tree and ‘n’ is the year of tree ring formation. The BAI chronology was produced using the bai.out function in the dplR package in R as some tree cores had missed pith and almost every core had intact bark (R Core Team 2015). We produced BAI series from each site from the period which has at least 10 trees to represent the site population, and to avoid idiosyncrasies at the individual tree level. We analyzed the trend of BAI series at each site for the time period of 1990–2014 ad, that represented the growth trend after the growth release phase, thereby avoiding periods of juvenile growth that are likely to be substantially determined by competitive interactions rather than climate (Jump et al. 2006).
Results
Climatic trends
Mustang (Lete and Thakmarpha) showed significant warming with a consistent increase of mean and maximum annual temperature during recent decades but without any trend in minimum temperatures (Fig. 2). The moist site (Lete) showed a significant increase in annual rainfall and a stable trend of spring season (March–May) rainfall, whereas the dry site (Thakmarpha) showed no rainfall trend. Hence, considering the overall balance of temperature trends and total rainfall, the moist site (MT) is getting wetter and warmer whereas the dry site (DT) is getting drier and warmer in the recent decades (Figs. 2, 3a, b). The amount and duration of snow cover is an important determinant of soil moisture in high mountains in Himalayas (Müller et al. 2016). While we do not have direct measurements of snow cover at each study site, local people report substantial decrease in snow fall around Chimang (Tiwari, 2015 personal communication) (Fig. 4).
Ring width chronology and growth–climate response
We produced three well-replicated tree ring with chronologies of Betula utilis, from the moist treeline sites, and semi-arid timberline site. The location of sampling sites and chronology statistics are summarized in Table 1.
The growth climate analysis of B. utilis revealed a significant positive relationship (p < 0.05) between the ring width series and monthly rainfall during spring season (March–May) both at moist treeline (MT1, MT2) and dry timberline (DT) (Fig. 5d–f). At least 1 month during spring season showed significant positive correlation (p < 0.05) of total rainfall with ring width indices in each site. Previous year’s summer rainfall was found to have a stronger positive impact on tree radial growth at DT, while this effect was observed more weakly at MT1 and MT2. A significant negative relationship (p < 0.05) between radial growth and mean temperature (T mean) for the spring season was observed at both treeline sites (MT1 and MT2), although this relationship was weaker at DT. T mean of October also showed a significant negative relationship with radial growth (p < 0.05) at MT1 and DT (Fig. 5a, c). Further, the minimum temperature (T min) showed stronger negative correlation in early growing season (Feb–May) at MT2, which was weaker at MT1 and DT. However, the maximum temperature (T max) showed stronger negative correlation to ring width indices at DT in comparison to MT1 and MT2. Overall, while similarities in response are clear, we also found a contrasting response of radial growth to monthly climate among sites.
Correlation coefficients of tree ring width indices, with total monthly maximum (T max), mean (T mean), and minimum temperature (T min); MT1 (a), MT2 (b), DT (c), and total monthly rainfall; MT1 (d), MT2 (e), DT (f); of June in the previous year to October of the current year, spring season and annual climate, dashed horizontal lines indicate significant correlation at 95% confidence limit for a two-tailed test
Temporal pattern of radial growth
BAI chronology at MT1 (1933–2014) and MT2 (1935–2014 ad) indicated a rapid increase followed by relatively stable (MT1) and weakly declining (MT2) trend in recent years, whereas at DT, BAI (1912–2014 ad) increased steadily at the beginning and declined abruptly in recent decades. BAI at DT showed a statistically significant declining trend, while that of MT1 and MT2 was without statistically significant trend for the same period (1990–2014 ad) as indicated by linear regression (Figs. 6, 7).
Discussion
Climatic trends
Climate data for the study regions at Mustang showed a consistent warming both at MT and DT, and high spatial variability in rainfall, as reported in Eastern Himalayas (Sharma et al. 2000; Shrestha et al. 2012). The moist region of Lete is getting wetter and warmer with increasing rainfall, whereas the semi-arid region of Thakmarpha is getting drier and warmer with very low and stable rainfall over recent decades. Such diverging trends of temperature and precipitation might have significant impacts on plant growth and forest productivity, given that the rapidly increasing maximum temperature enhances evapotranspiration and will negatively impact soil moisture availability in the drier spring season (Schickhoff et al. 2015). The increasing rainfall at Lete and almost stable rainfall at Thakmarpha is contrary to the enhanced frequency of winter and pre-monsoon drought reported from western Nepal (Wang et al. 2013). While the climatic trend of the Trans-Himalayan region (Mustang) showed the typical regional pattern of a rapid increase in maximum temperature and almost stable trend in minimum temperature, local precipitation trends show more variability. A consistent trend of increasing average rainfall was reported in the central Himalayas except the Northern Triangle temperate forest ecoregion (Shrestha et al. 2012), irrespective of decreasing precipitation trends in Western Himalayas (Kumar and Jain 2009). This contrast limits broad generalizations of climate trends and vegetation responses in the region (Schickhoff 2005; Miehe et al. 2015; Tiwari et al. 2016). We emphasize that the climate of the interior of the topographically complex Trans-Himalayan region is highly spatially variable and regionally complex so that extrapolation of future precipitation trends and forest responses for the region could be misleading.
Ring width chronology and growth–climate response
Our results demonstrated that the radial growth of B. utilis was mainly limited by temperature-induced moisture stress during the spring season both at treeline (MT) and timberline (DT), irrespective of substantial differences in total rainfall between sites. The spring season rainfall and maximum temperature were relatively more important than mean temperature in the dry timberline site (DT). Our findings are in agreement with the moisture sensitivity of birch reported from the upper timberline in central Himalaya (Dawadi et al. 2013; Liang et al. 2014; Gaire et al. 2016), and for Abies spectabilis from the semi-arid treeline in the Trans-Himalayan zone (Tiwari et al. 2016). However, tree growth at high altitudes and latitudes was also reported to have been favored by high summer temperature (Barber et al. 2004; Chen et al. 2011). In our results, the temperature sensitivity of birch at MT was seen in the negative correlation with mean temperature (T mean) of early spring (Feb–March), which was less strong at DT.
Our results revealed that spring season precipitation was more important for radial growth at DT in comparison with MT; this could be because of higher temperature and increased competition between trees at DT and also due to precipitation deficit in that area, in comparison to sparse trees and higher precipitation at MT, as has been reported in various studies in Himalaya and Tibet (Lv and Zhang 2012; Qi et al. 2015; Tiwari et al. 2016). Spring season climate is critical for B. utilis in the Himalayan highlands as shoot, leaf, and floral buds begin to sprout during April (Bisht et al. 2014), and warmer springs can contribute to elevated frost damage and reduced water availability, with consequent negative impacts on tree growth (Körner 2003). The early growing season (Feb–May) mean temperature (T mean) sensitivity in the treeline sites could be associated with snow pack accumulation, as most of the precipitation during this time falls in the form of snow. The timing and duration of snow accumulation are important factors in the treeline where early warming (increase of T mean) of growing season (Feb–March) could facilitate budding and shoot formation (Bisht et al. 2014) and radial growth by snow melt (Vaganov et al. 1999; Bekker 2005). However, there is a chance of heat-induced water deficit during the spring season and any such drought exposure could affect growth and seedling establishment at treelines, and may even cause retreating of treeline (Liang et al. 2014).
In this study, we reported more or less similar climate growth response at MT1, MT2, and DT, although the strength of the correlation differed. Tree radial growth showed a stronger negative response to maximum temperature (T max) at DT (Fig. 5), implying that projected warming will negatively impact future growth as drought stress intensifies at DT, given consistent warming and low precipitation in the region (Figs. 1, 2, 3). In the Himalayan region, moisture stress during spring season (March–May) was found to limit the radial growth of B. utilis even at the treeline (Gaire et al. 2016). Some studies in the Himalaya also reported radial growth of A. spectabilis to be temperature sensitive (Bräuning 2004; Borgaonkar et al. 2011; Gaire et al. 2014), and they explained a negative relationship between spring climate and radial growth as threshold effects of moisture or temperature. Further, they emphasized heat-induced water deficit associated with high velocity of wind and increased evapotranspiration, to have negative influence on tree growth (Cook et al. 2003; Gaire et al. 2014). We infer that the precipitation seasonality is highly important for systems with a short growing season in higher altitude, with a shorter time for plants to compensate for a dry period, and especially so when the season is also characterized by high temperature (Spence et al. 2015). However, we also emphasize here that, although the rainfall is increasing in the moist region like Lete Lekh, and it is stable in the semi-arid region like Chimang, if precipitation is increasingly falling as rainfall rather than snow, soil water recharge from snow could further decline. Hence, even where overall precipitation is increased, the site might even be drier in the summer since water from rain flows away quickly, while the water from snowmelt seeps gradually into the soil over a longer period (Müller et al. 2016). Consequently, accurate characterisation of snow fall is desirable to help understand if weaker spring precipitation and continuous warming will drive further decline of birch radial growth and vitality in the semi-arid region of Mustang.
Temporal pattern of radial growth
In mature forest stands, age-related trends of BAI are generally positive. BAI may continue to increase in healthy stands (LeBlanc et al.1992; Duchesne et al. 2003), or stabilize (LeBlanc et al. 1992), but it does not show a decreasing trend until trees begin to senesce or unless trees are subject to significant growth stress (LeBlanc et al. 1992; Weiner and Thomas 2001; Duchesne et al. 2003; Jump et al. 2006). Our results showed that the BAI of birch at MT1 and MT2 typically followed a sigmoidal pattern as it increased rapidly from young to middle age and remained almost stable during period of middle and older age as described by Spiecker et al. (1996) and Weiner and Thomas (2001). We report abruptly decreasing BAI of birch trees in the semi-arid timberline (DT) over recent decades in comparison to that of relatively moist treeline (MT). The negative influence of warm-day conditions (T max) to radial growth also indicated that the semi-arid high mountain biota is experiencing temperature-induced drought stress (Fig. 5c, f). This finding emphasizes that tree growth and productivity in the semi-arid high mountains might further decline under the projected warming scenario (IPCC 2013). The contrasting growth trend of birch at moist treeline and semi-arid timberline sites showed the strong influence of precipitation associated with increasing warm-day conditions. The decrease of BAI at DT should be interpreted with some caution, however, because of lower canopy exposure at DT than MT, and potentially higher competition between trees in this semi-arid zone. In contrast, MT is characterized by comparatively less competition and a more open canopy (Spiecker et al. 1996), which might contribute to increasing/stable BAI at MT. Furthermore, nutrient availability, soil quality, and topographic exposure also play important roles in modifying local growth patterns of birch forests in the region (Müller et al. 2016). Nonetheless, given the growth–climate relationships identified, we believe that structural differences and potential-localized site quality variation between the treelines do not adequately explain BAI decline at DT, since this site would be expected to show lower BAI with lower individual tree resource availability but not declining BAI over time.
Consequently, we interpret our results as demonstrating that declining BAI at the semi-arid site is an early signal of growth decline at DT over the recent decades, due to increasing drought stress. Although growth climate analysis revealed DT to be more sensitive to precipitation than temperature, we emphasize that the increasing temperature in the future (IPCC 2013) would further intensify any temperature-induced drought stress in the region. B. utilis is an important early successional species in the Himalayan treelines (Shrestha et al. 2007) and therefore a highly significant species in terms of forest ecosystems in the Himalayan highlands, declining growth in this species might alter competitive relationships, and hence, future forest composition at the lower range edge in the semi-arid forest of Chimang Lekh.
Conclusion
The spatial variability of climate in the interior of high mountain regions like the Himalayas is high due to interactions of complex topography and rain shadow effects. Although climate warming is generally expected to have an ameliorating influence on plant growth at high elevation, leading to upward elevational shifts of tree species, the positive influence of increasing temperature is not always universal in such regions. Strong gradients of precipitation over short distances result in high-altitude forests showing varying sensitivity to temperature and moisture. We find that spring precipitation and temperature are critical for radial growth of Himalayan birch, yet precipitation is more important than temperature at drier sites. Birch radial growth declined significantly at the dry site investigated here, where the rainfall trend is almost stable despite increasing temperatures over the recent decades. No such growth decline is seen in the wet site, where moisture availability remains adequate. Decreasing birch radial growth at its lower range edge demonstrates the negative influence of increasing temperatures in contrast with the moist upper treeline. Our results highlight the strongly spatially variable response of Himalayan birch to elevated temperatures and changing precipitation patterns in the region and can refine our understanding of likely responses of mountain forests to climate change.
References
Allen CD, Macalady AK, Chenchouni H, Bachelet D, McDowell N, Vennetier M, Kitzberger T, Rigling A, Breshears DD, Hogg ET, Gonzalez P, Fensham R, Zhang Z, Castro J, Demidova N, Lim JH, Allard G, Running SW, Semerci A, Cobb N (2010) A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For Ecol Manag 259:660–684. doi:10.1016/j.foreco.2009.09.001
Aryal A, Hipkins JJW, Raubeinhimer D, Brunton D (2012) Distribution and diet of brown bear in Annapurna Conservation Area, Nepal. Ursus 23:231–236
Barber VA, Juday GP, Finney BP, Wilmking M (2004) Reconstruction of summer temperatures in interior Alaska from tree-ring proxies: evidence for changing synoptic climate regimes. Clim Change 63:91–120
Bekker MF (2005) Positive feedback between tree establishment and patterns of subalpine forest advancement, Glacier National Park, Montana, U.S.A. Arct Antarct Alp Res 37(1):97–107. doi:10.1657/1523-0430(2005)037[0097:PFBTEA]2.0.CO;2
Beniston M (2003) Climatic changes in mountain regions: a review of possible impacts. Clim Change 59:5–31
Bisht VK, Kuniyal CP, Bhandari AK, Nautiyal BP, Prasad P (2014) Phenology of plants in relation to ambient environment in a subalpine forest of Uttarakhand, western Himalaya. Physiol Mol Biol Plants 20:399–403. doi:10.1007/s12298-014-0238-2
Borgaonkar HP, Sikder AB, Ram S (2011) High altitude forest sensitivity to the recent warming: a tree-ring analysis of conifers from Western Himalaya, India. Quat Int 236:158–166. doi:10.1016/j.quaint.2010.01.016
Bräuning A (2004) Tree-ring studies in the Dolpo-Himalya (western Nepal). In: Proceedings of the DENDROSYMPOSIUM 2003 2:8–12
Bunn AG, Graumlich LJ, Urban DL (2005) Trends in twentieth-century tree growth at high elevations in the Sierra Nevada and White Mountains, USA. Holocene 15:481–488
Cavin L, Jump AS (2016) Highest drought sensitivity and lowest resistance to growth suppression are found in the range core of the tree Fagus sylvatica L. not the equatorial range edge. Glob Change Biol. doi:10.1111/gcb.13366
Chen L, Wu S, Pan T (2011) Variability of climate-growth relationships along an elevation gradient in the Changbai Mountain, northeastern China. Trees 25:1133–1139
Cook ER (1985) A time-series analysis approach to tree-ring standardization. Ph.D Dissertation, The University of Arizona Press, Tucson
Cook ER, Krusic PJ, Jones PD (2003) Dendroclimatic signals in long tree-ring chronologies from the Himalayas of Nepal. Int J Climatol 23:707e732
Dawadi B, Liang E, Tian L, Devkota LP, Yao T (2013) Pre-monsoon precipitation signal in tree rings of timberline Betula utilis in the central Himalayas. Quat Int 283:72–77. doi:10.1016/j.quaint.2012.05.039
Duchesne L, Ouimet R, Morneau C (2003) Assessment of sugar maple health based on basal area growth pattern. Can J For Res 33:2074–2080
Esper J, Cook E, Schweingruber F (2002) Low-frequency signals in long tree-ring chronologies for reconstructing past temperature variability. Science 295:2250–2253
Fritts HC (1976) Tree rings and climate (Reprint, 2001). The Blackburn Press, Caldwell, NJ
Gaire NP, Koirala M, Bhuju DR, Borgaonkar HP (2014) Treeline dynamics with climate change at the central Nepal Himalaya. Clim Past 10:1277–1290. doi:10.5194/cp-10-1277-2014
Gaire NP, Koirala M, Bhuju DR, Carrer M (2016) Site- and species-specific treeline responses to climatic variability in eastern Nepal Himalaya. Dendrochronologia. doi:10.1016/j.dendro.2016.03.001
Government of Nepal (2010) Mustang District profile. District Statistical Office, Government of Nepal, Mustang District, Nepal, p 87
Grabherr G, Gottfried M, Pauli H (1994) Climate effects on mountain plants. Nature 369:448
Greenwood S, Chen JC, Chen CT, Jump AS (2014) Strong topographic sheltering effects lead to spatially complex treeline advance and increased forest density in a subtropical mountain region. Glob Change Biol 20:3756–3766. doi:10.1111/gcb.12710
Harsch MA, Hulme PE, McGlone MS, Duncan RP (2009) Are treelines advancing? A global meta-analysis of treeline response to climate warming. Ecol Lett 12:1040–1049. doi:10.1111/j.1461-0248.2009.01355.x
Hofgaard A (1997) Inter-relationships between treeline position, species diversity, land use and climate change in the central scandes mountains of Norway. Glob Ecol Biogeogr Lett 6:419–429. doi:10.2307/2997351
Holtmeier FK (2003) Mountain timberlines: ecology, patchiness and dynamics. Kluwer Academic Publishers, Dordrecht
Holtmeier FK, Broll G (2007) Treeline advance—driving processes and adverse factors. Landsc Online 1:1–33. doi:10.3097/LO.200701
IPCC (2013) Summary for policymakers. In: Stocker TF, Qin D, Plattner GK, Tignor M, Allen SK, Boschung J, Nauels A, Xia Y, Bex V, Midgley PM (eds) Climate change 2013: the physical science basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on climate change. Cambridge University Press, Cambridge, pp 3–32
Jump AS, Hunt JM, Peñuelas J (2006) Rapid climate change-related growth decline at the southern range edge of Fagus sylvatica. Glob Change Biol 12:2163–2174. doi:10.1111/j.1365-2486.2006.01250.x
Körner C (2000) Biosphere response to CO2 enrichment. Ecol Appl 10:1590–1619
Körner C (2003) Alpine plant life: functional plant ecology of high mountain ecosystems. Springer, Berlin
Kumar V, Jain SK (2009) Trends in seasonal and annual rainfall and rainy days in Kashmir valley in the last century. Quat Int 211:64–69
La Sorte FA, Jetz W (2010) Projected range contractions of montane biodiversity under global warming. Proc R Soc B 277:3401–3410
LeBlanc DC, Nicholas NS, Zedaker SM (1992) Prevalence of individual-tree growth decline in red spruce populations of the southern Appalachian Mountains. Can J For Res 22:905–914
Liang E, Dawadi B, Pederson N, Eckstein D (2014) Is the growth of birch at the upper timberline in the Himalayas limited by moisture or by temperature? Ecology 95:140307191613003. doi:10.1890/13-1904.1
Lv LX, Zhang QB (2012) Asynchronous recruitment history of Abies spectabilis along an altitudinal gradient in the Mt. Everest region. J Plant Ecol 5:147–156. doi:10.1093/jpe/rtr016
Miehe S, Miehe G, Miehe S, Böhner J, Bäumler R, Ghimire SK, Bhattarai K, Chaudhary RP, Subedi M (2015) Vegetation ecology. In: Miehe G, Pendry CA (eds) Nepal: an introduction to the natural history, ecology and human environment in the Himalayas—a companion to the Flora of Nepal, Chapter 16, 1st edn. The Royal Botanical Garden Edinburgh, Edinburgh, pp 385–472
Morgan JA, Pataki DE, Körner C, Clark H, Del Grosso SJ, Grünzweig JM, Knapp AK, Mosier AR, Newton PCD, Niklaus PA, Nippert JB, Nowak RS, Parton WJ, Polley HW, Shaw MR (2004) Water relations in grassland and desert ecosystems exposed to elevated atmospheric CO2. Oecologia 140:11–25
Morison JIL (1993) Response of plants to CO2 under water limited conditions. Vegetatio 104:193–209
Müller M, Schwab N, Schickhoff U, Böhner J, Scholten T (2016) Soil temperature and soil moisture patterns in a Himalayan alpine treeline ecotone. Arct Antarct Alp Res 48:501–521. doi:10.1657/AAAR0016-004
Nogués-Bravo D, Araújo MB, Errea MP, Martínez-Rica JP (2007) Exposure of global mountain systems to climate warming during the 21st century. Glob Environ Change 17:420–442
Norby RJ, DeLucia EH, Gielen B, Calfapietra C, Giardina CP, King JS, Ledford J, McCarthy HR, Moore DJP, Ceulemans R, De Angelis P, Finzi AC, Karnosky DF, Kubiske ME, Lukac M, Pregitzer KS, Scarascia-Mugnozza GE, Schlesinger WH, Oren O (2005) Forest response to elevated CO2 is conserved across a broad range of productivity. Proc Natl Acad Sci USA 102:18052–18056
Peñuelas J, Ogaya R, Boada M, Jump AS (2007) Migration, invasion and decline: changes in recruitment and forest structure in a warming-linked biome shift in Catalonia (NE Spain). Ecography 30:830–838
Peñuelas J, Hunt JM, Ogaya R, Jump AS (2008) Twentieth Century changes of tree-ring δ13C at the southern range-edge of Fagus sylvatica: increasing water-use efficiency does not avoid the growth decline induced by warming at low altitudes. Glob Change Biol 14:1076–1088. doi:10.1111/j.1365-2486.2008.01563.x
Peñuelas J, Canadell JG, Ogaya R (2011) Increased water-use efficiency during the 20th Century did not translate into enhanced tree growth. Glob Ecol Biogeogr 20:597–608. doi:10.1111/j.1466-8238.2010.00608.x
Phipps RL, Whiton JC (1988) Decline in long-term growth trends of white oak. Can J For Res 18:24–32
Qi Z, Liu H, Wu X, Hao Q (2015) Climate-driven speedup of alpine treeline forest growth in the Tianshan Mountains, Northwestern China. Glob Change Biol 21:816–826. doi:10.1111/gcb.12703
R Core Team (2015). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. http://www.R-project.org/
Rinn F (2003) TSAP-Win: time Series Analysis and presentation for dendrochronology and related applications. Version 0.55 user reference. Heidelberg. http://www.rimatech.com
Rubino DL, McCarthy BC (2000) Dendroclimatological analysis of white oak (Quercus alba L., Fagaceae) from an old-growth forest of southeastern Ohio, USA. J Torrey Bot Soc 127:240–250
Salzer MW, Hughes MK, Bunnb AG, Kipfmueller KF (2009) Recent unprecedented tree-ring growth in bristlecone pine at the highest elevations and possible causes. Proc Natl Acad Sci USA 106:20348–20353
Sano M, Furuta F, Kobayashi O, Sweda T (2005) Temperature variations since the mid-18th century for western Nepal, as reconstructed from tree-ring width and density of Abies spectabilis. Dendrochronologia 23:83–92
Schickhoff U (2005) The upper timberline in the Himalayas, Hindu Kush and Karakorum: a review of geographical and ecological aspects. In: Broll G, Keplin B (eds) Mountain ecosystems: studies in treeline ecology. Springer, Berlin, pp 275–354
Schickhoff U, Bobrowski M, Böhner J, Bürzle B, Chaudhary RP, Gerlitz L, Heyken H, Lange J, Müller M, Scholten T, Schwab N, Wedegärtner R (2015) Do Himalayan treelines respond to recent climate change? An evaluation of sensitivity indicators. Earth Syst Dyn 6:245–265. doi:10.5194/esd-6-245-2015
Sharma E, Tsering K (2009) Climate change in the Himalayas: the vulnerability of biodiversity. Sustain Mt Dev 55:10–12
Sharma K, Moore B, Vorosmarty C (2000) Anthropogenic, climatic, and hydrologic trends in the Koshi basin, Himalaya. Clim Change 47:141–165
Shrestha BB, Ghimire B, Lekhak HD, Jha PK (2007) Regeneration of treeline birch (Betula utilis D. Don) forest in a Trans-Himalayan dry valley in central Nepal. Mt Res Dev 27:259–267. doi:10.1659/mrdd.0784
Shrestha UB, Gautam S, Bawa KS (2012) Widespread climate change in the Himalayas and associated changes in local ecosystems. PLoS ONE 7:1–10. doi:10.1371/journal.pone.0036741
Spence LA, Liancourt P, Boldgiv B, Petraitis PS, Casper BB (2015) Short-term manipulation of precipitation in Mongolian steppe shows vegetation influenced more by timing than amount of rainfall. J Veg Sci. doi:10.1111/jvs.12349
Spiecker H, Mielikaeinen K, Kohl M, Skovsgaard P (eds) (1996) Growth trends in European forests: studies from 12 countries. Research report, European Forest Institute No. 5. Springer, Berlin
Stainton JDA (1972) Forests of Nepal. New York
Stokes MA, Smiley TL (1968) An introduction to tree-ring dating. The University of Chicago Press, Chicago, p 63
TISC (Tree Improvement and Silviculture Component) (2002) Forest and vegetation types of Nepal. TISC Document Series 105. Department of Forest, Kathmandu, Nepal
Tiwari A, Fan Z-X, Jump AS, Li S-F, Zhou Z-K (2016) Gradual expansion of moisture sensitive Abies spectabilis forest in the Trans-Himalayan zone of central Nepal associated with climate change. Dendrochronologia. doi:10.1016/j.dendro.2016.01.006
Vaganov EA, Hughes MK, Kirdyanov AV, Schweingruber FH, Silkin PP (1999) Influence of snowfall and melt timing on tree growth in subarctic Eurasia. Nature 400(6740):149–151. doi:10.1038/22087
Wang SY, Yoon JH, Gillies RR, Cho C (2013) What caused the winter drought in western nepal during recent years? J Clim 26:8241–8256. doi:10.1175/JCLI-D-12-00800.1
Wardle P (1974) Alpine timberlines. In: Lives JD, Barry RG (eds) Arctic alp environments. Methuen, London, pp 371–402
Weiner J, Thomas SC (2001) The nature of tree growth and the age-related decline in forest productivity. Oikos 94:374–376
Xu J, Grumbine RE, Shrestha A, Eriksson M, Yang X, Wang Y, Wilkes A (2009) The melting Himalayas: cascading effects of climate change on water, biodiversity, and livelihoods. Conserv Biol 23:520–530. doi:10.1111/j.1523-1739.2009.01237.x
Yang B, Kang XC, Liu JJ, Brauning A, Qin C (2010) Annual temperature history in Southwest Tibet during the last 400 years recorded by tree rings. Int J Climatol 30:962–971
Yang B, He M, Melvin TM, Zhao Y, Briffa KR (2013) Climate control on tree growth at the upper and lower treelines: a case study in the Qilian Mountains Tibetan Plateau. PLoS ONE 8(7):e69065
Zobel DB, Singh SP (1997) Himalayan forests and ecological generalizations. Bioscience 47:735–745
Acknowledgements
This research was supported by National Natural Science Foundation of China (NSFC) (U1502231). We thank two anonymous reviewers for their constructive comments. The authors are grateful to KP Sharma, Mr. Raju Bista, Binod Tulachan, Chandra, and Sagar for their contribution to field management and sample collection. We would also like to thank Bharat Babu Shrestha, Uttam Babu Shrestha, Su Tao, Li Shu Feng, Shalik Ram Sigdel, Julie Lebreton Anberree, Deepak Kharal, and Shankar Panthi for their guidance and support. Finally, we acknowledge Department of National Parks and Wildlife Reserve Government of Nepal and the Annapurna Conservation Area Project (ACAP, Nepal) for providing permission to carry out the field work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Jan Wunder.
Rights and permissions
About this article
Cite this article
Tiwari, A., Fan, ZX., Jump, A.S. et al. Warming induced growth decline of Himalayan birch at its lower range edge in a semi-arid region of Trans-Himalaya, central Nepal. Plant Ecol 218, 621–633 (2017). https://doi.org/10.1007/s11258-017-0716-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-017-0716-z