Abstract
Niche differentiation with respect to habitat has been hypothesized to shape patterns of diversity and species distributions in plant communities. African forests have been reported to be relatively less diverse compared to highly diversed regions of the Amazonian or Southeast Asian forests, and might be expected to have less niche differentiation. We examined patterns of structural and floristic differences among five topographically defined habitats for 494 species with stems ≥1 cm dbh in a 50-ha plot in Korup National Park, Cameroon. In addition, we tested for species–habitat associations for 272 species (with more than 50 individuals in the plot) using Torus translation randomization tests. Tree density and basal area were lowest in areas with negative convexity, which contained streams or were inundated during rainy periods and highest in moist well-drained habitats. Species composition and diversity varied along the topographical gradient from low flat to ridge top habitats. The low depression and low flat habitats were characterized by high diversity and similar species composition, relative to slopes, high gullies and ridge tops. Sixty-three percent of the species evaluated showed significant positive associations with at least one of the five habitat types. The majority of associations were with low depressions (75 species) and the fewest with ridge tops (8 species). The large number of species–habitat associations and the pronounced contrast between low (valley) and elevated (ridgetop) habitats in the Korup plot shows that niche differentiation with respect to edaphic variables (e.g., soil moisture, nutrients) contributes to local scale tree species distributions and to the maintenance of diversity in African forests.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Understanding the processes that generated and continue to maintain high levels of species diversity in tropical forests is a central challenge to community ecology (Hubbell and Foster 1986; Gentry 1988; Hubbell 2001; Wright 2002; Condit et al. 2005). A long-standing hypothesis for the maintenance of diversity within species-rich forests is that resource-based niche differentiation and other mechanisms of habitat specificity allow large numbers of species to coexist within communities (Ricklefs 1977; Webb and Peart 2000; Harms et al. 2001; Hardy and Sonké 2004; Potts et al. 2004; Valencia et al. 2004; Gunatilleke et al. 2006). Species distribution patterns that are biased with respect to environmental variables would support this hypothesis (Gartlan et al. 1986; Newbery et al. 1988; Clark et al. 1999; Webb and Peart 2000; Phillips et al. 2003; Hall et al. 2004; Paoli et al. 2006).
Topographic features greatly influence local conditions, especially soil processes and hydrology, that may in turn influence the demographic processes of growth, mortality, and recruitment (Gartlan et al. 1986; Clark et al. 1999; Daws et al. 2002; Miyamoto et al. 2003). Variation in demographic performance among habitats can translate into species’ associations with their preferred habitats (Comita and Engelbrecht 2009; Russo et al. 2008; Yamada et al. 2007). Thus, resolving the role of habitat partitioning in the maintenance of high species diversity in rainforests will depend in part on detecting species–habitat associations (Webb and Peart 2000; Harms et al. 2001; Hall et al. 2004; Potts et al. 2004). Differences among species in their habitat associations, coupled with habitat heterogeneity, will contribute to the maintenance of high diversity by allowing species to coexist by specializing on different habitats. In addition, individual species–habitat associations will shape spatial patterns of forest structure, species composition and diversity, as habitats considered more conducive will serve as concentration areas for a higher number of species (Potts et al. 2004).
Comparative studies of the main tropical forest ecosystems show that African rainforests have relatively poor α-diversity compared to that of the highest diversity regions of Asia and the Americas (Parmentier et al. 2007). In contrast to this overall pattern of diversity, current understanding of the local-scale community-assembly mechanisms for tropical African tree communities is very limited and complicated by previous sampling designs. For example, inventories based on 1-ha plots spread across a wide area capture fewer than half of the local species, with many represented only by a single individual (Hall and Swaine 1981). Furthermore, most of these inventories focus on large trees with dbh ≥ 10 cm (Hall and Swaine 1981; Gartlan et al. 1986; Newbery et al. 1986; Hardy and Sonké 2004) and in some cases only include selected taxa (Hall et al. 2004). These small plots limit the identification of habitats at scales that could provide meaningful inferences on plant populations and also preclude comparisons of degrees of habitat specificity with other tropical forests.
We established a 50-ha forest dynamics research plot (FDP) in southwestern Cameroon, with the largest single plot dataset from the African continent, to document the abundance of all tree species and evaluate species–habitat associations that reflect niche partitioning as a driving force for species coexistence. The plot includes varied terrain to assess the extent to which tree species in the community have biased distributions with respect to environmental variation defined by topographic habitat types. With the first enumeration of trees completed, we ask in this article the following questions: (i) To what degree do habitats differ in forest structure and species composition? (ii) Are pockets of high or low plant diversity associated with particular habitat types? (iii) To what extent are species’ distributions associated with habitat types defined by topographic features? (iv) Are habitats with many associated species the most diverse?
Materials and methods
Study area
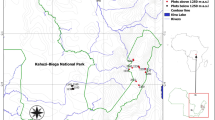
Korup National Park (5°10′ N, 8°70′ E; Fig. 1) is located in southwestern Cameroon and lies within the Guineo-Congolian forest of tropical Africa (White 1983), in a species-rich area that has been identified as a former Pleistocene refugium (Maley 1987). It constitutes one of the last remnants of the Atlantic coastal forest, i.e., la forêt biafriéene (Letouzey 1985), in an area that has been identified as a biodiversity hotspot, combining both high plant diversity with a high rate of deforestation (Küper et al. 2004). The climate of the southern part of Korup is strongly seasonal, with a single distinct dry season (average monthly rainfall < 100 mm) from December to February, followed by a long and intense wet season, with heaviest rainfall in August (Newbery et al. 1998; Chuyong et al. 2004a). Over a 17-year period from 1984 to 2001, the southern part of the park received an annual average of 5040 mm of rain (Newbery et al. 2004). The average monthly temperature ranges between 25.1 and 27.6°C and solar radiation ranges from 199 to 248 W/m2. The soils are derived from highly weathered syenite and other igneous rocks; because of the high rainfall they are skeletal and sandy at the surface, highly leached, and poor in nutrients (Newbery et al. 1998; Chuyong et al. 2002).
The 50-ha Korup FDP is located in the southern part of Korup National Park and covers an area with diverse terrain (Fig. 1). The plot is 1000 m × 500 m, with the longer axis running north–south and its elevation ranges from 150 to 240 m asl. The southern part of the plot is fairly flat, with a valley bottom that contains a permanent stream flowing westward, while the northern part is steeper, with distinct gullies and large boulders. Further description of the Korup FDP can be found in Thomas et al. (2003), Chuyong et al. (2004b), and Kenfack et al. (2007).
Plot enumeration
The first enumeration of the Korup FDP was carried out from November 1996 to June 1999, following the Center for Tropical Forest Science (CTFS) standardized methods described in Condit (1998). Initially, the 50-ha plot was surveyed and demarcated into 1,250 20 × 20 m quadrats. Elevation was measured to the nearest 0.1 m at each corner post and the value added to the absolute elevation got from the published survey map of the area. Standard geostatistical kriging procedures were then used to produce the topographic map based on a 5-m grid spanning the entire plot (see Fig. 2; kriged points at 5-m intervals were used only to produce the map for visualization purposes—all tests were conducted with the 20-m interval elevation values).
All trees ≥ 1 cm in stem diameter in each quadrat were measured, tagged with unique identification numbers, mapped to the nearest 50 cm, and sorted into morphospecies. Diameter was measured at breast height (DBH, i.e., 1.3 m above the ground) except for irregular stems (swollen and buttressed stems), in which case the point of measurement was just below the swelling, or 0.5 m above buttresses and stilt roots. Most stems were measured at breast height, so we use DBH to refer to all diameter measurements in this article. The stems were mapped relative to the 20 × 20 m grid in the field and their respective xy-coordinates were obtained by digitizing these mapped points on a Summersketch® tablet (Thomas et al. 2003).
Herbarium specimens were collected for every morphospecies. Taxonomic identification was conducted using regional floras and by matching specimens at SCA, K, MO, and YA (herbarium abbreviations following Holmgren et al. 1990). Specimens were deposited at the respective herbaria where matchings were done.
Close to 80% of the species have been confidently identified and some new to science have been described (see Sonké et al. 2002; Kenfack et al. 2003, 2004, 2006). Family classifications followed the Angiosperm Phylogeny Group (APG II, 2003). Species were grouped into four life-forms defined by their maximum attainable heights as follows; treelets/small trees (<10 m), understorey (10–20 m), lower canopy (20–30 m), and upper canopy (>30 m). Maximum attainable heights were obtained from the literature supplemented by field observations (Thomas et al. 2003).
Habitat classification
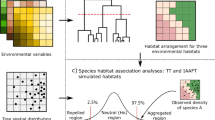
Each 20 × 20 m quadrat was assigned to a habitat category based on its topographic attributes, elevation, slope and convexity, using the methods described in Yamakura et al. (1995), Harms et al. (2001), and Valencia et al. (2004). For each quadrat, elevation was calculated as the mean of the elevation at its four corners. Slope was the mean angular deviation from the horizontal of each of the four triangular planes formed by connecting three corners at a time. Convexity was calculated as the difference between the mean elevation of the focal quadrat and the mean elevation of the eight surrounding quadrats. For edge quadrats, convexity was calculated as the difference between the elevation of the focal quadrat center and the mean elevation of the four corners. The transition from valley to slope coincides with the uppermost flood level of the stream that flows through the plot and was defined as 165 m, based on observations of high water levels. This elevation is convenient since it is close to the median elevation of the plot. Using thresholds of 165 m for mean elevation, 15° for slope (the median) and zero for convexity, the 1,250 quadrats were classified into five topographic habitats (Fig. 2) as follows: low depression (mean elevation < 165 m, slope < 15° and convexity < 0); low flat (mean elevation < 165 m, slope < 15° & convexity ≥ 0); high gully (mean elevation ≥ 165 m, slope ≥ 15°, and convexity < 0); Slopes (mean elevation ≥ 165 m, slope ≥ 15° and convexity ≥ 0); and Ridge top (mean elevation ≥ 165 m and slope < 15°).
Structural and floristic differences among habitats
To compare forest structure among habitats, stem density, and mean basal area were calculated in each 20 × 20 m quadrat and then the mean and standard deviation were calculated over quadrats assigned to each habitat type. To compare diversity among habitat types, we calculated species richness as the total number of species found in each habitat, as well as the mean number of species per 20 × 20 m quadrat. To control for differences in stem density and area among habitat types, we also calculated Fisher’s α (following the routine given in Condit 1998) for each 20 × 20 m quadrat and obtained the mean for each habitat type, as well as the overall Fisher’s α for each habitat type (i.e., all quadrats combined). Confidence limits were calculated based on variances across quadrats within the different habitat categories.
Pairwise comparisons of species stem density between habitats were used to assess inter-habitat similarity. A straight line through the origin with slope = 1 would signify that all species had consistent densities in the two habitats. We fitted no-intercept linear regressions to the log-transformed densities and used the corresponding r 2 values as similarity indices. The mean Sørensen similarity index “with cover” (Valencia et al. 2004) was also calculated for pairs of 20 × 20 m quadrats in habitats i and j for within and between-habitat comparisons in species composition. Between-habitat means were standardized as in Valencia et al. (2004), but without correction for geographic distance using:
where SORij is the mean Sørensen similarity index between quadrat pairs (one in habitat i and the other in habitat j). Since the most abundant species are often used to define forest composition (e.g., Valencia et al. 2004), the top 10 ranking species in density in each habitat were considered as dominants and used in assessing compositional differences.
Species–habitat associations
One of our primary objectives was to determine the prevalence of topographic habitat association in tree species in the Korup plot. The challenge to determining how many species and to what extent they show habitat associations is the inherent spatial autocorrelation that occurs both in species distributions and in topography, which is often characterized by large areas of similar features such as large flat areas, or slopes up to a ridge top (Condit et al. 2000; Webb and Peart 2000; Harms et al. 2001; Philips et al. 2003). Therefore, many ordinary parametric and nonparametric analyses are not appropriate and can lead to spurious associations when they assume independence among non-independent sample units (Legendre 1993). Therefore, we adopted torus-translation tests (Harms et al. 2001; Hall et al. 2004; Gunatilleke et al. 2006) to assess patterns of association between trees and our topographically defined habitats.
Our torus-translation tests incorporated the spatial structure of the trees and most of the spatial autocorrelation of the topographically defined habitats. Each test required translating the habitat map in four cardinal directions, moving the entire habitat map one column or row of 20 × 20 m quadrats at a time. The positions of the individual stems were not altered as the habitats were shifted, but the character of the habitat assigned to a given stem changed with the shifts of the habitat map. The number of individuals of each species was counted in each quadrat for each translation of the habitat map, and the relative density for each focal species in each focal habitat was calculated as the density of the focal species divided by the density of all species combined in the focal habitat, i.e., the proportion of all stems in a given habitat represented by the focal species. Each of the 1,249 unique translations of the habitat map (not including the 0,0 translation, i.e., the true habitat map) provided a new estimate of the expected relative density of each species in each habitat if species distributions had appeared on the landscape at random with respect to habitats (Harms et al. 2001).
The observed association of a species with a given habitat (on the true habitat map) was compared to the frequency distribution of expected values. In torus translation tests with a significance level of α = 0.05, significant associations are those in which the observed level of association (measured by relative density) are ≤2.5 or ≥97.5% of the expected values. However here, we only consider positive associations, and therefore our tests are essentially one-tailed tests with α = 0.025. Rare species with <1 individual/ha were excluded from the analysis, leaving a total of 272 species for which we tested for significant positive association with the five habitat types.
Results
Habitat differences in stand structure, species diversity and composition
The low depressions had the lowest stem density and high gullies had the lowest basal area, whereas the low flat habitat had the highest stem density and slopes had the highest basal area (Table 1). High stem density in the low flat habitat was mostly due to small sized individuals. High basal area on the slopes matched the higher proportion of large stemmed individuals (dbh > 60 cm) in that habitat.
Species richness varied among habitat types and declined from the lower part of the plot to the ridge top (Table 1). Based on Fisher’s α, the low flat habitat was the most diverse and the high gullies the least diverse (Table 1). Three significant diversity groups were obtained in the following decreasing order: low flat and low depression > slopes > high gully and ridge tops.
In terms of species composition, low depression and low flat areas contained similar densities of all species. This was evident in the high r 2 value (0.80) obtained from the pairwise regression of species density between the two habitats. High gullies, slopes and ridge tops were also very similar; with r 2 values ranging between 0.76 and 0.86 (see Table 2). The low habitats (low depression and low flat) differed in densities of all species relative to the high habitats (high gullies, slopes and ridge top), as indicated by low r 2 values for pairwise habitat comparisons of density (Table 2). This pattern was also evident in the disparity of the Sørensen similarity indices between the low and high habitats. The low depression and low flat were relatively similar to each other, as were high gullies, slopes and ridge tops (Table 2). The greatest contrast was between low flat areas and high gullies.
Dominant species were inconsistent across the different habitats (Table 3). For example, Phyllobotryon spathulatum, the dominant species in the low flat with 921 individuals/ha, had 144 individuals/ha and was ranked 8th in the high gully habitat; Rinorea gabunensis was dominant on the slopes with 383 individuals/ha compared to only 3 individuals/ha in the low flat habitat (ranked 177). Even so, similarities in dominant species occurred between low depression and low flat habitats. Overall, all of the 10 forest-wide dominant species had densities >50 individuals/ha in each habitat, except Rinorea lepidobotrys, which had low densities in the high gullies and slopes (Table 3).
Species–habitat associations
Of the 272 species with >50 individuals in the plot, 172 (63%) showed significant positive associations with at least one of the five habitat types (Table 4). The majority of associations were with low depressions (75 species) and the fewest with the ridge tops (8 species). Only 8 species showed multiple positive associations (even the pair of habitats with the most species positively associated with both, i.e., high gullies and slopes, had only five positively associated species in common). The 172 species that showed significant positive associations represented 63 upper canopy, 46 sub-canopy, 48 understorey, and 15 treelets species, which represent 84, 55, 43, and 7% of species in each category, respectively. Selected distribution maps illustrate some of these patterns of association (Fig. 3). For example, Beilschmiedia sp. showed strong association to the low depressions (Fig. 3a) Cola semecarpophylla and Lecomtedoxa klaineana (Fig. 3b) to low flat areas (Fig. 3). Drypetes staudtii showed strong association to both high gullies and slope. Hymenostegia afzelii appears to be a generalist in that it is relatively evenly distributed among habitats (Fig. 3C). Significant habitat association was not restricted to dominant species with relatively high densities in their preferred habitat, but included rarer species as well. For example, Homalium sarcopetalum and Rauvolfia mannii were significantly associated to the low depression but had densities <10 individuals ha−1 in that habitat.
Discussion
Forest structure, composition and diversity across topographically defined habitats
Our results reveal clear differences in forest structure, diversity, and species composition among the five topographic habitat types in the Korup forest dynamics plot. Tree density and basal area were lowest in habitats with negative convexity values (low depressions and high gullies, respectively). Such areas contain streams or are likely to be inundated during rainy periods, which reduces the area available for tree recruitment and may also lower growth and survival, resulting in fewer, smaller trees. In contrast, the habitats with the highest tree density and basal area (low flat and slopes, respectively) are both well-drained, but relatively moist areas, suggesting that such habitats are better for overall tree performance than inundated or dry habitats. This is consistent with topography-related patterns of forest structure in central Panama, where tree densities were found to be lowest in inundated (swamp) and streamside habitats and highest in low plateau and slope habitats (Harms et al. 2001).
Interestingly, patterns of diversity and composition among habitats differed from those of forest structure. Diversity per quadrat was highest in low-lying habitats, despite the low tree density in low depressions, and decreased from slopes to high gully and ridge top habitats. Low depressions and low flat habitats also had similar species composition, which differed from the composition in steeper and elevated habitats. These trends in species composition and diversity at Korup follow the ridge-valley contrast that has been reported for other tropical forest sites in Ecuador (Valencia et al. 2004) and Sri Lanka (Gunatilleke et al. 2006). Despite clear compositional differences among those habitat groups, species that were dominant in one habitat tended to be fairly common across all habitats in the forest, suggesting that species–habitat preferences are relatively weak (i.e., growth and survival are only slightly reduced outside of the preferred habitat; Yamada et al. 2007) or that preferred habitats serve as a source of propagules for less optimal ‘sink’ habitats.
We found mixed support for our initial hypothesis that habitats with many associated species would be the most diverse. Low depressions had the most associated species, nearly twice as many as any other habitat, and also had the highest diversity per quadrat. At the other end of the spectrum, ridge tops, one of the least diverse habitats, had the fewest species associations. However, the low flat habitat, which had the highest species richness per quadrat, had fewer species associations than the less diverse slope and high gully habitats.
The observed patterns of diversity are likely related to variation in soil moisture availability among habitats. The Korup FDP receives very high annual rainfall (>5000 mm/year), but experiences a severe, 3-month dry season. The well-drained soils of the ridge top are most likely to be associated with water-stress in trees during the dry season, which may result in lower species diversity since drought-sensitive species typically have low survival in such habitats during the dry season (Comita and Engelbrecht 2009). In contrast, the lower habitats probably experience less water stress during the dry season, and may therefore support a higher diversity of trees. This is consistent with regional-scale trends of lower diversity of tropical trees in areas with lower moisture and longer seasonal droughts (Gentry 1988), and is thought to be because fewer species can physiologically tolerate stressful drought conditions. This idea is supported by our finding that very few species were associated with ridge tops, the driest habitat type. However, we also cannot rule out effects of soil nutrients, which vary spatially in the Korup plot (Chuyong, unpublished data) and can also influence tree species distributions and diversity (Newbery and Proctor 1984; Paoli et al. 2006; John et al. 2007).
Habitat associations and species coexistence in plant communities
We found that the majority of species in the Korup plot were significantly associated with at least one of the five topographic habitat types. Although partitioning at the coarse scale of habitats examined in this, and similar studies would clearly be insufficient for explaining the coexistence of the hundreds of species typically present in tropical tree communities, our results suggest that habitat partitioning plays a role in shaping species distributions and contributes to the maintenance of diversity in the Korup forest. Given the apparent variation in soil moisture of topographic habitats within the plot, the observed species–habitat associations provide indirect support for the idea of hydrologic niche partitioning (Silvertown et al. 1999; Webb and Peart 2000; Gibbons and Newbery 2003). Studies in a meadow community in Europe and a fynbos community in South Africa have revealed niche segregation along fine-scale hydrological gradients, due to a trade-off between species tolerance of aeration stress and drought stress (Silvertown et al. 1999; Araya et al. 2010). In tropical forests, experimental work has shown that tree species vary widely in their drought sensitivity (Engelbrecht and Kursar 2003), and that variation drives species distributions across both local and regional moisture gradients (Engelbrecht et al. 2007; Baltzer et al. 2008). Similarly, species habitat associations within the Korup plot may be related to species distributions at regional scales. For example, the tree species Oubanguia alata, which was associated with low depressions in the plot, is mostly limited to wet forests of the Korup-Mount Cameroon area (Chuyong et al. 2004a, b). By contrast, both Hymenostegia afzelii (Fig. 3) and Annickia affinis are widely distributed in the plot and are also common in both wet and dry forests in the region (Aubréville 1970; Versteegh & Sosef 2007).
In Korup, strong habitat associations were shown predominantly by canopy and upper canopy species. This contrasts with findings from elsewhere. At Yansuni, Ecuador, significant habitat association was shown predominantly by understory species (Valencia et al. 2004), while in the dipterocarp forest at Sinharaja, Sri Lanka, all growth forms including most of the abundant species and most of the canopy dominants were habitat specialists (Gunatilleke et al. 2006).
Of the 172 species we tested, 63% showed significant positive habitat associations based on the conservative torus-translation test of association. This proportion is similar to the 64 and 79% obtained through similar torus-translation tests for Barro Colorado Island, Panama (Harms et al. (2001) and Sinharaja FDP, Sri Lanka (Gunatilleke et al. 2006), respectively. Philips et al. (2003) also reported an exceptionally high degree of habitat specialization (nearly 80%) for relatively well-sampled species in the Amazon, the highest record to date for tropical forest studies. Differences in the proportion of species showing significant habitat associations may be due either to variation in the strength of niche partitioning or to differences in the amount of topographic heterogeneity in the landscape captured within the plot area. Thus, it is difficult to make inferences about different processes acting in different forests based solely on proportion of associated species within a plot. Nonetheless, the high proportion of species associated with habitats in the Korup plot indicates that the lower diversity of African forests is unlikely to be due to lower topographic variation or less niche partitioning in these forests relative to forests in other tropical regions.
In conclusion, similar to patterns reported for tropical forests in Asia and the Americas, we found that topographic variation drives tree species distributions on local scales and plays a significant role in shaping forest structure, species composition and diversity in this African forest plot. These results support the idea that niche partitioning contributes to the maintenance of diversity in species rich plant communities.
References
Araya YN, Silvertown J, Gowing DJ, McConway KJ, Linder HP, Midgely G (2010) A fundamental, eco-hydrological basis for niche segregation in plant communities. New Phytol. doi: 10.1111/j.1469-8137.2010si.03475.x
Aubréville A (1970) Flore du Cameroun. In: Aubréville A, Leroy J-F (eds) Légumineuses-Césalpinoïdées, vol 9. Muséum National d’Histoire Naturelle, Paris
Baltzer JL, Davies SJ, Bunyavejchewin S, Noor NSM (2008) The role of desiccation tolerance in determining tree species distributions along the Malay-Thai Peninsula. Funct Ecol 22:221–231
Chuyong GB, Newbery DM, Songwe NC (2002) Litter breakdown and mineralization in a central African rain forest dominated by ectomycorrhizal trees. Biogeochemistry 61:73–94
Chuyong GB, Newbery DM, Songwe NC (2004a) Rainfall input, throughfall and stemflow of nutrients in a central African rain forest dominated by ectomycorrhizal trees. Biogeochemistry 67:73–91
Chuyong GB, Condit R, Kenfack D, Losos E, Sainge M, Songwe NC, Thomas DW (2004b) Korup forest dynamics plot, Cameroon. In: Losos EC, Leigh EG Jr (eds) Forest diversity and dynamism: findings from a large-scale plot network. University of Chicago Press, Chicago, pp 506–516
Clark DB, Palmer MW, Clark DA (1999) Edaphic factors and the landscape-scale distributions of tropical rain forest trees. Ecology 80:2662–2675
Comita LS, Engelbrecht BMJ (2009) Seasonal and spatial variation in water availability drive habitat associations in a tropical forest. Ecology 90:2755–2765
Condit R (1998) Tropical forest census plots: methods and results from Barro Colorado Island, Panama and a comparison with other plots. Springer-Verlag, Berlin
Condit R, Ashton P, Baker P et al (2000) Spatial patterns in the distribution of tropical tree species. Science 288:1414–1418
Condit R, Ashton P, Baslev H et al (2005) Tropical tree α-diversity: results from a worldwide network of large plots. Biol Skrif 55:565–582
Daws MI, Mullins CE, Burslem DFRP, Paton SR, Dalling JW (2002) Topographic position affects the water regime in a semideciduous tropical forest in Panama. Plant Soil 238:79–90
Engelbrecht BMJ, Kursar TA (2003) Comparative drought-resistance of seedlings of 28 species of co-occurring tropical woody plants. Oecologia 136:383–393
Engelbrecht BMJ, Comita LS, Condit R, Kursar TA, Tyree MT, Turner BL, Hubbell SP (2007) Drought sensitivity shapes species distribution patterns in tropical forests. Nature 447:80–82
Gartlan JS, Newbery DC, Thomas DW, Waterman PG (1986) The influence of topography and soil phosphorus on the vegetation of Korup forest reserve, Cameroun. Vegetatio Acta Geobot 65:131–148
Gentry AH (1988) Changes in plant community diversity and floristic composition on environmental and geographical gradients. Ann Mo Bot Gard 75:1–34
Gibbons JM, Newbery DM (2003) Drought avoidance and the effect of local topography on trees in the understorey of Bornean lowland rain forest. Plant Ecol 43:63–75
Gunatilleke CVS, Gunatilleke IAUN, Esufali S, Harms KE, Ashton PMS, Burslem DFRP, Ashton PS (2006) Species-habitat associations in a Sri Lankan dipterocarp forest. J Trop Ecol 22:371–384
Hall JB, Swaine MD (1981) Distribution and ecology of vascular plants in a tropical rain forest. Dr W Junk Publishers, The Hague
Hall JS, McKenna JJ, Ashton PMS, Gregoire TG (2004) Habitat characterizations underestimate the role of edaphic factors controlling the distribution of Entandrophragma. Ecology 85:2171–2183
Hardy OJ, Sonke B (2004) Spatial pattern analysis of tree species distribution in a tropical rain forest of Cameroon: assessing the role of limited dispersal and niche differentiation. Forest Ecol Manag 197:191–202
Harms KE, Condit R, Hubbell SP, Foster RB (2001) Habitat associations of trees and shrubs in a 50-ha neotropical forest plot. J Ecol 89:947–959
Holmgren PK, Holmgren NH, Barnett LC (1990) Index herbariorum, part I: the herbaria of the world, 8th edn. New York Botanical Garden, Bronx
Hubbell SP (2001) The unified neutral theory of biodiversity and biogeography. Princeton University Press, Princeton
Hubbell SP, Foster RB (1986) Biology, chance, and history and the structure of tropical rain forest tree communities. In: Diamond J, Case TJ (eds) Community ecology. Harper and Row, New York, pp 314–329
John R, Dalling JW, Harms KE, Yavitt JB, Stallard RF, Mirabello M, Hubbell SP, Valencia R, Navarrete H, Vallejo M, Foster RB (2007) Soil nutrients influence spatial distributions of tropical tree species. Proc Natl Acad of Sci USA 104:864–869
Kenfack D, Gosline G, Gereau RE, Schatz G (2003) The genus Uvariopsis (Annonaceae) in Tropical Africa, with a recombination and one new species from Cameroon. Novon 13:443–449
Kenfack D, Ewango CEN, Thomas DW (2004) Manilkara lososiana, a new species of Sapotaceae from Cameroon. Kew Bull 59:609–612
Kenfack D, Sainge NM, Thomas DW (2006) A new species of Cassipourea (Rhizophoraceae) from western Cameroon. Novon 16:61–64
Kenfack D, Thomas DW, Chuyong G, Condit R (2007) Rarity and abundance in a diverse African forest. Biodivers Conserv 16:2045–2074
Küper W, Sommer JH, Lovett JC, Mutke J, Linder HP, Beentje HJ, Van Rompaey R, Chatelain C, Sosef M, Barthlott W (2004) Africa’s hotspots of biodiversity redefined. Ann Mo Bot Gard 91:525–535
Legendre P (1993) Spatial autocorrelation: trouble or new paradigm? Ecology 74:1659–1673
Letouzey R (1985) Notice de la carte phytogéographique du Cameroun au 1:500.000. Institut de la Carte Internationale de la Végétation, Toulouse
Maley J (1987) Fragmentation de la forêt dense humide ouest-africaine et extension des biotopes montagnards au quaternaire récent: nouvelles données polliniques et chronologiques: implications paléoclimatiques et biogéographiques. Paleoecol Afr 18:307–334
Miyamoto K, Suzuki E, Kohyama T, Seino T, Mirmanto E, Simbolon H (2003) Habitat differentiation among tree species with small-scale variation of humus depth and topography in a tropical heath forest of Central Kalimantan, Indonesia. J Trop Ecol 19:43–54
Newbery DM, Proctor J (1984) Ecological studies in four contrasting lowland rain forests in Gunung Mulu National Park, Sarawak IV Associations between tree distribution and soil factors. J Ecol 72:475–493
Newbery DM, Gartlan JS, Mckey DB, Waterman PG (1986) The influence of drainange and soil phosphorus on the vegetation of Douala-Edea Forest Reserve, Cameroon. Vegetatio 65:149–162
Newbery DM, Alexander IJ, Thomas DW, Gartlan JS (1988) Ectomycorrhizal rain-forest legumes and soil phosphorus in Korup National Park, Cameroon. New Phytol 109:433–450
Newbery DM, Songwe NC, Chuyong GB (1998) Phenology and dynamics of an African rainforest at Korup, Cameroon. In: Newbery DM, Prins HHT, Brown ND (eds) Dynamics of tropical communities. Blackwell Science, Oxford, pp 177–224
Newbery DM, van der Burgt XM, Moravie MA (2004) Structure and inferred dynamics of a large grove of Microberlinia bisulcata trees in central African rain forest: the possible role of periods of multiple disturbance events. J Trop Ecol 20:131–143
Paoli GD, Curran LM, Zak DR (2006) Soil nutrients and beta diversity in the Bornean Dipterocarpaceae: evidence for niche partitioning by tropical rain forest trees. J Ecol 94:157–170
Parmentier I, Malhi Y, Senterre B et al (2007) The odd man out? Might climate explain the lower tree alpha-diversity of African rain forests relative to Amazonian rain forests? J Ecol 95:1058–1071
Phillips OL, Vargas PN, Monteagudo AL, Cruz AP, Chuspe Zans M-E, Sanchez WG, Yli-Halla M, Rose S (2003) Habitat association among Amazonian tree species: a landscape-scale approach. J Ecol 91:757–775
Potts MD, Davies SJ, Bossert WH, Tan S, Nur Supardi MN (2004) Habitat heterogeneity and niche structure of trees in two tropical rain forests. Oecologia 139:446–453
Ricklefs RE (1977) Environmental heterogeneity and plant species diversity: a hypothesis. Am Nat 111:376–381
Russo SE, Brown P, Tan S, Davies SJ (2008) Interspecific demographic trade-offs and soil-related habitat associations of tree species along resource gradients. J Ecol 96:192–203
Silvertown J, Dodd ME, Gowing DJG, Mountford JO (1999) Hydrologically defined niches reveal a basis for species richness in plant communities. Nature 400:61–63
Sonké B, Kenfack D, Robbrecht E (2002) A new species of the Tricalysia atherura group (Rubiaceae) from southwestern Cameroon. Adansonia 24:173–177
Thomas DW, Kenfack D, Chuyong GB, Sainge NM, Losos EC, Condit RS, Songwe NC (2003) Tree species of Southwestern Cameroon: tee distribution maps, diameter tables and species documentation of the 50-ha Korup forest dynamics plot. Center for Tropical Forest Science, Washington
Valencia R, Foster RB, Muñoz GV, Condit R, Svenning J-C, Hernandez C, Romoleroux K, Losos E, Magard E, Balslev H (2004) Tree species distributions and local habitat variation in the Amazon: large forest plot in eastern Ecuador. J Ecol 92:214–229
Versteegh CPC, Sosef MSM (2007) Revision of the African genus Annickia (Annonaceae). Syst Geogr Pl 77:91–118
Webb CO, Peart DR (2000) Habitat associations of trees and seedlings in a Bornean rain forest. Ecology 88:464–478
White F (1983) The vegetation of Africa. UNESCO, Paris
Wright SJ (2002) Plant diversity in tropical forests: a review of mechanisms of species coexistence. Oecologia 130:1–14
Yamada T, Zuidema PA, Itoh A, Yamakura T, Ohkubo T, Kanzaki M, Tan S, Ashton PS (2007) Strong habitat preference of a tropical rain forest tree does not imply large differences in population dynamics across habitats. J Ecol 95:332–342
Yamakura T, Kanzaki M, Itoh A, Ohkubo T, Ogino K, Ernest Chai OK, Hua Seng L, Ashton PS (1995) Topography of a large-scale research plot established within a tropical rain forest at Lambir, Sarawak. Tropics 5:41–56
Acknowledgments
We thank the Ministry of Environment and Forests, Cameroon for permission to conduct the field program in the Korup National Park. The Korup FDP is affiliated with the Center for Tropical Forest Science, a global network of large-scale demographic tree plots. Analyses were partially supported by U.S. National Science Foundation award DEB-9806828 to the Center for Tropical Forest Science of the Smithsonian Institution. KEH acknowledges support from NSF (DEB 0211004 and OISE 0314581) that contributed toward the completion of this manuscript. LSC acknowledges support from the National Center for Ecological Analysis and Synthesis, a Center funded by NSF (Grant #EF-0553768), the University of California, Santa Barbara, and the State of California.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chuyong, G.B., Kenfack, D., Harms, K.E. et al. Habitat specificity and diversity of tree species in an African wet tropical forest. Plant Ecol 212, 1363–1374 (2011). https://doi.org/10.1007/s11258-011-9912-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-011-9912-4