Abstract
We studied the soil seed bank in a possible scenario of fire regime shift and asked: (1) Does high fire frequency impact the density of seeds stored, species richness and evenness? (2) Overall, does high fire frequency produce changes in the presence–absence and abundance of species? The study was implemented in a Mediterranean Basin ecosystem in plots with increasing fire frequency (unburned, burned once and burned twice in the last 66 years). The number of seeds increased with fire frequency for all life forms (shrub, scrub, perennial forb, annual forb and perennial graminoid). Species richness of annual forbs also increased. Evenness of shrubs diminished because the number of seeds in all the species decreased, except C. albidus, which increased. Overall, differences in the abundance of species were found, mainly by depleting shrubs and increasing forbs. There were no differences in the presence–absence data. In conclusion, high fire frequencies act as a filtering factor for species of a larger size and advanced maturity age. In contrast, life forms of small size and rapid onset of reproductive maturity can be enhanced. This community conversion from woody to herbaceous soil seed banks is fundamental to identify vegetation changes in future regimes of high fire frequency.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Studies on soil seed banks are fundamental for predicting potential changes in vegetation composition in response to environmental changes and recurrent disturbances (Harper 1977). Mediterranean ecosystems have been typically considered resilient to fire, and disturbances are often essential for maintaining them in optimal diversity conditions (Faivre et al. 2011). This vegetation, however, is not adapted to fire per se, but to a particular fire regime (Keeley et al. 2011). Species that exhibit good performance under a particular regime can be threatened if it changes. Fire regime is a complex concept that includes five different components: (1) frequency; (2) fire intensity and severity; (3) fuel consumption and fire spread patterns; (4) burn patch size and distribution; (5) fire seasonality (Gill 1973; Keeley 2009). Nowadays, the abrupt shift of fire regimes in many fire-prone areas as a result of human activity and ongoing climate change is a major concern (Westerling et al. 2006; Pitman et al. 2007; Pausas and Fernández-Muñoz 2012). These changes may threaten ecosystem stability and severely impact species persistence (Lavorel et al. 2007).
Species composition in fire-prone areas is driven mainly by the interaction of persistence mechanisms and life history traits of species with fire regime (Bradstock et al. 1997; Pausas 1999). Disturbance–persistence mechanisms in Mediterranean ecosystems consist mainly of three different strategies: (1) resprouting from persistent biomass (resprouters); (2) recruiting new individuals from a persistent seed bank (seeders); (3) combining both strategies (facultative resprouters, Keeley 1986). Resprouters persist by burying their resprouting structures or by protecting them with specialised structures. In contrast, seeder species have mainly hard seed coats that enable seeds to resist fire temperatures (Keeley et al. 2011). This coat is also responsible for physical dormancy, which permits seed accumulation in soil seed banks during the periods between disturbances (Clemente et al. 2007). Seeder species may outlive parent plants by means of persistent soil seed banks until new disturbances or favourable conditions appear (Noble and Slatyer 1980). In fact, many of these species present germination promoted by disturbance-related cues that produces a flush of germination and establishment in initial post-fire stages (Santana et al. 2012). Possession of these persistence mechanisms is responsible chiefly for the resilience observed in Mediterranean vegetation.
Human activities and ongoing climate change have synergistically promoted a fire regime shift in the Mediterranean Basin (Pausas and Fernández-Muñoz 2012). Fire frequency has increased in the last few decades and forecasts predict further future accentuation (Moriondo et al. 2006). Consequently, the regenerative capacity of species may be compromised. Depletion effects have been observed in some resprouting species after consecutive fires (Delitti et al. 2005). However, obligate seeders are probably the most sensitive functional group, and can even undergo extinction locally. These species may fail to successfully replenish their seed bank if the inter-fire interval is shorter than the time taken to reach first reproduction. In the Mediterranean Basin, such local extinctions have been clearly documented for species with a canopy seed bank; for example, Pinus halepensis. This species needs an inter-fire interval of at least 15 years to develop a sufficient seed bank (Eugenio and Lloret 2006; Santana et al. 2010). In addition, previous studies on aboveground vegetation have described that consecutive fires can modify vegetation composition to practically eliminate obligate seeder shrubs with persistent soil seed banks (Zedler et al. 1983; Haidinger and Keeley 1993; Vilà-Cabrera et al. 2008). However, above-ground vegetation does not generally represent the whole community composition since most Mediterranean Basin species are sheltered as propagules in soil seed banks (Paula et al. 2009). Therefore, direct studies that focus on seed banks are required to understand if obligate seeder communities are vulnerable to altered fire frequency (Ooi 2012). Frequent fires may lead to losses of species diversity and can drive ecosystems towards more simplified compositions dominated by short life span, fast growth and early reproductive life forms (Santana et al. 2010; Keeley and Brennan 2012).
In this paper, we assess the effect of fire regime shift in driving seed bank composition in Mediterranean ecosystems dominated by obligate seeders. For this purpose, we selected nine plots with a history of increasing fire frequency (i.e. unburned, burned once and burned twice). We checked the responsiveness of the soil seed bank by assessing changes in (1) the amount of seeds stored, and (2) species richness and evenness. At the community level, we also checked significant changes in composition by a multivariate analysis for both (3) the presence–absence and (4) abundance of species. Species were classified according to their life form and resprouting ability (i.e. obligate seeders and facultative resprouters). We hypothesised that high fire frequency can filter those species of larger size, slower growth and higher reproductive maturity age. In contrast, life forms of small size and fast reproductive maturity would be enhanced under this regime. We also hypothesised that obligate seeders would be more affected than facultative resprouters because their persistence depends exclusively on accumulating a seed bank during inter-fire intervals. In the Mediterranean Basin, seeders are the most abundant functional group and their persistence may prove fundamental for preserving the diversity and functioning of these ecosystems (Paula et al. 2009).
Methods
Study sites
The soil seed bank was assessed at three sites in inland areas of the Valencia region (SE Spain), namely: Onil (38°39′05.20″N–0°39′46.65″W), Pardines (38°40′20.28″N–0°39′20.45″W) and Ayora (39°06′55.56″N–0°56′55.29″W). In all cases, the study sites were old field terraces abandoned ca. 60–70 years ago, and have a well-documented history of exploitation and fire occurrence (Santana et al. 2010). Their altitude ranges between 900 and 1,050 m a.s.l., and their climate is typically Mediterranean. Mean annual rainfall ranges between 466 mm (Onil) to 537 mm (Ayora). There is a pronounced summer drought from June to August, with usually no more than 65 mm of rainfall. The mean annual temperature is ~14 °C, and the mean maximum temperature for the hottest month (July) is 30 °C. To minimise inter-site environmental variability, all sites were oriented to the north and located on marls. Soils are Calcaric cambisols (Anon 1988).
Each site consisted of three areas that had been burned at different frequencies in the last few decades. Specifically at each site, there were areas that have remained unburned since their abandonment (No-Fire), areas burned once (1-Fire) and areas burned twice (2-Fires). Approximately, 66 years have elapsed without fire in the No-Fire areas until the sampling date (Fig. 1). The 1-Fire areas have resulted from the various wildfires that occurred in 1984 in Onil and Pardines, and in 1979 for Ayora. Thus, 22–27 years have elapsed since the occurrence of fire to the sampling date. Finally, the 2-Fires areas for Onil and Pardines proceeded from two different experimental fires performed in 1994 over part of the 1-Fire areas (see Baeza et al. 2002 for more details). Similarly, part of the area burned once in Ayora was burned again by another wildfire in 1996. Therefore, the 2-Fires areas had an inter-fire interval lasting 10–17 years between the first and second fire, and lasting 10–12 years since the second fire to the sampling date (Fig. 1).
Little is known about the fire frequency regime in the study area. A fire cycle of 49 years (i.e. time to burn an area equal to all the burnable land) has been suggested for the whole Valencia region (Pausas and Fernández-Muñoz 2012). Abdel-Malak and Pausas (2006) calculated a 22-year fire cycle for the Ayora site. This ad hoc experiment, therefore, allows us to assess possible seed bank shifts under future regimes of increasing fire frequency and short inter-fire intervals.
The areas with a different fire frequencies were located no farther than 500 m from one another at each site. We assumed that they shared the same soil and climatic characteristics. At the onset of the study, the vegetation in the No-Fire areas consisted mainly of woodlands dominated by P. halepensis as a result of the natural colonisation of abandoned fields. The 1-Fire areas were dominated by shrublands of Rosmarinus officinalis, while the 2-Fires areas consisted of a mixed shrubland of Cistus albidus and Ulex parviflorus. A complete description of the aboveground vegetation can be found in Santana et al. (2010). This study focuses on the response of species with a significant soil seed bank; i.e. shrub, scrub and herbaceous species (see below). P. halepensis was not considered because it forms a canopy seed bank. Obligate resprouters such as Quercus ilex, Quercus coccifera and Juniperus oxycedrus were also present, but they were very scarce and they do not form persistent soil seed banks. The nomenclature follows de Bolòs et al. (1993).
Soil seed bank sampling
Soil samples were taken in mid-September 2006 after the seed dispersal of summer and before germination during the wet season (autumn). A 10 m × 10 m plot was laid out within each area, and 20 points within the plots were randomly chosen from a 1 m × 1 m grid. Finally, one soil core (7 cm in diameter and 5 cm deep) was collected at each random point to determine seed bank composition. When present, leaf litter was carefully removed before each sampling. The seeds included within the litter were not included in our study because they were negligible. Throughout the study, 180 soil samples were collected. Soil samples were stored under dry conditions for a few days until they were prepared for germination. Seed bank composition was assessed indirectly in a glasshouse by monitoring seedling emergence under optimal conditions for germination. Initially, soil samples were sieved and washed through meshes of 5 and 0.2 mm to remove coarse and fine fractions. Consequently, the species whose seeds were smaller than 0.2 mm were not included in this study. Soil samples were spread evenly onto plastic trays (7 cm × 5 cm × 5 cm) filled with sterilised sand. The layer of soil in the trays did not exceed a 1-cm depth in any case. Ten trays filled only with sterilised sand were used to detect seed contamination, although none was detected. Trays were kept moist by a micro-sprinkler irrigation system that watered samples every 2–3 days. Emerging seedlings were counted regularly, identified and removed. Unidentified taxa were transplanted to pots until the growth and flowering stages to allow their identification. Germination was monitored over 20 months (from October 2006 to June 2008). Most of the species stored in the Mediterranean seed bank were physically dormant; i.e. they have a seed coat that avoids embryo imbibition. Therefore in order to detect these dormant seeds, trays were kept dry and exposed to summer temperature fluctuations during the summer months of 2007 (July, August and September). Santana et al. (2013a) observed that these summer soil temperature fluctuations are a good germination cue for Mediterranean Basin species with physical dormancy. After disconnecting the glasshouse temperature control, the mean maximum temperatures reached values of over 45 °C during the summer months of 2007 (°C, Mean ± SD: July 52.3 ± 6.9; August 51.7 ± 2.4; September 45 ± 5.3). However, it is worth noting that the seed bank can be slightly underestimated because it is likely that seed dormancy was not broken in all the seeds. In any case, this treatment would allow to detect the most part of seeds sampled; there are not Mediterranean Basin species which depend exclusively on fire-related cues for breaking their seed dormancy (i.e. high temperature or smoke), although in some cases can enhance germination (Luna et al. 2007; Santana et al. 2013a).
Data analysis
The detected species were classified into two functional groups according to their resprouting ability: obligate seeders (OS) or facultative resprouters (F). The two groups were then subdivided into life forms as follows: obligate seeders into shrubs, scrub, perennial forbs and annual forbs, and facultative species into shrubs + scrubs, perennial graminoids and perennial forbs. Shrubs and scrubs were considered together in the facultative group because of their low number. The scrubs group included all the dwarf woody plants (shorter than 50 cm in height). Species classification was done following the database of Paula et al. (2009) and personal observations (see Electronic Supplementary Material 1, ESM1, for the complete list of species and classification).
For each functional trait (resprouting ability and life forms), we analysed: (1) the total amount of seeds; (2) species richness and (3) evenness in response to fire frequency. We calculated Pielou’s evenness index (J = H/log (S); where H is the Shannon’s diversity index and S is species richness; Alday et al. 2009). For this purpose, we used generalised linear mixed models (GLMM) by considering fire frequency (No-fire, 1-Fire and 2-Fires) as a fixed factor and site (Onil, Pardines and Ayora) nested, with plot as a random factor to account for spatial autocorrelation. Each model was performed twice by changing its baseline to compare the three fire frequencies between them. The data counting models (total amount of seeds and species richness) were fitted using a Poisson error distribution and a log-link function (Crawley 2007).
The effect of fire frequency on community composition was tested by permutational multivariate analysis of variance (PMAV, the “adonis” function; Oksanen 2011). These analyses were first performed for all the species and then by discerning between obligate seeders and facultative resprouters. Quantitative shifts on the soil seed bank were tested using the species-abundance log-transformed (log(x + 1)) matrix. The aim was to curb the influence of the more abundant species. For the data analysis, the 20 subsamples from the same site and fire recurrence were pooled to reduce the spatial heterogeneity of the seed banks. In a second analysis, the qualitative shifts in the species composition were analysed using the species presence–absence matrix. In the quantitative and qualitative analyses, Bray-Curtis and Jaccard distances were respectively used with 999 permutations. The spatial homogeneity of variances was checked in all the PMAV analyses using the “betadisper” function and the data met all the requisites. The overall differences between fire frequencies (P > 0.05) and all the pair-wise comparisons were tested by adjusting the level of significance to P ≤ 0.017 between contrasts (Bonferroni correction; Sokal and Rohlf 1995). Finally, non-metric multidimensional scaling (NMDS, the “metaMDS” function with the Bray-Curtis and Jaccard similarity matrix; Oksanen 2011) was used to observe the seed bank compositional differences produced by fire frequency in the ordination space. To help the interpretation, the standard deviational ellipses of each fire frequency were then used to illustrate the position of each frequency on the biplots (the “ordiellipse” function; Oksanen 2011).
All the analyses were performed using the packages “lme4” (GLMM, Bates et al. 2011) and “vegan” (evenness and all the multivariate analyses, Oksanen et al. 2011) in the R statistical environment (version 2.14.2., Development Core Team 2012, Vienna, Austria).
Results
Number of seeds, species richness and evenness
The seed bank size included 4,578 germinated seeds of 62 species corresponding to an overall seed density of 6,693 ± 3,122 seeds m−2 (mean ± SD, n = 3). However, seed densities varied considerably between fire frequencies with 1,272 ± 488 seeds m−2 for No-Fire, 4,377 ± 1,177 seeds m−2 for 1-Fire and 14,430 ± 8,489 seeds m−2 for 2-Fires.
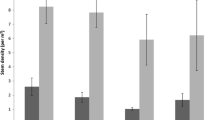
The number of seeds significantly increased with fire frequency for both OS and F (Table 1; Fig. 2a). However, OS were globally more abundant than F (6,251 ± 2,612 vs. 168 ± 95, Fig. 2a). In addition, most of the life forms composing OS (shrubs, scrubs and annual forbs) significantly increased with fire frequency, while only perennial graminoids increased significantly in F (Fig. 2a and ESM2 for a detailed GLMM response of functional groups to fire frequency).
Seed number (a), species richness (b) and evenness (c) for life forms with a different regeneration strategy (OS = Obligate seeder, F = Facultative resprouter). Columns and error bars show mean ± SE (n = 3). Different letters show significant differences among sites with different fire recurrence at P < 0.017
Of the 62 species detected in this study, 41 were OS and 21 were F (see ESM1 for the complete list of species). Fire frequency affected species richness for OS but not F (Table 1; Fig. 2b). This increase was mainly due to annual species since the rest of the life forms did not change with fire frequency (Fig. 2b). In contrast, species richness in F was not affected by fire frequency (Fig. 2b).
Fire frequency did not produce significant differences in obligate seeders evenness. However, differences were found if they were split into life forms. Shrubs evenness significantly decreased with fire frequency (Table 1; Fig. 2c), but annual forbs and perennial graminoids increased significantly (Fig. 2c). However for F, fire frequency only increased significantly evenness in perennial graminoids (Fig. 2c).
Community composition
Fire frequency affected species composition when the abundance of all species was considered (PMAV P = 0.022) and accounted for 43 % of variance in the species data. Specifically, these significant differences were between No-Fire and 2-Fires (PMAV, R 2 = 0.29, P < 0.01; Table 2). NMDS ordination (stress: 0.05; Fig. 3a) and SD-ellipses, based on species abundance, clearly showed that the three levels of fire frequency occupied different regions of the ordination space along axis 1. No-Fire was located on the right of axis 1 (+ve); 2-Fires was displayed on the left of axis 1 (−ve), showing an obvious separation from No-Fires; finally, 1-Fires was centred between these two extremes and considerably overlapped them, showing an intermediate composition. The same pattern was observed when analysing OS and F independently (Table 2). In contrast, the presence–absence data gave no significant differences when all the species were considered together (PMAV, R 2 = 0.29, P = 0.123; Table 2). NMDS ordination (stress: 0.08; Fig. 3b) and SD-ellipses, based on the presence–absence data, clearly showed this lack of difference with the three levels of fire frequency overlapping around the origin.
Non-metric multidimensional scaling (NMDS) ordination of seed bank plots with different fire recurrence in Mediterranean Basin ecosystems (stress value = 0.05). a Plots and distribution of fire occurrences expressed as bivariate-deviational ellipses (95 % confidence intervals) using a species-abundance matrix (quantitative analysis), b plots and distribution of fire occurrences expressed as bivariate-deviational ellipses (95 % confidence intervals) using the presence–absence data (qualitative analysis), c most frequent species using a species-abundance matrix (quantitative analysis). Species code: Aar Anagallis arvensis, Agr Arenaria grandiflora, Bre Brachypodium retusum, Cma Centaurea mariolensis, Cpu Cerastium pumilum, Cse Chaenorhinum serpyllifolium, Cal Cistus albidus, Ccl Cistus clusii, Dgl Dactylis glomerata, Dpe Dorycnium pentaphyllum, Fer Fumana ericoides, Gva Galium valentinum, Hap Helianthemum apenninum, Hci Helianthemum cinereum, Hsy Helianthemum syriacum, Hfi Helictotrichon filifolium, Hpe Hornungia petraea, Omi Ononis minutissima, Prh Papaver rhoeas, Pse Plantago sempervirens, Psp Poa sp., Rph Reseda phyteuma, Rva Reseda valentina, Rof Rosmarinus officinalis, Ss1 Silene sp.1, Ste Sonchus tenerrimus, Sof Stipa offneri, Upa Ulex parviflorus
After analysing the dominant species independently, the results showed different behaviours within the same functional group (OS and F; Table 3). In obligate seeder shrubs, C. albidus was the only dominant species whose density increased with fire frequency. Other species, such as Cistus clusii, R. officinalis and U. parviflorus, presented a maximum in 1-Fire, but decreased in 2-Fires (Table 3). The scrub Helianthemum cinereum increased with fire frequency, while Fumana ericoides decreased. In F, the perennial graminoid Dactylis glomerata increased with fire frequency, but Helictotrichon filifolium presented a maximum in 1-Fire (Table 3). The NMDS species plot for the species-abundance data (Fig. 3c) reflected these trends. Those species with increasing abundance in 2-Fires occupied the left hand part of axis 1 (+ve). Specifically, species such as Arenaria grandiflora, Galium valentinum, Cerastium pumilum, Hornungia petraea (annual and perennial forbs) and D. glomerata (perennial graminoid) were found there. The species presenting a maximum in 1-Fire or No-Fire were either centred on the origin or occupied the right hand part of the axis. For example, we found obligate seeder shrubs (C. clusii, R. officinalis, and U. parviflorus) and the scrub F. ericoides.
Discussion
Based on the evidence presented here, fire regime shifts in the form of short-fire intervals have the capacity to modify soil seed banks in Mediterranean Basin ecosystems. Changes in species composition and density are clearly observed between unburned areas and areas burned twice in the last few decades (with a 10–17-year inter-fire interval), while areas which have suffered only one fire exhibit intermediate compositional and density values between these two extremes. This shift mainly results from a significant increase in perennial and annual forbs and perennial graminoids in burned areas, all in detriment to shrub species. These results, thus, support our hypothesis of high fire frequency being a filtering factor for species of larger size, slower growth and higher age of reproductive maturity (i.e. shrubs such as U. parviflorus and R. officinalis). In contrast, life forms of a small size and fast reproductive maturity are enhanced (i.e. annual and perennial forbs such as H. petraea and A. grandiflora, and perennial graminoids such as D. glomerata). This community conversion from woody to herbaceous soil seed banks is consistent with the patterns observed in the aboveground vegetation in Mediterranean Basin ecosystems showing similar shifts after frequent fires (Delitti et al. 2005; Vilà-Cabrera et al. 2008; Santana et al. 2010, 2013b).
As expected in fire-prone ecosystems (Paula et al. 2009), OS are dominant in the soil seed bank under study. In addition, the abundance of all OS life forms is modulated by fire frequency. In contrast, the F group is quite scarce and less affected by fire frequency. Only perennial graminoids significantly increase with fire recurrence. In fact, this supports the hypothesis that OS sustain more impact than F because their persistence depends exclusively on filling their seed banks during inter-fire intervals.
Indeed, the seed density of shrub species increased with fire frequency, but this is the result of an increase in only one species, C. albidus. The remaining shrub species were depleted, which clearly leads to diminished evenness with fire frequency (Fig. 2c). Probably, the time to reach reproductive maturity and to successfully fill the seed bank in C. albidus is shorter than the known inter-fire interval (10–17 years between fires). Roy and Sonié (1992) observed that C. albidus reproductive maturity begins 1–2 years after its establishment and that its full reproductive capacity is reached about 5 years later. The increase in C. albidus may also be stimulated by the disturbance-promoted germination of this species (Santana et al. 2012, 2013a). However, it is worth noting that the remaining obligate seeder shrub species (U. parviflorus, R. officinalis and C. clusii) also display disturbance-promoted germination, and that they were not enhanced, but depleted. This failure is interesting because a fitness advantage for these species in case of frequent fires has been proposed (Pausas et al. 2012). These species are highly flammable and their disturbance-promoted germination suggests a co-evolution that benefits them in high fire frequencies (Saura-Mas et al. 2010). Our finding of seed bank changes contradicts the idea that these species benefit from high fire frequencies. In the present study, with fire frequencies of 10–17-year inter-fire intervals, these species may fail to replenish their soil seed bank, which compromises their regenerative capacity and can lead to local extinction. In line with this, Vilà-Cabrera et al. (2008) observed in a hot-spot of fire frequency (de Creus Cape, NE Iberian Peninsula) that the cover of obligate seeder shrubs decreased with fire frequency intervals, and that even C. albidus drastically diminished after four fires over three decades. However, it should be taken into account that the immaturity risk (i.e. the inter-fire interval is shorter than the time taken to reach the first reproduction) may not be the only factor that explains the seed bank patterns in this study. There were different times since fire for each fire frequency (ca. 66 years for No-Fire, 22–27 years for 1-Fire and 10–12 years for 2-Fires), and both species and seed longevity can also influence the soil seed bank dynamics. Individuals of many species may become senescent and die with time, leading to a subsequent decline in seed output. This decline may be especially important for herbs and forbs, which are short-lived like their seeds.
The depletion of woody plants by fire opens up resources that are exploited by other life forms to help them grow and fill their seed bank. Annual, perennial forbs and perennial graminoids can benefit from declining competition with perennial vegetation. These species are capable of maturing in year 1 or 2 after the disturbance, thus allowing them to successfully fill their seed bank. Perennial graminoids may also be promoted by their ability to resprouting capacity, and by the fact that their sexual reproduction is enhanced during year 1 post-fire (e.g. Brachypodium retusum and Ampelodesmos mauritanica; Caturla et al. 2000; Vilà et al. 2001). Here, our findings follow this pattern and clearly illustrate how fire regimes with short inter-fire intervals may potentially modify the balance of the soil seed bank from shrub-dominated to herb-dominated communities. Similar patterns with changes from woody to herbaceous systems have been observed in Californian chaparral (Zedler et al. 1983; Haidinger and Keeley 1993), even with significant invasions on non-native herbs (Keeley and Brennan 2012). In Australian ecosystems, short inter-fire intervals can also lead to biodiversity losses and to extirpations of some species (Bradstock et al. 1995).
One interesting result is that short-fire intervals shift the species composition in the ecosystems studied. Nonetheless, they affect only species abundance and do not promote significant species extinctions. This is followed by non-significant losses of species richness. Soil seed banks in Mediterranean Basin ecosystems may, therefore, be considered resistant to species loss in the fire frequency regime studied. However, further studies over longer periods of time and experiencing high fire frequencies would be needed to confirm this resistance. These results contrast with canopy seed banks species such as P. halepensis, which became completely extinct in the inter-fire interval studied (Eugenio and Lloret 2006; Santana et al. 2010). This resistance to extinction may be conferred by the bet-hedging strategy used by many species (especially shrubs and scrubs with physical dormancy). A reservoir of seeds remains ungerminated in soil after the post-disturbance flush of germination, thus buffering the probability of extinction if two consecutive fires occur (Auld and Denham 2006). In fact, the high fire frequency studied is far from negative, but enhances the species richness and evenness of some functional groups, mainly annual forbs and perennial graminoids. These species have a short-term persistent seed bank that can suffer depletions if the fire interval exceeds their life span (Wright and Clarke 2009). Otherwise, it is worth to note that fire frequency is not the only factor modulating seed bank composition in this work. The effect of fire frequency (No-Fire, 1-Fire and 2-Fires) is also mingled with vegetation age; i.e. experimental plots also differed in time since the last fire (ca. 66, 22–27 and 10–12 years).
Finally, further research which takes into account soil seed banks is needed to monitor threats in ecosystem biodiversity. Seed banks shelter the majority of species in Mediterranean ecosystems from disturbances (Paula et al. 2009) and such approaches can preclude failures in detecting losses of species that are not present in aboveground vegetation. Despite the strong soil seed bank resistance to increasing fire frequency observed in these ecosystems, this threat may be accentuated if this frequency increases with shorter inter-fire intervals. In addition, the persistence of soil seed banks in future climatic conditions can sustain the impact of the interaction of other complex mechanisms beyond altered fire regimes. Rising temperatures will amplify the temperature fluctuations taking place in bare soils after disturbances. This will release the physical dormancy of many seeds which remain ungerminated, thus compromising the efficiency of the bet-hedging mechanism (Ooi 2012). At the same time, higher air temperatures may also increase seedling mortality by increasing soil moisture evaporation with the subsequent net loss of seeds from seed banks and species richness (del Cacho et al. 2012). The inclusion of these factors in future soil seed bank experiments in fire-prone areas shall be fundamental to identify the inter-fire intervals thresholds and the relative importance of those factors limiting the potential recovery of seed bank species.
References
Abdel-Malak D, Pausas JG (2006) Fire regime and post-fire normalized difference vegetation index changes in the eastern Iberian península (Mediterranean basin). Int J Wildland Fire 15:407–413
Alday JG, Martinez-Ruiz C, Bravo F (2009) Evaluating different harvest intensities over understory plant diversity and pine seedlings, in a Pinus pinaster Ait. natural stand of Spain. Plant Ecol 201:211–220
Anon (1988) Soil map of the world. FAO-UNESCO, Revised legend. World Soil resources. FAO Report 60, Rome
Auld TD, Denham AJ (2006) How much seed remains in the soil after a fire? Plant Ecol 187:15–24
Baeza MJ, De Luis M, Raventós J, Escarré A (2002) Factors influencing fire behaviour in shrublands of different stand ages and the implications for using prescribed burning to reduce wildfire risk. J Environ Manag 65:199–208
Bates D, Maechler M, Bolker B (2011) lme4: Linear mixed-effects models using S4 classes. R package version 0.999375-42. http://CRAN.R-project.org/package=lme4
Bradstock RA, Keith DA, Auld TD (1995) Fire and conservation: imperatives and constraints on managing for diversity. In: Bradstock RA, Auld TD, Keith DA, Kingsford RT, Lunney D, Silversten DP (eds) Conserving biodiversity: threats and solutions. Surrey Beatty and Sons, Sydney, pp 323–333
Bradstock RA, Tozer MG, Keith DA (1997) Effects of high frequency fire on floristic composition and abundance in a fire-prone heathland near Sydney. Aust J Bot 45:641–655
Caturla RN, Raventós J, Guàrdia R, Vallejo VR (2000) Early post-fire regeneration dynamics of Brachypodium retusum Pers. (Beauv.) in old fields of the Valencia region (eastern Spain). Acta Oecol 21:1–12
Clemente AS, Rego FC, Correia OA (2007) Seed bank dynamics of two oblígate seeders, Cistus monspeliensis and Rosmarinus officinalis, in relation to time since fire. Plant Ecol 190:175–188
Crawley MJ (2007) The R Book. Wiley, Chichester
de Bolòs O, Vigo J, Masalles RM, Ninot JM (1993) Flora manual dels Paisos Catalans. Portic, Barcelona
del Cacho M, Saura-Mas S, Estiarte M, Peñuelas J, Lloret F (2012) Effect of experimentally induced climate change on the seed bank of a Mediterranean shrubland. J Veg Sci 23:280–291
Delitti W, Ferran A, Trabaud L, Vallejo VR (2005) Effects of fire recurrence in Quercus coccifera L. shrublands of the Valencia Region (Spain): I. plant composition and productivity. Plant Ecol 177:57–70
Eugenio M, Lloret F (2006) Effects of repeated burning on Mediterranean communities of the northeastern Iberian Peninsula. J Veg Sci 17:755–764
Faivre N, Roche P, Boer MM, McCaw L, Grierson PF (2011) Characterization of landscape pyrodiversity in Mediterranean environments: contrast and similarities between south-western Australia and south-eastern France. Landsc Ecol 26:557–571
Gill AM (1973) Effects of fire on Australia’s native vegetation. CSIRO, Division of Forest Research, Canberra
Haidinger TL, Keeley JE (1993) Role of high fire frequency in destruction of mixed chaparral. Madrono 40:141–147
Harper JL (1977) Populations biology of plants. Academic Press, London
Keeley JE (1986) Resilience of Mediterranean shrub communities to fires. In: Dell B, Hopkins AJM, Lamont BB (eds) Resilience in Mediterranean-type ecosystems. Dr W. Junk Publishers, Dordrecht, pp 95–112
Keeley JE (2009) Fire intensity, fire severity and burn severity: a brief review and suggested usage. Int J Wildland Fire 18:116–126
Keeley JE, Brennan TJ (2012) Fire-driven alien invasion in a fire-adapted ecosystem. Oecologia 169:1043–1052
Keeley JE, Pausas JG, Rundel PW, Bond WJ, Bradstock RA (2011) Fire as an evolutionary pressure shaping plant traits. Trends Plant Sci 16:406–411
Lavorel S, Flannigan MD, Lambin EF, Scholes MC (2007) Vulnerability of land systems to fire: interactions among humans, climate, the atmosphere, and ecosystems. Mitig Adapt Strateg Glob Chang 12:33–53
Luna B, Moreno JM, Cruz A, Fernández-González F (2007) Heat-shock and seed germination of a group of Mediterranean plant species growing in a burned área: an approach based on plant functional types. J Exp Bot 60:324–333
Moriondo M, Good P, Durao R, Bindi M, Giannakopoulos C, Corte-Real J (2006) Potential impact of climate change on fire risk in the Mediterranean. Clim Res 31:85–95
Noble IR, Slatyer RO (1980) The use of vital attributes to predict successional changes in plant communities subject to recurrent disturbances. Vegetatio 43:5–21
Oksanen J (2011) Multivariate Analysis of Ecological Communities in R: vegan tutorial. http://cc.oulu.fi/~jarioksa/opetus/metodi/vegantutor.pdf. Accessed 18 March 2014
Oksanen J, Blanchet F, Kindt R, Legendre P, O’Hara R, Simpson G, Solymos P, Stevens M, Wagner H (2011) Vegan: Community Ecology Package. R package version 1.17-8. http://CRAN.R-project.org/package=vegan. Accessed 18 March 2014
Ooi MKJ (2012) Seed bank persistence and climate change. Seed Sci Res 22:S53–S60
Paula S, Arianoutsou M, Kazanis D, Tavsanoglu Ç, Lloret F, Buhk C, Ojeda F, Luna B, Moreno JM, Rodrigo A, Espelta JM, Palacio S, Fernández-Santos B, Fernandes PM, Pausas JG (2009) Fire-related traits for plant species of the Mediterranean Basin. Ecology 90:1420
Pausas JG (1999) Response of plant functional types to changes in the fire regime in Mediterranean ecosystems: a simulation approach. J Veg Sci 10:717–722
Pausas JG, Fernández-Muñoz S (2012) Fire regime changes in the Western Mediterranean Basin: from fuel-limited to drought-driven fire regime. Clim Chang 110:215–226
Pausas JG, Alessio G, Moreira B, Corcobado G (2012) Fires enhance flammability in Ulex parviflorus. New Phytol 193:18–23
Pitman AJ, Narisma GT, McAnemey J (2007) The impact of climate change on the risk of forest and grassland fires in Australia. Clim Chang 84:383–401
Roy J, Sonié L (1992) Germination and population dynamics of Cistus species in relation to fire. J Appl Ecol 29:647–655
Santana VM, Baeza MJ, Marrs RH, Vallejo VR (2010) Old-field secondary succession in SE Spain: can fire divert it? Plant Ecol 211:337–349
Santana VM, Baeza MJ, Maestre FT (2012) Seedling establishment along post-fire succession in Mediterranean shrublands dominated by obligate seeders. Acta Oecol 39:51–60
Santana VM, Baeza MJ, Blanes MC (2013a) Clarifying the role of fire heat and daily temperature fluctuations as germination cues for Mediterranean Basin obligate seeders. Ann Bot Lond 111:127–134
Santana VM, Baeza MJ, Marrs RH (2013b) Response of woody and herbaceous fuel to repeated fires in Mediterranean gorse shrublands. Int J Wildland Fire 22:508–514
Saura-Mas S, Paula S, Pausas JG, Lloret F (2010) Fuel loading and flammability in the Mediterranean Basin woody species with different post-fire regenerative strategies. Int J Wildland Fire 19:783–794
Sokal RR, Rohlf FJ (1995) Biometry. W.H. Freeman and Co., New York
Vilà M, Lloret F, Ogheri E, Terradas J (2001) Positive fire-grass feedback in Mediterranean Basin woodlands. For Ecol Manag 147:3–14
Vilà-Cabrera A, Saura-Mas S, Lloret F (2008) Effects of fire frequency on species composition in a Mediterranean shrubland. Ecoscience 15:519–528
Westerling AL, Hidalgo HG, Cayan DR, Swetnam TW (2006) Warming and earlier spring increase western US forest wildfire activity. Science 313:940–943
Wright BR, Clarke PJ (2009) Fire, aridity and seed banks. What does seed bank composition reveal about community processes in fire-prone desert? J Veg Sci 20:663–674
Zedler PH, Clayton RG, McMaster GS (1983) Vegetation change in response to extreme events: the effect of a short interval between fires in California chaparral and coastal scrub. Ecology 64:809–818
Acknowledgments
We thank Segundo Ríos for his help in seedling identification. V. M. Santana has been supported by a VAli+d post-doctoral grant awarded by the Generalitat Valenciana. J. G. Alday has been supported by Basque Country Government post-doctoral grant (DEUI BFI-2010-245). We would like to thank again the reviewers and the Editor for the time dedicated to our manuscript and for their numerous, constructive and helpful comments provided. This research has been carried out as part of the RESILIEN (CGL 2011-30515-C02-02) and the Consolider-Ingenio 2010 (GRACCIE CSD2007-00067) projects. CEAM is supported by the Generalitat Valenciana.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Neal J. Enright.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Santana, V.M., Alday, J.G. & Baeza, M.J. Effects of fire regime shift in Mediterranean Basin ecosystems: changes in soil seed bank composition among functional types. Plant Ecol 215, 555–566 (2014). https://doi.org/10.1007/s11258-014-0323-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-014-0323-1