Abstract
The questions “Will the environment surrounding moorlands become refugia for a Japanese subalpine coniferous species, Abies mariesii Mast., after climate change?” and “How does the spatial resolution of a species distribution model affect the global warming predictions?” have been discussed in this study. This study was conducted at Hakkoda Mountains, the northern side of Honshu Island, Japan. We constructed 50-m mesh model using a climate variable, two topography variables and two variables relating to moorlands. We applied the model to eight global warming scenarios, including decreasing or non-decreasing scenarios of moorlands. We also constructed a coarse-resolution model at approximately 1-km resolution and compared the model predictions with the fine ones. The results showed that the coarse-resolution model tended to overestimate the range of suitable habitats for A. mariesii. On the other hand, some suitable habitats around moorlands could only be predicted by the fine-resolution model. The fine-resolution model indicated that the peripheries of the moorlands are the most important potential refugia for A. mariesii on Hakkoda Mountains. Although these suitable areas were notable in the +2°C scenario, all suitable habitats completely disappeared in the +4°C scenario. We concluded that it would be effective to conserve the A. mariesii populations around moorlands which are likely to persist after global warming, as well as moorlands themselves. This assessment could only be achieved by fine-resolution models that incorporate non-climatic variables including topography and moorland-related variables with climatic variables. In contrast, a coarse-resolution model overestimated the suitable habitats whilst underestimating potential local refugia. Thus, fine-resolution models are more effective for developing practical adaptation of conservation measures.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Recent global warming is strongly affecting terrestrial ecosystems by shifting the ranges of plant and animal species poleward and upward (IPCC 2007). An increasing number of studies on the upward shift of the range of plants in alpine ecotones have been conducted in Europe and North America (Beckage et al. 2008; Lenoir et al. 2008). Climate change is expected to decrease or even eliminate the habitats of alpine and subalpine plants because these plants are isolated on high mountains (Horikawa et al. 2009). Thus, these plants are particularly sensitive to global warming (Smith et al. 2009).
Many studies have predicted the effects of climate change on the distributions of wild plants and vegetation (Araújo et al. 2005; Huntley et al. 1995; Iverson and Prasad 1998; Thuiller et al. 2005). However, most of these predictions are based on coarse-grained (e.g. 50 km) climate surfaces or idealized scenarios with uniform warming, which fail to consider spatially heterogeneous warming at local and landscape scales (Ashcroft et al. 2009). Trivedi et al. (2008) also suggested that recent large-scale modelling studies may overestimate the ability of montane plant species to adapt to global warming because the input climate data had coarse resolution and were biased against their cold, high altitude habitats. Finer resolution studies in Japan have used a Japanese grid coding system, referred to as ‘Third Mesh’ in Japan, to predict potential refugia for some dominant species (e.g. stone pine: Horikawa et al. 2009; beech-dominant forests: Matsui et al. 2009; dwarf bamboo: Tsuyama et al. 2011). Each of Third Mesh cells measures 30″ latitude by 45″ longitude (~1 × 1 km2), which was defined by the Geospatial Information Authority of Japan (Japan Map Center 1998). However, because elevational variation in a 1-km cell is significantly large in Japanese complex and precipitous mountainous areas, the variance of the temperature in a 1-km cell must be large. Therefore, 1-km resolution (i.e. using one representative value per 1-km cell) is still insufficient for practical conservation management including adaptive measures at a regional or local scale. In addition, the influences of geographical factors, such as topography, are hardly ever incorporated (Matsui et al. 2004b). In general, higher resolution spatial analysis is more realistic. However, at a certain point, high-resolution analysis becomes difficult due to constraints in the availability of high-resolution data. Nevertheless, since global warming is inevitable (IPCC 2007), high-resolution predictions at a regional or local scale are needed to develop practical adaptive measures.
In this study, we predicted the effect of global warming on a subalpine coniferous species, Abies mariesii, on Hakkoda Mountains, Japan. According to Nogami (1994), who estimated the changes in vegetation zones according to a warmth index (WI) (Kira 1977), a temperature increase of 2°C would reduce the area of Japanese subalpine coniferous forests to about a quarter of their current range. This drastic change is due to the lower elevation of the Japanese mountains compared with the other continents, which limits the upward shift of the trees. Consequently, it is important to determine the effects of global warming on these trees to develop conservation measures.
Abies mariesii is the dominant subalpine coniferous species in subalpine areas of Honshu Island. Since A. mariesii has extended its distribution into the moorland below its lower elevation range limit (Yamanaka et al. 1988), moorlands may be local refugia for A. mariesii in a warmer climate. A fine-resolution model is needed to verify this type of refugia. We determine potential refugia as the locations where species would survive after climate change according to fine-resolution species distribution models. We address the following questions: (1) Can the peripheries of the moorlands be potential refugia for A. mariesii? (2) Is the fine-resolution model effective to predict such local refugia?
Methods
Study area and distribution data
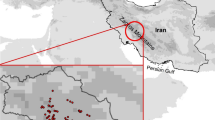
The study area was 20 × 15 km2 and extended across the Hakkoda Mountains that are south of Aomori City in Aomori Prefecture, Japan (latitude: 40°33′ to 40°44′ and longitude: 140°47′ to 140°57′) (Fig. 1). The elevation of this area ranged from 183 to 1,585 m. Japanese beech (Fagus crenata), a deciduous broadleaf tree, is the dominant tree species up to about 900 m. A. mariesii is the dominant tree species in the subalpine zone at elevations above 1,000 m. The Japanese stone pine (Pinus pumila) grows at elevations over 1,400 m. Many patches of moorlands are found in depressions of volcanic ash from a massive eruption in the Pleistocene epoch (Koike et al. 2005; Murach and Ulrich 1988). The Hakkoda Mountains have some of the deepest snow in the world. The maximum snow depth, annual mean temperature and annual precipitation at the Sukayu station (Fig. 1) are 3–5 m, 4–6°C and 1,300–2,300 mm, respectively (Japan Meteorological Agency; http://www.jma.go.jp/jma/indexe.html). Most of the land above 700 m has been designated as a national park, although there are also a few farms or tree plantations (Fig. 2a). Human impact on the natural vegetation has been minimal, particularly for the subalpine forests.
Horizontal and vertical distribution of presence cells (a dotted polygons, b black dots) and absence cells (a outside of the black dotted polygons, b grey dots) for A. mariesii based on aerial photographs. The number of presence or absence cells was 106 and 120 at Third Mesh resolution, respectively, and 18,200 and 70,975 at the 50-m mesh resolution, respectively. a The brown colour shows the topography of the study area; darker shades indicate higher elevations. Hatched areas depict the areas of farms or tree plantations. (Color figure online)
Abies mariesii is a subalpine coniferous species endemic to Japan that has adapted to habitats with heavy snowfall (Kaji 1982; Sugita 1990). Although A. mariesii was not dominant during the last glacial period, when snowfall was light, its distribution has expanded since snowfall began to increase in the Hypsithermal period (Morita 1985). Since other subalpine coniferous species gradually became extinct, many subalpine forests in Japan, particularly those in snowy regions, now consist only of A. mariesii. Therefore, if local extinction of A. mariesii occurs on Hakkoda Mountains, no other subalpine coniferous species could replace it. However, the distribution of A. mariesii also extends into the moorlands at lower elevations (Yamanaka et al. 1988), suggesting that these sites could be refugia in warmer climates.
We created a distribution map of A. mariesii on Hakkoda Mountains (Hakkoda A. mariesii map, HAmap: Fig. 2a) based on aerial photographs taken in October 1996 and September 2003 as detailed in Online Resource S1. We transformed the HAmap to presence absence data at 50-m mesh and Third Mesh resolutions. We excluded the areas of farms or tree plantations (Fig. 2a) from them and used as the response variables at each scale.
Environmental variables for the Third Mesh model
Although plant distribution is mainly affected by temperature and soil moisture content, Japanese trees usually do not suffer serious water deficits because there is sufficient rainfall throughout most of Japan. Therefore, since temperature is the most important factor for plant distribution in Japan (Nogami 1994), the WI proposed by Kira (1977) is frequently used in studies of Japanese plants. WI, defined as the annual sum of positive differences between monthly mean temperatures and +5°C, is a measure of the effective warmth for plants during their growing season. Summer (May–September) precipitation (PRS) is an index of the water supply during the growing season. Likewise, winter (December–March) precipitation (PRW) is an index of the accumulation of snow in cold regions. Third Mesh model was constructed only by WI (33.5–71.5), PRS (599–1,155 mm) and PRW (548–1,009 mm) because, at this resolution, topography was not described meaningfully (Matsui et al. 2004b). Current climatic data for Third Mesh cells were obtained from the Japan Meteorological Agency (1996), which provides monthly climatic temperature and precipitation for 1953–1982 and 1953–1976, respectively.
Environmental variables for the 50-m mesh model
In the 50-m mesh model, WI was the only climatic variable, which was based on downscaled temperatures from the Third Mesh climatic data sets as followed (Online Resource S2 Fig. 5). Temperature data at the Third Mesh resolution (Japan Meteorological Agency 1996) were downscaled to 50-m mesh resolution by using the method of Zimmermann and Kienast (1999). In brief, we first calculated the monthly linear lapse rates at the study area, based on the average monthly temperature and elevation data at four meteorological stations (Fig. 1) Second, the elevation and temperature data at Third Mesh resolution were resampled to 50-m mesh resolution and smoothed with a circular filter having a 450 m (nine 50-m mesh cells) radius, respectively (i.e. Temp3rd and Elev3rd). Finally, we calculated 50-m mesh resolution temperature data (Temp50) for each month according to the following equation:
where Temp3rd is the temperature at 50-m mesh resolution derived from Third Mesh temperature, L is lapse rates of the month, Elev50 is the 50-m mesh elevation data obtained from the Digital elevation model (DEM) (Geographical Survey Institute 2000) and Elev3rd are the elevation at 50-m mesh resolution derived from Third Mesh DEM. We did not use precipitation data in the 50-m mesh model because a method for downscaling them has not been established yet (Charles et al. 2007) and predicting future precipitation has high uncertainty (Mearns et al. 1997).
In addition to the WI, we used two topographical variables, slope and aspect, calculated from the Elev50. To test our working hypothesis that the peripheries of the moorlands can be potential refugia for A. mariesii, we added the Euclidean distance from each cell to the nearest moorlands as the explanatory variable in the 50-m mesh model. In this procedure, the current distribution of the moorlands was identified in the same way as the HAmap (Online Resource S3 Fig. 6a). According to our pre-analysis using the variables mentioned above, high omission errors occurred inside the moorlands. Thus, we added another explanatory variable that indicates the location inside or outside the moorlands. Namely, WI (29.1–76.3), slope (0°–48°), aspect (eight categorical data), distance from moorland (50–7,900 m) and moorland P/A (presence/absence) were used in the 50-m mesh model. These variables were generated by using the Spatial Analyst tools in ArcGIS version 9.2 (ESRI, Redlands, USA).
Modelling framework
To construct a species distribution model, we used a classification tree (CT) (Clark and Pregibon 1992), a binary partitioning algorithm that recursively splits a data set into subsets based on a single predictor variable, which is chosen to minimize the deviance of the response variables, until each subset is relatively homogeneous. The advantages of CT include (De’ath and Fabricius 2000) (1) the ability to use different types of response variables; (2) the capacity for interactive exploration, description, and prediction; (3) invariance to transformations of explanatory variables and (4) straightforward graphical interpretation of complex results involving interactions.
The most appropriate tree size in CT was obtained after cross-validation to avoid overfitting or underfitting the model (Clark and Pregibon 1992). Deviance weighted scores (DWS), defined as the sum of the reduction of deviance between parent nodes and the children nodes generated by each predictor variable (Matsui et al. 2004a), were calculated to evaluate the contribution of each predictor variable to the model. For these analyses, we used R 2.8.0 (R Development Core Team 2008).
The area under the curve (AUC) value, which was derived from the receiver-operating characteristic (ROC) analysis (Metz 1978), was used for validating model performance. AUC was calculated according to bootstrap methods (Efron 1979). That is, we compared the predicted presence absence data with validation data that randomly selected from the study data with replacement for 100 times and calculated the mean and SD values of AUC. AUC values range between 0.5 (the model has no discrimination ability) and 1.0 (perfect discrimination) (Zweig and Campbell 1993). Although AUC has been commonly used for validating SDMs, some problems were pointed out (e.g. Lobo et al. 2008) including that the approach tend to overestimate the model fit because spatial autocorrelation inherently exists between the training and validation data sets (Morin and Thuiller 2009).
ROC analysis can also provide useful information for selecting an appropriate probability threshold by describing the trade-off between correctly predicting the occurrence of a species (true positive) and incorrectly predicting the presence of the species (false positive) (Pearce and Ferrier 2000). Pearson et al. (2004) stressed the importance of minimizing the number of sites with observed presence predicted as being unsuitable, and proposed three response categories: suitable, marginal and unsuitable habitats. We divided the predicted potential habitats into the following three categories defined by Tsuyama et al. (2008), which are similar to those of Pearson et al. (2004); (1) suitable habitats, defined as the areas where the predicted probability of occurrence was greater than the probability threshold; (2) marginal habitats, defined as the areas with a probability of occurrence greater than or equal to 0.01 but less than the probability threshold and (3) non-habitats, defined as areas that were neither suitable nor marginal.
Climate change scenarios
We analysed four temperature increase scenarios at the Third Mesh and 50-m mesh resolutions. Each scenario only differed in the increase of the daily average temperature of each month (+1, +2, +3 or +4°C). Because future climate scenarios based on general circulation model simulations have too coarse spatial resolutions (ca. at 100–450-km resolutions) to use for a local study, we did not use them. To make more realistic predictions in the 50-m mesh model, we prepared two moorland scenarios, namely fixed and decreased. In the fixed scenario, we assumed that the current distributions of moorlands do not change during climatic warming. In contrast, the decreased scenario modelled reductions in moorland distribution during climatic change. We developed the latter scenario by constructing a CT model of moorlands as detailed in Online Resource 3. As a result, we modelled eight future scenarios for the 50-m mesh model and four future scenarios for the Third Mesh model.
Results
Distribution data of A. mariesii
The study area was divided into 294 cells of the Third Mesh and 117,261 cells of the 50-m mesh resolution. There were 106 presence cells and 120 absence cells at the Third Mesh resolution and 18,200 presence cells and 70,975 absence cells at the 50-m mesh resolution. The areas of farms or tree plantations that were excluded from analysis comprised 68 cells of the Third Mesh and 28,086 cells of the 50-m mesh resolution (Fig. 2a).
Performance of the Third Mesh and 50-m mesh models of A. mariesii
The CT diagram for A. mariesii based at the Third Mesh resolution had three terminal nodes (TNs) (Fig. 3a). The mean and standard deviation for the AUC value of the CT were 0.966 and 0.0119, respectively. The AUC value was considered to be ‘excellent’ according to the criteria established by Swets (1988) and Thuiller et al. (2003). The ROC analysis indicated that the optimal threshold probability was 0.689. The WI variable accounted for all of the DWS.
Diagrams of the CT for A. mariesii at the Third Mesh (a) (~1 × 1 km2) and 50-m mesh (b) resolutions. The conditions, occurrence probabilities, and number of Third Mesh cells (n) are shown at all nodes. If the condition is met, the left branch is followed; otherwise the right branch is followed. The length of the vertical lines below each true–false split corresponds to the change in the magnitude of deviance between parent and children nodes. a Three TNs are labelled A–C. The WI was used as an explanatory variable; however, other climatic variables were not included in this model. b Nine TNs are labelled A–I. The WI, slope, aspect, distance from moorland and moorland presence/absence (P/A) were used as explanatory variables; however, other environmental variables were not included in this model
The CT diagram for A. mariesii at the 50-m resolution had nine TNs (Fig. 3b). The mean and standard deviation for the AUC value of the CT were 0.930 and 0.000768, respectively, which was considered to be ‘excellent’ (Swets 1988; Thuiller et al. 2003). The optimal threshold probability was 0.384. The DWS were as follows: WI (86.8%), slope (5.4%), aspect (3.0%), distance from moorlands (2.4%) and moorland P/A (presence/absence) (2.3%).
Current potential habitats and their changes with climate change scenarios
For the current climate (Fig. 4a), the Third Mesh model showed higher probabilities of occurrence and larger suitable and marginal habitats than those of the 50-m mesh model. The suitable habitats for A. mariesii were predicted to shift to higher altitudes and be divided into two populations in the northern and southern areas (Fig. 4b–d). In the +1°C scenario (Fig. 4b), the Third Mesh model predicted larger suitable habitats than those of the 50-m mesh model. In the +2°C scenario (Fig. 4c), the 50-m mesh model predicted some areas of suitable habitats at TN D that were outside those of the Third Mesh model. These areas were located in the vicinity of the moorlands. In the +3°C scenario (Fig. 4d), the summits of high mountains were predicted to be suitable habitats by both models. However, all suitable habitats completely disappeared in both models in the +4°C scenario.
Predicted distributions of potential habitats for A. mariesii at the 50-m mesh and Third Mesh resolutions in the current climate (a) and future climate change scenarios [+1°C (b), +2°C (c), and +3°C (d)] with fixed (top) and decreased (bottom) moorland scenarios. The key in the lower left corner shows the type of habitat, occurrence probability, and area corresponding to the habitat for each TN at the Third Mesh resolution (Fig. 3a) and the 50-m mesh resolution (Fig. 3b). For the 50-m mesh resolution, the habitats are shown in different colours. For the Third Mesh resolution, the habitats corresponding to TNs A and B are hatched or solid, respectively, whilst that for TN C covers the remaining area. (Color figure online)
We compared the areas of potential habitats predicted by the 50-m mesh model in the fixed and decreased moorland scenarios (Fig. 4; Table 1). In the +2°C scenario, the area of suitable habitat for A. mariesii was predicted to be 126.1 ha in the fixed scenario and 76.4 ha in the decreased scenario (Table 1). In the +1, +2 and +3°C scenarios, the suitable area in the fixed scenario was larger than the decreased scenario by 26.9 ha (8.63%), 49.7 ha (39.4%) and 0.3 ha (20%), respectively. In the +3 and +4°C scenarios, the suitable areas were predicted to become very small in both moorland scenarios (0.0–1.5 ha).
Discussion
Effects of environmental variables on the distribution of A. mariesii
WI was the only explanatory variable in the Third Mesh model (Fig. 3a) because neither PRS nor PRW were effective. This may have been the result of the limited sample size in this model that prevented any significant difference from being found in the presence or absence cells. Another possible explanation is that the precipitation data at regional scales were less accurate than those at the Japanese Archipelago scale.
In the 50-m mesh model, the most important explanatory variable was also WI. In the CT (Fig. 3b), the first divergence at WI = 46.5 was the most important and this value is in close agreement with that estimated by Nogami and Ohba (1991) (WI = 47) for the boundary between the distributions of A. mariesii and beech (F. crenata) in the Japanese Archipelago scale.
The second most important explanatory variable was slope. Our result that gentler slopes were associated with higher distribution probabilities (Fig. 3b) may be related to snow gliding, the process by which snow on a slope moves along the ground. Snow gliding, which occurs on open slopes with inclinations >15° (Leitinger et al. 2008), causes great pressure on trees. The divergence of the slope variable in the 50-m mesh model was 17.5°, which suggests that snow gliding pressure affects the distribution of A. mariesii.
The third most important explanatory variable in the 50-m mesh model was aspect (Fig. 3b). Our result that the distribution probability was lower on the east slope may be due to the detrimental effect of snowdrifts, which are formed by winter monsoons that approach from the northwest. A. mariesii cannot survive the extreme pressure of a snowdrift because it cannot adopt a creeping growth form (Shidei 1956). In addition, the growth periods of A. mariesii shorten in areas with deep snow deposits.
Finally, the suitable habitats of TN D in the CT for the 50-m mesh model (Fig. 3b) merits discussion. The location of these habitats suggested that the peripheries of the moorlands are potential refugia for A. mariesii on Hakkoda Mountains, even if it is located in the warm area where beech (F. crenata) dominates. Previously, Yamanaka et al. (1988) suggested that if A. mariesii did not have any competitors at lower elevations, it might have extended its range. In fact, A. mariesii has a higher tolerance for the perhumid soil environment of the moorland than beech (Sugita 1992), which would enable A. mariesii to thrive around the moorland even at higher WI values. Murach and Ulrich (1988) observed a similar relationship between the European beech (F. sylvatica) and Norway spruce (Picea abies) and showed that the root growth of F. sylvatica is much more sensitive to low pH than that of P. abies. Thus, the growth of beech trees is strongly inhibited by acid mineral soils (Marschner 1991). In our study, the periphery of the moorlands prevented the growth of beech trees and consequently provided the potential habitats for A. mariesii.
Resolution of species distribution models
The estimates of the probability of occurrence by Third Mesh model were greater than those of the 50-m mesh model, particularly in the current climate (Fig. 4a). Coarse-resolution models are susceptible to this type of overestimation because cells with even a small area of occurrence of a species are treated as presence cells. On the other hand, the Third Mesh model partly underestimated the suitable habitats of A. mariesii, especially in the +2°C scenario (Fig. 4c). In this scenario, some predicted suitable habitats of TN D in the 50-m mesh model were located outside those of the Third Mesh model (Fig. 4c). The fine-resolution models could involve the effects of topography and some very local environmental gradients like the peripheries of the moorlands, which enabled us to detect local refugia that coarse-resolution models have overlooked. The Third Mesh model also underestimated the locations near the summit (Fig. 4d). Coarse-resolution models tend to represent the altitude of the summit lower than the fine-resolution ones, causing cool areas to be overlooked as suitable habitats. Incorporating various factors into fine-resolution models according to each species, more potential refugia may be found.
Although most subalpine areas in Japan are protected as nature conservation areas, their fragile ecosystems are very sensitive to climate warming (Nogami 1994; Tanaka et al. 2009). One of the most important adaptation measures for these ecosystems would be to protect the refugia. Coarse-resolution models may not be able to address such issues on a regional or local scale.
Abbreviations
- AUC:
-
Area under curve
- CT:
-
Classification tree
- DEM:
-
Digital elevation model
- DWS:
-
Deviance weighted scores
- HAmap:
-
Hakkoda Abies mariesii map
- PRS:
-
Summer (May–September) precipitation
- PRW:
-
Winter (December–March) precipitation
- ROC:
-
Receiver-operating characteristics
- TN:
-
Terminal node
- WI:
-
Warmth index
References
Araújo MB, Thuiller W, Williams PH, Reginster I (2005) Downscaling European species atlas distributions to a finer resolution: implications for conservation planning. Glob Ecol Biogeogr 14:17–30
Ashcroft MB, Chisholm LA, French KO (2009) Climate change at the landscape scale: predicting fine-grained spatial heterogeneity in warming and potential refugia for vegetation. Glob Change Biol 15:656–667
Beckage B, Osborne B, Gavin DG, Pucko C, Siccama T, Perkins T (2008) A rapid upward shift of a forest ecotone during 40 years of warming in the Green Mountains of Vermont. PNAS 105:4197–4202
Charles SP, Bari MA, Kitsios A, Bates BC (2007) Effect of GCM bias on downscaled precipitation and runoff projections for the Serpentine catchment, Western Australia. Int J Climatol 27:1673–1690
Clark LA, Pregibon D (1992) Tree-based models. In: Chambers JM, Hastie TJ (eds) Statistical models in S. Wadsworth & Brooks/Cole Advanced Books & Software, Pacific Grove, pp 377–419
De’ath G, Fabricius KE (2000) Classification and regression trees: a powerful yet simple technique for ecological data analysis. Ecology 81:3178–3192
Efron B (1979) Bootstrap methods: another look at the jackknife. Ann Stat 7:1–26
Geographical Survey Institute (2000) Digital map 50 m grid (elevation). Geographical Survey Institute, Tsukuba
Horikawa M, Tsuyama I, Matsui T, Kominami Y, Tanaka N (2009) Assessing the potential impacts of climate change on the alpine habitat suitability of Japanese stone pine (Pinus pumila). Landsc Ecol 24:115–128
Huntley B, Berry PM, Cramer W, McDonald AP (1995) Special paper: modelling present and potential future ranges of some European higher plants using climate response surfaces. J Biogeogr 22:967–1001
IPCC (2007) In: Parry ML, Canziani OF, Palutikof JP, Linden PJ, Hanson CE (eds) Climate Change 2007: impacts, adaptation and vulnerability. Contribution of Working Group II to the fourth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge, p 976
Iverson LR, Prasad AM (1998) Predicting abundance of 80 tree species following climate change in the eastern United States. Ecol Monogr 68:465–485
Japan Map Center (1998) Numerical map user guide, 2nd version. Japan Map Center, Tokyo
Japan Meteorological Agency (1996) Climate normals for Japan. Japan Meteorological Agency, Tokyo
Kaji M (1982) Studies on the ecological geography of subalpine conifers: distribution pattern of Abies mariesii in relation to the effect of climate in the postglacial warm period. Bull Tokyo Univ For 72:31–120
Kira T (1977) A climatological interpretation of Japanese vegetation zone. In: Miyawaki A, Tuexen R (eds) Vegetation science and environmental protection. Maruzen, Tokyo, pp 21–30
Koike K, Toshikazu T, Chinzei K, Miyagi T (2005) Regional geomorphology of Japanese Islands, geomorphology of Tohoku region, vol 3. University of Tokyo Press, Tokyo
Leitinger G, Höller P, Tasser E, Walde J, Tappeiner U (2008) Development and validation of a spatial snow-glide model. Ecol Model 211:363–374
Lenoir J, Gegout JC, Marquet PA, de Ruffray P, Brisse H (2008) A significant upward shift in plant species optimum elevation during the 20th century. Science 320:1768–1771
Lobo JM, Jiménez-Valverde A, Real R (2008) AUC: a misleading measure of the performance of predictive distribution models. Global Ecol Biogeogr 17:145–151
Marschner H (1991) Mechanisms of adaptation of plants to acid soils. Plant Soil 134:1–20
Matsui T, Nakaya T, Yagihashi T, Taoda H, Tanaka N (2004a) Comparing the accuracy of predictive distribution models for Fagus crenata forests in Japan. Jpn J For Environ 46:93–102
Matsui T, Yagihashi T, Nakaya T, Taoda H, Yoshinaga S, Daimaru H, Tanaka N (2004b) Probability distributions, vulnerability and sensitivity in Fagus crenata forests following predicted climate changes in Japan. J Veg Sci 15:605–614
Matsui T, Takahashi K, Tanaka N, Hijioka Y, Horikawa M, Yagihashi T, Harasawa H (2009) Evaluation of habitat sustainability and vulnerability for beech (Fagus crenata) forests under 110 hypothetical climatic change scenarios in Japan. Appl Veg Sci 12:328–339
Mearns LO, Rosenzweig C, Goldberg R (1997) Mean and variance change in climate scenarios: methods, agricultural applications, and measures of uncertainty. Clim Change 35:367–396
Metz CE (1978) Basic principles of ROC analysis. Semin Nucl Med 8:283–298
Morin X, Thuiller W (2009) Comparing niche- and process-based models to reduce prediction uncertainty in species range shifts under climate change. Ecology 90:1301–1313
Morita Y (1985) The vegetational history of the subalpine zone in northeast Japan II. The Hachimantai mountains. Jpn J Ecol 35:411–420
Murach D, Ulrich B (1988) Destabilization of forest ecosystems by acid deposition. GeoJournal 17:253–259
Nogami M (1994) Thermal condition of the forest vegetation zones and their potential distribution under different climates in Japan. J Geogr 103:886–897
Nogami M, Ohba H (1991) Japanese vegetation seen from warmth index. Kagaku 61:39–49
Pearce J, Ferrier S (2000) Evaluating the predictive performance of habitat models developed using logistic regression. Ecol Model 133:225–245
Pearson RG, Dawson TP, Liu C (2004) Modelling species distributions in Britain: a hierarchical integration of climate and land-cover data. Ecography 27:285–298
R Development Core Team (2008) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Shidei T (1956) A view on the cause of the lack of coniferous forest zone in subalpine area on some mountains in the Japan sea side. J Jpn For Soc 38:356–358
Smith W, Germino M, Johnson D, Reinhardt K (2009) The altitude of alpine treeline: a bellwether of climate change effects. Bot Rev 75:163–190
Sugita H (1990) Consideration on the history of the development of the Abies mariesii forest during postglacial time based on its distributional character. Jpn J Hist Bot 6:31–37
Sugita H (1992) Ecological geography of the range of the Abies mariesii forest in northeast Honshu, Japan, with special reference to the physiographic conditions. Ecol Res 7:119–132
Swets JA (1988) Measuring the accuracy of diagnostic systems. Science 240:1285–1293
Tanaka N, Nakazono E, Tsuyama I, Matsui T (2009) Assessing impact of climate warming on potential habitats of ten conifer species in Japan. Glob Environ Res 14:153–164
Thuiller W, Araújo MB, Lavorel S (2003) Generalized models vs. classification tree analysis: predicting spatial distributions of plant species at different scales. J Veg Sci 14:669–680
Thuiller W, Lavorel S, Araújo MB, Sykes MT, Prentice IC (2005) Climate change threats to plant diversity in Europe. PNAS 102:8245–8250
Trivedi MR, Berry PM, Morecroft MD, Dawson TP (2008) Spatial scale affects bioclimate model projections of climate change impacts on mountain plants. Glob Change Biol 14:1089–1103
Tsuyama I, Nakao K, Matsui T, Higa M, Horikawa M, Kominami Y, Tanaka N (2011) Climatic controls of a keystone understory species, Sasamorpha borealis, and an impact assessment of climate change in Japan. Ann For Sci 68:689–699
Yamanaka M, Sugawara K, Ishikawa S (1988) A historical study of the Abies mariesii forest found in the montane zone in the south Hakkoda mountains, northeast Japan. Jpn J Ecol 38:147–157
Yonekura K, Kajita T (2003) BG Platns wamei-gakumei (Japanese–Latin) index (YList). http://bean.bio.chiba-u.jp/bgplants/index.html (in Japanese). Accessed 28 July 2011
Zimmermann NE, Kienast F (1999) Predictive mapping of alpine grasslands in Switzerland: species versus community approach. J Veg Sci 10:469–482
Zweig MH, Campbell G (1993) Receiver-operating characteristic (ROC) plots: a fundamental evaluation tool in clinical medicine. Clin Chem 39:561–577
Acknowledgments
The authors are grateful to K. Yonekura for advice about species identification as well as K. Hikosaka, T. Sasaki, C. Kamiyama, A. Yoshida, H. Daimaru and M. Yasuda for their valuable suggestions. This study was supported by the Global Environment Research Fund (grant Nos. F-092, S-4 and S-8) of the Ministry of the Environment, Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
Nomenclature: Yonekura and Kajita (2003).
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shimazaki, M., Tsuyama, I., Nakazono, E. et al. Fine-resolution assessment of potential refugia for a dominant fir species (Abies mariesii) of subalpine coniferous forests after climate change. Plant Ecol 213, 603–612 (2012). https://doi.org/10.1007/s11258-012-0025-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-012-0025-5