Abstract
Knowledge of the response of litter mass loss to climate warming and litter quality in alpine ecosystems is still sparse. Here, we conducted a 507-day litter decomposition experiment along an elevation gradient from 3200 to 3800 m using different litter types to determine the influences of litter quality and climate change on the elemental mass losses and on the temperature sensitivity of litter mass losses (annual percentage decomposition (%) per 1°C temperature difference). Mass losses of C, nitrogen (N), phosphorus (P), potassium (K), sodium (Na), calcium (Ca), and Magnesium (Mg) decreased with an increase in elevation. In general, N and Na concentrations in litter and ratios of C:N and lignin:N were the best predictors of C mass losses. A higher N concentration and C:N ratio in litter caused greater C mass losses, but higher lignin:N ratio in litter resulted in lower C mass losses. Litter decomposition occurred in a two-stage process. Carbon mass loss in litter was mainly limited by soil temperature in the first growing season of the decomposition period, whereas N concentration and ratios of C:P and N:P limited carbon mass loss in the remaining litter during the second growing season of the decomposition period. Soil moisture appeared not to affect litter mass loss and the temperature sensitivity of litter mass loss of grass litter was greater than that of shrub litter in the alpine region.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Litter nutrient concentration, the concentration of carbon fractions (e.g., lignin), carbon:nitrogen (C:N) and lignin:N ratios have been widely used as litter quality variables due to their influence on microbial activity and litter decomposition rates (Melillo et al. 1982; Taylor et al. 1989; Berg et al. 1996; Murphy et al. 1998). These variables are also used in some ecosystem carbon models (Running and Hunt 1993; Parton et al. 1994). However, there is still no universally accepted litter-quality variable (see citations in Murphy et al. 1998) because several sources of variation obscure our understanding of the effects of different variables, and because the process of litter mass loss depends on complex interactions between soil properties, climatic factors, and litter types (Meentemeyer 1978; Harmon et al. 1990; Berg et al. 1993; Aerts 1997; Murphy et al. 1998; Hobbie 2000; Hobbie and Vitousek 2000; Fierer et al. 2005). There is a particular lack of knowledge about how litter quality and its variables affect litter mass loss in alpine regions (Cornelissen et al. 2007).
A number of studies have shown that the temperature sensitivity of litter mass loss can vary with litter type and extent of mass loss (Kirschbaum 1995; Katterer et al. 1998; Dalias et al. 2001; McTiernan et al. 2003; Fierer et al. 2005). Global warming and associated environmental changes are predicted to affect most regions of the northern hemisphere and will be particularly pronounced at high northern latitudes during this century (ACIA 2005; IPCC 2007). As the largest grassland area on the Eurasian continent, the Qinghai-Tibetan plateau is mostly situated at 3,500 m or more above sea level, and covers an area of ~2.5 million km2 (Zheng et al. 2000). Evidence shows that the Qinghai-Tibetan plateau is experiencing climatic warming (Thompson et al. 1993, 2000). Moreover, the region is predicted to experience “much greater than average” increases in surface temperature in the future (IPCC 2007). Despite the importance of this region, little is known about the effect of global warming on litter decomposition, which could in turn affect carbon storage in these grasslands.
In this study, we conducted a 2-year litter decomposition experiment along an elevation gradient from 3200 to 3800 m. We collected samples of four litter types from alpine meadows free-grazed by sheep (two grass litter samples, a shrub litter sample, and a mixed litter sample) in May 2007. The aims of this study were to investigate for the first time (1) the effect of litter quality and climate change on element mass losses, and (2) the temperature sensitivity of litter mass loss on the Tibetan plateau.
Materials and methods
Experimental site
The experimental site is located at the Haibei Alpine Meadow Ecosystem Research Station (HBAMERS), a facility run by the Northwest Institute of Plateau Biology of the Chinese Academy of Sciences. HBAMERS is situated at latitude 37º 37′N, longitude 101º 12′E. The station lies in the northeast of the Tibetan plateau in a large valley surrounded by the Qilian Mountains (Fig. 1). The station experiences a typical plateau continental climate which is dominated by the southeast monsoon from May to September in summer and high pressure from Siberia in winter. Summers are short and cool, and winters are long and severely cold. Mean annual temperature is −2°C, and mean annual precipitation is 500 mm, over 80% of which falls during the summer monsoon season. Mean elevation of the valley bottom is 3,200 m. The soil is a Mat-Gryic Cambisol (Chinese Soil Taxonomy Research Group, 1995), corresponding to Gelic Cambisol (WRB 1998). Basic soil properties were organic carbon 5.5 and 3.3%, pH 7.3 and 7.4, bulk density 0.75 and 1.11 g cm−3 at 10 and 20 cm soil depths, respectively (Cao et al. 2008). A detailed site description can be found in Zhao and Zhou (1999).
Litter sample collection and decomposition
The plant community at the experimental site at 3,200 m is dominated by Elymus nutans (Griseb.), Poa pratensis (Linn.), Festuca ovina (Linn.), Gentiana straminea (Maxim.), Kobresia humilis (C. A. Mey. ex Trautv.) (Serg.), Carex scabrirostris (Kükenth.), Scripus distigmaticus (Kükenth.) (Tang et Wang.), Potentilla nivea (Linn.), Gentiana grumii (Kusnez.), Blysmus sinocompressus (Tang et Wang.), Leontopodium nanum (Hook. f. et Thoms.) (Hand.-Mazz.), and Dasiphora fruticosa (Linn.) (Rydb.). Litter samples of E. nutans, B. sinocompressus, D. fruticosa (shrub), and mixed litter of the community were collected randomly from this meadow community grazed by sheep in May 2007, air-dried, and stored indoors until the beginning of the decomposition experiment.
About 3 km from HBAMERS, one 10 × 20 m plot was fenced in autumn 2005 at each elevation of 3200, 3600, and 3800 m above sea level along the southern side of the Qilian Mountains. Air-dried litter samples (20 g oven-dry mass at 80°C) of single species and mixed litter samples of the community were placed in 10 × 20 cm litterbags constructed from 1-mm mesh nylon cloth. We placed a total of 360 litter bags at the three sites at 3200, 3600, and 3800 m at spacings of 10–20 cm on June 1 2007, and 5 litterbags each species/community mixture were randomly selected to take back to the laboratory on July 24 and October 14 in 2007, and April 24, June 24, July 24 and October 24 in 2008, respectively, to measure litter mass loss (i.e., 5 replicates × 4 litter types × 3 elevations × 6 sample dates).
All the samples at each sampling period were dried at 80°C to measure their mass losses, and then ground to pass through a 1-mm sieve to determine concentrations of carbon (C), nitrogen (N), phosphorus (P), potassium (K), calcium (Ca), and magnesium (Mg) as per the methods described in AOAC (1984). Litter chemistry was measured by sequentially digesting material into fractions that correspond with soluble cell contents, cellulose, hemicellulose, lignin, and acid insoluble ash (Van Soest 1963; Ryan et al. 1990) on a forage fiber analyzer (ANKOM 200, Macedon, New York, USA). All the nutrient concentrations were calculated on the basis of organic matter (i.e., dry matter minus crude ashes).
Soil temperature and soil moisture along the elevation gradient
At the center of each plot, weather stations (Onset Computer Corporation, Japan) were used to monitor soil temperature and soil moisture at 5-cm soil depth. The sensors were connected to a CR1000 datalogger, and soil temperature and soil moisture were measured every 1 min, and then 30-min averages were stored.
Data calculation and analysis
Repeated-measures analysis of variation of mass losses of different elements in litter was used with elevation and species as between-subject effects and with sampling date as the within-subject effect, using GLM analysis in SPSS (Version 12, SPSS Inc. Chicago, Illinois, USA, 2005). For each species and sampling date, the significant difference between elevations was assessed by One-way ANOVA and least significance difference (LSD) tests.
Temperature sensitivity of litter mass loss was defined as the relative differences in litter mass losses (%) between two different elevations divided by the annual average temperature difference (oC) of the corresponding elevations. Linear regression analysis was performed to test the dependency of the annual average percentage mass losses of litter on differences in annual average soil temperature at 5-cm depth among elevations. The significance of temperature sensitivity of mass losses (i.e., the slopes of the regression equations) among different litter types was tested using paired comparisons.
Partial correlation analysis was performed to test the dependency of C mass losses in each sampling period on the proposed litter quality variables and average soil temperature and soil moisture at 5 cm. All the significances mentioned in the text were at the 0.05 level.
Results
Soil temperature and moisture along the elevation
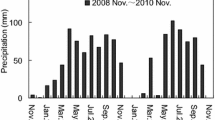
In general, although there was no significant difference between 3200 and 3600 m for annual soil temperature and soil moisture at 5 cm, they decreased with an increase in elevation (P < 0.05). Their annual average values over two years were 3.3, 2.8, and 0.1°C for soil temperature (Fig. 2a), and 18.6, 17.8, and 8.1% for soil moisture at 3200, 3600, and 3800 m, respectively (Fig. 2b). However, the patterns of the differences in soil temperature and soil moisture at 5 cm among elevations differed significantly between the growing season (May to October) and nongrowing season. For example, during the growing season, the soil temperatures at 5 cm were 9.5, 7.5, and 5.8°C at 3200, 3600, and 3800 m, respectively, whereas during the nongrowing season they were −2.9, −1.8, and −5.6°C, respectively, indicating that during the nongrowing season, the average soil temperature at 5 cm was significantly higher at 3,600 m due to temperature inversion on the foot of the mountain. During the growing season, soil moisture was 26.2, 27.6, and 14.4% at 3200, 3600, and 3800 m, respectively, indicating that the mean soil moisture at 5 cm was lowest at 3,800 m during the growing season, especially from June to July in 2007 and 2008 (Fig. 2b) due to the effect of topography on rainfall.
Initial litter chemical components and their mass losses
The initial litter chemistry of E. nutans, B. sinocompressus, D. fruticosa, and mixed litters of the communities showed significant differences (Table 1). When sorting the species and mixed litters from high to low litter quality, each of the potential litter-quality variables classified the litter types differently. The shrub species, D. fruticosa, was an exception, having a significantly lower litter quality when measured by all of the variables except percentages of phosphorus and calcium. The other litter types displayed more variation in relation to one or another variable. E. nutans had the lowest N concentration and highest C:N ratio (low litter quality), but the most favorable litter quality relative to several other variables (e.g., percentage lignin, lignin:N ratio). Mixed litter had the highest litter quality according to N concentration and C:N ratio. However, when evaluating the other variables (e.g., percentage of cellulose and hemicellulose), the litter quality of the mixed litter diminished compared to the other litter types. Values for B. sinocompressus were generally intermediate among the litter types. Since each of the litter-quality variables classifies the litter quality differently in relation to each other, there exists a range of possibilities for utilizing litter quality as a predictor of mass losses.
All element (C, N, P, K, Na, Ca, and Mg) mass losses were affected significantly by elevation, sampling date, litter type, and their interactions (Table 2 and Figs. 3, 4 and 5). In general, their mass losses decreased significantly with an increase in elevation, but the differences became smaller with decomposition time. For example, the average cumulative C mass losses for all litter types were 28.1, 19.9, and 14.5%, and 60.8, 57.3, and 54.7% during 54-day and 507-day decompositions at 3200, 3600, and 3800 m, respectively. The average relative differences of C mass losses were greater by 93.8 and 37.2% at 3200 and 3600 m compared with that at 3,800 m during 54-day decomposition, whereas the differences were only 11.2 and 4.8% during 507-day decomposition, indicting that the potential effect of climate variables (temperature, moisture) on litter decomposition decreases with decomposition time (Table 3). The average cumulative mass losses of grass litter (i.e., E. nutans and B. sinocompressus) were greater than that of shrub litter (D. fruticosa) for C, P, K, Na, and Mg, whereas the opposite was observed for N during the decomposition period. The average cumulative mass losses of C and Na of the mixed litter were the greatest during the decomposition period. The effect of litter quality on litter decomposition varied with decomposition time. For example, the average cumulative C mass losses for all elevations were 18.2, 21.0, 27.1, and 17.1%, and 58.6, 59.5, 63.2, and 49.1% for E. nutans, B. sinocompressus, mixed litter, and D. fruticosa during 54-day and 507-day decompositions, respectively. The relative differences of C mass losses were 6.4, 22.8, and 58.5% during 54-day decomposition, and 19.3, 21.2, and 28.7% during 507-day decomposition compared with that of D. fruticosa (Table 3), indicating that the potential effect of litter quality on C mass loss may be controlled by different litter quality variables with decomposition time. In the early stages of litter decomposition (i.e., during the 54-day decomposition period), the effect of climate variables on litter decomposition was greater than that of litter quality based on the average relative differences (%) of C mass losses at different elevations compared with that of 3,800 m and the average relative differences (%) of C mass losses of different litter types compared with that of D. fruticosa, whereas the opposite was the case over the longer decomposition period (i.e., during 507-day decomposition) (Table 3).
In order to determine whether litter decomposition occurs in a two-stage process, we evaluated the differences in C mass loss of each species/community mixed litter between elevations during the first growing season, during the nongrowing season and during the second growing season (Fig. 6). We found that for all litter types, the relative cumulative C mass losses during the first growing season as a proportion of total cumulative C mass losses over the 507-day decomposition period significantly decreased with an increase in elevation. During the second growing season, however, it was lowest at 3,200 m and even significantly increased with an increase in elevation for all litter types except D. fruticosa, although there was no significant difference between 3600 and 3800 m (Fig. 6).
Cumulative carbon mass loss under specified decomposition periods as a percentage of total cumulative carbon mass loss over the whole decomposition period. FGS first growing season (114 days), NGS nongrowing season (181 days), and SGS second growing season (212 days). Different letters mean significant difference at 0.05 level and bars in the figure show standard errors
The average cumulative mass losses of C, N, P, K, Na, Ca, and Mg for all litter types and elevations during the 507-day decomposition period were 57.6, 46.2, 78.3, 58.9, 75.0, 76.6, and 63.8%, respectively, indicating that N mass loss was less than that of other elements.
Factors affecting carbon mass loss
In general, based on partial correlation analysis of results from 6 samplings over the 507-day decomposition period, we found that high C concentration and high ratios of N:P and lignin:N in initial and remaining litter reduced C mass losses, whereas positive correlations were observed between C mass losses and concentrations of N, P, Na, cellulose, and ratios of C:N and N:P in initial and remaining litters. Soil temperature and soil moisture did not affect C mass losses (Table 4). However, if one excludes the nongrowing season from 24 October 2007 to 24 June 2008, C mass losses increased significantly with increases in soil temperature and concentrations of N, P, Na, cellulose and C:N ratio (Table 4). There were no significant partial correlations between C mass losses and any variables in Table 1 during the nongrowing season (data not shown). During the first growing season of the decomposition experiment, soil temperature, N concentration and ratios of C:N and lignin:N were the main factors that affected C mass losses in litter, whereas partial correlation was not significant between C mass losses and soil temperature during the second growing season of the decomposition experiment (Table 4). These results indicate that C mass losses in litter was mainly controlled by soil temperature during the early phase of the decomposition period and then by litter quality during the later phase of the decomposition period, and that soil moisture appeared to have no significant effect on C mass losses in litter.
Temperature sensitivity of litter mass losses
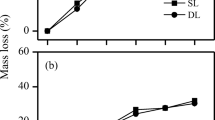
We measured the relationships between the percentages of the litter mass losses during the first year (i.e., over a 386-day decomposition period) and the differences of annual soil temperature at 5 cm across the elevation gradient. The relationships were weak for the mixed litter (P = 0.052) and D. fruticosa (P = 0.090). The temperature sensitivities of the litter mass loss of the mixed litter, B. sinocompressus, E. nutans, and D. fruticosa were approximately 4.9, 5.6, 8.7, and 3.4, respectively (Fig. 7), which were significantly different (P = 0.016).
Relationships between litter mass loss (%) for a 386 day decomposition period across the elevation gradient and differences in annual average soil temperature at 5 cm depth among elevations. The regression equations are EN: y = 8.6989x − 2.2244 (r 2 = 0.99, P = 0.002); BS: y = 5.6351x − 0.1115 (r 2 = 0.99, P = 0.013); M: y = 4.7472x − 0.2373 (r 2 = 0.97, P = 0.052); and DF: y = 3.4451x + 2.9538 (r 2 = 0.86, P = 0.090), respectively, where y is the litter mass loss for a 386-day decomposition period across the elevation gradient, and x is the difference in annual average soil temperature at 5 cm among elevations. M: mixed litter, BS: Blysmus sinocompressus, EN: Elymus nutans, DF: Dasiphora fruticosa
Discussion
Many decomposition studies have identified lignin concentration and the ratios of lignin:N and C:N as the most reliable predictors of decomposition (Berg et al. 1982; Stohlgren 1988; Taylor et al. 1989; Running and Hunt 1993; Parton et al. 1994; Murphy et al. 1998). In particular, Murphy et al. (1998) suggested that litter mass loss was limited by C substrates rather than by nutrient content, including N concentration. However, our results indicated that N concentrations and ratios of C:N and lignin:N in litter are the best predictors of C mass losses, although the relationships between C mass loss and litter nutrient concentration or ratios of C:N, C:P, N:P, and lignin:N varied with decomposition periods (Table 4). Significant effects of litter N content on litter mass loss have been observed in other mesic systems (Taylor et al. 1989). High N concentration in litter consistently promoted a faster C mass loss during the summer decomposition periods (Table 4), whereas the chemical components in litter did not affect C mass losses during the winter decomposition period due to slow decomposition with low temperature. Along with the changes in decomposition dynamics, some factors controlling C mass loss switched from the ratios of C:N and lignin: N during the first growing season to the ratios of C:P and N:P during the second growing season (Table 4). It is likely that variation of resource ratios in litter with decomposition time and stoichiometric constraints result in variation of the decomposer community (Cherif and Loreau 2007), because variation in the elemental composition of bacteria and fungi is very small (Makino et al. 2003). It is known, for example, that bacteria and fungi have different C:P and C:N ratios and that the substrate C:N ratio can have an influence on the fungal/bacteria biomass ratio (Eiland et al. 2001). We found that N mass loss was less than that of other elements in litter, causing lower C:N ratios and higher N:P ratios in the remaining litter over decomposition time. Similarly, the vast number of limiting resources created by the combination of various essential elements (e.g., P and Na) in organic resources with different possible elemental ratios may affect the structure of the decomposer community (Cherif and Loreau 2007). Inconsistent with previous reports that a low C:N ratio promotes a faster rate of decomposition (Berg et al. 1982), or reports that there is no relationship between the C:N ratio in litter and mass losses (Murphy et al. 1998), we found that there were positive correlations between C:N ratios and C mass losses during different decomposition periods, except during winter and the second growing season (Table 4). Two reasons can be proposed to explain this difference. First, the statistical analysis method used was different. Significant negative correlations (P < 0.05) were found between C mass losses and C:N ratios in litter when simple correlation analysis was used (data not shown). However, positive partial correlations were found between C mass losses and C:N ratios in litter during the decomposition periods when partial correlation analysis was used (Table 4), indicating that simple negative correlations between C mass losses and C:N ratios in litter were caused by other correlated variables rather than the C:N ratio itself (Tilman and Downing 1994). Second, although initial a higher C:N ratio for Dasiphora fruticosa (Table 1) caused lower C mass loss (i.e., a negative correlation), C:N ratios in the remaining litter decreased with decomposition time due to N mass losses which matched the variation in C mass losses (i.e., a positive correlation). Therefore, it is important to exercise caution when directly comparing the relationship between C mass loss and C:N ratio in litter in the literature since the analysis method used to calculate the correlations can vary significantly across studies.
The effect of soil temperature on C mass losses varied with decomposition period (Table 4). Soil temperature explained approximately 67% of variation in C mass loss during the first growing season, but it did not affect C mass loss during the second growing season. These results indicate that with the decomposition time the effect of quality of the remaining litter on C mass loss may override the effect of soil temperature during the second growing season. Soil moisture did not affect litter mass losses in the region. However, Murphy et al. (1998) found at below 2,500 m elevation in the Southwest USA that litter mass loss was mainly controlled by lignin concentration and precipitation rather than temperature. Therefore, in our colder and wetter region at more than 3,200 m elevation, temperature may be an important factor controlling C mass loss during the first summer, and future summer warming may increase C mass loss from litter.
Litter mass loss and its temperature sensitivity for the low litter quality shrub, D. fruticosa, was low compared with grasses (Fig. 7), which is consistent with previous reports (Cornelissen et al. 2007). As warming increases invasion of shrubs to the community in the alpine and tundra regions (Klein et al. 2007, 2008; Post et al. 2008), future warming may reduce the amount of carbon released to the atmosphere from litter in the region. The temperature sensitivity of litter mass loss will influence rates of ecosystem carbon sequestration in a warmer world (Jones et al. 2003; Fierer et al. 2005), in particular in our study area. Bosatta and Agren (1999) have hypothesized that the temperature sensitivity of litter mass loss is governed by microbial enzyme kinetics and will be influenced by the quality of the litter C being consumed by microorganisms. This has been called the “C quality-temperature” hypothesis by Fierer et al. (2005), who first determined the hypothesis and suggested that the temperature sensitivity of microbial decomposition should be inversely related to litter C quality. However, we found that there were no significant correlations between the temperature sensitivity of litter mass loss and the initial chemical components in litter, even though a shrub with the highest lignin concentration (Table 1) had the lowest temperature sensitivity (Fig. 6). This is probably due to different approaches used to characterize temperature sensitivity (Q10 used in Fierer et al. 2005).
Conclusion
Higher temperatures significantly increased element litter mass losses, which suggests that global warming will decrease C storage in the study site, unless shrub encroachment is extensive. The N concentrations and ratios of C:N and lignin:N in litter were the best predictors of C mass loss. The effect of soil temperature on C mass losses was greater than that of litter nutrient quality during the first growing season of the decomposition period, whereas C mass loss in litter was mainly controlled by N concentration and the ratios of C:P and N:P in remaining litter during the second growing season of the decomposition period. The mass losses of grass litter and their temperature sensitivity were greater than that of shrub litter. Therefore, our study on the temperature sensitivity of litter mass losses may have important implications for predictions about future contributions of alpine and possibly also other cold regions to carbon dynamics worldwide.
References
ACIA (2005) Arctic climate impact assessment. Cambridge University Press, Cambridge
Aerts R (1997) Climate, leaf litter chemistry, and leaf litter decomposition in terrestrial ecosystems: a triangular relationship. Oikos 79:439–449
AOAC (1984) Official methods of analysis of the Association of Official Analytical Chemists, 14th edn. Association of Official Analytical Chemists, Washington, DC
Berg B, Wessen B, Ekbohm G (1982) Nitrogen level and decomposition in Scots pine litter. Oikos 38:291–296
Berg B, Berg MP, Bottner P, Box EO et al (1993) Litter mass loss rates in pine forests of Europe and eastern United States: some relationships with climate and litter quality. Biogeochemistry 20:127–159
Berg B, Ekbohm G, Johansson ME, McClaugherty CA, Rutigliano F, Santo AV (1996) Maximum decomposition limits of forest litter types: a synthesis. Can J Bot 74:659–672
Bosatta E, Agren GI (1999) Soil organic matter quality interpreted thermodynamically. Soil Biol Biochem 31:1889–1891
Cao G, Xu XL, Long RJ, Wang QL, Wang CT, Du YG, Zhao XQ (2008) Methane emissions by alpine plant communities in the Qinghai-Tibet plateau. Biol Lett 4:681–684
Cherif M, Loreau M (2007) Stoichiometric constraints on resource use, competitive interactions, and elemental cycling in microbial decomposers. Am Nat 169:709–724
Chinese Soil Taxonomy Research Group (1995) Chinese soil taxonomy. Science Press, Beijing, pp 58–147
Cornelissen JHC, van Bodegom PM, Aerts R et al (2007) Global negative vegetation feedback to climate warming responses of leaf litter decomposition rates in cold biomes. Ecol Lett 10:619–627
Dalias P, Anderson J, Bottner P, Couteaux M (2001) Temperature responses of carbon mineralization in conifer forest soils from different regional climates incubated under standard laboratory conditions. Glob Chang Biol 6:181–192
Eiland F, Klamer M, Lind AM, Baath E (2001) Influence of initial C/N ratio on chemical and microbial composition during long term composting of straw. Microb Ecol 41:272–280
Fierer N, Craine JM, McLauchlan K, Schimel JP (2005) Litter quality and the temperature sensitivity of decomposition. Ecol 86:320–326
Harmon ME, Baker GA, Spycher G, Greene E (1990) Leaf-litter decomposition in the Picea/Tsuga forests of Olympic National Park, Washington, USA. For Ecol Manage 31:55–66
Hobbie SE (2000) Interactions between litter lignin and soil nitrogen availability during leaf litter decomposition in a Hawaiian montane forest. Ecosystems 3:484–494
Hobbie SE, Vitousek PM (2000) Nutrient limitation of decomposition in Hawaiian forests. Ecol 81:1867–1877
IPCC (2007) Climate Change 2007: summary for policymaker. Valencia, Spain
Jones CD, Cox P, Huntingford C (2003) Uncertainty in climate-carbon-cycle projections associated with the sensitivity of soil respiration to temperature. Tellus B Chem Phys Meteorol 55:642–648
Katterer T, Reichstein M, Andren O, Lomander A (1998) Temperature dependence of organic matter decomposition: a critical review using literature data analysed with different models. Biol Fert Soil 27:258–262
Kirschbaum MUF (1995) The temperature dependence of soil organic matter decomposition, and the effect of global warming on soil organic C storage. Soil Biol Biochem 27:753–760
Klein JA, Harte J, Zhao XQ (2007) Experimental warming, not grazing, decreases rangeland quality on the Tibetan plateau. Ecol Appl 17:541–557
Klein JA, Harte J, Zhao X-Q (2008) Decline in medicinal and forage species with warming is mediated by plant traits on the Tibetan plateau. Ecosystems 11:775–789
Makino W, Cotner JB, Sterner RW, Elser J (2003) Are bacteria more like animals than plants? growth rate and resource dependence of bacterial C:N:P stoichiometry. Funct Ecol 17:121–130
McTiernan KB, Coüteaux MM, Berg B et al (2003) Changes in chemical composition of Pinus sylvestris needle litter during decomposition along a European coniferous forest climatic transect. Soil Biol Biochem 35:801–812
Meentemeyer V (1978) Macroclimate and lignin control of litter decomposition rates. Ecol 59:465–472
Melillo JM, Aber JD, Muratore JF (1982) Nitrogen and lignin control of hardwood leaf litter decomposition dynamics. Ecol 63:621–626
Murphy KL, Klopatek JM, Klopatek CC (1998) The effects of litter quality and climate on decomposition along an elevation gradient. Ecol Appl 8:1061–1071
Parton WJ, Ojima DS, Cole CV, Schimel DS (1994) A general model for soil organic matter dynamics: sensitivity to litter chemistry, texture, and management. In: Quantitative modeling of soil forming processes. Soil Science Society of American Special Publication 39, pp 147–167. Madison, Wisconsin, USA
Post E, Pedersen C, Wilmers CC, Forchhammer MC (2008) Phenological sequences reveal aggregate life history response to climatic warming. Ecol 89:363–370
Running SW, Hunt ER Jr (1993) Generalization of a forest ecosystem process model for other biomes, BIOME-BGC, and an application for global-scale models. In: Ehleringer JE, Field CB (eds) Scaling physiological processes: leaf to global. Academica Press, New York, pp 141–158
Ryan M, Melillo J, Ricca A (1990) A comparison of methods for determining proximate carbon fractions of forest litter. Can J For Res 20:166–171
Stohlgren TJ (1988) Litter dynamics in two Sierran mixed conifer forests. II. Nutrient release in decomposing leaf litter. Can J For Res 18:1136–1144
Taylor BR, Parkinson D, Parsons WFJ (1989) Nitrogen and lignin content as predictors of litter decay rates: a microcosm test. Ecol 70:97–104
Thompson LG, Mosley-Thompson E, Davis M et al (1993) Recent warming: ice core evidence from tropical ice cores with emphasis on Central Asia. Glob Planet Change 7:145–156
Thompson LG, Yao T, Mosley-Thompson E et al (2000) A high-resolution millennial record of the South Asian monsoon from Himalayan ice cores. Science 289:1916–1919
Tilman D, Downing JA (1994) Biodiversity and stability in grasslands. Nature 367:363–365
Van Soest PJ (1963) Use of detergents in analysis of fibrous feeds: a rapid method for the determination of fiber and lignin. J Assoc Off Anal Chem 46:829–835
WRB (1998) World reference base for soil resources. FAO/ISRIC/ISSS, Rome
Zhao XQ, Zhou XM (1999) Ecological basis of alpine meadow ecosystem management in Tibet: Haibei alpine meadow ecosystem research station. Ambio 28:642–647
Zheng D, Zhang QS, Wu SH (2000) Mountain geoecology and sustainable development of the Tibetan plateau. Kluwer Academic, Norwell
Acknowledgments
This research was funded by the “100-Talent Program” of the Chinese Academy of Sciences, the Knowledge Innovation Programs (KZCX2-YW-N-040/XB2-06-01), the Chinese National Natural Science Foundation Commission (30871824), and Qinghai Science and Technology Bureau.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xu, G., Hu, Y., Wang, S. et al. Effects of litter quality and climate change along an elevation gradient on litter mass loss in an alpine meadow ecosystem on the Tibetan plateau. Plant Ecol 209, 257–268 (2010). https://doi.org/10.1007/s11258-009-9714-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-009-9714-0