Abstract
The management of patients with midgut neuroendocrine tumors (MNET) is rapidly evolving. Current preoperative detection rates of primary tumor sites are higher than ever and progression-free survival in patients with already advanced disease is expanding due to the implementation of novel efficacious treatment strategies. This survival benefit may potentially translate into a need for a multidisciplinary approach to an even more heterogenous variety of clinical conditions, among these, carcinoid syndrome (CS) and carcinoid heart disease (CHD). The latter often triggers substantial morbidity and mortality, hence a systematic screening, an accurate diagnosis, as well as effective interventions are critically important. The rarity of the disease has result in a relative lack of statistically powerful evidence, which in turn may have rendered significant variability between practices. In this regard, despite recent guidelines, the optimal follow-up of patients with CHD remain debatable to some authors, perhaps due to the preponderance of certain schools throughout the manuscript. Herein, we present a concise and practical guidance document on clinical screening and echocardiographic surveillance of patients with CHD based on a comprehensive review of the literature, and complemented by our experience at the Center for Carcinoid and Neuroendocrine Tumors at The Mount Sinai Hospital.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Without doubt, diagnosis and management of midgut neuroendocrine tumors is currently, if not the most, one of the most dynamic areas of gastroenterology. The field has evolved from one characterized by limited treatment options to one identified by an exponentially increasing number of clinical trials and available novel therapeutic agents [1] These major advances include a) the effective combination of multiple imaging techniques such as PET/CT [2], SRS, and endoscopy to achieve higher identification rates of primary tumor sites [3] b) a more precise tumor classification or taxonomy [4] c) a shift towards a more aggressive interventional and surgical approach d) completion of major trials that will surely assign to Telotristat Ethyl [5], Everolimus [6] and 177Lu-Dotate [7] pivotal roles in an immediate future treatment protocols. Consequently, the current preoperative detection rate of primary tumor sites has been demonstrated to be as high as 80%, the incidence of MNETs has increased almost seven-fold, and progression-free survival in patients with already advanced disease is expected to importantly expand.
Among patients with MNETs, survival benefit will certainly translate into a need for a stepwise multidisciplinary approach with a broader collection of treatment strategies to face an unusual variety of clinical conditions, among these, CS and CHD. The latter often triggers substantial morbidity and mortality, hence a systematic screening, an accurate diagnosis, as well as effective interventions are critically important. However, the rarity of the disease has led to a relative lack of statistically powerful evidence, which in turn may have rendered significant variability in opinions regarding the optimal management of these patients [8] In addition, the latest expert statement, although promoted by an integrative leadership, presents a geographical and institutional bias and ignores part of the contemporary carcinoid literature [9] The following is a concise and practical guidance document on clinical screening and echocardiographic surveillance of CHD based on a comprehensive review of the literature, and complemented by the authors’ experience at the Center for Carcinoid and Neuroendocrine Tumors at The Mount Sinai Hospital.
1 From carcinoid syndrome to carcinoid heart disease
Midgut neuroendocrine tumors are generally slow-growing, silent and relatively indolent [10] Symptoms often result from mechanical complications (incidental finding due to gastrointestinal bleeding or obstruction) or from their inherent capacity of synthesizing, storing, and releasing large amounts of bioactive substances (prominently serotonin) into the systemic bloodstream [11] Normally, serotonin is mostly recaptured, inactivated, and metabolized to 5-hydroxyindolacetic acid (5-HIAA) by hepatic enzymes for subsequent renal clearance [12] In the presence of hepatic metastases, ectopic functional foci secrete copious amounts of serotonin that will exceed and bypass the degradation capacity of the liver [13] and then reach the systemic circulation, the inferior vena cava and ultimately the right heart. Exceptions to this pathophysiologic pathway are very rare cases of extensive retroperitoneal lymph node metastases (<1%) with drainage to the thoracic duct [14] (Fig. 1).
The overproduction and dissemination of serotonin into the systemic circulation triggers a wide constellation of signs and symptoms known as CS [15,16,17], mainly characterized by vasovagal lability (cutaneous flushing and hypotension), gastrointestinal hypermotility, cardiac involvement and bronchospasm. Cardiac manifestations, known as CHD, are secondary to a plaque-like endocardial deposition of fibrous tissue (myofibroblasts, smooth muscle cells, and extracellular matrix) that often extends to the endocardial surface of the valve leaflets or cusps (and subvalvular apparatus) leading to different patterns of valve dysfunction and later ventricular failure [18].
Although the action mechanisms of serotonin have been extensively studied by the pharmaceutical industry for the past decade [19], the etiology for myocardial and valve injury remains poorly understood [20]. Contemporary publications have unanimously suggested a more concrete and selective link between serotonin 5-HT2B receptors [21] and heart valve disease secondary to a mitogenic and secretory response in ventricular and valvular fibroblasts [22] The activation of 5-HT2B receptors [23] has been observed to directly promote the proliferation and fibrosis of the leaflets by accumulation of extracellular matrix secreted by the valvular interstitial cells [24] Additionally, this signal transduction has been shown to upregulate the cytokine transforming growth factor β1 (TGF-β1) which then acts as a key mediator in the appearance of leaflet changes due to myofibroblasts deposition and increased production of collagen and glycosaminoglycans [25] Carcinoid heart disease involves primarily the right-side of the heart in over 90% of the patients [26] In the absence of a primary endobronchial carcinoid tumor (additional satellite production of serotonin), classic publications have demonstrated that pulmonary endothelial cells might be able to metabolize large amounts of circulating serotonin. Therefore, the development of left-sided CHD mandates the presence of a persistent cardiac shunt (commonly a patent foramen ovale or atrial septal defect) or a very severe or poorly controlled tumor activity [27] that may overcome the pulmonary potential to deactivate serotonin [28] (Table 1).
Development of carcinoid heart disease secondary to the presence of foregut (bronchopulmonary, thymus, stomach, pancreas, proximal duodenum, ovaries), midgut (distal duodenum, jejunum, ileum, appendix, ascending colon, 2/3 transverse colon), and hindgut (1/3 of transverse colon, descending colon, rectum, cecum) neuroendocrine tumors. 5-HIAA: 5-Hydroxyindoleacetic acid
5-HIAA: 5-Hydroxyindoleacetic acid, APhos: alkaline phosphatase, Bb: bilirubin, BP: blood pressure, CHD: carcinoid heart disease, CMR: cardiac magnetic resonance, CTS: cardiothoracic surgeon, EKG: electrocardiogram, GGT: gamma-glutamyl transferase, LFT: liver function test, LV: left ventricle, M: months, MNET: midgut neuroendocrine tumor, MOD: moderate, NT-proBNP: N-terminal pro b-type natriuretic peptide, PET-CT: Positron emission tomography-computed tomography, Q: every, RV: right ventricle, RVLS: right ventricular longitudinal strain by speckle tracking echocardiography TEE: transesophageal echocardiogram, TTE: transthoracic echocardiogram, Y: year
AV: aortic valve, AR: aortic regurgitation, AS: aortic stenosis, CMR: cardiac magnetic resonance, LV: left ventricle, MR: mitral regurgitation, MS: mitral stenosis, MV: mitral valve, PR: pulmonic regurgitation, PV: pulmonic valve, RV: right ventricle, RVLS by STE: right ventricular longitudinal strain by speckle tracking echocardiography, TR: tricuspid regurgitation, TV: tricuspid valve
2 Screening for carcinoid heart disease
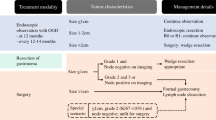
After tumor grading, the identification of hepatic metastases and CS, the existence of extrahepatic disease (e.g. lymph nodes, peritoneum, lung, bone, brain, or ovaries), and cardiac involvement are considered the most unfavorable prognostic factors in patients with MNETs [29] Regarding CHD, the accelerated adoption of novel systemic medical therapies has correlated with a significant improved control of hormonal syndromes and consequently, with an important prevalence decrease [30] Furthermore, CHD is a very unpredictable condition, and even patients with advanced degrees of valve pathology might remain asymptomatic until clinical decompensation rapidly triggers signs and symptoms of right ventricular failure (peripheral edema > ascites > pleural effusions > hepatic congestion) [31] Taking into consideration its ominous impact on survival, screening for early detection of valve disease or ventricular dysfunction is beyond imperative to monitor those asymptomatic patients at risk [32] (Fig. 2).
2.1 The role of cognitive cardiology - physical examination and symptoms
Direct and indirect actions of abnormally elevated levels of serotonin correlate with a number of adverse cardiovascular effects including vasovagal lability (vasodilatation and frequent variations in blood pressure), arrhythmias, pulmonary hypertension, myocardial fibrosis, and valve disease [33] Accordingly, every patient with a MNET should be seen and followed by a clinical cardiologist with expertise in structural valve disease (additional expertise in CHD when feasible) in order to have a baseline transthoracic echocardiography (TTE). This is more and more relevant since CHD might be the first manifestation of CS in up to 20% of patients [34].
Although clinical diagnosis of early CHD mandates a high index of suspicion, experienced clinicians may detect subtle abnormalities during cardiac auscultation. Careful examination of the jugular venous pressure is essential as an early prominent V-wave of tricuspid regurgitation may be observed. A coincident V-wave with carotid pulse is also characteristic of at least moderate tricuspid regurgitation. Tricuspid regurgitation usually yields a pansystolic murmur which increases in intensity during inspiration. Similarly, an early diastolic murmur or a systolic murmur can be heard in pulmonic position in the presence of pulmonic regurgitation or stenosis, respectively.
2.2 Appropriateness of biochemical markers and transthoracic echocardiography
During the last decade, the utility and applicability of biochemical markers in clinical practice has been strongly endorsed by some groups, not only to detect cardiac involvement, but also to assess disease progression and to evaluate prognosis in patients with CHD [35] For example, plasma 5-HIAA and NT-proBNP have been found to have a pivotal prognostic and diagnostic significance, respectively, Chromogranins have been associated with disease progression and recurrence, and Activin A has been linked to disease severity, especially in patients with MNETs [36] Moreover, to some authors’ surprise, some of these biomarkers have been suggested to direct a more “rational or responsible” use of echocardiographic screening. This has been further accentuated by administrators who assume a brisk increment of the cost-effectiveness ratio with frequent or regular scanning. As cardiovascular specialists, we are obliged to admit that the consistent incorporation of more manageable imaging devices to the routine cardiovascular practice might have resulted in a trend towards favoring the use of tests or procedures (such as echocardiography) for the diagnosis and interrogation of certain cardiac conditions [37] However, we do not believe this is or should be subject of debate in patients with or with potential to experience CHD. As we have previously pointed out in editorials and academic events, patients with MNETs, CS and CHD still represent a management challenge, and several assumptions deserve attention and perhaps further discussion regarding the use of biomarkers and their implication in current and future consensus statements.
A screening test (whether a quantitative measurement or a qualitative interrogation of an imaging study by a trained observer) aims to reduce morbimortality by early detection of disease (and early intervention). The target population must be very well defined to have a significant prevalence and therefore a meaningful predictive value. Scenarios where a screening test may not be appropriate include the study of a rare disease (consequently with an unknown or low prevalence), an unclear benefit of early treatment, or data extracted from a small sample size which might be difficult to extrapolate to a larger number of screening centers. Despite publication of ambitious and thorough research manuscripts, we should not forget that MNETs are rare and represent a very heterogenous cohort with multitude of disparities (primary location, tumor grading, duration of the disease, metastatic disease, functional status, therapeutic approach including or not primary tumor resection) [38] If we further scrutinize those patients who develop CS and subsequent CHD, the comparison among subjects is even more demanding (variation of serotonin levels based on individual tumor activity represent a major bias) and achieving significant statistical power may become utopic.
Many reports have consistently emphasized the discordance among official guidelines for the management of MNETs regarding the optimal modality and timing for cardiac screening [39,40,41] As expected, the alluded documents are mostly led and developed by neuroendocrine specialists. Interestingly, although the first expert statement on diagnosis and management of CHD certainly involves a more extensive panel of cardiovascular specialists [9], some recommendations seem to remain driven and supported by neuroendocrine literature and expertise. According to some of the referenced articles of the manuscript, clinical examination may be an unreliable screening method for identifying patients with cardiac involvement [42] and echocardiography is a laborious and costly screening tool that is currently overused and entails a visit to the cardiologist [36] This particular position is not surprising since data from the United Kingdom have shown that 50% of centers do not include a cardiologist in their multidisciplinary team [43] Our institutional protocol reinforces the presence of a cardiovascular specialist (with expertise in structural and carcinoid heart disease) early on at diagnosis of a MNET to proceed with a baseline physical examination and TTE (experienced clinical cardiologists are able to detect faint murmurs and subtle changes in volume status). This seems critical given the erratic clinical variability among patients with CHD, ranging from well compensated asymptomatic valve disease to rapidly deteriorating right ventricular failure [44] With regard to the suitability of TTE, several points can be made: 1) Portable ultrasonography is becoming a routine low-cost tool to complement and improve the diagnostic yield of clinical examination across medical specialties [45] 2) Technical improvement and miniaturization of ultrasound devices permits cardiovascular screening by non-experts [46] 3) A focused cardiac ultrasound should be differentiated from a limited TTE interrogation or a formal TTE study [47] 4) Having a cardiologist as part of a multidisciplinary team allows easy access to comprehensive point-of-care echocardiography, particularly in the setting of atrial fibrillation [48, 49] 5) Midgut neuroendocrine tumors have been associated with 5-year survival rates below 50% in the setting of metastatic disease [50]; this evidence together with the unpredictable clinical behavior of these patients may preclude cost-effective strategies 6) The idiosyncrasy of CHD has led to an unsuccessful standardization of disease severity (mild, moderate, or severe remain undefined) and echocardiographic scores.
Exposure to high plasma levels of serotonin has been well established as the main precursor of cardiac involvement and subsequent valve disease [51] In turn, worsening degrees of valve dysfunction (mostly tricuspid regurgitation and stenosis in this setting) may result in atrial dilatation, ventricular dilatation, and further (functional or secondary) tricuspid regurgitation. Successive volume overload of the cardiac chambers triggers the release of neurohormones like N-terminal pro-B-type natriuretic peptide (NT-proBNP) in response to wall dilatation and stretching. Based on these tenets, the latest consensus statement has deemed measurement of plasma (preferred) or urinary 5-HIAA levels as mandatory for diagnosis (and follow up) of CS as well as for identification of those patients at risk of developing CHD. Alternatively, NT-proBNP has been proposed as the best screening tool for CHD in patients with CS. However, several points can be raised regarding the use of NT-proBNP for screening: 1) Serotonin may induce pulmonary hypertension in about 25% of patients with CS, which in turn may result in a baseline degree of right ventricular overload or dysfunction [52] 2) Ongoing institutional research has demonstrated ventricular abnormalities in patients without echocardiographic evidence of CHD 3) Similarly to aortic insufficiency, isolated pulmonary regurgitation can be very well tolerated without causing ventricular overload 4) There is a coherent discrepancy among authors regarding the most accurate cutoff level (235 pg/ml vs. 260 pg/ml vs. 280 pg/ml) and sensitivity (92% vs. 69% for a cutoff level of 260 pg/ml) [35, 53] 5) Up to a third of patients with no progression to CHD may present a > 50% increase in NT-proBNP from baseline value [54] 6) Overlapping pathologies [55] do exist and need to be ruled out 7) Interindividual variation and reference change value need to be considered in such a unique population [56].
The optimal timing of intervention and current trends for the use of cardiac surgery have been recently questioned [57], mainly based on an ambiguous interpretation of the absence of independent association of certain markers of heart failure and postoperative outcomes [30] In this context, current ACC/AHA guidelines for the management of patients with valvular heart disease [58] recommend tricuspid valve surgery in the following scenarios: 1) Isolated symptomatic severe tricuspid stenosis - Class I 2) Presence of symptoms due to severe primary tricuspid regurgitation that are unresponsive to medical therapy - Class IIA 3) Asymptomatic or minimally symptomatic patients with severe primary tricuspid regurgitation and progressive degrees of moderate or greater right ventricular dilatation and/or systolic dysfunction - Class IIB. The European guidelines [59], slightly more aggressive, endorse tricuspid valve surgery in the following scenarios: 1) Symptomatic patients with severe tricuspid stenosis - Class I 2) Symptomatic patients with severe isolated primary tricuspid regurgitation without severe right ventricular dysfunction - Class I 3) Asymptomatic or mildly symptomatic severe isolated primary tricuspid regurgitation and progressive right ventricular dilatation or deterioration of right ventricular function - Class IIA. Most of all, both manuscripts recognize that outcomes of tricuspid valve surgery are dependent on right ventricular function, and that surgery needs to be considered before patients become ineligible due to advanced right ventricular failure [60] With this factors in mind, it seems prudent to establish an even closer cardiologic and echocardiographic surveillance in patients who can experience a rapid progression of valve disease and/or deterioration of right ventricular function [61].
2.3 Echocardiographic characterization of carcinoid heart disease
Transthoracic echocardiography is the gold standard imaging modality for the assessment of presence, extent, and severity of CHD [62]. Interrogation should include a morphological appraisal of the valves (with an emphasis on the right-sided ones), atrial septum and ventricular septum (patency should be ruled out at baseline), as well as a functional evaluation of all cardiac chambers (Fig. 3). The morphologic criteria to establish a diagnosis is based on the study of (1) leaflet thickening (echocardiographic enhancement); (2) leaflet mobility (abnormal motion due to restriction); and (3) subvalvular apparatus (thickening, fusion and shortening of chordal structures leading to leaflet retraction). Functional assessment of the cardiac cavities should include dimensions, volumes, and longitudinal speckle tracking strain when available (Appendix).
The right-sided valves (tricuspid and pulmonic) are typically thickened, shortened, and retracted. In the early stages, there is mild thickening with changes of the normal concave leaflet curvature [34]. Disease progression results in further thickening, which results in additional restriction. Chordae are commonly fused and various patterns of “plaque” deposition can be found. In advanced stages, the manifest deterioration of valvular architecture leads to progressive retraction and non-coaptation [63] (Fig. 4). Chronic degrees of tricuspid and additional pulmonary regurgitation result in continuous right atrial and ventricular enlargement secondary to volume overload. This in turn leads to worsening right ventricular dysfunction in patients with a potentially abnormal baseline ventricular function [64].
Although left-sided pathology is not as frequent and have not been as well characterized as right-sided lesions, several reports have described a less aggressive and more diffuse pattern of disease [65]. Characteristic echocardiographic features in the aortic position include decreased cusp mobility due to thickening and restriction as well as various degrees of leaflet doming (loss of normal concavity during diastole) with rolling of the free edge mainly leading to aortic regurgitation [66]. The mitral leaflets might be thickened and the subvalvular apparatus is most frequently affected resulting in chordal thickening and fusion, leading to retraction and restricted leaflet motion (chordal fusion has been only described in isolated cases of severe untreated disease). Most patients present with regurgitation combined with minimal (rare cases) degrees of stenosis.
The right ventricle has a complex semilunar shape, hence the assessment with two-dimensional methods based on geometrical models for volume estimation potentially results in random variable accuracy [67] To date, echocardiographic techniques to evaluate right ventricular function have included fractional area change (FAC), tricuspid annular plane systolic excursion (TAPSE), myocardial performance index (MPI), and rate of pressure rise. Although all these methods have been considered to have “good” correlation with radionuclide angiography ejection fraction and cardiac magnetic resonance [68], limitations have been ever present and they all require advanced echocardiographic skills. However, the inclusion of speckle tracking echocardiography has transformed the acquisition of right ventricular dimensions and volumes, as well as the calculation of right ventricular ejection fraction. Transthoracic echocardiography might be performed using a commercially available echocardiography system, and speckle tracking analysis is performed offline using a dedicated software. Several publications have reported on the increasing feasibility and applicability of STE as a predictor of right ventricular failure, clinical deterioration, and mortality in patients with CHD and pulmonary hypertension. Excluding patients with overt CHD, right ventricular strain was reduced in patients with 5-HIAA levels above the normal range compared to those with normal range levels [69].
3 Conclusion
Remarkable progress in the management of MNETs has generated multiple questions regarding optimal timing, sequencing, and selection of interventions [70] Surgery is at present indicated regardless of metastatic status, and tumor debulking (± adjuvant procedures) is considered to address mechanical complications, local disease, and particular hepatic lesions. This favoritism towards more contentious surgical strategies will in turn expand the use of baseline TTE and the requirement of cardiology clearance in newly diagnosed patients.
The increasing availability of efficacious treatment options to control the neuroendocrine tumor burden will surely introduce therapeutic dilemmas. Disease management may become longer and more complex, cardiac surgery may have a different role, and the incidence of CS and subsequent CHD may significantly decrease. If supplementary data further corroborate the efficacy of tryptophan hydroxylase inhibitors, the clinical profile of patients with CHD may drastically change. In consequence, physicians will potentially face more challenging patient profiles (non-responders) with refractory carcinoid syndrome, and perhaps more aggressive forms of cardiac involvement. Regardless, but more so if we observe this transition, patients at significant risk should be centralized to reference centers and followed by a multidisciplinary team that includes a cardiologist with expertise in structural and carcinoid heart disease. Furthermore, this change is also expected to complicate standardization of cardiac screening and surveillance.
As we reflect on our algorithm, the screening method for CHD should be chosen based on physician’s experience, initial test results, availability of techniques, and ultimately overall costs. Early involvement of an experienced cardiologist is essential in establishing an accurate baseline cardiovascular examination (often enriched by point-of-care echocardiography). Subsequently, initial test results should dictate follow up intervals and future appointments (i.e. symptomatic disease, poorly controlled tumor burden, the presence of valve disease or ventricular abnormalities, 5-HIAA levels, NT-proBNP levels). The availability of techniques (cardiac imaging) and workforce (multidisciplinary team or tumor board) certainly imposes the next management step; in the absence of a trained cardiovascular specialist (and other technical resources), the use of NT-proBNP for cardiac screening or monitoring of CHD seems fairly judicious. Overall costs should not represent a major concern at present (portable devices provide a very low-cost screening method). However, in a near future, survival improvement may certainly result in longer therapeutic and follow up periods.
References
Yordanova A, Ahmadzadehfar H, Gonzalez-Carmona M, Strassburg C, Mayer K, Feldmann G, et al. A step-by-step clinical approach for the management of neuroendocrine tumours. Horm Metab Res. 2017;49(2):77–85.
Mussig K, Oksuz MO, Dudziak K, Ueberberg B, Wehrmann M, Horger M, et al. Association of somatostatin receptor 2 immunohistochemical expression with [111In]-DTPA octreotide scintigraphy and [68Ga]-DOTATOC PET/CT in neuroendocrine tumors. Horm Metab Res. 2010;42(8):599–606.
Keck KJ, Maxwell JE, Menda Y, Bellizzi A, Dillon J, O'Dorisio TM, et al. Identification of primary tumors in patients presenting with metastatic gastroenteropancreatic neuroendocrine tumors. Surgery. 2017;161(1):272–9.
Grillo F, Albertelli M, Annunziata F, Boschetti M, Caff A, Pigozzi S, et al. Twenty years of gastroenteropancreatic neuroendocrine tumors: is reclassification worthwhile and feasible? Endocrine. 2016;53(1):58–62.
Kulke MH, Horsch D, Caplin ME, Anthony LB, Bergsland E, Oberg K, et al. Telotristat ethyl, a tryptophan hydroxylase inhibitor for the treatment of carcinoid syndrome. J Clin Oncol. 2017;35(1):14–23.
Pavel ME, Hainsworth JD, Baudin E, Peeters M, Horsch D, Winkler RE, et al. Everolimus plus octreotide long-acting repeatable for the treatment of advanced neuroendocrine tumours associated with carcinoid syndrome (RADIANT-2): a randomised, placebo-controlled, phase 3 study. Lancet. 2011;378(9808):2005–12.
Strosberg J, El-Haddad G, Wolin E, Hendifar A, Yao J, Chasen B, et al. Phase 3 trial of 177Lu-Dotatate for midgut neuroendocrine tumors. N Engl J Med. 2017;376(2):125–35.
Warner RR, Castillo JG. Carcinoid heart disease: the challenge of the unknown known. J Am Coll Cardiol. 2015;66(20):2197–200.
Davar J, Connolly HM, Caplin ME, Pavel M, Zacks J, Bhattacharyya S, et al. Diagnosing and managing carcinoid heart disease in patients with neuroendocrine tumors: an expert statement. J Am Coll Cardiol. 2017;69(10):1288–304.
Oberg K, Couvelard A, Delle Fave G, Gross D, Grossman A, Jensen RT, et al. Antibes consensus conference p. ENETS consensus guidelines for standard of care in neuroendocrine tumours: biochemical markers. Neuroendocrinology. 2017;105(3):201–11.
Kema IP, de Vries EG, Muskiet FA. Clinical chemistry of serotonin and metabolites. J Chromatogr B Biomed Sci Appl. 2000;747(1–2):33–48.
Manolopoulos VG, Ragia G, Alevizopoulos G. Pharmacokinetic interactions of selective serotonin reuptake inhibitors with other commonly prescribed drugs in the era of pharmacogenomics. Drug Metabol Drug Interact. 2012;27(1):19–31.
Lundin L, Norheim I, Landelius J, Oberg K, Theodorsson-Norheim E. Carcinoid heart disease: relationship of circulating vasoactive substances to ultrasound-detectable cardiac abnormalities. Circulation. 1988;77(2):264–9.
Bernheim AM, Connolly HM, Pellikka PA. Carcinoid heart disease in patients without hepatic metastases. Am J Cardiol. 2007;99(2):292–4.
Druce M, Rockall A, Grossman AB. Fibrosis and carcinoid syndrome: from causation to future therapy. Nat Rev Endocrinol. 2009;5(5):276–83.
Hannah-Shmouni F, Stratakis CA, Koch CA. Flushing in (neuro)endocrinology. Rev Endocr Metab Disord. 2016;17(3):373–80.
Koch CA, Azumi N, Furlong MA, Jha RC, Kehoe TE, Trowbridge CH, et al. Carcinoid syndrome caused by an atypical carcinoid of the uterine cervix. J Clin Endocrinol Metab. 1999;84(11):4209–13.
Castillo JG, Filsoufi F, Rahmanian PB, Anyanwu A, Zacks JS, Warner RR, et al. Early and late results of valvular surgery for carcinoid heart disease. J Am Coll Cardiol. 2008;51(15):1507–9.
Cosyns B, Droogmans S, Rosenhek R, Lancellotti P. Drug-induced valvular heart disease. Heart. 2013;99(1):7–12.
Jann H, Wertenbruch T, Pape U, Ozcelik C, Denecke T, Mehl S, et al. A matter of the heart: myocardial metastases in neuroendocrine tumors. Horm Metab Res. 2010;42(13):967–76.
Hutcheson JD, Setola V, Roth BL, Merryman WD. Serotonin receptors and heart valve disease--it was meant 2B. Pharmacol Ther. 2011;132(2):146–57.
Yabanoglu S, Akkiki M, Seguelas MH, Mialet-Perez J, Parini A, Pizzinat N. Platelet derived serotonin drives the activation of rat cardiac fibroblasts by 5-HT2A receptors. J Mol Cell Cardiol. 2009;46(4):518–25.
Rothman RB, Baumann MH, Savage JE, Rauser L, McBride A, Hufeisen SJ, et al. Evidence for possible involvement of 5-HT(2B) receptors in the cardiac valvulopathy associated with fenfluramine and other serotonergic medications. Circulation. 2000;102(23):2836–41.
Etienne N, Schaerlinger B, Jaffre F, Maroteaux L. The 5-HT2B receptor: a main cardio-pulmonary target of serotonin. J Soc Biol. 2004;198(1):22–9.
Li B, Zhang S, Zhang H, Nu W, Cai L, Hertz L, et al. Fluoxetine-mediated 5-HT2B receptor stimulation in astrocytes causes EGF receptor transactivation and ERK phosphorylation. Psychopharmacology. 2008;201(3):443–58.
Castillo JG, Milla F, Adams DH. Surgical management of carcinoid heart valve disease. Semin Thorac Cardiovasc Surg. 2012;24(4):254–60.
Riechelmann RP, Pereira AA, Rego JF, Costa FP. Refractory carcinoid syndrome: a review of treatment options. Ther Adv Med Oncol. 2017;9(2):127–37.
Connolly HM, Schaff HV, Mullany CJ, Rubin J, Abel MD, Pellikka PA. Surgical management of left-sided carcinoid heart disease. Circulation. 2001;104(12 Suppl 1):I36–40.
Modlin IM, Gustafsson BI, Pavel M, Svejda B, Lawrence B, Kidd M. A nomogram to assess small-intestinal neuroendocrine tumor (‘carcinoid’) survival. Neuroendocrinology. 2010;92(3):143–57.
Connolly HM, Schaff HV, Abel MD, Rubin J, Askew JW, Li Z, et al. Early and late outcomes of surgical treatment in carcinoid heart disease. J Am Coll Cardiol. 2015;66(20):2189–96.
Bhattacharyya S, Davar J, Dreyfus G, Caplin ME. Carcinoid heart disease. Circulation. 2007;116(24):2860–5.
Castillo JG, Silvay G, Solis J. Current concepts in diagnosis and perioperative management of carcinoid heart disease. Semin Cardiothorac Vasc Anesth. 2013;17(3):212–23.
Mekontso-Dessap A, Brouri F, Pascal O, Lechat P, Hanoun N, Lanfumey L, et al. Deficiency of the 5-hydroxytryptamine transporter gene leads to cardiac fibrosis and valvulopathy in mice. Circulation. 2006;113(1):81–9.
Bhattacharyya S, Toumpanakis C, Burke M, Taylor AM, Caplin ME, Davar J. Features of carcinoid heart disease identified by 2- and 3-dimensional echocardiography and cardiac MRI. Circ Cardiovasc Imaging. 2010;3(1):103–11.
Dobson R, Burgess MI, Banks M, Pritchard DM, Vora J, Valle JW, et al. The association of a panel of biomarkers with the presence and severity of carcinoid heart disease: a cross-sectional study. PLoS One. 2013;8(9):e73679.
Korse CM, Bonfrer JM, Aaronson NK, Hart AA, Taal BG. Chromogranin a as an alternative to 5-hydroxyindoleacetic acid in the evaluation of symptoms during treatment of patients with neuroendocrine tumors. Neuroendocrinology. 2009;89(3):296–301.
Marijon E, Ou P, Celermajer DS, Ferreira B, Mocumbi AO, Jani D, et al. Prevalence of rheumatic heart disease detected by echocardiographic screening. N Engl J Med. 2007;357(5):470–6.
Cives M, Soares HP, Strosberg J. Will clinical heterogeneity of neuroendocrine tumors impact their management in the future? Lessons from recent trials. Curr Opin Oncol. 2016;28(4):359–66.
Kunz PL, Reidy-Lagunes D, Anthony LB, Bertino EM, Brendtro K, Chan JA, et al. North American neuroendocrine tumor S. consensus guidelines for the management and treatment of neuroendocrine tumors. Pancreas. 2013;42(4):557–77.
Niederle B, Pape UF, Costa F, Gross D, Kelestimur F, Knigge U, et al. Vienna consensus conference p. ENETS consensus guidelines update for neuroendocrine neoplasms of the jejunum and ileum. Neuroendocrinology. 2016;103(2):125–38.
Ramage JK, Ahmed A, Ardill J, Bax N, Breen DJ, Caplin ME, et al. Guidelines for the management of gastroenteropancreatic neuroendocrine (including carcinoid) tumours (NETs). Gut. 2012;61(1):6–32.
Bhattacharyya S, Toumpanakis C, Caplin ME, Davar J. Analysis of 150 patients with carcinoid syndrome seen in a single year at one institution in the first decade of the twenty-first century. Am J Cardiol. 2008;101(3):378–81.
Dobson R, Valle JW, Burgess MI, Poston GJ, Cuthbertson DJ. Variation in cardiac screening and management of carcinoid heart disease in the UK and Republic of Ireland. Clin Oncol (R Coll Radiol). 2015;27(12):741–6.
Moller JE, Connolly HM, Rubin J, Seward JB, Modesto K, Pellikka PA. Factors associated with progression of carcinoid heart disease. N Engl J Med. 2003;348(11):1005–15.
Moore CL, Copel JA. Point-of-care ultrasonography. N Engl J Med. 2011;364(8):749–57.
Ploutz M, Lu JC, Scheel J, Webb C, Ensing GJ, Aliku T, et al. Handheld echocardiographic screening for rheumatic heart disease by non-experts. Heart. 2016;102(1):35–9.
Spencer KT, Kimura BJ, Korcarz CE, Pellikka PA, Rahko PS, Siegel RJ. Focused cardiac ultrasound: recommendations from the American Society of Echocardiography. J Am Soc Echocardiogr. 2013;26(6):567–81.
Changizi S, Khori V, Alizadeh AM. The effects of serotonin on the electrophysiological properties of atrioventricular node during an experimental atrial fibrillation. Naunyn Schmiedeberg’s Arch Pharmacol. 2015;388(8):843–52.
Langer C, Piper C, Vogt J, Heintze J, Butz T, Lindner O, et al. Atrial fibrillation in carcinoid heart disease: the role of serotonin. A review of the literature. Clin Res Cardiol. 2007;96(2):114–8.
Yao JC, Hassan M, Phan A, Dagohoy C, Leary C, Mares JE, et al. One hundred years after “carcinoid”: epidemiology of and prognostic factors for neuroendocrine tumors in 35,825 cases in the United States. J Clin Oncol. 2008;26(18):3063–72.
Selim AM, Sarswat N, Kelesidis I, Iqbal M, Chandra R, Zolty R. Plasma serotonin in heart failure: possible marker and potential treatment target. Heart Lung Circ. 2017;26(5):442–9.
Hood KY, Mair KM, Harvey AP, Montezano AC, Touyz RM, MacLean MR. Serotonin signaling through the 5-HT1B receptor and NADPH oxidase 1 in pulmonary arterial hypertension. Arterioscler Thromb Vasc Biol. 2017;37(7):1361–70.
Bhattacharyya S, Toumpanakis C, Caplin ME, Davar J. Usefulness of N-terminal pro-brain natriuretic peptide as a biomarker of the presence of carcinoid heart disease. Am J Cardiol. 2008;102(7):938–42.
Dobson R, Burgess MI, Valle JW, Pritchard DM, Vora J, Wong C, et al. Serial surveillance of carcinoid heart disease: factors associated with echocardiographic progression and mortality. Br J Cancer. 2014;111(9):1703–9.
Killu AM, Newman DB, Miranda WR, Maleszewski JJ, Pellikka P, Schaff HV, et al. Carcinoid heart disease without severe tricuspid valve involvement. Cardiology. 2016;133(4):217–22.
Meijers WC, van der Velde AR, Muller Kobold AC, Dijck-Brouwer J, Wu AH, Jaffe A, et al. Variability of biomarkers in patients with chronic heart failure and healthy controls. Eur J Heart Fail. 2017;19(3):357–65.
Bonou M, Kapelios CJ, Kaltsas G, Perreas K, Toutouzas K, Barbetseas J. Cardiac surgery for carcinoid heart disease: a weapon not to be misused. Cardiology. 2017;136(4):243–51.
Nishimura RA, Otto CM, Bonow RO, Carabello BA, Erwin JP 3rd, Fleisher LA, et al. 2017 AHA/ACC focused update of the 2014 AHA/ACC guideline for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on clinical practice guidelines. J Am Coll Cardiol. 2017;70(2):252–89.
Vahanian A, Alfieri O, Andreotti F, Antunes MJ, Baron-Esquivias G, Baumgartner H, et al. Guidelines on the management of valvular heart disease (version 2012): the joint task force on the management of valvular heart disease of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS). Eur J Cardiothorac Surg. 2012;42(4):S1–44.
Bhattacharyya S, Raja SG, Toumpanakis C, Caplin ME, Dreyfus GD, Davar J. Outcomes, risks and complications of cardiac surgery for carcinoid heart disease. Eur J Cardiothorac Surg. 2011;40(1):168–72.
Castillo JG, Filsoufi F, Adams DH, Raikhelkar J, Zaku B, Fischer GW. Management of patients undergoing multivalvular surgery for carcinoid heart disease: the role of the anaesthetist. Br J Anaesth. 2008;101(5):618–26.
Zoghbi WA, Enriquez-Sarano M, Foster E, Grayburn PA, Kraft CD, Levine RA, et al. Recommendations for evaluation of the severity of native valvular regurgitation with two-dimensional and Doppler echocardiography. J Am Soc Echocardiogr. 2003;16(7):777–802.
Pellikka PA, Tajik AJ, Khandheria BK, Seward JB, Callahan JA, Pitot HC, et al. Carcinoid heart disease. Clinical and echocardiographic spectrum in 74 patients. Circulation. 1993;87(4):1188–96.
Castillo JG, Adams DH, Fischer GW. Absence of the tricuspid valve due to severe carcinoid heart disease. Rev Esp Cardiol. 2010;63(1):96.
Castillo JG, Filsoufi F, Rahmanian PB, Adams DH. Quadruple valve surgery in carcinoid heart disease. J Card Surg. 2008;23(5):523–5.
Bhattacharyya S, Schapira AH, Mikhailidis DP, Davar J. Drug-induced fibrotic valvular heart disease. Lancet. 2009;374(9689):577–85.
Dorosz JL, Bolson EL, Waiss MS, Sheehan FH. Three-dimensional visual guidance improves the accuracy of calculating right ventricular volume with two-dimensional echocardiography. J Am Soc Echocardiogr. 2003;16(6):675–81.
Ueti OM, Camargo EE, Ueti Ade A, de Lima-Filho EC, Nogueira EA. Assessment of right ventricular function with Doppler echocardiographic indices derived from tricuspid annular motion: comparison with radionuclide angiography. Heart. 2002;88(3):244–8.
Haugaa KH, Bergestuen DS, Sahakyan LG, Skulstad H, Aakhus S, Thiis-Evensen E, et al. Evaluation of right ventricular dysfunction by myocardial strain echocardiography in patients with intestinal carcinoid disease. J Am Soc Echocardiogr Off Publ Am Soc Echocardiogr. 2011;24(6):644–50.
Mehrvarz Sarshekeh A, Halperin DM, Dasari A. Update on management of midgut neuroendocrine tumors. Int J Endocr Oncol. 2016;3(2):175–89.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest statement
The authors declare that they have no conflict of interest at present.
Informed consent
For this type of study formal consent is not required.
Human and animal rights
This review does not contain any studies with human participants or animals performed by any of the authors.
Appendix
Appendix
APPROPRIATE USE CRITERIA FOR TRANSTHORACIC ECHOCARDIOGRAPHY (Maximum Score)a | EXTRAPOLATION TO PATIENTS WITH MNETs AND CARCINOID SYDNROME |
|---|---|
Symptoms or conditions such as chest pain, dyspnea, palpitations, TIA, stroke, or peripheral embolic event | Unusual fatigue, dyspnea, palpitations and changes in blood pressure (carcinoid crisis and potential dehydration) |
Prior testing that is concerning for SHVD such as chest X-ray, ECG, or cardiac biomarkers | Non-specific ST changes, STach, p pulmonale or RBBB on the EKG. Elevation of carcinoid ± cardiac biomarkers |
Frequent or exercise-induced ventricular premature complexes, atrial fibrillation, supraventricular or ventricular tachycardia | Restricted HR variability, AFib, SVT and AFib due to heart failure, carcinoid crisis |
Clinical symptoms or signs consistent with a cardiac diagnosis known to cause lightheadedness / presyncope / syncope | Carcinoid syndrome and uncontrolled diarrhea may lead to dehydration and lightheadedness / presyncope / syncope |
Syncope when there are no other symptoms or signs of cardiovascular disease | Acute changes in blood pressure and arrhythmias (carcinoid crisis) may lead to syncopal episodes |
Evaluation (and re-evaluation) of suspected pulmonary hypertension including evaluation of right ventricular function | Evaluation of suspected pulmonary hypertension including evaluation of right ventricular function |
Increasing dyspnea, respiratory failure or hypoxemia of uncertain etiology | Increasing fatigue and dyspnea (hypoxemia) due to frequent bronchospasm and/or congestive heart failure |
Initial evaluation (and re-evaluation) when there is a reasonable suspicion of valvular or structural heart disease | Initial evaluation after diagnosis of MNET or re-evaluation after positive screening for CHD (see algorithm) |
Routine surveillance (≥3y) of mild valvular stenosis or regurgitation without a change in clinical status or cardiac exam | Routine surveillance (Q6M - Q1y) of mild valvular regurgitation w/o a change in symptoms or cardiac exam |
Routine surveillance (≥1y) of moderate or severe valvular stenosis or regurgitation without a change in clinical status or cardiac exam | Routine surveillance (Q3M - Q6M) of ≥ moderate valvular regurgitation w/o a change in symptoms or cardiac exam |
Suspected cardiac mass, suspected cardiovascular source of embolus, suspected pericardial conditions | Suspected cardiac mass, suspected cardiovascular source of embolus, suspected pericardial conditions |
Initial evaluation (and re-evaluation) of known or suspected HF based on symptoms, signs, or abnormal test results | Evaluation (and re-evaluation) of known or suspected HF based on symptoms, signs, or abnormal test results |
Routine surveillance (≥1y) of HF when there is no change in clinical status or cardiac exam | Routine surveillance (Q6M) of HF (always after using more advanced imaging techniques to have a baseline assessment) |
Initial evaluation (and re-evaluation) of known or suspected cardiomyopathy | Evaluation (and re-evaluation) of known or suspected CMP (always after using more advanced imaging techniques) |
Baseline and serial re-evaluations in a patient undergoing therapy with cardiotoxic agents | Baseline and serial re-evaluations in a patient undergoing therapy with cardiotoxic agents |
Initial evaluation of known or suspected adult congenital heart disease | Initial evaluation of the atrial septum mandates to perform a “bubble test” during the first TTE assessment |
Rights and permissions
About this article
Cite this article
Castillo, J.G., Naib, T., Zacks, J.S. et al. Echocardiography in functional midgut neuroendocrine tumors: When and how often. Rev Endocr Metab Disord 18, 411–421 (2017). https://doi.org/10.1007/s11154-017-9434-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11154-017-9434-z