Abstract
Purpose
The purpose of this study was to explore the ability of Distress Thermometer (DT) scores to discern important differences in quality of life scores among women with breast cancer.
Methods
The National Comprehensive Cancer Network’s DT, the Functional Assessment of Cancer Therapy—Breast (FACT–B), and a demographic questionnaire were completed by 111 women recently diagnosed with breast cancer.
Results
Patients considered moderately to severely distressed (score ≥ 4 on DT) scored significantly lower on FACT–B QOL scales and subscales when compared to those in the group scoring 3 or below. For those scales for which minimally important differences (MIDs) have been established, differences between the two groups were 2–3 and a half times the established MID.
Conclusions
Moderately to severely distressed patients have significantly lower QOL than those with expected or mild distress. The DT provides a quick and easy screening tool to alert the healthcare team to clinically relevant alterations in patients’ QOL.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
According to the World Health Organization, breast cancer is the most prevalent cancer affecting women worldwide [1]. In the United States, an estimated 207,090 women will be diagnosed with breast cancer in 2011 [2]. Those affected by cancer often encounter a trajectory filled with potential for worry, stress, and loss. The national comprehensive cancer network (NCCN) advocates use of the term distress to describe the negative emotional experience of individuals diagnosed with cancer [3]. Distress is a common response to cancer and can result in diminished health-related quality of life (QOL) [4].
Distress and quality of life (QOL) are most often negatively correlated and distress has been studied as a predictor of QOL [5–7]. Yet, according to Carlson and Bultz [8], the distinction between distress and QOL needs to be delineated, as the terms are often used interchangeably and perhaps inaccurately in the literature. QOL encompasses a broad spectrum of issues, including physical, social, cognitive, spiritual, emotional, and role functioning, as well as other psychological symptoms and physical symptoms such as pain, nausea and vomiting, and fatigue [8]. While acknowledging QOL’s multidimensionality and subjectivity, Cella defined the construct as the patients’ appraisal of and satisfaction with their current level of functioning as compared to what they perceive to be possible or ideal [9]. Although an Institute of Medicine (IOM) [10] committee found no consensus regarding a conceptual model for QOL, the most common domains measured include the psychological, social, physical, and spiritual dimensions [11–13].
Comparing the NCCN [3] definition of distress to Cella’s definitions may provide insight into the relationship between the two constructs. As defined by NCCN [3], distress is a “… multifactorial unpleasant emotional experience of a psychological (cognitive, behavioral, emotional), social, and/or spiritual nature that may interfere with the ability to cope effectively with cancer, its physical symptoms and its treatment” (p. DIS–2). In contrast to QOL, distress refers primarily to symptoms of anxiety, depression, and adjustment disorders related to the cancer experience [8]. Distress may result from the person’s struggle to adjust to or cope with the impact of cancer on his or her expected physical, emotional, and social well-being.
Distress is a common problem in cancer patients with studies reporting a prevalence rate of 26–62% in patients with a variety of cancer diagnoses at different stages [14–21]. However, it has been estimated only 5% of distressed patients are recognized and receive treatment [22]. The significance of these findings has led some to propose distress as the sixth vital sign [22, 23]. Clinicians are often focused on treating the cancer and its physical manifestations, and may be too busy to assess for distress, or may assume patients would express psychosocial needs if they had them [4]. Given psychosocial factors play a role in QOL and psychological adjustment [24], comprehensive assessment and holistic care planning are arguably beneficial for patients and providers.
The relationship between distress and QOL has been studied in cancer samples to a limited degree. Manning Walsh [7] found symptom distress and QOL to be inversely correlated in women with breast cancer diagnosed within the past 2 years (mean months since diagnosis = 10) and that symptom distress explained 34% of the variance in QOL. In a study of high-grade glioma patients, Fox, Lyon, and Farace [5] found that symptoms of distress (depression, fatigue, and sleep disturbance) explained 29% of the variance in QOL. In another study of brain tumor patients, researchers found that distress (symptoms of depression) explained more than half of the variance in QOL [6].
Heightened levels of distress have been associated with poorer QOL across multiple domains [25]. A study of high-grade brain tumor patients exploring relationships between distress using the Distress Thermometer (DT), and QOL using the Functional Assessment of Cancer Therapy–Brain (FACT–Br) found significant negative correlations between distress ratings and emotional well-being (EWB) and social well-being (SWB) subscales and a lesser degree of correlation with the physical well-being (PWB) subscale [26]. While the correlation between distress and QOL has been established, no studies to date have examined the differences in QOL scores between those considered minimally distressed and those considered moderately to severely distressed.
To address the lack of knowledge regarding the relationship between degree of distress and QOL in women with newly diagnosed breast cancer, this study explored the ability of DT scores to discern important differences in QOL scores.
Methods
Procedure
Data were collected as part of a larger project designed to examine the implementation of a chronic disease care model applied to multidisciplinary breast cancer care (Centers for Disease Control and Prevention, grant number H75/CCH424696-01). The major focus of the larger project involved providing psychosocial support services within a multidisciplinary breast cancer clinic based upon evidence-based practice in psychosocial oncology. Data used for this analysis included measures of patient demographic characteristics, distress, distress-related problems, and QOL, which were collected at baseline prior to intervention.
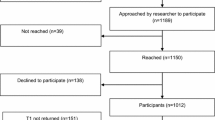
Prior to data collection, the study was approved by the University of Louisville Human Subjects Protection Program and the James Graham Brown Cancer Center’s (JGBCC) Clinical Scientific Review Committee. A research nurse was present during clinic operation to screen candidates for eligibility. All patients presenting to the JGBCC Multidisciplinary Breast Clinic for treatment between November 29th, 2005 and October 3rd, 2006 were invited to participate if they met the following inclusion criteria: (a) age 21 or older; (b) diagnosed with locally invasive or metastatic breast cancer; and (c) able to respond to surveys in written or spoken English. Following preamble consent, research instruments were self-administered with assistance offered if needed. Although some chose to return the instruments by mail, most participants (65%) completed the surveys during their clinic visit. Data were collected from 111 patients diagnosed with breast cancer within the previous 6 months.
Measures
Research instruments included a basic demographic questionnaire, the DT [27], and the Functional Assessment of Cancer Therapy—Breast (FACT–B) [28]. See Table 1 for a comparison of measures used to operationalize study constructs.
Demographic characteristics
The demographic questionnaire included questions concerning age, race, marital status, education in years, income, employment status (before and after cancer), ages of children, number of people in the household, and insurance status.
Overall distress and problem list
The DT [27] was used to measure overall distress. Participants rated overall distress during the prior week on an 11-point scale with zero indicating “no distress” and 10 signifying “extreme distress.” Studies have shown the DT to compare favorably with other measures of distress including: the hospital anxiety and depression scale (HADS) [25, 29–31]; the center for epidemiological studies depression scale (CES-D) [32]; and the brief symptom inventory (BSI-18) [25]. The cut-off score for determining sensitivity to adjustment disorders and major depression on the DT, when compared to these other instruments, has varied from 4 [32] to 5 [29, 30]. The NCCN distress management guidelines state a score of 4 or greater indicates moderate or severe distress and should trigger further evaluation while a score less than 4 signifies mild distress or “expected distress” to be routinely managed by the primary oncology team. In a pooled analysis of nine studies involving 1,477 patients, Mitchell found the DT’s sensitivity to be 77.1%, specificity to be 66.1%, positive predictive value to be 55.6%, and negative predictive value to be 84.0% [33], suggesting that a two-stage screening process may be indicated to identify possible cases of distress.
A problem list accompanies the DT [27] and can be used to identify causes for distress. Patients indicate whether or not they have experienced specific problems in the past week. The list of problems includes practical, family, emotional, spiritual/religious and physical issues. Hoffman et al. studied the internal consistency of the problem groupings and found strong alphas for physical (0.92) and emotional (0.88) subscales but low alphas for the practical (0.42) and spiritual (0.31) groupings [34].
Quality of life
Health-related QOL was measured using the Functional Assessment of Cancer Therapy–Breast (FACT–B) [28]. The FACT–B includes the four FACT–General (FACT–G) subscales: PWB, EWB, SWB, and functional well-being (FWB) and the breast cancer subscale (BCS). BCS items have been demonstrated to be uniquely important for women with breast cancer and were included in this subscale based upon input from patients and practitioners. These items address issues such as: shortness of breath, sexuality, hair loss, weight change, pain and swelling. Initial validation of the FACT–B estimated the alpha coefficient for the total score to be 0.88 with subscale alpha coefficients ranging from 0.63 to 0.90; estimates of test–retest reliability, convergent and divergent validity also supported the utility of this instrument [35]. For this study population, alpha coefficients were 0.89 for the PWB, 0.84 for the EWB, 0.87 for the FWB, 0.85 for the SWB, and 0.77 for the BCS. Alphas for the total FACT–G and total FACT–B were 0.93 and 0.94, respectively (see Table 3 for a comparison of alphas from normative data and alpha performance in this study).
For the FACT subscales, respondents used a Likert scale to indicate how true statements related to various aspects of their illness had been for them during the prior week. Items were rated on a five-point scale with zero indicating that an item applied “not at all” and four indicating “very much.” To calculate subscale scores, items were reverse coded as necessary so that lower ratings represented a more negative state. Values for subscale items were then summed to produce the subscale score. To deal with missing subscale item values, the scoring algorithm described in the Manual of the Functional Assessment of Chronic Illness Therapy (FACIT Scales), Version 4, was used [28]. A trial outcome index (TOI) score (considered to be a sensitive indicator of outcomes [36]), was obtained by adding the individual’s scores on the PWB, FWB, and BCS. An overall FACT–G score was obtained by adding scores on the four subscales of that instrument; the overall FACT–B score included scores on the FACT–G subscales plus the BCS. Higher scores on the FACT–G are indicative of higher QOL.
Normative reference values for the FACT–B were developed via a 3 year validation study of the FACT Measurement System (the Bilingual Intercultural Oncology Quality of Life project) [37].
Minimally Important Differences (MIDs) for the FACT–B were established by Eton et al. using both distribution- and anchor-based methods [36]. According to the Clinical Significance Consensus Meeting Group, “the MID is the smallest difference in score in the domain of interest that patients perceive as important, either beneficial or harmful, and which would lead the clinician to consider a change in the patient’s management” [36]. The following MID estimates were supported by the Eton study: BCS = 2–3 points; TOI = 5–6 points; FACT–G total = 5–6 points; and FACT–B = 7–8 points.
Analysis plan
Statistical Package for the Social Sciences (SPSS), Version 19 was used for data management and analysis. Pearson’s product-moment correlations were used to examine correlations between distress and the QOL subscales and overall scores. One sample t tests were used to compare the sample results to normative data for the FACT–B. The sample was then subdivided into two groups: (1) those scoring 4 or above (moderately to severely distressed); and (2) those scoring below four (mildly distressed). Independent samples t tests were used to evaluate differences on QOL scales and subscales between these two groups. Mean scores on the TOI, BCS, FACT–G, and FACT–B were determined; the mean score differences were calculated and compared to established MIDs. Cohen’s d was calculated as a measure of effect size.
Results
Sample characteristics
Two of the 113 eligible patients invited to participate opted out of the study due to survey burden, resulting in a 98% participation rate. Demographic information for the 111 participants is summarized in Table 2. The average age of women in the sample was 52.7 years (SD = 11.7, median = 51). Participants had an average of 13 years of education (SD = 2.84, median = 12 years). Most participants were Caucasian (72%) and either married or living with a partner (59%). Most participants had children (92%), and about one-third had one or more children age 18 or younger. A large proportion (35%) lived in households with incomes under $10,000. Approximately 23% were receiving Medicaid and 13% had no insurance. Thirty-two percent of women reported having stage I breast cancer, 12% stage II, 26% stage III, 12% stage IV, and 17% were unknown.
Distress
The mean DT score for study participants was 4.87 (SD = 3.2, median = 5.0) (see Table 3). As shown in Table 4, the most frequently identified problems on the problem list were either emotional or physical with over half of participants reporting fatigue (57%), worry (55%), nervousness (55%), sleep-related problems (55%), or sadness (54%).
Health-related quality of life
Mean FACT–B total and subscale scores for study participants are reported and compared to normative or comparative data in Table 3. Statistically significant differences were found between our data and the comparative data on FACT–B total scores, FACT–G total scores, BCS scores, FWB scores, EWB score, and PWB scores.
QOL differences by degree of distress
Patients considered moderately to severely distressed (score ≥ 4 on DT) scored significantly lower on all QOL scales and subscales when compared to those in the group scoring 3 or below. Mean differences between the two groups compared to established minimally important differences (MIDs) are displayed in Table 5. Differences between the two groups were 2–3.5 times higher than the established MIDs. Using Cohen’s d, moderate effect sizes were found when scores on the BCS, TOI, FACT–G, and FACT–B were compared.
Discussion
The primary aim of this study was to explore the ability of Distress Thermometer scores to differentiate important differences in QOL scores in women recently diagnosed with breast cancer. In comparing QOL scores between participants with moderate to severe distress (score of 4 or above) and those with mild or “expected” distress (3 or less), we found overall scores on the FACT–B to be 27 points lower and scores on the BCS scale to be 6 points lower for those with moderate to severe distress. The mean differences between the two groups were two to three and a half times the MID with moderate effect sizes. This finding is significant in that (1) it is clinically relevant and demonstrates that the cut-off score for determining distress on the DT reflects significant clinical differences in patients’ QOL; and (2) it provides further validation for the utility of the DT as a meaningful, brief screening tool.
Comprehensive patient assessment is time consuming and burdensome to both patients and staff [38]. Therefore, brief screening tools are needed to identify those in need of further assessment and assistance. Because of its brevity and ease of administration, the DT is well-suited for alerting the treatment team to the intensity of the patient’s reaction to the disease. Indeed, Mitchell et al. [39] explored the acceptability of common screening methods used to detect distress and related mood disorders among cancer care clinicians and found 77% of clinicians would be prepared to utilize an ultra-short method, providing some evidence to support the acceptability of a tool such as the DT. Ultra-short measures such as the DT have proven value in screening and identifying patients in need of further assessment but also have a number of limitations. Specifically, ultra-short measures lack specificity in identifying particular problems such as depression; problems with determining the face-validity of single-item measures and patient’s difficulty scaling themselves on single-item measures further complicate their use [40]. Patients have also indicated that single-item evaluations do not completely capture their situation [40].
Used alone, the DT does not provide insight related to the factors contributing to the distress. While the dichotomous problem list can provide additional information as to sources of distress and suggest referral needs, “Yes” or “No” answers are less likely to yield clinically useful information regarding response to an intervention and may not adequately represent the patient’s total response to a specific problem area or the weight that problem carries for them in terms of distress. In order to address this issue, Akizuki et al. added an Impact Thermometer to the DT to increase specificity and evaluate impairment of social functioning [41], and researchers in Belgium combined the DT with the colored complaint scale to create the “Distress Barometer” with the goal of improving the specificity of distress screening without decreasing the sensitivity of the DT [42].
Lower QOL scores have been associated with worse clinical outcomes. A recent meta-analysis of 30 randomized controlled trials found health-related QOL scores to predict survival [43]. Further assessment of QOL of distressed patients may assist the clinical team to identify intervention strategies targeting problematic domains. Unfortunately, QOL data has often been collected without being used to impact treatment decision-making [44]. When patients are assessed using the DT and QOL instruments, it is important to incorporate findings into care planning and treatment outcome goals. As an early proponent of QOL measurement, Calman challenged those using QOL measures not only to identify the “gap” between the individual’s expectations and experience but also to direct efforts toward narrowing or eliminating the gap and improving a person’s QOL [45].
Our high participation rate (98%) among all eligible patients provided us with a representative sample of women treated at our multidisciplinary breast cancer clinic. This accrual success was made possible by having a research nurse present and actively recruiting on clinic days. Our sample does differ from the general population of breast cancer patients on several parameters. First, the median age of 51 in our sample is younger than the national median age of women diagnosed with breast cancer [46] making it a comparatively young sample. Second, the majority of psychosocial oncology research has involved upper middle class participants [47]. In this sample, 35% of participants had household incomes of $10,000 or less, perhaps making this sample more representative of the breast cancer population with regard to socioeconomic resources. Third, the mean distress score of 4.87, in this sample, met the NCCN criteria for further evaluation [3]. The NCCN–recommended cutoff for moderate to severe distress is 4, making this a relatively distressed sample. Fourth, QOL subscale and total scale scores for this study sample were lower than such scores reported from the normative sample. Similar to the Eton breast cancer study cited earlier, Brucker et al. [37] identified increments of at least two points for subscales and at least five points for the FACT-G total scores as minimally important differences to be used in comparing samples or multiple administrations of the scales in the same population. Using this standard, the scores of our sample were notably lower than those for the normative population on the FACT–G total (−8.0 difference), FACT–B total (−14.89), and the PWB (−2.7), FWB (−3.16) and BCS (−2.59) subscales. Numerous studies have reported younger breast cancer patients experience greater distress than older women with the same diagnosis [48–51] and lower income cancer patients have higher distress and lower QOL [47, 52]. Therefore, it is not surprising that our younger, lower income sample experienced higher distress and lower QOL than the cancer population at large.
These results should be interpreted in light of some study limitations including a relatively small sample size and reliance on cross-sectional data. Additionally, the sample was derived from a tertiary cancer care facility and a dedicated multidisciplinary breast cancer clinic; therefore, the results are best generalized to women with breast cancer receiving treatment in similar settings. The sample also demonstrated some unique characteristics worthy of mentioning, including generally lower levels of physical and functional well-being, greater concerns related to breast cancer, and lower overall QOL as compared to established norms. Finally, results of this study are based on self-reported distress and QOL. Cross validation of such findings with collateral assessments from health care providers, caregivers, or significant others might add another dimension to understanding distress and QOL in such a population.
Conclusion
Based on study findings, moderately to severely distressed patients have significantly lower QOL than those with expected or mild distress. The DT provides a quick and easy screening tool to alert the healthcare team to specific patients experiencing negative alterations in their QOL. Adding a QOL measure is one way to gain a more comprehensive picture of the areas or domains contributing to that distress.
This research was supported by a grant from the Centers for Disease Control and Prevention; grant number H75/CCH424696-01. Ms. Schapmire was supported by a Doctoral Training Grant from the American Cancer Society.
References
World Health Organization. (2011). Breast cancer: Prevention and control. Retrieved May 10th, 2011, from http://www.who.int/cancer/detection/breastcancer/en/.
Howlader, N., Noone, A. M., Krapcho, M., Neyman, N., Aminou, R.,& Waldron, W. et al. (2011). SEER stat fact sheets: Breast. Retrieved May 10th, 2011, from http://www.seer.cancer.gov/statfacts/html/breast.html.
National Comprehensive Cancer Network. (2010). NCCN clinical practice guidelines in oncology: Distress management. (Vol. v.1.2010). Jenkintown, PA: NCCN.
Madden, J. (2006). The problem of distress in patients with cancer: More effective assessment. Clinical Journal of Oncology Nursing, 10(5), 615–619.
Fox, S. W., Lyon, D., & Farace, E. (2007). Symptom clusters in patients with high-grade glioma. Journal of Nursing Scholarship, 39(1), 61–67.
Pelletier, G., Verhoef, M. J., Khatri, N., & Hagen, N. (2002). Quality of life in brain tumor patients: The relative contributions of depression, fatigue, emotional distress, and existential issues. Journal of Neuro-oncology, 57(1), 41–49.
Manning Walsh, J. K. (2005). Social support as a mediator between symptom distress and quality of life in women with breast cancer. Journal of Obstetric, Gynecologic, and Neonatal Nursing, 34(4), 482–493.
Carlson, L. E., & Bultz, B. D. (2003). Cancer distress screening: Needs, models, and methods. Journal of Psychosomatic Research, 55(5), 403–409.
Cella, D. F., & Cherin, E. A. (1988). Quality of life during and after cancer treatment. Comprehensive Therapy, 14(5), 69–75.
Institute of Medicine, National Research Council. (2005). From cancer patient to cancer survivor: Lost in transition. Washington, D.C.: The National Academies Press.
Ferrell, B. R., Grant, M. M., Funk, B., Otis-Green, S., & Garcia, N. (1997). Quality of life in breast cancer survivors as identified by focus groups. Psycho-Oncology, 6(1), 13–23.
Juarez, G., Ferrell, B. R., Uman, G., Podnos, Y., & Wagman, L. (2008). Distress and quality of life concerns of family caregivers of patients undergoing palliative surgery. Cancer Nursing, 31(1), 2–10.
Institute of Medicine [IOM]. (2008). Cancer care for the whole patient: Meeting psychosocial health needs. Washington, D.C.: The National Academies Press.
Dabrowski, M., Boucher, K., Ward, J. H., Lovell, M. M., Sandre, A., Bloch, J., et al. (2007). Clinical experience with the NCCN distress thermometer in breast cancer patients. Journal of the National Comprehensive Cancer Network, 5(1), 104–111.
Carlson, L. E., Angen, M., Cullum, J., Goodey, E., Koopmans, J., Lamont, L., et al. (2004). High levels of untreated distress and fatigue in cancer patients. British Journal of Cancer, 90(12), 2297–2304.
Fallowfield, L., Ratcliffe, D., Jenkins, V., & Saul, J. (2001). Psychiatric morbidity and its recognition by doctors in patients with cancer. British Journal of Cancer, 84(8), 1011–1015.
Graves, K. D., Arnold, S. M., Love, C. L., Kirsh, K. L., Moore, P. G., & Passik, S. D. (2007). Distress screening in a multidisciplinary lung cancer clinic: Prevalence and predictors of clinically significant distress. Lung Cancer, 55(2), 215–224.
Keir, S. T., Calhoun-Eagan, R. D., Swartz, J. J., Saleh, O. A., & Friedman, H. S. (2008). Screening for distress in patients with brain cancer using the NCCN’s rapid screening measure. Psycho-Oncology, 17(6), 621–625.
Sellick, S. M., & Edwardson, A. D. (2007). Screening new cancer patients for psychological distress using the Hospital Anxiety and Depression Scale. Psycho-Oncology, 16(6), 534–542.
Zabora, J., Brintzenhofeszoc, K., Curbow, B., Hooker, C., & Piantadosi, S. (2001). The prevalence of psychological distress by cancer site. Psycho-Oncology, 10(1), 19–28.
Hegel, M. T., Moore, C. P., Collins, E. D., Kearing, S., Gillock, K. L., Riggs, R. L., et al. (2006). Distress, psychiatric syndromes, and impairment of function in women with newly diagnosed breast cancer. Cancer, 107(12), 2924–2931.
Bultz, B. D., & Holland, J. C. (2006). Emotional distress in patients with cancer: The sixth vital sign. Community Oncology, 3(5), 311–314.
Bultz, B., & Carlson, L. (2006). Emotional distress: The sixth vital sign—Future directions in cancer care. Psycho-Oncol, 15, 93–95.
Shapiro, S., Lopez, A., Schwartz, G., Bootzin, R., Figueredo, A., Braden, C., et al. (2001). Quality of life and breast cancer: Relationship to psychosocial variables. Journal of Clinical Psychology, 57(4), 501–519.
Jacobsen, P. B., Donovan, K. A., Trask, P. C., Fleishman, S. B., Zabora, J., Baker, F., et al. (2005). Screening for psychologic distress in ambulatory cancer patients: A multicenter evaluation of the Distress Thermometer. Cancer, 103(7), 1494–1502.
Kvale, E., Murthy, R., Taylor, R., Lee, J., & Nabors, L. (2009). Distress and quality of life in primary high-grade brain tumor patients. Supportive Care in Cancer, 17(7), 793–799.
National Comprehensive Cancer Network. (2003). Distress management practice guidelines. Journal of the National Comprehensive Cancer Network, 1, 344–374.
Cella, D. F. (1997). Manual of the Functional Assessment of Chronic Illness Therapy (FACIT Scales) (Version 4 ed.). Evanston, IL: Evanston Northwestern Healthcare.
Cohen, M., Hoffman, R. G., Cromwell, C., Schmeidler, J., Ebrahim, F., Carrera, G., et al. (2002). The prevalence of distress in persons with human immunodeficiency virus infection. Psychosomatics, 43(1), 10–15.
Roth, A., Kornblith, A. B., Batel-Copel, L., Peabody, E., Scher, H. I., & Holland, J. C. (1998). Rapid screening for psychologic distress in men with prostate carcinoma: A pilot study. Cancer, 82(10), 1904–1908.
Trask, P., Paterson, A., Riba, M., Brines, B., Griffith, K., Parker, P., et al. (2002). Assessment of psychological distress in prospective bone marrow transplant patients. Bone Marrow Transpl, 29, 917–925.
Ransom, S., Jacobsen, P. B., & Booth-Jones, M. (2006). Validation of the distress thermometer with bone marrow transplant patients. Psycho-Oncology, 15(7), 604–612.
Mitchell, A. J. (2007). Pooled results from 38 analyses of the accuracy of distress thermometer and other ultra-short methods of detecting cancer-related mood disorders. Journal of Clinical Oncology, 25(29), 4670–4681.
Hoffman, B. M., Zevon, M. A., D’Arrigo, M. C., & Cecchini, T. B. (2004). Screening for distress in cancer patietns: The NCCN rapid-screening measure. Psycho-Oncology, 13, 792–799.
Cella, D. F., Tulsky, D. S., Gray, G., Sarafian, B., Linn, E., Bonomi, A., et al. (1993). The functional assessment of cancer therapy scale: Development and validation of the general measure. Journal of Clinical Oncology, 11(3), 570–579.
Eton, D. T., Cella, D., Yost, K. J., Yount, S. E., Peterman, A. H., Neuberg, D. S., et al. (2004). A combination of distribution-and anchor-based approaches determined minimally important differences (MIDs) for four endpoints in a breast cancer scale. Journal of Clinical Epidemiology, 57, 898–910.
Brucker, P., Yost, K., Cashy, J., Webster, K., & Cella, D. F. (2005). General population and cancer patient norms for the Functional Assessment of Cancer Therapy-General (FACT-G). Evaluation And The Health Professions, 28(2), 192–211.
Butt, Z., Wagner, L. I., Beaumont, J. L., Paice, J. A., Peterman, A. H., Shevrin, D., et al. (2008). Use of a single-item screening tool to detect clinically significant fatigue, pain, distress, and anorexia in ambulatory cancer practice. Journal of Pain and Symptom Management, 35(1), 20–30.
Mitchell, A. J., Kaar, S., Coggan, C., Herdman, J., Mitchell, A. J., Kaar, S., et al. (2008). Acceptability of common screening methods used to detect distress and related mood disorders-preferences of cancer specialists and non-specialists. Psycho-Oncology, 17(3), 226–236.
Vodermaier, A., Linden, W., & Siu, C. (2009). Screening for emotional distress in cancer patients: A systematic review of assessment instruments. Journal of the National Cancer Institute, 101(21), 1464–1488.
Akizuki, N., Yamawaki, S., Akechi, T., Nakano, T., & Uchitomi, Y. (2005). Development of an impact thermometer for use in combination with the distress thermometer as a brief screening tool for adjustment disorders and/or major depression in cancer patients. Journal of Pain and Symptom Management, 29(1), 91–99.
Bauwens, S., Baillon, C., Distelmans, W., & Theuns, P. (2009). The ‘Distress Barometer’: Validation of method of combining the Distress Thermometer with a rated complaint scale. Psycho-Oncology, 18(5), 534–542.
Quinten, C., Coens, C., Mauer, M., Comte, S., Sprangers, M., Cleeland, C., et al. (2009). Baseline quality of life as a prognostic indicator of survival: A meta-analysis of individual patient data from EORTC clinical trials. The Lancet, 10, 865–871.
Goodwin, P. J., Black, J. T., Bordeleau, L. J., & Ganz, P. A. (2003). Health-related quality-of-life measurement in randomized clinical trials in breast cancer–taking stock. Journal of the National Cancer Institute, 95(4), 263–281.
Calman, K. C. (1984). Quality of life in cancer patients–an hypothesis. Journal of Medical Ethics, 10(3), 124–127.
Horner, M. J., Reis, L. A. G., Krapcho, M., Neyman, N., Aminou, R., & Howlader, N. et al. (Eds.). (2009). SEER cancer statistics review, 1975–2006 National Cancer Institute. Retrieved June 30, 2009, from http://seer.cancer.gov/csr/1975_2006/.
Meyerowitz, B. E., & Oh, S. (2009). Psychosocial response to cancer diagnosis and treatment. In S. M. Miller, D. J. Bowen, R. T. Croyle, & J. H. Rowland (Eds.), Handbook of cancer control and behavioral science: A resource for researchers, practitioners, and policymakers (pp. 361–377). Washington D.C.: American Psychological Association.
Baucom, D., Porter, L., Kirby, J., Gremore, T., & Keefe, F. (2005/2006). Psychosocial issues confronting young women with breast cancer. Breast Disease, 23(1), 103–113.
Allen, S., Shah, A., Nezu, A., Nezu, C., Ciambrone, D., Hogan, J., et al. (2002). A problem solving approach to stress reduction among younger women with breast carcinoma: A randomized controlled trial. Cancer, 94(12), 3089–3100.
Avis, N., Crawford, S., & Manuel, J. (2005). Quality of life among younger women with breast cancer. Journal of Clinical Oncology, 23(15), 3322–3330.
Wong-Kim, E., & Bloom, J. (2005). Depression experienced by young women newly diagnosed with breast cancer. Psycho-Oncology, 14, 564–573.
Parker, P. A., Baile, W. F., De Moor, C., & Cohen, L. (2003). Psychosocial and demographic predictors of quality of life in a large sample of cancer patients. Psycho Oncology, 12(2), 183–193.
Cella, D. F. (1994). Quality of life: Concepts and definition. Journal of Pain and Symptom Management, 9(3), 186–192.
Overcash, J., Extermann, M., Parr, J., Perry, J., & Balducci, L. (2001). Validity and reliability of the FACT-G scale for use in the older person with cancer. American Journal of Clinical Oncology, 24(6), 591–596
Brady, M. J., Cella, D. F., Mo, F., Banomi, A. E., Tulsky, D. S., Lloyd, S. R., Deasy, S., Cobleigh, M., & Shiomoto, G. (1997). Reliability and validity of the Functional Assessment of Cancer Therapy-Breast quality-of-life instrument. Journal of Clinical Oncology, 15(3), 974–986.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Head, B.A., Schapmire, T.J., Keeney, C.E. et al. Use of the Distress Thermometer to discern clinically relevant quality of life differences in women with breast cancer. Qual Life Res 21, 215–223 (2012). https://doi.org/10.1007/s11136-011-9934-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11136-011-9934-3