Abstract
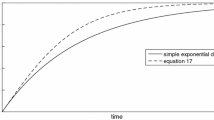
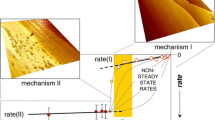
The kinetics and mechanism of calcite dissolution at various pH are investigated. A scheme of the ion distribution in various states of the medium is presented. The kinetic curves of dissolution are modeled, taking into account the structure of the electrical double layer and the acid-base characteristics of calcite.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
REFERENCES
Legrand, L., Savoye, S., Chausse, A., and Messina, R., Study of Oxidation Products Formed on Iron in Solutions Containing Bicarbonate/Carbonate, Electrochim. Acta, 2000, vol. 46, p. 111.
Videm, K. and Dugstad, A., Corrosion of Carbon Steel in an Aqueous Carbon Dioxide Environment, Mater. Perform., 1989, vol. 28, no.4, p. 46.
Sjoberg, E.L., A Fundamental Equation for Calcite Dissolution Kinetics, Geochim. Cosmochim. Acta, 1976, vol. 40, p. 441.
Jensen, D.L., The Solubility of Rhodochrosite (MnCO3) and Siderite (FeCO3) in Anaerobic Aquatic Environments, Appl. Geochem., 2002, vol. 17, p. 503.
Lim, T., Hwang, E.R., and Ha, H., Effects of Temperature and Partial Pressure of CO2/O2 on Corrosion Behaviour of Stainless-Steel in Molten Li/Na Carbonate Salt, J. Power Sources, 2000, vol. 89, p. 1.
Fredd, C.N. and Fogler, H.S., The Kinetics of Calcite Dissolution in Acetic Acid Solutions, Chem. Eng. Sci., 1998, vol. 53, p. 3863.
Liang, Y., Baer, D.R., and McCoy, J.M., Dissolution Kinetics at the Calcite-Water Interface, Geochim. Cosmochim. Acta, 1996, vol. 60, p. 4883.
Sjoberg, E.L. and Rickard, D., Calcite Dissolution Kinetics: Surface Speciation and the Origin of the Variable pH Dependence, Chem. Geol., 1984, vol. 42, p. 119.
Sjoberg, E.L. and Rickard, D., Temperature Dependence of Calcite Dissolution Kinetics between 1 and 62°C at pH 2.7 to 8.4 in Aqueous Solutions, Geochim. Cosmochim. Acta, 1984, vol. 48, p. 485.
Morse, J.W., The Kinetics of Calcium Carbonate Dissolution and Precipitation, in Carbonates: Mineralogy and Chemistry, Reeder, R.J., Ed., vol. 11 of Reviews in Mineralogy, Mineralogical Society of America, 1983, p. 227.
Rickard, D. and Sjoberg, E.L., Mixed Kinetic Control of Calcite Dissolution Rates, Am. J. Sci., 1983, vol. 283, p. 815.
Plummer, L.N., Wigley, T.M.L., and Parkhurst, D.L., A Critical Review of the Kinetics of Calcite Dissolution and Precipitation, in Chemical Modelling of Aqueous Systems, Jenne, E.A., Ed., Am. Chem. Soc. Symp. Ser., 1979, vol. 36, p. 538.
Plummer, L.N., Wigley, T.M.L., and Parkhurst, D.L., The Kinetics of Calcite Dissolution in CO2-Water Systems at 5–10°C and 0.0–1.0 atm CO2, Am. J. Sci., 1978, vol. 278, p. 179.
Sjoberg, E.L., Kinetics and Mechanism of Calcite Dissolution in Aqueous Solutions at Low Temperatures, Stockholm Contrib. Geol., 1978, vol. 32, p. 1.
Plummer, L.N. and Wigley, T.M.L., The Dissolution of Calcite in CO2-Saturated Solutions at 25°C and One Atmosphere Total Pressure, Geochim. Cosmochim. Acta, 1976, vol. 40, p. 191.
Sjoberg, E.L., A Fundamental Equation for Calcite Dissolution Kinetics, Geochim. Cosmochim. Acta, 1976, vol. 40, p. 441.
Morse, J.W., Dissolution Kinetics of Calcium Carbonate in Seawater: 3. A New Method for the Study of Carbonate Reaction Kinetics, Am. J. Sci., 1974, vol. 274, p. 97.
Tarasevich, M.R., Khrushcheva, E.I., and Filinovskii, V.Yu., Vrashchayushchiisya diskovyi elektrod s kol'tsom (Rotating-Ring Disk Electrode), Moscow: Nauka, 1987.
Pleskov, Yu.V. and Filinovskii, V.Yu., Vrashchayushchiisya diskovyi elektrod (Rotating Disc Electrode), Moscow: Nauka, 1972.
Damaskin, B.B. and Petrii, O.A., Vvedenie v elektrokhimicheskuyu kinetiku (Introduction to Electrochemical Kinetics), Moscow: Vysshaya Shkola, 1983.
Fredd, C.N. and Fogler, H.S., The Influence of Chelating Agents on the Kinetics of Calcite Dissolution, J. Colloid Interface Sci., 1998, vol. 204, p. 187.
Hales, B. and Emerson, S., Evidence in Support of First-Order Dissolution Kinetics of Calcite in Seawater, Earth Planet. Sci. Lett., 1997, vol. 148, p. 317.
Gorichev, I.G., Kutepov A.M., Gorichev, A.I., Izotov, A.D., Kinetika i mekhanizm rastvoreniya oksidov i gidroksidov zheleza v kislykh sredakh (Kinetics and Mechanism of Dissolution of Iron Oxides and Hydroxides in Acidic Media), Moscow: Ross. Univ. Druzhby Narodov, 1999.
Charlot, G., Les methodes de la chimie analytique, Paris: Masson, 1961. Translated under the title Metody analiticheskoi khimii, Moscow: Khimiya, 1965.
Bezdenezhnykh, A.A., Inzhenernye metody sostavleniya uravnenii skorostei reaktsii i rascheta kineticheskikh konstant (Engineering Methods for Formulating the Reaction Rate Equations and Calculating the Kinetic Constants), Leningrad: Khimiya, 1973.
Wiese, G.R., James, R.O., Yates, D.E., and Healy, T.W., Electrochemistry of the Colloid/Water Interface, Int. Rev. Sci.,, 1976, vol. 6, p. 53.
Westall, J. and Hohl, H., A Comparison of Electrostatic Models for the Oxide/Solution Interface, Adv. Colloid. Interface Sci., 1980, vol. 12, no.2, p. 265.
Davis, J.A., James, R.D., and Leckie, J.O., Surface Ionization and Complexation at the Oxide/Water Interface, J. Colloid Interface Sci., 1978, vol. 63, no.3, p. 480.
Davis, J.A. and Leckie, J.O., Surface Properties of Amorphous Iron Oxyhydroxide and Adsorption of Metal Ions, J. Colloid Interface Sci., 1978, vol. 67, no.1, p. 90.
Davis, J.A. and Leckie, J.O., Adsorption of Anions, J. Colloid Interface Sci., 1980, vol. 74, no.1, p. 32.
Hayes, K.F., Papelis, C., and Leckie, J.O., Modeling Ionic Strength Effects on Anion Adsorption at Hydrous Oxide/Solution Interfaces, J. Colloid Interface Sci., 1988, vol. 125, no.2, p. 717.
Hayes, K.F. and Leckie, J.O., Modeling Ionic Strength Effects on Cation Adsorption at Hydrous Oxide/Solution Interfaces, J. Colloid Interface Sci., 1987, vol. 115, no.2, p. 564.
Barrow, N.J. and Bowden, J.W., A Composition of Models for Describing the Adsorption of Anions on a Variable Change Mineral Surface, J. Colloid Interface Sci., 1987, vol. 119, no.1, p. 236.
Barrow, N.J., Effect of Surface Heterogeneity on Ion Adsorption by Metal Oxide and by Soils, Langmuir, 1993, vol. 9, no.10, p. 2606.
Gorichev, I.G., Batrakov, V.V., and Shaplygin, I.S., Complexation on the Surface of Iron Hydroxides: I. Investigation Methods and Model Description of the Acid-Base Properties at the Iron Oxide-Electrolyte Interface, Neorg. Mater., 1994, vol. 30, no.10, p. 330.
Gorichev, I.G., Batrakov, V.V., and Shaplygin, I.S., Complexation on the Surface of Iron Hydroxides: II. Experimental Data on Ion Adsorption and Surface Complexation, Neorg. Mater., 1994, vol. 30, no.10, p. 346.
Moulin, P. and Roques, H., Zeta Potential Measurement of Calcium Carbonate, J. Colloid Interface Sci., 2003, vol. 261, p. 115.
Huang, Y.C., Fowkes, F.M., Lloyd, T.B., and Sanders, N.D., Adsorption of Calcium Ions from Calcium Chloride Solutions onto Calcium Carbonate Particles, Langmuir, 1991, vol. 7, no.8, p. 1742.
Author information
Authors and Affiliations
Additional information
__________
Translated from Teoreticheskie Osnovy Khimicheskoi Tekhnologii, Vol. 39, No. 6, 2005, pp. 651–659.
Original Russian Text Copyright © 2005 by Dolgaleva, Gorichev, Izotov, Stepanov.
Rights and permissions
About this article
Cite this article
Dolgaleva, I.V., Gorichev, I.G., Izotov, A.D. et al. Modeling of the Effect of pH on the Calcite Dissolution Kinetics. Theor Found Chem Eng 39, 614–621 (2005). https://doi.org/10.1007/s11236-005-0125-1
Received:
Issue Date:
DOI: https://doi.org/10.1007/s11236-005-0125-1