Abstract
Given that ecological knowledge of large urban river systems is extremely sparse due to logistics and accessibility an occupancy modeling study was conducted on the resident birds of Delhi. River Yamuna passing through Delhi is a refuge for several resident and migratory species of birds but its water quality has been deteriorating due to ever growing population, contributing most of its pollution load in the urban center. The objectives of this study were 1) to determine whether the occupancy of common waterbirds varied between differential polluted sections of the river, 2) to better understand the associations between habitat variables and use of the river habitat by common waterbirds and 3) to identify whether any species or groups of species could be useful as indicators for habitat quality. The occupancy parameter Ψ was estimated for 17 common resident species among 68 species, recorded from 176 sampling locations during 2018, on the banks of river. Species occupancy was estimated as a function of habitat variables such as physico-chemical characteristic of water, solid waste, percent area of surface and emergent vegetation and human disturbance at each sampling site. The results suggest that Little Grebe (Tachybaptus ruficollis), Grey Heron (Ardea cinerea), Grey-headed Swamphen (Porphyrio poliocephalus), Eurasian Coot (Fulica atra), Common Moorhen (Gallinuala chloropus), Purple Heron (Ardea purpurea) and Little Cormorant (Microcarbo niger) are negatively associated with the Total Dissolved Solute (TDS). Interestingly, occupancy of Black-winged Stilt (Himantopus himantopus) is negatively associated with pH but positively with solid waste strongly suggesting its preference for the polluted sections of the river, possibly due to the increased availability of food. For river Yamuna, estimates of Ψ for various birds forms a baseline to study future trends. This study also indicates that species like Black-winged Stilt, can serve as an indicator of contaminated water bodies. The challenges posed by increasing urbanization and pollution to riparian bird habitats in urban areas can be effectively dealt with by incorporating such ecological knowledge with habitat restoration and conservation efforts.
Similar content being viewed by others

Avoid common mistakes on your manuscript.
Introduction
The rapid and often unplanned growth of urban centers throughout India has led to a significant decline in remnant habitat patches. The increasing trend of urbanization has resulted in considerable loss of biodiversity (Czech and Krausman 1997; Melliger et al. 2018), including birds (Aronson et al. 2014; Beissinger and Osborne 1982; Mills et al. 1988), leading to either local extinctions (Slabbekoorn 2013) or various species adapting towards their altered habitats (Møller et al. 2012). Being highly visible and active, birds are considered as excellent indicators of environmental changes (Bibby et al. 1992; Savard et al. 2000) and a robust assessment of mere presence/ absence information pertaining to a species or groups of species can be extremely meaningful. Wetlands are among the first to be affected by intensifying urbanization, encroachment, pollution, and other factors. Studies on urban rivers as habitat for biodiversity, particularly birds, is fast emerging as an area of interest, worldwide. Many urban waterbirds are intimately tied to local waterbodies for foraging (McClure et al. 2015; Urfi 2010) and breeding (Zipkin et al. 2009). Among studies attempting to evaluate the influence of habitat features on species diversity, is one on estuarine marshes along Connecticut River which indicated that breeding birds richness was positively associated with marshy areas and birds that used the habitat primarily for feeding preferred environmental heterogeneity, water cover and proximity to marsh habitat (Craig and Beal 1992). Indicator river birds have been linked to local and catchment-scale effects, such as river flow, water channel character, bank morphology, and land use of rivers (Sinha et al. 2019). Nagarajan and Thiyagesan (1996) found a correlation between water quality and waterbirds richness and abundance. Linkages between heavy metal pollution and breeding behaviour were observed in Great Tits Parus major (Janssens et al. 2003). All these studies indicate that the presence or absence of certain groups or species can provide meaningful information about wetland quality as they are intimately tied to their habitat. By and large, information regarding importance of urban riverine habitats for bird conservation is scarce as the vital issue of detectability is largely missing from them, raising concerns about the reliability of the studies. To assess the impacts of urbanization on wetland birds, conservation monitoring programs that incorporate ecological knowledge about diverse avian taxa are urgently required (Niemela 1999; Savard et al. 2000).
A robust estimation of detectability is the key to a proper assessment of distribution and abundance patterns of birds across a wide range of habitats (MacKenzie et al. 2006). Many multi-species monitoring studies (such as Khera et al. 2009; Sinha et al. 2019) rely on raw counts and thus incorporate an inadvertent assumption of complete detection or constant detection across the survey sites, which is problematic. In recent years occupancy modeling has established itself as an important tool to estimate site occupancy values in a robust manner and establish habitat relationships (Ametller et al. 2017; De Wan et al. 2009; Gibbs et al. 2017; Webb et al. 2017). Several recent studies have tended to highlight its utility in studies of urban biodiversity such as utilization of urban parks and forest by bird communities (Canedoli et al. 2018; Chibesa and Downs 2017), by taxa as diverse as mammals and salamanders (Weaver and Barrett 2018; Eakin et al. 2018) and, most recently, domestic cats (Vanek et al. 2021).
Delhi state in North India, which has a rich and well documented avian diversity (Urfi 2010) is undergoing rapid loss of natural habitats particularly wetlands (Urfi 2010). Among some recent studies dealing with impacts of urbanization on the birds of Delhi is a field investigation on Black Kite (Milvus migrans) which revealed that it preferred poorly managed waste dumping sites, high human density areas and a specific type of road configuration (Kumar et al. 2018). A study on House Sparrow (Passer domesticus) showed its preference for agricultural sites (Khera et al. 2009) over others in Delhi. Occupancy modeling studies on the terrestrial birds of the state revealed interesting differences in birds of different guilds between the central older parts of the capital city, with horizontal city expansion (known as Lutyen’s Delhi) characterized by low rise buildings, lower physical densities and high ground coverage, and peripheral areas with newer and vertical city expansion with lesser vegetation, high rise buildings, compact development, higher physical densities, small open spaces and low ground coverage (Tiwary and Urfi 2016).
Wetland birds of Delhi are at risk too and studies have documented a decline in species diversity, and the local extinction of flagship species such as the Sarus Crane (Grus antigone) (Urfi 2010). Different species of birds appear to be remarkably diverse in their ability to withstand pollution and how this may be linked to their food, foraging behavior and diet needs a detailed investigation. While the river Yamuna that flows through Delhi provides an excellent habitat for waterbirds in this region it is also regarded as one of the most polluted rivers in India with the Delhi stretch being the major contributor to its pollution load. There is a striking difference in water quality as soon as it enters through Wazirabad, on its northern periphery. Hence, the River can be divided into two sections with pollution drastically increasing south of Wazirabad. Since, robust scientific information on wetland birds is largely missing from the state as it is often difficult to put a finger on a precise cause for their decline i.e. loss of habitat, pollution or something else and to formulate mitigation strategies or to identify which species are most at risk. Although there have been attempts to assess the pollution load (Misra 2010; CPCB 2016) of the river Yamuna in Delhi, which is the life line of the city, a complete and thorough estimation of the factors affecting riverine birds is missing. Hence, the stark differences in water quality and other parameters in the two segments of the river i.e. before entry into Delhi and in Delhi stretch, offers us a unique opportunity to address some of these questions.
In an earlier study (Tiwary and Urfi 2016), we used occupancy modeling to study the distribution of terrestrial birds within the urban matrix of Delhi. In an extension of this study here we explore the occupancy of wetland birds along the river Yamuna passing through Delhi State with habitat specific covariates. We attempt to obtain a robust estimation of occupancy probability using single-season occupancy models for different species. The key objectives of this study were 1) to determine whether the occupancy of common waterbirds differed between the two river sections i.e. before and after Wazirabad, 2) to better understand the associations between habitat variables and use of the river habitat by common waterbirds in River Yamuna and 3) to determine whether any species or groups of species could be useful as indicators for habitat quality. Since occupancy data are of vital importance in devising conservation strategies the robust estimation of this information can greatly determine the validity and effectiveness of various conservation schemes. The occupancy estimates of various species of birds that emerge from the present study will serve as baseline information for conservation monitoring of the common wetland birds of the Delhi region. We discuss our results in context of the potential of occupancy modeling for conservation studies and city planning, emphasizing the importance of wetland habitats with respect to birds (Walker et al. 2008).
Materials and methods
Study area
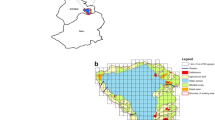
The Delhi region broadly falls in the area lying at the confluence of the semi-arid Punjab plains and upper Gangetic plains (Tiwary and Urfi 2016; Urfi 2010) in north India (Fig. 1). Delhi’s urban area with neighboring satellite cities of Ghaziabad, Gurugram, Faridabad and Noida constitute the National Capital Region (NCR) with an overall population of 26 million (United Nations 2016). The river Yamuna which is 1376 km long, from its starting point in Yamunotri, to its merger with the river Ganges in Prayagraj has its most polluted stretch in the state of Delhi (Urfi 2010). We sampled a 102 km stretch starting from Sonipat in Haryana state till Greater Noida in Uttar Pradesh state, including the 48 km long stretch flowing through Delhi. From the point when the river enters Delhi (at Wazirabad) to the point where it exits the state at Okhla, there is a marked deterioration in its quality (CPCB 2016). Based on this observation, we have divided the sampled region into two sections (See Fig. 1). Section 1 stretch is the one which lies upstream of Wazirabad in which the river is comparatively cleaner (N = 76) and Section 2, following it is the visibly deteriorated river with poor water quality (N = 100). Recent construction activities on the floodplain of the river Yamuna, especially those during the Commonwealth Games during 2010 (Urfi 2010) have resulted in considerable loss of natural habitats, particularly in Section 2 (National Wetland Atlas 2011).
Study Area showing sampling sites throughout the stretch of River Yamuna passing through Delhi. Section 1 (▲) comprises the stretch of river before it enters the state of Delhi at Wazirabad and section 2 (●) comprises the stretch beyond, through the territory of Delhi state, including sampling areas downstream lying outside Delhi. Inset shows the location of Delhi in India
Field study design and bird survey
Our study area was a 102-km stretch of the Yamuna River, including the Delhi section as well as contiguous sections up and downstream (Fig. 1). We randomly placed multiple sites along the stretch of river using maps obtained from Google Earth pro and selected 176 sites for data recording based on their accessibility. Each site was placed at least 300–500 m apart in order to avoid repeat counting of a single bird occupying multiple sites (Latif et al. 2016). We used the distance function in Google Earth pro to confirm the distance between the marked sampling sites. Two primary surveys were conducted during 10 January to 5 April in 2018, each 2 to 3 days apart to obtain temporal replicates, treating replicate visits within a year as repeated surveys. Waterbird species within 50 m around the point of observation (including both river stream and river bank habitat) were recorded using a binocular (15 × 25) and camera, for 10 min at each point (Bibby et al. 1992). All points were covered by a single observer between 07.00–11.00 AM on clear days. The scientific names of birds (Gill et al. 2020) included in this study and their food habits (Ali and Ripley 1987) provided in Online Resource 1.
Estimation of habitat variables
We assumed that water quality of the river will influence the utilization of habitat by birds in the region. We were particularly interested in understanding what role it plays in presence or absence of bird species in the habitat. Since we assumed that species would avoid polluted waters or sub-optimal habitat, depending upon its food preferences therefore we measured physio-chemical parameters of water including Dissolved Oxygen (DO) (mg/l), pH (PH), Total Dissolved Solutes (TDS) (ppm), Conductivity (CON) (mS/cm), Resistivity (RES)(Ωcm), Oxidation-reduction potential (ORP)(mV) using a handheld water analysis meter (HI 769828 Multiparameter Probe, Hanna Instruments, USA). These water quality parameters were recorded on both the surveys and finally averaged to be used as a single covariate to model occupancy (Nagarajan and Thiyagesan 1996). The instrument was calibrated using standard solutions before use.
The percent cover of aquatic vegetation (VEG) was estimated in a 50 m plot surrounding the survey location during each visit (Conway 2009; Darrah and Krementz 2010). Aquatic vegetation described as emergent vegetation and floating vegetation was visually ascertained as survey specific covariate. Human disturbance (HD) index was also calculated as survey specific covariate in which the total number of people were counted on the river bank, during each visit to the site. The count was done for a duration of 10 min during the transect by an additional observer. We finally averaged the recordings of VEG and HD of the two visits to be used as a single covariate in the analysis. We also observed that often the river was used as a dumping site (municipal waste), cremation ground and sometimes waste disposal from cultural rituals (flowers, earthen pots, sculptures) and recreational purposes. This waste often gets washed off the banks of the river. Hence, we calculated solid waste (SW) as a categorical variable with varying scores, where 0 stands for no solid waste, 1 for moderate and 2 for high solid waste at the site in which most of the bank was covered by it. Before using these covariates in our analysis, we checked all of them for any possible correlation (Pearson’s Correlation, r > 0.8). Only those covariates were retained in the final analysis that were not correlated with others. Ultimately, DO, PH, TDS, VEG, HD and SW were used as covariates for further analysis.
Occupancy modeling
We used single season occupancy model (MacKenzie et al. 2002) in Program Presence ver. 5.8 (Hines 2006) for modeling occupancy and detection probabilities. For each species, we created an encounter history matrix (176 site rows by 2 survey columns) with binary detection/non-detection data summarized for each site x survey combination. We used single-species occupancy modeling in Presence with maximum likelihood and logit link to model relationships between covariance and occupancy (psi) and detectability (p). The present study was restricted only to the resident species of Delhi and all winter and summer migrants were excluded from the analysis, thus avoiding any possibility of change in occupancy within a sampling site during the study period, due to immigration or emigration. This is a step towards meeting the closure assumption of occupancy modeling as explained in Mackenzie et al. (2002) i.e. the system must be demographically closed to changes in occupancy status during the sampling period. Replicate surveys were conducted as close in time as possible to minimize violations of the closure assumption (MacKenzie et al. 2006; Rota et al. 2009).
To account for the effect of habitat variables we modeled occupancy as a function of various covariates recorded from the sampling sites (DO, PH, TDS, VEG, HD, SW).
Model selection
We first developed the baseline model for each species where both occupancy probability (ψ) and detection probability (p) were assumed to be constant across all sampling sites [Constant model ψ(.) p(.)]. To account for the variation in detection probability, we modeled p as a function of human disturbance (HD) and solid waste (SW) at the sampling site because we assumed that disturbance and the presence of solid waste will affect the site visibility and could influence detection probability. We also assumed that the variation in survey-specific percent vegetation (VEG) may also affect the probability of detecting the species (Mackenzie et al. 2006). Then, we developed competing models to assess the factors responsible for variations in the detection probability. Among the several models built, in some p was assumed to be affected by VEG, HD, SW variables singly (univariate model) and in different combinations (multivariate model). We fitted all univariate models and multivariable models for p. Models assuming constant p across sampling sites were often ranked among the best models, indicating homogeneity and detection probability <1.
The best performing model from this analysis was used for modeling ψ as a function of various habitat covariates dissolved oxygen (DO), pH (PH), total dissolved solute (TDS), percentage of vegetation (VEG), human disturbance (HD) and solid waste (SW). First, we fitted all univariate models and then fitted multivariate models with select combinations using best performer covariate models during univariate model analysis. We used a multi-model approach to assess if one or more models stood out to be the best, based on Akaike Information Criteria (Δ AIC < 2) (Burnham and Anderson 2002), and then derived the estimates of covariates using the top model if there was no evidence of model uncertainty. In cases where model uncertainty was evident, model-averaging was employed for all the models with Δ AIC < 2 (Table 2). Site occupancy estimates for before and after Wazirabad (Fig. 1) were separately averaged from top model (Fig. 2).
We conducted the analysis for all the bird species except migratory species (closure assumption) detected during the sampling. However, it was observed that due to insufficient presence/ absence data of some species, the occupancy value was inestimable and the beta values could not be interpreted. Hence, those species were dropped from the result presentation.
Results
As previous studies have shown, most of the water quality parameters deteriorate sharply once the river Yamuna enters the state of Delhi (from section 1 to section 2, before Delhi and Delhi & beyond stretches, respectively). These include a sharp decline in the dissolved oxygen content of the water, which was also observed in our study (Table 1). During our study a total of 68 species of birds were recorded from the 176 sampling locations along the river Yamuna (Online Resource 1). Due to insufficient detection history, analysis could not be conducted for a few rare resident species. Hence, occupancy parameter (Ψ) and coefficients of covariates showing the relationship of species occupancy with habitat variables could be estimated for 17 species of resident birds. Top model/s for all the species is provided in Table 2 along with their model AIC weight/s (sum of all the model weights in case of model uncertainty) and extended details such as number of parameters, −2 logLikelihood, delta_AIC, Individual model wt are provided in Online Resource 3. The model averaged estimates of occupancy generated for the waterbird species were highly variable Table 3. Species like Black-winged Stilt (Himantopus himantopus), Common Sandpiper (Actitis hypoleucos), Grey Heron (Ardea cinerea) and Common Redshank (Tringa totanus) were the most abundant and widespread waterbirds in the study area (ψ ≥ 0.75). Other species with relatively high occupancy were Red-wattled Lapwing (Vanellus indicus) and Little Cormorant (Microcarbo niger) whose ψ value ranged between 0.50 to 0.75. The occupancy of Little Grebe (Tachybaptus ruficollis), Great Cormorant (Phalacrocorax carbo), Red-naped Ibis (Pseudibis papillosa), Purple Heron (Ardea purpurea), Common Moorhen (Gallinula chloropus) and White-throated Kingfisher (Halcyon smyrnensis) was considerably low (ψ ≤ 3.0) (Table 3).
We separately averaged site level occupancy estimates from the top models of some species for the two different sections of the river (Fig. 2). The occupancy value of species like Black-winged Stilt, Common Redshank, Red-naped Ibis, Red-wattled lapwing and Indian Pond Heron (Ardeola grayii) was higher in Section 2 of the river suggesting that they are more associated with polluted waterbodies. Although the occupancy of Red-naped Ibis, (Table 3) was quite low, it showed preference towards the polluted waters (Fig. 2). Interestingly Black-winged Stilt whose occupancy was quite high (Table 3) throughout the stretch showed a similar trend. This species also frequented contaminated aquatic habitats in the lower sections of the river i.e. section 2. Remaining 13 birds showed preference towards comparatively clean water, among them Eurasian Coot (Fulica atra), Great Cormorant and Little Cormorant both essentially fish dependent species (Online Resource 1), showed considerable difference in the occupancy (Fig. 2).
Among the 17 species, interesting relationships were observed between modeled occupancy and habitat covariates. The occupancy value of Black-winged Stilt exhibited a decreasing trend in relation to PH of the sampled habitat but an increasing trend with SW around the sampling site (Fig. 4). Eurasian Coot shows an increasing occupancy with rise in DO level and decreasing occupancy with PH and TDS (Fig. 3). Occupancy of Common Sandpiper shows an increasing trend with DO of the river water and SW around the sampling site, however decreasing trend with PH (Fig. 3). Little Cormorant exhibited a negative association with PH, HD and TDS and positive trend towards DO (Fig. 3).
Indian Pond Heron showed a positive association with percent vegetation and human disturbance whereas Grey-headed Swamphen (Porphyrio poliocephalus) exhibited positive association with VEG and negative with HD suggesting that it avoided crowded areas (Fig. 4). Great Cormorant on the other hand exhibited positive association with DO and negative association with SW implying that it avoided polluted areas (Fig. 4). In our study, the pH at all the sampling sites ranged from 6.63 to 10.70 (Table 1) and we found that while in the case of Common Moorhen occupancy was positively associated with PH, preferring close to neutral waters, occupancy of Black winged Stilt, River Lapwing (Vanellus duvaucelii), Common Sandpiper, Eurasian Coot, Red-naped Ibis, Little Cormorant was negatively associated with PH (Table 3). Interestingly, Little Grebe, Purple Heron and Little Egret (Egretta garzetta) is negatively associated with TDS (Fig. 5) which was quite high in the water body (Table 1) suggesting that they avoid polluted waters. However, White-throated Kingfisher was not associated with any covariate (Table 3) according to the top model.
A forest plot showing beta coefficient ± SE (x-axis), the association of the species occupancy ± SE with the habitat variable for select species. (DO = Dissolved Oxygen of water, PH = pH of water, TDS = Total Dissolved Solute of water, VEG = % area covered by Emergent and Floating Vegetation, HD = Human Disturbance, SW = Solid Waste)
Discussion
Our results demonstrate apparent differences in occupancy of select species of birds in the two different sections of the river viz. the relatively clean section 1 and the polluted section 2, lying in the Delhi and further downstream stretch. This suggests that some species which appear to be resilient to polluted waters in urban systems could serve as ecological indicators, the prime example of which, as it emerges from our study is the Black-winged Stilt (Fig.5).
Various top models for different species indicated that occupancy of those birds might be dependent on the water quality of the river, though other factors could be important. Availability of food often drives birds towards the waterbody (McNicol and Wayland 1992; McClure et al. 2015). The positive correlation of habitat use with comparatively high DO in water by piscivorous birds like Little Cormorant and Great Cormorant suggests the same, as the occurrence of fishes in water is greatly influenced by its oxygen content (Pink and Abrahams 2018). Food occurrence related factors are likely to explain the positive correlation between DO and the local presence of Eurasian Coot and Common Sandpiper. While the former feeds on aquatic vegetation the latter probes in the mud and picks up invertebrates. However, Red-naped Ibis and Red-wattled Lapwing, negatively associated with DO, are birds that are often found in open agricultural fields and not entirely dependent on water bodies for food (Ali and Ripley 1987). In the present investigation, TDS was negatively associated with the use of wetland by Little Grebe, Common Moorhen, Grey-headed Swamphen, Purple Heron, Grey Heron, Eurasian Coot and Little Cormorant. TDS influences the salinity and conductivity of the water body (Rameshkumar et al. 2019). It can also affect the taste of water and soil structure, aeration and permeability which can be responsible for indirect effect on the growth of aquatic flora along the coastline (Ali et al. 2019). The nature of relationships between habitat covariates affecting food supplies can be complex. For instance, in a study by Pink and Abrahams (2018) variations in DO levels affected feeding activity by Forster’s Terns (Sterna forsteri). They postulated that the energy budgets for adult Forster’s terns and subsequent fledging of the chicks is dependent on the DO content of the water body as their prey use deeper waters as a refuge from predation. Hence, the terns avoided hypoxic water. However, in the present context more studies would be required before the exact relationship between the food resources of individual species of birds and water quality parameters can be established.
Sites with pH extremes, which is likely to adversely impact the food supply and hence will be avoided by birds would be a reasonable expectation. Significant correlation of pH with waterbird richness, abundance and density have been reported in Pichavaram Wetlands, Southern India (Nagarajan and Thiyagesan 1996). Waterbird use of wetland and pH has been correlated in Lakes of Sadbury Ontario (McNicol and Wayland 1992) in which pH influenced the habitat selection by insectivorous birds. In our study, most of the birds showed negative correlation with pH (Fig. 3). This would be logical to expect given that birds would avoid basic waters as such sites are likely to be those where sewage content is high, and likely to influence presence of phytoplankton, macro and micro invertebrates, amphibians and fish (McNicol and Wayland 1992; Pierce 1993; Brown 1982). Waterbird species should respond to variation in pH in case they depend on these organisms as prey.
In four species the amount of vegetation present at the sites was found to be the factor influencing use of the water body. In the case of Indian Pond Heron and Great Cormorant the relationship with vegetation was unexpectedly found to be positive but in the case of River Lapwing it was negative. The latter being a wader feeding on invertebrates in the ground is likely to prefer cleaner, non-vegetated sites. However, the preference towards vegetated areas in case of Grey-headed Swamphen is expected as the species use such habitats for breeding and feeding (Fig. 3). While Black-winged Stilt, Indian Pond Heron and Purple Heron were positively associated with human disturbance, both the Little Egret and Grey-headed Swamphen seemed to avoid high human density areas (Table 3). Black-winged Stilt, Common Sandpiper, Common Redshank and Little Egret were found to be positively associated with the solid waste probably because of the availability of food, which in turn helps them to survive in these habitats.
Different species of birds themselves appear to be remarkably diverse in their ability to withstand urbanization. Also, while some birds are regarded as synanthropic or human-tolerant species (Blair 1996; Marzluff 2001; Tratalos et al. 2007), others are reportedly less so. Over the years, increase in high proportion of some species can be attributed to an adaptation to disturbances within the habitat (Gray and Mirza 1979). Although disturbance there seem to be variations in terms of species susceptibility to human presence as well as its ability to get used to disturbance. For instance, in a study of Oystercatchers (Haematopus ostralegus) overwintering on the Exe estuary Urfi et al. (1996) found that in high human density areas such as the Exmouth beach Oystercatchers had larger reactive distances compared to those areas of the estuary (say mussel beds on the estuary) where they did not frequently encounter humans and so allowed closer approach. Susceptibility to disturbance can also vary at over wintering and nesting grounds in the case of migratory birds. In this regard the Ruddy Sheldrake (Tadorna ferruginea) which breeds in Ladakh in north India but overwinters across many parts of the Indian plains is a case in point. While it is extremely tame and allows close human approach in Ladakh, in the wintering grounds it is extremely vary and takes off at the slightest provocation (Ali and Ripley 1987).
With respect to the question, whether water quality parameters have a direct bearing on birds, the existing paradigm is that in most cases they influence the food resources. However, birds can be found in polluted water bodies because eutrophication increases the food available in specific cases (Van Impe 1985), a fact borne out by several studies (Campbell 1984; Pounder 1976; Shutes 2001). But another matter which needs investigation is the extent to which the polluted food is affecting the metabolism of the birds (and eventually fitness) in the long run, even though in the short run the birds seem to be fulfilling their dietary or energetic requirements.
Methodological caveats
We have estimated a wide range of habitat variables related to water quality, human disturbance and habitat quality (percent vegetation and visible solid waste) and modeled the species occupancy as a function of these. However, there may be a case that food availability for waterbirds belonging to different feeding guilds can also affect their occupancy. Measuring this parameter was a major logistic challenge for a large river system in a multispecies study. We believe that water quality can explain the variation in food availability up to some extent and the occupancy estimates and habitat associations arising out of this study remains free from any major confounding factor and are meaningful in better understanding both this study system and urban river systems in general. The water quality parameters could be considered as a snapshot measurement of covariates but given the nature of the study the same could not be estimated in any other manner (one recalls a statement by a Greek philosopher, Heraclitus that one cannot step into the same river twice). Other studies have used traffic noise as covariate in estimating occupancy patterns of forest birds (Goodwin and Shriver 2011), another study has used relative abundance index of humans as snapshot measurement to estimate occupancy of Trumpeter Hornbill (Bycanistes bucinator) (Chibesa and Downs 2017). As mentioned, in our study we have taken an average of parameters measured during the repeat surveys.
Conservation
The demarcation into pre and post Wazirabad phase with respect to water quality parameters is really stark and to a large extent it reflects in occupancy estimates of different species of birds. As our study shows, factors related to human disturbance, loss of habitat due to encroachment of riparian habitat and water quality are important in explaining occurrence of different species of birds. Much of the differences in the water quality of the river are due to the presence of drains that pour their effluents into the river. Normally, with the flow of the river, the pollutants would get diluted but the flow of the river is under the control of the irrigation department and is erratic (Urfi 2003). Very often the flow of the river is extremely slow which does not cause the flushing out of the industrial effluent that is poured into the river. While the Government of Delhi has designed comprehensive plans to establish many waste water treatment plants for cleaning River Yamuna, it remains to be seen how effective such steps will prove in the future. The encroachments on the river by way of creating built up areas, bridges etc. is also contributing to the disappearance of natural habitats for birds and other biodiversity and there is a need to check this (Urfi 2010).
References
Ali S, Ripley SD (1987) Handbook of the birds of India and Pakistan. Oxford University Press, New Delhi
Ali N, Khan MU, Khoso SA (2019) Identification of biological threats zones along the coastline of Karachi. Int J Environ Sci Technol 16:3557–3564. https://doi.org/10.1007/s13762-018-1884-7
Ametller HT, Hernandez-Matias A, Pretus JLL, Real J (2017) Landfills determine the distribution of an expanding breeding population of the endangered Egyptian vulture Neophron percnopterus. Ibis 159:757–768
Aronson MFJ, La Sorte FA, Nilon CH, Katti M, Goddard MA, Lepczyk CA, Warren PS, Williams NG, Cilliers S, Clarkson B, Dobbs C, Dolan R, Hedblom M, Klotz S, Kooijmans JL, Kühn I, MacGregor-Fors I, McDonnell M, Mörtberg U, Pyšek P, Siebert S, Sushinsky J, Werner P, Winter M (2014) A global analysis of the impacts of urbanization on bird and plant diversity reveals key anthropogenic drivers. Proc R Soc B281:20133330
Beissinger SR, Osborne DR (1982) Effects of urbanization on avian community organization. Condor 84:75–83
Bibby C, Burgess ND, Hill DA (1992) Bird Census Techniques. Academic Press, London
Blair RB (1996) Land use and avian species diversity along an urban gradient. Ecol Appl 6:506–519
Brown DJA (1982) The effect of pH and calcium on fish and fisheries. Water Air Soil Pollut 18:343–351. https://doi.org/10.1007/BF02419422
Burnham KP, Anderson DR (2002) Model selection and multi-model inference. A practical information-theoretic approach, 2nd edn. Springer, New York. https://doi.org/10.1007/b97636
Campbell LH (1984) The impact of changes in sewage treatment on seaducks wintering in the Firth of Forth, Scotland. Biol Conserv 28(2):173–180
Canedoli C, Manenti R, Padoa-Schioppa E (2018) Birds biodiversity in urban and periurban forests: environmental determinants at local and landscape scales. Urban Ecosyst 21:779–793. https://doi.org/10.1007/s11252-018-0757-7
Chibesa M, Downs CT (2017) Factors determining the occupancy of trumpeter hornbills in urban-forest mosaics of KwaZulu-Natal, South Africa. Urban Ecosystems 20:1027–1034. https://doi.org/10.1007/s11252-017-0656-3
Conway CJ (2009) Standardized north American marsh bird monitoring protocols, version 2009-2. Wildlife research report #2009-02. U.S. Geological Survey. Arizona cooperative fish and wildlife research unit, Tuscon
CPCB (Central Pollution Control Board) (2016) Water quality data of River. Central Pollution Control Board, Delhi https://cpcb.nic.in Accessed on 05 August 2019
Craig RJ, Beal KG (1992) The influence of habitat variables on marsh bird communities of the Connecticut River estuary. Wilson Bulletin 104(2):295–311 www.jstor.org/stable/4163153
Czech B, Krausman PR (1997) Distribution and causation of species endangerment in the United States. Science 277:1116–1117
Darrah AJ, Krementz DG (2010) Occupancy and habitat use of the least bittern and pied-billed grebe in the Illinois and upper Mississippi River valleys. Waterbirds 33(3):367–375. https://doi.org/10.1675/063.033.0314
De Wan AA, Sullivan PJ, Lembo AJ, Smith CR, Maerz JC, Lassoie JP, Richmond ME (2009) Using occupancy models of forest breeding birds to prioritize conservation planning. Biol Conserv 142(5):982–991
Eakin CJ, Hunter M, Calhoun A (2018) Bird and mammal use of vernal pools along an urban development gradient. Urban Ecosyst 21:1029–1041. https://doi.org/10.1007/s11252-018-0782-6
Gibbs JP, Rouhani S, Shams L (2017) Scale-dependence in polychlorinated biphenyl (PCB) exposure effects on waterbird habitat occupancy. Ecotoxicology 26:762–771
Gill F, Donsker D, Rasmussen P (2020) IOC world bird list (v10.1). https://doi.org/10.14344/ioc.ml.10.1
Goodwin SE, Shriver WG (2011) Effects of traffic noise on occupancy patterns of forest birds. Conserv Biol 25(2):406–411 https://www.jstor.org/stable/27976476
Gray JS, Mirza FB (1979) A possible method for detecting pollution induced disturbance on marine benthic communities. Mar Pollut Bull 10:142–146. https://doi.org/10.1016/0025-326X(79)90082-1
Hines J (2006) Presence. Software to estimate patch occupancy and related parameters. USGS, Patuxent wildlife research center, Laurel, http://www.mbr-pwrc.usgs.gov/software/presence.html accessed 15 may 2013
Janssens E, Dauwe T, Pinxten R, Eens M (2003) Breeding performance of great tits (Parus major) along a gradient of heavy metal pollution. Environ Toxicol Chem 22(5):1140–1145
Khera N, Mehta V, Sabata BC (2009) Interrelationship of birds and habitat features in urban greenspaces in Delhi, India. Urban Forestry Urban Green 8:187–196
Kumar N, Gupta U, Jhala YV, Qureshi Q, Gosler AG, Sergio F (2018) Habitat selection by an avian top predator in the tropical megacity of Delhi: human activities and socio-religious practices as prey-facilitating tools. Urban Ecosyst 21:339–349. https://doi.org/10.1007/s11252-017-0716-8
Latif QS, Ellis MM, Amundson CL (2016) A broader definition of occupancy: comment on Hayes and Monfils. J Wildl Manag 80(2):192–194
MacKenzie DI, Nichols JD, Lachman GB, Droege S, Royle JA, Langtimm CA (2002) Estimating site occupancy rates when detection probabilities are less than one. Ecology 83:2248–2245
MacKenzie DI, Nichols JD, Royle JA, Pollock KH, Bailey LA, Hines JE (2006) Occupancy modeling and estimation. Academic Press, San Diego
Marzluff JM (2001) Worldwide urbanisation and its effects on birds. In: Marzluff JM, Bowman R, Donnelly R (eds) Avian ecology and conservation in an urbanising world. Springer Science, New York, pp 19–47
McClure CJ, Korte AC, Heath JA, Barber JR (2015) Pavement and riparian forest shape the bird community along an urban river corridor. Global Ecol Conserv 4:291–310. https://doi.org/10.1016/j.gecco.2015.07.004
McNicol DK, Wayland M (1992) Distribution of waterfowl broods in Sudbury Area Lakes in relation to fish, macroinvertebrates, and water chemistry. Can J Fish Aquat Sci 49(S1):122–133. https://doi.org/10.1139/f92-307
Melliger RL, Braschler B, Rusterholz HP, Baur B (2018) Diverse effects of degree of urbanisation and forest size on species richness and functional diversity of plants and ground surface-active ants and spiders. PLoS One 13:15
Mills G, Dunning J, Bates J (1988) Effects of urbanization on breeding bird community structure in Southwestern Desert habitats. Condor 91:416–428
Misra AK (2010) A river about to die: Yamuna. J Water Resource Protect 2:489–500
Møller AP, Diaz M, Flensted-Jensen E, Grim T, Ibáñez-Álamo JD, Jokimäki J, Mänd R, Markó G, Tryjanowski P (2012) High urban population density of birds reflects their timing of urbanization. Oecologia 170:867–875. https://doi.org/10.1007/s00442-012-2355-3
Nagarajan R, Thiyagesan K (1996) Waterbirds and substrate quality of Pichavaram wetlands, southern India. Ibis 138:710–721
National Wetland Atlas (2011) SAC/EPSA/ABHG/NWIA/ATLAS/34/2011, space applications Centre (ISRO), Ahmedabad, India 310p http://saconenvis.nic.in/publication%5CNWIA_National_atlas.pdf accessed on 24 June, 2020
Niemela J (1999) Ecology and urban planning. Biodivers Conserv 8:119–131
Pierce BA (1993) The effects of acid precipitation on amphibians. Ecotoxicology 2:65–77. https://doi.org/10.1007/BF00058215
Pink M, Abrahams MV (2018) In shallow water ecosytems the abiotic environment is more important than prey abundance for foraging terns. Environ Biol Fish 101:355–362
Pounder B (1976) Wintering flocks of goldeneyes at sewage outfalls in the Tay estuary. Bird Study 23:121–131
Rameshkumar S, Radhakrishnan K, Aanand S, Rajaram R (2019) Influence of physicochemical water quality on aquatic macrophyte diversity in seasonal wetlands. Appl Water Sci 9:12. https://doi.org/10.1007/s13201-018-0888-2
Rota CT, Fletcher RJ Jr, Dorazio RM, Betts MG (2009) Occupancy estimation and the closure assumption. J Appl Ecol 46(6):1173–1181. https://doi.org/10.1111/j.1365-664.2009.01734.x
Savard JPL, Clergeau P, Mennechez G (2000) Biodiversity concepts and urban ecosystems. Landsc Urban Plan 48:131–142
Shutes RBE (2001) Artificial wetlands and water quality improvement. Environ Int 26(5):441–447
Sinha A, Chatterjee N, Ormerod SJ, Adhikari BS, Krishnamurthy R (2019) River birds as potential indicators of local- and catchment-scale influences on Himalayan river ecosystems. Ecosyst People 15(1):90–101. https://doi.org/10.1080/26395916.2019.1591508
Slabbekoorn H (2013) Songs of the city: noise-dependent spectral plasticity in the acoustic phenotype of urban birds. Anim Behav 85(5):1089–1099. https://doi.org/10.1016/j.anbehav.2013.01.021
Tiwary NK, Urfi AJ (2016) Spatial variations of bird occupancy in Delhi: the significance of woodland habitat patches in urban centres. Urban For Urban Green 20:338–334
Tratalos J, Fuller RA, Warren PH, Davies RG, Gaston K (2007) Urban form, biodiversity potential and ecosystem services. Landscape Urban Plann 83:308–317
United Nations, Department of Economic and Social Affairs, Population Division (2016) The World’s Cities in 2016 – Data Booklet (ST/ESA/SER.A/392) https://www.un.org/en/development/desa/population/publications/pdf/urbanization/the_worlds_cities_in_2016_data_booklet.pdf Accessed on 06 June 2020
Urfi AJ (2003) The birds of Okhla barrage bird sanctuary, Delhi, India.Forktail 19:39–50
Urfi AJ (2010) Using heronry birds to monitor urbanization impacts: a case study of painted stork Mycteria leucocephala nesting in the Delhi zoo, India. Ambio 39:190–193
Urfi AJ, Goss-Custard JD, Durell SEA (1996) The ability of oystercatchers Haematopus ostralegus to compensate for lost feeding time: field studies on individually marked birds. J Appl Ecol 33:873–883
Van Impe J (1985) Estuarine pollution as a probable cause of increase of estuarine birds. Marine Pollution Bull 16:271–276
Vanek JP, Rutter AU, Preuss TS, Jones HP, Glowacki GA (2021) Anthropogenic factors influence the occupancy of an invasive carnivore in a suburban preserve system. Urban Ecosyst 24:113–126. https://doi.org/10.1007/s11252-020-01026-x
Walker JS, Balling RC Jr, Briggs JM, Katti M, Paige SW, Wentz EA (2008) Birds of a feather: interpolating distribution patterns of urban birds. Comput Environ Urban Syst 32:19–28
Weaver N, Barrett K (2018) In-stream habitat predicts salamander occupancy and abundance better than landscape-scale factors within exurban watersheds in a global diversity hotspot. Urban Ecosyst 21:97–105. https://doi.org/10.1007/s11252-017-0694-x
Webb MH, Terauds A, Tulloch A, Bell P, Stojanovic D, Heinsohn R (2017) The importance of incorporating functional habitats into conservation planning for highly mobile species in dynamic systems. Conserv Biol 31(5):1018–1028. https://doi.org/10.1111/cobi.12899
Zipkin EF, DeWan A, Royle A (2009) Impacts of forest fragmentation on species richness: a hierarchical approach to community modelling. J Appl Ecol 46:815–822. https://doi.org/10.1111/j.1365-2664.2009.01664.x
Acknowledgements
Sana Rehman thanks the Waterbird Society, USA for partial funds to attend the Waterbird Society 43rd Annual Meeting 2019 to discuss this work and colleagues at Rajdhani College for their encouragement. Nawin Kumar Tiwary thanks Indraprastha College for Women, University of Delhi for the award of a research project enabling him to complete some parts of the fieldwork. A J Urfi thanks the University of Delhi for funds under various schemes (Department of Science & Technology, India-purse grant, Research & Development grant and Teaching & Research grant). We thank Praveen Thakur and Paritosh Ahmed for their help in field work. We also thank the two anonymous referees and the associate editor for their constructive comments in the earlier versions of the manuscript.
Availability of data and materials
‘Not applicable’.
Funding
The research leading to these results received funding from University of Delhi under various schemes (‘Department of Science & Technology, India-purse grant’, ‘Research & Development grant’ and ‘Teaching & Research grant’.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Data collection and analysis were performed by Sana Rehman. The first draft of the manuscript was written by Sana Rehman and all authors commented on previous versions of the manuscript. Nawin K. Tiwary provided assistance in the analysis and creating the final draft. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The authors have no relevant financial or non-financial interests to disclose. All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript.
Consent for publication
On behalf of all the co-authors, the ‘corresponding author gives the consent for publication of this manuscript.
Competing interests
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Rights and permissions
About this article
Cite this article
Rehman, S., Tiwary, N.K. & Urfi, A.J. Conservation monitoring of a polluted urban river: an occupancy modeling study of birds in the Yamuna of Delhi. Urban Ecosyst 24, 1399–1411 (2021). https://doi.org/10.1007/s11252-021-01127-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11252-021-01127-1