Abstract
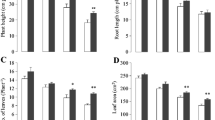
The effects of La3+ on the antioxidant enzyme activities and the relative indices of cellular damage in cucumber seedling leaves were studied. When cucumber seedlings were treated with low concentrations of LaCl3 (0.002 and 0.02 mM), peroxidase (PO) activity increased, and catalase (CAT) activity was similar to that of control leaves at 0.002 mM La3+ and increased at 0.02 mM La3+, whereas superoxide dismutase (SOD) activity did not change significantly. The increase in the contents of chlorophyll (including chlorophylls a and b), carotenoids in parallel with the decrease in the level of malondialdehyde (MDA) suggested that low concentration of La3+ promoted plant growth. However, except the increase in SOD activity at 2 mM La3+, CAT and PO activities and the contents of pigments decreased at high concentrations of La3+ (0.2 and 2 mM), leading to the increase of MDA content and the inhibition of plant growth. It is suggested that lanthanum ion is involved in the regulation of active oxygen-scavenging enzyme activities during plant growth.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- CAT:
-
catalase
- MDA:
-
malondialdehyde
- PO:
-
peroxidase
- SOD:
-
superoxide dismutase
REFERENCES
Bouvier, F., Backhaus, R.A., and Camara, B., Induction and Control of Chloroplast-Specific Carotenoid Genes by Oxidative Stress, J. Biol. Chem., 1998, vol. 273, pp. 30651–30659.
Asada, K. and Takahashi, M., Production and Scavenging of Active Oxygen in Photosynthesis, Photoinhibition: Topics in Photosynthesis, Kyle, D.J., Osmond, C.B., and Artzen, C.J., Eds., Amsterdam: Elsevier, 1987, pp. 227–287.
Gille, G. and Singler, K., Oxidative Stress in Living Cells, Folia Microbiol., 1995, vol. 2, pp. 131–152.
Alia Saradhi, P.P. and Mohanty, P., Involvement of Proline in Protecting Thylakoid Membranes against Free Radical-Induced Photodamage, J. Photochem. Photobiol. B: Biol., 1997, vol. 38, pp. 253–257.
Asada, K., Production and Action of Active Oxygen Species in Photosynthetic Tissues, Causes of Photooxidative Stress and Amelioration of Defense Systems in Plants, Foyer, C.H. and Mullineaux, P.M., Eds., Boca Raton: CRC, 1994, pp. 77–104.
Davies, K.J., Intracellular Proteolytic Systems May Function as Secondary Antioxidant Defense: A Hypothesis, J. Free Rad. Biol. Med., 1986, vol. 2, pp. 55–173.
Foyer, C.H., Protection against Oxygen Radicals: An Important Defense Mechanism Studied in Transgenic Plants, Plant Cell Environ., 1994, vol. 17, pp. 507–523.
He, Y.W. and Loh, C.S., Cerium and Lanthanum Promote Floral Initiation and Reproductive Growth of Arabidopsis thaliana, Plant Sci., 2000, vol. 159, pp. 117–124.
Wu, J., Wang, C., and Mei, X., Stimulation of Taxol Production and Excretion in Taxus spp. Cell Cultures by Rare-Earth Chemical Lanthanum, J. Biotechnol., 2001, vol. 85, pp. 67–73.
Ouyang, J., Wang, X., Zhao, B., Yuan, X., and Wang, Y., Effects of Rare-Earth Elements on the Growth of Cistanche deserticola Cells and the Production of Phenylethanoid Glycosides, J. Biotechnol., 2003, vol. 102, pp. 129–134.
Zeng, F.L., An, Y., Zhang, H.T., and Zhang, M.F., The Effects of La3+ on the Peroxidation of Membrane Lipids in Wheat Seedling Leaves under Drought Stress, Biol. Trace Element Res., 1999, vol. 69, pp. 141–150.
Pang, X., Wang, D.H., Xing, X.Y., Peng, A., Zhang, F.S., and Li, C.J., Effect of La3+ on the Activities of Antioxidant Enzymes in Wheat Seedlings under Lead Stress in Solution Culture, Chemosphere, 2002, vol. 47, pp. 1033–1039.
Zhang, L., Zeng, F., and Xiao, R., Effect of Lanthanum Ions (La3+) on the Reactive Oxygen Species Scavenging Enzymes in Wheat Leaves, Biol. Trace Element Res., 2003, vol. 91, pp. 243–255.
Luck, H., Catalase, in Methods of Enzymatic Analysis, New York: Academic, 1963, vol. 3, pp. 885–894.
Reuveni, R., Shimoni, M., Karchi, Z., and Kuc, J., Peroxidase Activity as a Biochemical Marker for Resistance of Muskmelon (Cucumis melo) to Pseudoperenospora cubensis, Phytopathology, 1992, vol. 82, pp. 749–753.
Arnon, D.I., Copper Enzymes in Isolated Chloroplasts, Polyphenol Oxidase in Beta vulgaris, Plant Physiol., 1949, vol. 24, pp. 1–15.
Liaaen-Jensen, S. and Jensen, A., Quantitative Determination of Carotenoids in Photosynthetic Tissue, in Methods in Enzymology, New York: Academic, 1971, vol. 23, pp. 586–602.
Dhindsa, R.S. and Matowe, W., Drought Tolerance in Two Mosses: Correlated with Enzymatic Defense against Lipid Peroxidation, J. Exp. Bot., 1981, vol. 32, pp. 79–91.
McCord, J.M. and Fridovich, I., Superoxide Dismutase: An Enzymic Function for Erythrocuprein (Hemocuprein), J. Biol. Chem., 1969, vol. 244, pp. 6049–6055.
Monk, L.S., Fagerrstedt, K.V., and Crawford, R.M., Oxygen Toxicity and Superoxide Dismutase as an Antioxidant in Physiological Stress, Physiol. Plant., 1989, vol. 76, pp. 456–459.
Elroy, S.O., Bernstein, Y., and Groner, Y., Overproduction of Human Cu-Zn-Superoxide Dismutase in Transfected Cells: Extenuation of Paraquat-Mediated Cytotoxicity and Enhancement of Lipid Peroxidation, EMBO J., 1986, vol. 5, pp. 615–622.
Davidson, J., Kettle, A.J., and Fatur, D.J., Mechanism of Inhibition of Catalase by Ascorbate, J. Biol. Chem., 1986, vol. 261, pp. 1193–1200.
Larson, R.A., The Antioxidants of Higher Plants, Phytochemistry, 1988, vol. 27, pp. 969–978.
Zeng, F.L., Zhang, M.F., Zhou, S.M., Wu, J.G., and Deng, R.W., The Effects of Lanthanide Chloride on Abscisic Acid and Electron Transport Activity of Some Crops, Biol. Trace Element Res., 1999, vol. 67, pp. 277–284.
Author information
Authors and Affiliations
Additional information
__________
From Fiziologiya Rastenii, Vol. 52, No. 3, 2005, pp. 338–342.
Original English Text Copyright © 2005 by Shi, Chen, Huang.
This article was submitted by the authors in English.
Rights and permissions
About this article
Cite this article
Shi, P., Chen, G.C. & Huang, Z.W. Effects of La3+ on the Active Oxygen-Scavenging Enzyme Activities in Cucumber Seedling Leaves. Russ J Plant Physiol 52, 294–297 (2005). https://doi.org/10.1007/s11183-005-0044-3
Received:
Issue Date:
DOI: https://doi.org/10.1007/s11183-005-0044-3