Abstract
A new nematode species, Dujardinascaris mormyropsis n. sp. (Anisakidae), is described from specimens found in the stomach and intestine of the cornish jack Mormyrops anguilloides (Linnaeus) (Osteoglossiformes; Mormyridae) in the Sangha River (Congo River basin) in Dzanga-Sangha Protected Areas, Central African Republic. Based on light and scanning electron microscopical examination, the new species differs from the only other congeneric species parasitising fishes in Africa, D. malapteruri (Baylis, 1923), mainly in the presence of dentigerous ridges on lips, absence of lateral caudal alae in the cloacal region, in the anteriorly curved, non-bifid distal tip of the gubernaculum and larger eggs (60–90 × 52–78 vs 45 × 30 μm); males of D. mormyropsis are characterised by the presence of a ventral precloacal cuticular ornamentation not observed in other Dujardinascaris spp. Dujardinascaris mormyropsis and D. malapteruri also differ in the order of their fish hosts (Osteoglossiformes vs Siluriformes). Specimens previously reported as Dujardinascaris graberi Troncy, 1969, a junior synonym of D. malapteruri, from Mormyrops engystoma Boulenger in Chad belong to the new species D. mormyropsis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Mormyrops anguilloides (Linnaeus) (Mormyridae) is a large carnivorous mormyrid reaching lengths up to 150 cm. It is widely distributed throughout the Nilo-Sudan, Congo, northern Lower Guinea and Zambezi ichthyological provinces, including Lakes Malawi and Tanganyika (Stiassny et al., 2007). To date, only two helminth species, a cestode larva and the nematode Procamallanus laeviconchus (Wedl, 1862), have been recorded from this host species in the Sudan (Khalil & Polling, 1997).
During recent helminthological investigations of some freshwater fishes in the Central African Republic, nematodes referable to Dujardinascaris Baylis, 1947 were recovered from the stomach and intestine of Mormyrops anguilloides collected in the Sangha River (Congo River basin) in Dzanga-Sangha Protected Areas. Their detailed study using both light and scanning electron microscopy showed that they represent a new species, which is described below.
Materials and methods
Fish were obtained from local fishermen operating on the Sangha River in the vicinity of Sangha Lodge (2°59′7″N, 16°13′58″E), within Dzanga-Sangha Special Dense Forest Reserve located in the forest zone of the Central African Republic. The nematodes recovered were washed in physiological saline and then fixed in hot 4% formalin. For light microscopical examination, the nematodes were cleared with glycerine. Drawings were made with the aid of a Zeiss drawing attachment. Specimens used for scanning electron microscopy (SEM) were postfixed in 1% osmium tetroxide (in phosphate buffer), dehydrated through a graded acetone series, critical-point-dried and sputter-coated with gold; they were examined using a JEOL JSM-7401F scanning electron microscope at an accelerating voltage of 4 kV (GB low mode). All measurements are in micrometres unless otherwise indicated. Fish were identified using photographs by Melanie L. J. Stiassny of the Department of Ichthyology, American Museum of Natural History (AMNH), New York, USA. The names of fishes follow FishBase (Froese & Pauly, 2014).
Family Anisakidae Railliet & Henry, 1912
Dujardinascaris mormyropsis n. sp.
Type-host: Cornish jack Mormyrops anguilloides (Linnaeus) (Osteoglossiformes: Mormyridae).
Site in host: Stomach, rarely also intestine.
Type-locality: Sangha River adjacent to Sangha Lodge (2°59′7″N, 16°13′58″E) (Congo River Basin), Dzanga-Sangha Protected Areas, Central African Republic (collected 11 September 2012).
Prevalence and intensity: 2 fish infected/3 fish examined; 1–16 specimens per fish.
Type-material: Holotype, allotype and paratypes are deposited in the Helminthological Collection of the Institute of Parasitology, Biology Centre of the Academy of Sciences of the Czech Republic, České Budějovice (Cat. No. N-1046).
Etymology: The specific name of this nematode relates to the genitive form of the generic name of the host.
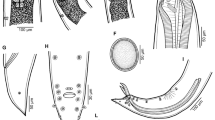
Dujardinascaris mormyropsis n. sp. A, Anterior end of male, lateral view; B, cephalic end of gravid female, dorsal view; C, cephalic end of male, ventral view; D, tail of female, lateral view; E, F, caudal end of male, ventral and lateral views; G, gubernaculum, lateral view; H, posterior end of male, lateral view; I, J, eggs
General. Rather large nematodes with distinct whitish uteri and ovaries, clearly visible in live specimens; colour of preserved specimens brownish. Lips with distinct interlocking processes (Fig. 2A, B). Middle anterior region of lips semicircular, provided with row of prominent, anteriorly oriented teeth (1 large middle tooth and about 10 smaller teeth on either side) (Figs. 2A, B, D, 3A). Dorsal lip with symmetrical pulp and 2 double papillae, with 2 forked anteriorly directed prolongations (Fig. 1A) appearing as 4 lobes in SEM (Fig. 2A, D). Subventral lips somewhat asymmetrical, of similar structure as dorsal lip, bearing 1 double and 1 single papillae (Fig. 2B); lateral amphids indistinct. Interlabia well developed, triangular, reaching anteriorly mid-length of lips (Figs. 1A–C, 2A, B). Cervical alae absent. Oesophagus rather long, ventriculus small, almost spherical (Fig. 1A). Long anterior dorsal intestinal caecum present. Nerve-ring encircling oesophagus at about 1/4 of its length. Excretory pore approximately at level of nerve-ring (Fig. 1A). Deirids not observed.
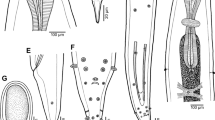
Dujardinascaris mormyropsis n. sp., scanning electron micrographs. A, cephalic end, apical view; B, ventrolateral lip, sublateral view; C, region of cloacal opening, ventral view (arrow indicates median preanal papilla); D, dorsal lip, subdorsal view; E, tail of male, subventral view (arrow indicates median preanal papilla); F, posterior part of male tail, lateral view (arrow indicates phasmid). Abbreviations: c, cloacal aperture; dl, dorsal lip; dp, double labial papilla; i, interlabuim; o, ventral precloacal cuticular ornamentation; sp, single cephalic papilla; vl, ventrolateral lip; 1, 3, lateral postanal papillae; 2, 4, subventral postanal papillae
Dujardinascaris mormyropsis n. sp., scanning electron micrographs. A, row of labial denticles; B, posterior end of male, lateral view; C, tail of male, lateral view; D, posterior end of male, ventral view; E, tail of female, lateral view; F, tail of male, lateral view (arrow indicates phasmid). Abbreviations: a, anus; b, first three pairs of preanal papillae; c, cloacal aperture; 1, 3, lateral postanal papillae; 2, subventral postanal papilla
Male [Based on 3 specimens; measurements of holotype in parentheses.] Length of body 11.52–15.33 (14.96) mm, maximum width 380–544 (530). Lips and iterlabia 75–90 (87) and 30–45 (45) long, respectively. Length of oesophagus 1.32–1.84 (1.81) mm, maximum width 82–109 (95), representing 11–12 (12)% of body length; ventriculus 95–150 (150) long, 95–109 (109) wide. Nerve-ring and excretory pore 408–558 (558) and 422–558 (558) from anterior extremity, respectively. Intestinal caecum 680–1,115 (1,115) long. Preanal papillae: first 3 pairs large, well and almost equally spaced from each other; 2 pairs of smaller papillae and 1 unpaired median papilla present near cloaca. Postanal papillae: 1 pair of small papillae just posterior to cloacal aperture and 4 pairs of papillae (2 subventral and 2 dorsolateral) on narrowed part of tail (Figs. 1E, F, H, 2C, E, F, 3C, D, F). Pair of small lateral phasmids situated between third and last but one pairs of postanal papillae (Figs. 1E, F, 2F, 3F). No caudal alae or ridges present in region of cloacal aperture and all 3 paired papillae free, not projecting into alae (Figs. 2C, 3C, D); one postanal papilla of this group may be occasionally absent (Fig. 2E). Precloacal body surface with slightly outlined subventral oblique muscle bands (Fig. 1H). Ventral cuticular ornamentation forming median longitudinal band of distinctly elevated, transversely striated cuticle present in region anterior to anterior cloacal lip (Figs. 1E, 2C, E, 3D); posterior cloacal lip with distinct superficial sculpture (Figs. 2C, E, 3C, D). Spicules non-alate, very narrow, equal in length, 1.70–1.81 (1.81), representing 7.5–14.8 (7.5)% of body length; proximal ends slightly expanded, distal ends pointed (Fig. 1H). Gubernaculum186–204 (186) long, its proximal end expanded to form dorsal lobe, distal end rounded (Fig. 1G, H). Tail 120–171 (156) long, with sharply pointed tip (Figs. 1E, F, H, 2E, F, 3B, C, F).
Gravid female [Based on 5 specimens; measurements of allotype in parentheses.] Length of body 26.04–43.22 (43.22) mm, maximum width 897–1,346 (1,346). Lips and interlabia 135–159 (147) and 78–96 (96) long, respectively. Length of oesophagus 2.20–3.07 (2.68) mm, maximum width 95–122 (109), representing 6–8 (6)% of body length; ventriculus 109–136 (136) long, 95–136 (122) wide. Nerve-ring and excretory pore 585–775 (775) and 585–816 (816) from anterior extremity, respectively. Intestinal caecum 1.19–1.73 (1.12) mm long. Vulva 9.13–15.92 (15.92) mm from anterior end, situated at 27–37 (37)% of body length; anterior vulval lip elevated only in smallest specimen. Vagina directed posteriorly from vulva. Eggs from almost spherical to oval, thin-walled, with smooth surface, 60–90 × 52–78 (52–60 in diameter); content of eggs mostly uncleaved or cleaved at most into 2 blastomeres (Fig. 1I, J). Tail conical, 367–639 (612) long, with sharply pointed tip (Figs. 1D, 3E). Phasmids indistinct.
Nongravid female [Based on 3 specimens.] Length of body 14.08–26.71 mm, maximum width 408–775. Lips and interlabia 81–141 and 39–69 long, respectively. Length of oesophagus 1.46–2.54 mm, maximum width 82–109, representing 9–10% of body length; ventriculus 95–122 long, 95–136 wide. Nerve-ring and excretory pore 442–639 and 435–639 from anterior extremity, respectively. Intestinal caecum 612–1,496 long. Vulva 6.19–8.54 mm from anterior end, situated at 32–44% of body length. Uterus empty. Tail conical, 272–449 long.
Discussion
Most species of the genus Dujardinascaris Baylis, 1947 are parasites of crocodilians (Mozgovoy, 1953; Sprent, 1977; Hartwich, 2009). Baylis (1923) was the first to describe a representative of this genus from a fish host: Dujardinia (= Dujardinascaris) malapteruri Baylis, 1923 from Malapterurus electricus (Gmelin) (Siluriformes, Malapteruridae) in Khartoum, Sudan; this species was also reported from the same host species and locality by Khalil (1969). Later Sprent (1990) redescribed D. malapteruri based on Baylis’ type-specimens and those collected from M. electricus in Gabon. According to him, Dujardinascaris graberi Troncy, 1969, described from M. electricus in Chad (Troncy, 1969), is a junior synonym of D. malapteruri.
Vassiliadès & Troncy (1974) reported D. graberi (= D. malapteruri) also from Heterotis niloticus (Cuvier) (Osteoglossiformes, Arapaimidae) and Mormyrops engystoma Boulenger (Osteoglossiformes, Mormyridae) in Chad, but Sprent (1990) has mentioned that the identity of these nematodes with D. malapteruri is uncertain. Since another ascaridoid with a long anterior intestinal caecum, Multicaecum heterotis Petter, Vassiliadès & Marchand, 1979, is a specific parasite of H. niloticus in Senegal, Nigeria and the Sudan (Petter et al., 1979; Akinsanya et al., 2007; Mašová et al., 2010), it is highly probable that the nematodes found by Vassiliadès & Troncy (1974) in H. niloticus and designated as D. graberi (= D. malapteruri) belong, in fact, to M. heterotis. The latter species was unknown at the time when the paper of Vassiliadès & Troncy (1974) appeared.
It is apparent from the above discussion that D. malapteruri is the only known valid species of Dujardinascaris parasitising African freshwater fishes as adults. Khalil & Polling (1997) also listed D. helicina Baylis, 1927 [sic] collected by Baylis (1928) from the gut of Lates microlepis Boulenger in Lake Tanganyika, Tanzania. However, Baylis’ (1928) paper shows that the author had only larvae presumed to be Dujardinia (= Dujardinascaris) helicina (Molin, 1860), parasitic in crocodiles as adult. However, this is an evident species misidentification, because D. helicina is a parasite of crocodiles in the Neotropical Region (Sprent, 1977; Moravec, 2001); moreover, larval morphology does not allow identification to the species level. Therefore, the larvae recorded by Baylis (1928) from L. microlepis should be reported as Dujardinascaris sp.
The original description of D. malapteruri provided by Baylis (1923) is inadequate, but this species was redescribed by Sprent (1990) (see above), who has also examined two males, one damaged female and two fourth-stage larvae of Dujardinascaris from Mormyrops engystoma from Chad, previously reported by Vassiliadès & Troncy (1974). Because these specimens somewhat differed from those of D. malapteruri from the type-host (particularly in the shape and structure of the gubernaculum), he questioned the conspecificity of these two samples.
Specimens of the present material from M. anguilloides have the same form and structure of the gubernaculum as described and illustrated by Sprent (1990) based on specimens from M. engystoma; the body measurements of both forms are similar, their hosts are congeneric, therefore, we consider them representatives of the same species. The general morphology and measurements of D. mormyropsis n. sp. are rather similar to those of D. malapteruri. However, the new species differs in the presence (vs absence) of dentigerous ridges on lips and the absence (vs presence) of lateral caudal alae (ridges) in the cloacal region, in the shape and structure of the tip of the gubernaculum (tip curved anteriorly, not bifid vs tip straight, bifid) and larger eggs (60–90 × 52–78 vs 45 × 30 μm). The ventral precloacal ornamentation observed in D. mormyropsis has not been described in D. malapteruri, but the latter species has not yet been studied by SEM. Both species differ in the order of their fish hosts (Osteoglossiformes vs Siluriformes).
Dujardinascaris malapteruri and D. mormyropsis seem to be so far the only two species of this genus parasitising fishes as adults. Several Dujardinascaris spp. were poorly described mostly from marine fishes by workers in Pakistan and India (D. magna Khan & Begum, 1971, D. ritai Zaidi & Khan, 1975, D. quadrii Zubairi & Farooq, 1976, D. sciaenae Bilqees, Fatima & Rehana, 1977, D. cybii Arya & Johnson, 1978), but in fact, none of these species can be regarded as belonging to the genus Dujardinascaris (see Sprent, 1990).
References
Akinsanya, B., Hassan, A. A., & Otubanjo, O. A. (2007). A comparative study of the parasitic helminth fauna of Gymnarchus niloticus (Gymnarchidae) and Heterotis niloticus (Osteoglossidae) from Lekki lagoon Lagos, Nigeria. Pakistani Journal of Biological Sciences, 10, 427–432.
Baylis, H. A. (1923). On the classification of the Ascaridae. III. A revision of the genus Dujardinia Gedoelst, with a description of a new genus of Anisakinae from a crocodile. Parasitology, 15, 223–232.
Baylis, H. A. (1928). Some parasitic worms, mainly from fishes, from Lake Tanganyika. Annals and Magazine of Natural History, Ser. 10, 1, 552–562.
Froese, R., & Pauly, D. (Eds.) (2014). FishBase. World Wide Web electronic publication. http://www.fishbase.org, version 01/2014.
Hartwich, G. (2009). Ascaridida. In: Anderson, R. C., Chabaud, A. G., & Willmott, S. (Eds), Keys to the nematode parasites of vertebrates. Archival volume. Wallingford: CABI, pp. 309–323.
Khalil, L. F. (1969). Studies on the helminth parasites of freshwater fishes of the Sudan. Journal of Zoology, London, 158, 143–170.
Khalil, L. F., & Polling, L. (1997). Check list of the helminth parasites of African freshwater fishes. Second edition. Pietersburg: University of the North, 185 pp.
Mašová, Š., Moravec, F., Baruš, V., & Seifertová, M. (2010). Redescription, systematic status and molecular characterisation of Multicaecum heterotis Petter, Vassiliadès et Marchand, 1979 (Nematoda: Heterocheilidae), an intestinal parasite of Heterotis niloticus (Osteichthyes: Arapaimidae) in Africa. Folia Parasitologica, 57, 280–288.
Moravec, F. (2001). Some helminth parasites from Morelet’s crocodile, Crocodylus moreletii, from Yucatan, Mexico. Folia Parasitologica, 48, 47–62.
Mozgovoy, A. A. (1953). [Ascaridata of animals and man and the diseases caused by them.] Osnovy Nematodologii Vol. 2. Moscow: Publishing House of the Academy of Sciences of the USSR, 616 pp. (In Russian).
Petter, A. J., Vassiliadès, G., & Marchand, B. (1979). Une nouvelle espèce de Multicaecum (Ascarididae, Nematoda) parasite de poisson en Afrique. Bulletin de l’Institut Fondamental d’Afrique Noire, Sér. A, 41, 324–329.
Sprent, J. F. A. (1977). Ascaridoid nematodes of amphibians and reptiles: Dujardinascaris. Journal of Helminthology, 51, 251–285.
Sprent, J. F. A. (1990). Some ascaridoid nematodes of fishes: Heterocheilinae. Systematic Parasitology, 16, 149–161.
Stiassny, M. L. J., Teugels, G. G., & Hopkins, C. D. (2007). The fresh and brackish water fishes of Lower Guinea, West-Central Africa. Vol. 1. Paris and Tervuren: IRD, MNHN, MRAC, 800 pp.
Troncy, P.-M. (1969). Description de deux nouvelles espèces de nématodes parasites de poissons. Bulletin du Muséum National d’Histoire Naturelle, Sér. 2, 41, 598–605.
Vassiliadès, G., & Troncy, P.-M. (1974). Nématodes parasites des poissons du bassin tchadien. Bulletin de l’Institut Fondamental d’Afrique Noire Sér. A, 36, 670–681.
Acknowledgements
We would like to thank the government of the Central African Republic, namely the Ministre de l’Education Nationale, de l’Alphabetisation, de l’Enseignement Superieur, et de la Recherche, for providing research permits to conduct our work in the Central African Republic; World Wildlife Fund and administration of Dzanga-Sangha Protected Areas for granting research approval and assistance with obtaining permits; and the Primate Habituation Programme for providing logistical support in the field. We are also greatly indebted to Rod Cassidy and his family for generous help in the field and for allowing M. J. to work in and use facilities of the Sangha Lodge. Thanks go also to Melanie L. J. Stiassny of the Department of Ichthyology, AMNH, New York, USA for the identification of examined fish. The authors’ thanks are also due to the staff of the Laboratory of Electron Microscopy, Institute of Parasitology, Biology Centre of the AS CR, České Budějovice for their technical assistance, and to Blanka Škoríková of the same Institute for help with illustrations. This study was partly supported by the Czech Science Foundation (Grant Nos. P505/12/G112 and P506/10/2330) and by institutional support (RVO:60077344, Institute of Parasitology, BC AS CR).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moravec, F., Jirků, M. Dujardinascaris mormyropsis n. sp. (Nematoda: Anisakidae) from the osteoglossiform fish Mormyrops anguilloides (Linnaeus) (Mormyridae) in Central Africa. Syst Parasitol 88, 55–62 (2014). https://doi.org/10.1007/s11230-014-9479-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11230-014-9479-2