Abstract
Aims
Silicon (Si) uptake and accumulation improves plant resilience to environmental stresses, but most studies examining this functional role of Si have focussed on grasses (Poaceae) and neglected other important plant groups, such as legumes (Fabaceae). Legumes have evolved a symbiotic relationship with nitrogen-fixing bacteria (rhizobia) housed in root nodules. Our study determined the impacts of silicon (Si) supplementation on Medicago truncatula inoculated with Ensifer meliloti rhizobial strains that differed in their capacity for nitrogen fixation: Sm1021 (‘low-efficiency’) or Sm1022 (‘high-efficiency’).
Methods
We examined how Si and rhizobial efficacy influence nodule and plant functional traits, including their chemical aspects. These combinations were supplied with or without Si in a glasshouse experiment, where we quantified nodule flavonoids and foliar chemistry (free amino acids, soluble protein, elemental C, N and Si).
Results
Si supply increased nodule number per plant, specific nodule flavonoid concentrations, contents of foliar nitrogenous compounds and foliar C, but not foliar Si. We also demonstrated that rhizobial efficacy altered the magnitude of Si effects on certain traits. For example, Si significantly promoted concentrations of foliar N and soluble protein in the plants associated with the ‘low-efficiency’ strain only, and this was not the case with the ‘high-efficiency’ one.
Conclusions
Collectively, our study indicates that Si generates positive effects on M. truncatula, particularly when the association with rhizobia is relatively inefficient, and may play a more prominent role in rhizobial functionality than previously thought.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Silicon (Si) uptake and accumulation in plants (silicification) confers a range of benefits, especially amelioration from biotic and abiotic stresses (Debona et al. 2017). For example, silicification can provide physical protection against herbivores (Massey and Hartley 2009) and pathogens (Wang et al. 2017). Silicification can also allow plants to tolerate nutrient deficiencies (Miao et al. 2010; Hernandez-Apaolaza 2014) and increase yield (Detmann et al. 2012). However, despite the manifold roles of Si in plant biology, most Si studies focus on Si-high accumulator plants, mainly grasses - Poaceae (Katz 2014), and overlook other plant functional groups, such as legumes - Fabaceae (Putra et al. 2020). Some legume species, such as pigeonpea (Cajanus cajan) and soybean (Glycine max) can accumulate a considerable amount of Si, but other species, such as Medicago spp. are known to accumulate a low amount of Si in the foliage (Hodson et al. 2005). However, low silicification may not necessarily reflect low benefits of Si in plant functions as this idea has also been suggested by Katz (2014). For example, Si supply promoted resistance in Arabidopsis thaliana against a fungal pathogen, even though this model plant was a low Si accumulator (Fauteux et al. 2006). More recently, Johnson et al. (2018) showed that Si could increase plant growth and root nodulation in lucerne (M. sativa), including under combined elevated CO2 and temperature, which mimicked projected climate change scenarios.
The family of leguminous plants is comprised of more than 20,000 species (Kenicer 2005), including some which are ecologically and agriculturally significant (Foyer et al. 2016). Legumes have evolved distinct symbiotic associations with nitrogen-fixing bacteria (rhizobia) inside root nodules (Hirsch 1992). Rhizobia convert atmospheric nitrogen (N2) via nitrogenase into available ammonium (Vessey 1994) in exchange for carbon-based photosynthates from the host plant (Checcucci et al. 2017). However, along a symbiotic continuum, legumes are exposed to a plethora of rhizobia whose relationships with their host ranges from mutualistic to parasitic (Sachs et al. 2018). In the latter case, which is believed to be more common in nature than previously understood, the result can be rhizobia with low efficiency that provide fewer benefits (e.g. fixed nitrogen) for their hosts (Gano‐Cohen et al. 2019). While host legumes can regulate or resist unfavourable rhizobia (Westhoek et al. 2021), some rhizobia can persist inside the root nodules by manipulating the host’s ability to regulate the symbiosis (Sachs et al. 2018). This may ultimately reduce plant fitness.
Cooke and Leishman (2016) hypothesised that the beneficial effects of Si on plant performance are greatest when plants are subjected to various environmental stresses, such as drought and salinity, or antagonistic biotic stresses, such as insect herbivory or pathogen attack. It is possible that symbiotic microorganisms may impose similar stresses on their host when their interactions with the plant is disadvantageous. This may be the case, for example, in the legume-rhizobia symbiosis where a rhizobial strain does not provision available N to the host plant efficiently (Terpolilli et al. 2008). For example, the latter study found that a model legume, barrel medic (M. truncatula genotype A17) possessed a lower symbiotic effectiveness when associated with a model rhizobial strain Ensifer meliloti Sm1021, but much higher of that when associated with a closely related strain E. meliloti Sm1022 (Terpolilli et al. 2013). Furthermore, a low symbiotic effectiveness was indicated by the production of small and pale (inactive: lack of leghaemoglobin) nodules as opposed to that of relatively large and pink (active: containing more leghaemoglobin) nodules for a high symbiotic effectiveness (Terpolilli et al. 2008). The former was often accompanied by decreases in nitrogen fixation, resulting in relatively low plant biomass and N content whereas the opposite was true for the latter (Terpolilli et al. 2008).
A growing number of studies suggest that Si may have positive impacts on the legume-rhizobia symbiosis (reviewed by Putra et al. 2020), potentially improving the efficacy of ‘low-efficiency’ rhizobia. Nelwamondo and Dakora (1999) demonstrated that Si supply promoted root nodulation and nitrogen fixation in symbiotic cowpea (Vigna unguiculata) with a Bradyrhizobium strain. Similarly, Johnson et al. (2017) also reported that Si supply benefitted root nodulation in symbiotic M. sativa with a commercial rhizobial strain and shoot biomass. However, the underlying mechanisms underpinning these benefits remain unclear. It was recently reported that Si supply promoted nitrogenase activity in the model legume M. truncatula, which was positively associated with silicification in either the foliage or the root nodule, depending on host genotype (Putra et al. 2021). Furthermore, some studies hypothesised that Si may promote root nodulation by increasing symbiotic chemical signals, such as flavonoids (Nelwamondo and Dakora 1999; Johnson et al. 2017). To date, no studies have investigated this hypothesis (reviewed by Putra et al. 2020).
Flavonoids are a specialised class of plant metabolites, fulfilling a wide range of physiological and ecological functions, such as UV protection, defence against herbivores (Simmonds 2003) and pathogens (phytoalexins) and microbial signalling in the rhizosphere (Dixon and Pasinetti 2010; Hassan and Mathesius 2012). In the legume-rhizobia symbiosis, specific (iso-)flavonoids can act as Nod (‘Nodulation’) gene inducers or repressors for compatible rhizobia (Cooper 2004). In addition, studies with Rhizobium leguminosarum found that more efficient rhizobial strains responded more strongly to Nod gene-inducing flavonoids (Maj et al. 2010). The Nod gene-inducers of Ensifer meliloti include 2’-O-methylliquiritigenin (4,4'-dihydroxy-2'-methoxychalcone), 7,4’-dihydroxyflavone, 7,4’-dihydroxyflavanone, luteolin and chrysoeriol (Maxwell et al. 1989; Hartwig et al. 1990). The effects of Si supply on flavonoid production in the legume-rhizobia symbiosis is largely unknown, but Fawe et al. (1998) reported that Si supply enhanced the synthesis of a flavonol aglycone rhamnetin in cucumber (Cucumis sativus), which was an effective antifungal to powdery mildew.
Silicon-induced root nodulation could also indirectly affect host plant primary metabolites, such as amino acids, and hence ecological interactions aboveground (Johnson et al. 2017). There are well-characterised interactions between Si and other elemental components of plants, such as carbon (C) and nitrogen (N) (Cooke and Leishman 2011; Schaller et al. 2012; Klotzbücher et al. 2018; Quigley et al. 2020), although few studies have been done in legumes. In grasses, there is generally a negative relationship between Si vs C, reflecting a ‘trade-off’ between Si and C as cell structural components in which the incorporation of the former is postulated to be metabolically cheaper than the synthesis of the latter (Raven 1983), but the relationship between Si vs N is less clear cut (Klotzbücher et al. 2018). Understanding potential changes in host chemistry caused by Si supply is crucial to predict relative cost and benefit of accumulating Si and its potential consequences not only on plant antagonists (Massey et al. 2007), but also mutualists, such as symbiotic microbes (Frew et al. 2017; Putra et al. 2020).
A previous study by Putra et al. (2021) indicated the significant differences in plant growth and nodule function might be related to the level of symbiotic efficacy of the rhizobial strain depending on the Medicago plant species (M. sativa vs M. truncatula) and their genotypes. Therefore, to examine this hypothesis, we specifically used the model legume M. truncatula genotype A17 in association with either rhizobial strain Sm1021 (‘low-efficiency’) or Sm1022 (‘high-efficiency’), grown under N-limited conditions. Besides its significance in molecular studies of legumes (Young et al. 2011), M. truncatula is also one of the most important forage crops worldwide (Kenicer 2005).
We measured plant growth, root nodulation and changes in metabolites within nodules (flavonoids) and plant shoots (free amino acids, soluble protein, and elemental C, N and Si) in response to Si supply and rhizobial association. Additionally, we determined if rhizobial efficacy affected the outcomes because the relationship between Si and rhizobia could potentially depend on the efficiency of the strain as discussed in Putra et al. (2021). For example, Si might have more beneficial effects in plants with low-efficiency strains because these are more likely to be under (symbiotic) stress. Therefore, we hypothesised that Si supply:
-
(i)
increases nodule number which may be linked to increases in flavonoid synthesis,
-
(ii)
resulting in higher foliar amino acids, soluble protein and thus higher N content,
-
(iii)
will have the biggest impacts on plants associated with the low-efficiency rhizobial strain due to those plants being more likely to experience N stress and this may corroborate with the ‘stress hypothesis’ suggested by Cooke and Leishman (2016).
Materials and methods
Plant material and rhizobial inoculation
The model legume species, barrel medic (Medicago truncatula Gaertn., Jemalong A17, hereafter ‘JM’) was chosen for this study. Barrel medic is an annual species and symbiotically associated with rhizobia (Terpolilli et al. 2008). Seeds of this genotype were obtained from the Australian Pasture Genebank, Adelaide, Australia. Seeds were surface-sterilised in 70% ethanol (v/v), 40% sodium hypochlorite (v/v) and then washed in a sterile MQ-water following methods from Putra et al. (2021) and Terpolilli et al. (2008). Afterwards, seeds were singly inoculated either with Ensifer meliloti (previously known as Sinorhizobium meliloti) Sm1021 or E. meliloti Sm1022 strains. It was reported that barrel medic associates more effectively with E. meliloti Sm1022 than Sm1021, e.g. better nodulation, higher plant biomass and shoot N content (Terpolilli et al. 2013). For simplicity, hereafter we refer to ‘low-efficiency strain’ (LE) and ‘high-efficiency strain’ (HE) for E. meliloti Sm1021 and Sm1022, respectively.
Both strains were originally provided by the Rhizobium Stock Centre, Murdoch University, Australia and subsequently sub-cultured on yeast mannitol agar (YMA) according to Howieson and Dilworth (2016). Growing conditions and inoculations of rhizobia (OD595nm = 0.1 or 108 CFUs ml−1) were similarly adopted from (Putra et al. 2021) prior to sowing seeds.
Soil media
An equal mixture of soil and sand (1:1 ratio by mass, hereafter ‘soil’) were obtained from Australian Native Landscapes Pty Ltd (NSW, Australia) and γ-sterilised (50 kGy; Steritech, NSW, Australia). Soil had low concentrations of bioavailable Si (11 mg kg−1). See Table S1, for all soil chemical properties.
Experimental design
We conducted a full factorial experiment using one-litre pots to grow 80 individuals of JM plants (see full details in Fig. S1). Each plant was singly inoculated either by LE or HE rhizobial strains. Half of the plants were supplemented with potassium silicate (+ Si) and the other half supplemented with potassium chloride (-Si or control solution), in the form of liquid solutions. Due to the alkaline nature of potassium silicate, the pH of + Si solution was adjusted to ~ 7.00 by adding HCl (Sigma-Aldrich). Therefore, to balance the added Cl− and K+ in + Si solution, KCl (Sigma-Aldrich) was used as a control solution (-Si) (see materials and methods reported in Putra et al. 2021). During seed germination and seedling growth, only tap water (~ 30 ml) was given for 17 days once a day prior to Si supplementation. Afterwards, plants were supplemented either with + Si or -Si (~ 65 ml) once every other day for 12 weeks (a 4-month-old). To avoid position bias, all plants were randomly assigned and rotated carefully on a weekly basis. Plants were grown in a glasshouse with a controlled humidity at 55% and temperature (day/night) 26/18 °C.
Plant harvest
Shoots and roots were harvested by gently taking out the whole plant, cutting it at the soil surface, and washing it with running water. Fresh nodules were excised from the roots. All plant parts including nodules were separately and immediately stored in a 50 ml Falcon tube, snap-frozen in liquid nitrogen and directly freeze-dried at -60 °C for 72 h. Freeze-dried plant and nodule samples were weighed for dry mass, and freeze-dried nodules per plant were counted by eye. Freeze-dried foliar samples were finely ground (ball-milled), stored inside a closed 1.5 ml tube at room temperature, whereas freeze-dried nodules inside a closed 1.5 ml tube were stored at -80 °C. Both samples were used for further chemical analyses.
Foliar elemental chemistry (Si, C and N)
A fine powder of freeze-dried foliar samples was processed for determining Si concentrations with an X-ray fluorescence spectrometer (Epsilon-3x; PANalytical-Almelo, The Netherlands). We followed procedures described in Reidinger et al. (2012) and Hiltpold et al. (2017) using a standard Si calibration from a certified plant reference material (i.e. citrus leaves SRM 1572). An automated dry combustion method (Dumas) using Elementar-Vario EL Cube Analyser (Elementar Analysensysteme GmbH, Hanau, Germany) was used to determine foliar concentrations of C and N from the same samples (burnt at 950 °C). Concentrations in % of dry mass were used to express foliar concentrations of Si, C and N.
Total soluble protein in the foliage
Soluble protein concentrations were measured in an identical fashion to Johnson et al. (2020). In brief, 1 mL of 0.1 M NaOH was added to approximately 23 mg of freeze-dried foliar tissue and homogenised for 30 min at 25 °C. Afterwards, the mixture was centrifuged for 5 min at 12,000 RPM. Dilutions (1:4) of each extract were measured in technical triplicate on a CLARIOstar High Performance Monochromator multimode microplate reader (BMG labtech, Offenburg, Germany) using the Bradford assay modified for a 96-well plate (Bradford 1976; Jones et al. 1989). A standard curve of bovine serum albumin was used to calculate protein concentrations.
Foliar free amino acids
Approximately 35 mg of freeze-dried foliar tissue was extracted in 245 µL 80% MeOH. Samples were then centrifuged at 25 °C for 15 min (15,000 RPM) and 100 µL of supernatant was removed and added to a glass vial insert placed within a 1.5 mL microtube. Samples were combined with 20 µL of 10 µg mL−1 DL-norvaline (internal standard) and placed in a vacuum concentrator for 60 min at 30 °C until all liquid was evaporated. In order to derivatise AAs, 50 µL of N-tert-butyldimethylsilyl-N- methyltrifluoroacetamide standard mixed with acetonitrile in a 1:1 ratio (v:v) was added to the glass inserts and sealed immediately. Samples were vortexed for 30 s and then mixed at 100 °C for 120 min at 300 RPM. Samples were then cooled to room temperature and analysed using an Agilent 7890A series gas chromatography (GC) system and a 5975C mass spectrometer (MS) detector operating in selected ion monitoring (SIM) mode. The samples were analysed with a J&W Scientific HP-5 column (30 m × 25 mm × 0.25 µm) and a temperature program set to 70 °C for 2 min and then increased by 20 °C min−1 until reaching 230 °C. The flow rate was set to 1.2 mL min−1 with H2 as the carrier gas. Injection port and transfer line temperatures were set at 250 °C and 280 °C, respectively. The MS detector was run in electron ionisation (EI) mode with a collision energy of 70 eV and an ion source temperature of 230 °C. Analysis of clean AA standards was performed to determine high quality mass spectra of each compound. Due to their instability in their silylated (derivatised) form, arginine and glutamine were converted to and quantified as ornithine and pyroglutamic acid, respectively (Leimer et al. 1977). The most dominant ion for each amino acid was selected as the quantifying ion, however in some instances the strongest ion was identical to a highly abundant background ion. In those cases (methionine, asparagine, arginine/ornithine, glutamic acid, glutamine/pyroglutamic acid, serine, threonine and phenylalanine), the second most dominant ion was selected for quantification. Retention times of AAs ranged from 5.832 – 20.203 min. We detected 20 foliar free AAs which were grouped into 11 non-essential AAs and nine essential AAs. Non-essential AAs included alanine, glycine, proline, tyrosine, aspartic acid, glutamic acid, arginine, serine, cysteine, asparagine and glutamine whereas essential AAs included isoleucine, leucine, valine, phenylalanine, tryptophan, histidine, lysine, threonine and methionine. Additionally, we also grouped phenylalanine, tryptophan and tyrosine into aromatic AAs as precursors for plant defence. These detected AAs were used for further statistical analyses. All chemical reagents and standards used for this assay were purchased from Sigma-Aldrich, NSW, Australia.
Flavonoids
Flavonoid extraction was performed based on Ng et al. (2015) with some modifications. In summary, pre-weighed and frozen nodule samples (25 mg per sample) were powderised in a Qiagen TissueLyser LT with a pre-cooled holder. Twenty ng of umbelliferone (internal standard; Sigma-Aldrich) was added into each sample tube, followed by 1 mL of 80% (v/v) LC–MS grade methanol (Merck). Samples were vortexed, sonicated at 4 °C for 30 min, followed by 15 min centrifugation at 16,000 × g. The supernatant was concentrated to dryness in a Speedvac™ centrifuge. Samples were resuspended in 200 µL of 80% (v/v) LC–MS grade methanol, vortexed for 10 s and filtered through a 0.2 µm regenerated cellulose micro-spin filter (CIRO, USA) and resuspended in 50 µL 80% (v/v) LC–MS grade methanol.
Samples were subjected to targeted analysis in a Thermo QE Plus UPLC-Orbitrap at the Joint Mass Spectrometry Facility of the Australian National University following the procedure by Ng et al. (2015) with some modifications. Samples and standards were separated in an Agilent Zorbax Eclipse 1.8 mm XDB-C18 2.1 × 50 mm column that was maintained at 40 °C, and separated on a linear gradient from 5–90% of 0.1% aqueous formic acid to 99.9% methanol containing 0.1% formic acid at a flow rate of 200 µL min−1. Data were collected in the positive ion mode and collision energies optimised for each flavonoid. The heated electrospray ionisation (HESI-II) probe was operated with the following settings: Ultra-high purity nitrogen gas was used as the sheath gas (45 L min−1), auxiliary gas (10 L min−1) and sweep gas (2 L min−1); the spray voltage was 3.5 kV and capillary temperature 250 °C; the S-lens RF level was 50 V; the auxiliary gas heater temperature was 300 °C. Tandem mass spectrometry was performed using the parallel reaction monitoring mode with a mass resolution of 17,500 at 1.0 microscan. The Automatic Gain Control target value was set at 1.0 E + 05 counts, maximum accumulation time was 50 ms and the isolation window was set at m/z 4.0. Data were acquired and analysed using the Thermo Scientific Xcalibur 4.0 software.
Flavonoid standards were dissolved in 80% methanol at 1 ppm and analysed in the same analysis run. Flavonoids were sourced as follows: 2’-hydroxyflavone, 3’-hydroxyflavone, 6,7,4’-trihydroxyisoflavone, 7,3’,4’-trihydroxyflavone, 7,4’-dihydroxyflavone, Afromosin, 5,7-dihydroxyflavone (Chrysin), Daidzein-7-O-glucoside (Daidzin), Eriodyctiol, Esculetin, Genistin, Glycitein, Isoliquiritigenin, Luteolin, Madecassoside, Naringenin-7-O-glucoside (Prunin), Formononetin-7-O-glucoside (Ononin), Prunetin, Puerarin, Resveratrol, Rutin, Taxifolin (Indofine Chemical Company, Hillsborough NJ, USA); Genistein, Hesperitin, Kaempferol-7-O-glucoside, Kaempferol-3-O-glucoside (Astragalin); Liquiritigenin, Morin (Extrasynthese, Genay Cedex, France); Coumestrol, Daidzein, Kaempferol (Cayman Chemical Company, Ann Arbor MI, USA); Medicarpin, 2'-O-methylliquiritigenin (Carbosynth, Compton, UK), Apigenin, Apigenin-7-neohesperidoside, Naringenin-7-O-rhamnoglucoside (Naringin), 3',5,7-trihydroxy-4'-methoxyflavone 7-rutinoside, Biochanin A, Formononetin, Naringenin, Quercetin, Quercetin-3-glucoside (Isoquercetin) (Sigma-Aldrich, Castle Hill Australia). From this source, we detected 17 flavonoid compounds out of 43 compounds, which were grouped into eight isoflavones and nine non-isoflavones based on their known chemical structures. We detected eight isoflavones (Daidzein, Formononetin, 7,3',4'-trihydroxyisoflavone, 6,7,4-trihydroxyisoflavone, Glycitein, Afromosin, Daidzin and Ononin), and nine non-isoflavones (Resveratrol, 2'-hydroxyflavone, 3'-hydroxyflavone, 7,4-dihydroxyflavone, Chrysin, Liquiritigenin, 2'-O-methylliquiritigenin, Medicarpin and Naringenin). Changes on concentration of detected compounds were subjected to further statistical analyses.
Statistical analyses
All statistical analyses were computed in R version 4.0.3 (R Core Team 2021). To examine whether Si supply affected multiple plant traits in general, we employed multivariate analysis of variance (MANOVA) using ‘Manova’ function (type = ‘II’) from the ‘car’ package (Fox and Weisberg 2019) with Rhizobia [low-efficiency strain or high-efficiency strain] and Si [-Si or + Si] as main and interacting factors. To accommodate the large number of response variables, relative to available degrees of freedom (the number samples and terms in the model), traits as response variables were divided into categories based on plant phenotypic and chemical groups. The MANOVA revealed that traits differed in their responses to the treatments (see Table S2); therefore, we focus in the main text on interpreting the outcomes of univariate two-way ANOVAs calculated from each multivariate model to assess the individual traits. This was done using the ‘Anova’ function (type = ‘II’) from the ‘car’ package (Fox and Weisberg 2019). Moreover, when p-values were corrected using an ‘fdr’ inference, no apparent quantitative changes between unadjusted and adjusted p-values were observed, suggesting that the inference could be ignored. Normality (‘qqPlot’) and homogeneity of variance (‘residualPlot’) plots were visually assessed and if the assumptions were not met then data were either square-root or loge transformed. When the interactive effect between Rhizobia and Si on dependent variables was significant (p < 0.05), the Tukey’s post hoc multiple comparison test was further conducted using the ‘pairs’ and ‘cld’ function from the ‘multcomp’ package (Hothorn et al. 2021) based on the estimated marginal means in a fitted model using the ‘emmeans’ function from the ‘emmeans’ package (Russell et al. 2021). Additionally, to understand how flavonoids and amino acids covaried and were clustered in response to the combination of group treatments, we analysed them separately with the principal component analysis (PCA) using ‘prcomp’ (‘devtools’ Wickham et al. (2021) and ‘ggbiplot’ in Vu et al. (2011) packages). To understand whether certain dependent variables (e.g. Si-induced nodule number vs flavonoids and/or foliar C) were associated with each other between -Si and + Si plants, Pearson’s correlation tests (‘cor’) from the ‘stats’ package (R Core Team 2021) were conducted. Finally, the ‘ggboxplot’ function from the ‘ggpubr’ package (Kassambara 2018) was used for data visualisations.
Results
Silicon increased nodule number per plant and nodule flavonoids
Total nodule numbers were significantly higher in HE (Sm1022) compared to LE (Sm1021)-inoculated plants (Fig. 1a). We found that there was a significant effect of Si on nodule number per plant (F1,53 = 18.62; p < 0.001; Table 1). Nodule number was significantly increased in + Si relative to -Si plants, but it was even more when the plant associated with LE rather than HE by + 86% and + 59%, respectively (Fig. 1a).
The impacts of Si supply on: a) nodule number per plant and nodule concentrations (ng g−1 dry mass) of b) Liquiritigenin, c) 2’-O-methylliquiritigenin, d) Formononetin, e) Glycitein and f) total flavonoids in the plants associated with low-efficiency (Sm1021) and high-efficiency (Sm1022) rhizobial strains. Dots represent individual measurement per plant (n). Statistically significant factors, namely rhizobial strain (Rhizo), Si, and their interactions are indicated as: ns (non-significant), *p < 0.05, **p < 0.01 and ***p < 0.001. Different capital letters and the horizontal lines indicate significance at p < 0.05
Si supply significantly increased concentrations of nine nodule flavonoids (out of 17 detected compounds), including Liquiritigenin (F1,22 = 4.53; p = 0.046; Table 2), 2’-O-methylliquiritigenin (F1,22 = 7.51; p = 0.013; Table 2), Formononetin (F1,22 = 14.68; p = 0.001; Table 2), Glycitein (F1,22 = 9.56; p = 0.006; Table 2) as well as total detected flavonoids (F1,22 = 12.28; p = 0.002; Table 2). Specifically, Liquiritigenin was augmented in the + Si LE and + Si HE plants by + 33% and + 167%, respectively (Fig. 1b). 2’-O-methylliquiritigenin also increased, particularly in the + Si HE plants (+ 150%) (Fig. 1c). Moreover, Formononetin was augmented in the + Si LE and + Si HE plants by + 200% and + 53%, respectively (Fig. 1d). Glycitein also increased, particularly in the + Si LE plants (+ 157%) (Fig. 1e). Supply of Si increased total detected flavonoids, to a greater extent when the plant associated with LE rather than HE (+ 131% and + 47%, respectively) (Fig. 1f). Interestingly, concentrations of Formononetin, Glycitein and total detected flavonoids were also significantly higher in nodules formed after inoculation with HE compared to LE rhizobia (Fig. 1). The percentage increases are summarised in Table S3. Additionally, how individual flavonoids covaried and were clustered are shown in Fig. S2a.
Silicon altered free amino acids (AAs) and total soluble protein in the foliage
Non-essential AAs were strongly affected by Si supply (F1,23 = 19.106; p < 0.001; Table 3), increasing in the + Si LE (Sm1021) and + Si HE (Sm1022) relative to the -Si LE and -Si HE plants by 90% and 141%, respectively (Fig. 2a). Essential AAs were also significantly affected by Si supply (F1,23 = 8.19; p = 0.009; Table 3), increasing by 23% and 161% (Fig. 2b). Consequently, total AAs were affected by Si (F1,23 = 19.49; p < 0.001; Table 3) where they were augmented by 73% and 144%. In addition, aromatic AAs were significantly affected by Si supply (F1,23 = 5.98; p = 0.024; Table 3) and increased by 29% and 132% (Fig. 2c).
The impacts of Si supply on foliar concentrations of free amino acids (µg g−1 dry mass): a) non-essential, b) essential and c) aromatic, and of d) total soluble protein (mg g−1 dry mass) in the plants associated with low-efficiency (Sm1021) and high-efficiency (Sm1022) rhizobial strains. Dots represent individual measurement per plant (n). Statistically significant factors, namely rhizobial strain (Rhizo), Si, and their interactions are indicated as: ns (non-significant), *p < 0.05, **p < 0.01 and ***p < 0.001. Different capital letters and the horizontal lines indicate significance at p < 0.05
In terms of individual AAs, only five (i.e., tyrosine, cysteine, isoleucine, leucine and methionine) out of 20 AAs were not significantly affected either by Si or its interaction with rhizobia (Table 3). The percentage increases/decreases of how Si significantly altered individual AAs are summarised in Table S4. Additionally, how individual amino acids covaried and were clustered are explained in Fig. S2b.
Si had a significant impact on total soluble protein (F1,23 = 32.02; p < 0.001; Table 1). Its effect on that, however, depended on rhizobia (F1,23 = 9.34; p = 0.006; Table 1). Total soluble protein was increased by Si in the LE plants by 84% (Fig. 2d). In contrast, total soluble protein was not significantly affected by Si in the HE plants (Fig. 2d).
Silicon affected concentrations of elemental C and N
Si significantly affected foliar concentrations of C (F1,11 = 23.54; p = 0.001; Table 1), N (F1,11 = 28.83; p < 0.001; Table 1) and C/N (F1,11 = 29.88; p < 0.001; Table 1) and there was a significant interactive effect between rhizobia and Si on foliar concentrations of N (F1,11 = 30.02; p < 0.001; Table 1) and of C/N (F1,11 = 66.45; p < 0.001; Table 1). Foliar concentrations of C were increased by Si by 5% in both LE (Sm1021) and HE (Sm1022)-inoculated plants (Fig. S3a). Si increased foliar concentrations of N in LE plants by 36%, whereas no significant effect of Si was found in HE plants (Fig. S3b). Consequently, Si decreased foliar C/N under in LE plants by 22%, whereas no significant effect of Si on that was found in HE plants (Fig. S3c). Foliar N concentrations were also significantly higher in plants inoculated with HE compared to LE rhizobia, but only in the -Si treatment (Fig. S3).
The effects of Si on plant biomass
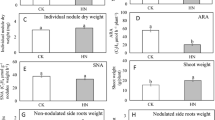
In terms of root biomass, there was a significant interactive effect between rhizobia and Si (F1,53 = 4.72; p = 0.03; Table 1). However, the multi-comparison test based on Tukey’s HSD showed that Si tended to increase root biomass in the plant associated with LE (Sm1021) by 14%, marginally non-significant with a 95% confidence interval (p = 0.076), whereas no significant difference on root biomass was found in the plants associated with HE (Sm1022) (Table 1).
Si supply did not significantly affect shoot biomass (F1,53 = 3.25; p = 0.077; Table 1), total plant biomass (F1,53 = 3.39; p = 0.07; Table 1) or nodule biomass (F1,52 = 3.05; p = 0.087; Table 1). Foliar concentrations of Si were not significantly affected by Si regardless of plant association with rhizobia (F1,29 = 0.83; p = 0.37; Table 1).
Silicon-enhanced nodule number was linked to increased nodule flavonoids and foliar concentrations of elemental C
There was a positive correlation in Si + (HE [Sm1022] and LE [Sm1021]) plants between nodule number and total flavonoid concentrations in Si + plants (r = 0.79; p = 0.002; Fig. 3a). The increase in nodule number was positively correlated with increased foliar % C in Si + plants only (r = 0.93; p = 0.008; Fig. 3b). However, no significant correlation was found between nodule number and foliar % N either in -Si (r = -0.61; p = 0.2) or + Si plants (r = 0.19; p = 0.7). Finally, mean and standard error (SE) values of all other quantified responses are provided in Tables S5 and S6.
Pearson’s correlation tests between nodule number per plant and: a) total flavonoids (ng g−1 dry mass) and b) foliar C (% dry mass). Light grey indicates -Si plants and dark grey indicates + Si plants, associated with low-efficiency (closed circle) or high-efficiency (closed triangle) rhizobial strains. Circles and triangles represent measurements from individual plants (n), regression lines represent a slope of the model and ribbons shows 95% CI. Statistical significance was set at p < 0.05 and indicated by an asterisk (*)
Discussion
This study provides novel evidence that Si supply substantially improves the functioning of the root nodulation in the model legume Medicago truncatula. One potential mechanism includes increasing concentrations of specific flavonoids that could act as Nod-gene regulators. Furthermore, we demonstrate that Si can improve root nodulation of a low-efficiency (LE: Sm1021) rhizobial strain. Besides these positive impacts of Si belowground, Si also strongly affects aboveground foliar primary metabolites, increasing free amino acids, total soluble protein and total N, possibly is facilitated by Si-enhanced root nodulation.
Consequences of Si supply on nodule number and nodule flavonoids
Previous studies demonstrated that Si enhanced nodule number in several legume species, for example cowpea Vigna unguiculata (Nelwamondo and Dakora 1999), lucerne M. sativa (Johnson et al. 2017) and soybean Glycine max (Steiner et al. 2018). A recent study also found that Si enhanced nitrogenase enzyme activity in M. truncatula (Putra et al. 2021). Despite these consistent findings, however, the mechanistic explanation for these impacts has not been identified. Our current findings suggest that increased production in nodule flavonoids resulting from Si supply may underpin increased root nodulation.
Si increased specific nodule flavonoids differently depending on plant association with rhizobial strains. In plants inoculated with the HE (Sm1022) strain, Si supply induced the concentrations of liquiritigenin and 2’-O-methylliquiritigenin by 167% and 150%, respectively. These flavonones are known to act as Nod-gene inducers in E. meliloti (Peck et al. 2006). In particular, 2’-O-methylliquiritigenin is a strong Nod gene inducer also identified from roots of alfalfa (Medicago sativa) (Maxwell et al. 1989), and its synthesis by chalcone O-methyltransferase occurs both during the early infection in the root hairs as well as in the infection zone of mature nodules in M. truncatula (Chen et al. 2015). In plants inoculated with the LE (Sm1021) strain, formononetin was strongly induced by Si (up to 200%). Local induction of this isoflavone was reported to accelerate auxin breakdown, regulating nodule organogenesis in white clover Trifolium repens cv. Haifa (Mathesius 2001). Formononetin is also active as an auxin transport inhibitor (Laffont et al. 2010) and could thus play a role in nodule initiation (Wasson et al. 2006). We also found that Si significantly increased another isoflavone, glycitein (up to 157%) in LE plants. However, this compound is an inactive precursor which has to be activated as a Nod-gene inducer for Bradyrhizobium infecting soybean (Pueppke et al. 1998) and its function as a Nod-gene inducer in E. meliloti is unknown. We did not detect the Nod-gene inducer luteolin in nodules; luteolin has been identified from seed washes of alfalfa but is not typically detected in roots (Maxwell et al. 1989). Overall, higher concentrations of Nod gene-inducing flavonoids in nodules might partly explain the better nodulation phenotype. This association is currently correlative and the mechanism will require further investigation. However, it was previously shown that Nod-gene induction in response to host flavonoids is stronger in more competitive compared to less competitive rhizobia (Maj et al. 2010), and that low concentrations of Nod gene inducing flavonoids can limit nodulation (Kapulnik et al. 1987).
Consequences of Si supply on foliar primary metabolites
Johnson et al. (2017) found that Si supplementation in lucerne (M. sativa) enhanced the production of essential, but not non-essential or total free amino acids (AAs), in the foliage, possibly was mediated via increases in nodule number. In support of their findings, we found that not only essential but also non-essential, aromatic and total foliar free AAs in its closely related species M. truncatula were augmented by Si. Changes in certain AAs may potentially alter host quality for herbivores, through changes in nutritional chemistry (Johnson et al. 2009; Ryalls et al. 2015) and specialised metabolites, such as flavonoids which are synthesised through the phenylpropanoid pathway (Simmonds 2003). We found that phenylalanine, the key precursor of that pathway (Dixon and Pasinetti 2010), was enhanced by Si. Moreover, the other aromatic AAs such as tryptophan and tyrosine are also main precursors for downstream defensive compounds, such as indole and alkaloids (Zeier 2013). Although Si promoted total free AAs to a much greater extent in plants with the HE (Sm1022) strain than those with the LE (Sm1021) strain, we found that the impacts of Si on individual AAs were compound specific depending on rhizobial strains. For example, Si enhanced proline, histidine and valine more highly in plants inoculated with the HE strain and asparagine, serine and arginine in plants with the LE strain. Variation of Si impacts on these individual AAs might influence different metabolic routes and signalling processes (Hildebrandt et al. 2015), and therefore plant functions. For example, the prominent increase in proline could help plants to better cope with environmental stresses (Hayat et al. 2012) as well as physiological activities, such as flowering and seed development (Mattioli et al. 2009). A higher accumulation of asparagine could contribute to increased plant nitrogen and protein contents (Lea et al. 2007).
We found total soluble protein, which is often used as a proxy for nutritional quality (Chapin 1980; Schwab and Broderick 2017; Johnson et al. 2020), was augmented by Si supply. Increased total soluble protein might be, in part, associated with Si-increased total AAs.
Interestingly, we found that total AAs were higher and soluble protein was lower in -Si LE plants, suggesting that LE plants might utilise AAs as precursors for other (defensive) metabolites as opposed to protein synthesis when there was a potential symbiotic stress from the LE strain. However, more crucially, these nitrogenous metabolites were drastically increased when LE plants were supplemented with Si, indicating that Si might alleviate the stress by improving the host plant quality when having symbioses with LE rhizobia.
Consequences of Si supply on foliar elemental chemistry
Si supply slightly increased foliar concentrations of C but greatly increased concentrations of N, resulting in a significantly lower C:N in plants associated with the LE (Sm1021) strain relative to those with the HE (Sm1022) strain. Moreover, we found the opposite trend in the current legume system relative to that in a grass systems; increasing foliar C was positively linked with Si-increased nodule number. This suggests that this positive relationship might be related to allocation of more organic compounds as a feedback of Si-enhanced nodulation in the foliage. Increased foliar N in + Si LE plants might be related to the fact that Si enrichment could promote nitrogenase activity (Putra et al. 2021). As a consequence, Si-enhanced nodule functionality could then contribute to higher foliar concentrations of foliar amino acids and soluble protein, resulting in higher concentrations of foliar N.
Unlike plant C and N, Si addition had no significant impact on foliar concentrations of Si in M. truncatula regardless of plant association with rhizobial strains. This might be explained by the fact that most Medicago-legumes are considered as low-Si accumulators relative to high-Si accumulators, such as grasses (Poaceae) in shoot by % dry mass (Hodson et al. 2005; Putra et al. 2020). However, a recent study found that Si addition in M. truncatula significantly increased Si accumulation in root nodules but not in foliar tissues (Putra et al. 2021), suggesting that silicification might occur in other plant organs besides leaves (Lux et al. 2020) more frequently than expected, especially in non-grasses (Katz 2014). A previous study by Fauteux et al. (2006) concluded that low Si uptake in Arabidopsis thaliana was sufficient to confer plant resistance against a fungal pathogen, suggesting that low silicification may not necessarily preclude Si functions, especially in non-grass taxa.
Negligible impacts of Si supply on nodule biomass and plant biomass
Silicon supply only had minor impacts on nodule biomass and plant biomass, though Si increased root and shoot biomass more in plants with the LE (Sm1021) strain and the HE (Sm1022) strain, respectively. Previous studies have shown that Si supply increased root growth in V. unguiculata and this might be related to increased concentrations of endogenous phytohormone abscisic acid (Dakora and Nelwamondo 2003; Mali and Aery 2009). Moreover, previous studies also found that Si supply increased shoot biomass in a symbiotic lucerne, a Medicago species closely related to barrel medic (Johnson et al. 2017, 2018; Putra et al. 2021).
Conclusions
In summary (Fig. 4), our findings point to the underlying biochemical mechanism whereby Si supply profoundly increases nodule number, which is positively correlated with increased concentrations of specific nodule flavonoids, likely leading to higher content of foliar nitrogenous chemistry in the model legume M. truncatula associated with two distinct rhizobial strains varying in their symbiotic efficacy. Intriguingly, Si may potentially improve host plant symbiosis with the low-efficiency rhizobial strain. In essence, a stimulating effect of Si in the production of flavonoids could enhance the symbiosis through stimulation of Nod-gene activity. Further investigation should explore and consider Si impacts on nodule properties and host plant quality (including defensive metabolites) in a broad range of legume species whose associations with their rhizobial symbionts are relatively poor to better understand whether Si is a key driver for a beneficial legume-rhizobia symbiosis and its consequences on plant–herbivore interactions.
Graphical summary of the key findings regarding the effects of silicon (Si) on a symbiotic legume Medicago truncatula associated with Ensifer meliloti rhizobial strains: Sm1021 (‘low-efficiency’) and Sm1022 (‘high-efficiency’). 1: Si supply promoted root nodulation (nodule number per plant) and nodule flavonoids, resulting in 2: increased foliar nitrogenous metabolites (e.g. free amino acids, soluble protein and total N). However, 3: beneficial effects of Si tended to be much greater when the plant associated with the ‘low-efficiency’ rhizobia (⨁) than with the ‘high-efficiency’ rhizobia (⊕) according to the ‘(symbiotic) stress hypothesis’
Data availability statement
Data can be obtained upon request.
References
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. https://doi.org/10.1016/0003-2697(76)90527-3
Chapin FSI (1980) The mineral nutrition of wild plants. Annu Rev Ecol Syst 11:233–260
Checcucci A, DiCenzo GC, Bazzicalupo M, Mengoni A (2017) Trade, diplomacy, and warfare: The quest for elite rhizobia inoculant strains. Front Microbiol 8:2207. https://doi.org/10.3389/fmicb.2017.02207
Chen D-S, Liu C-W, Roy S et al (2015) Identification of a core set of rhizobial infection genes using data from single cell-types. Front Plant Sci 6:575. https://doi.org/10.3389/fpls.2015.00575
Cooke J, Leishman MR (2016) Consistent alleviation of abiotic stress with silicon addition: A meta-analysis. Funct Ecol 30:1340–1357. https://doi.org/10.1111/1365-2435.12713
Cooke J, Leishman MR (2011) Silicon concentration and leaf longevity: Is silicon a player in the leaf dry mass spectrum? Funct Ecol 25:1181–1188. https://doi.org/10.1111/j.1365-2435.2011.01880.x
Cooper JE (2004) Multiple responses of rhizobia to flavonoids during legume root infection. Adv Bot Res 41:1–62
Dakora FD, Nelwamondo A (2003) Silicon nutrition promotes root growth and tissue mechanical strength. Funct Plant Biol 30:947–953. https://doi.org/10.1071/FP02161
Debona D, Rodrigues FA, Datnoff LE (2017) Silicon’s role in abiotic and biotic plant stresses. Annu Rev Phytopathol 55:85–107. https://doi.org/10.1146/annurev-phyto-080516-035312
Detmann KC, Araujo WL, Martins SC et al (2012) Silicon nutrition increases grain yield, which, in turn, exerts a feed-forward stimulation of photosynthetic rates via enhanced mesophyll conductance and alters primary metabolism in rice. New Phytol 196:752–762. https://doi.org/10.1111/j.1469-8137.2012.04299.x
Dixon RA, Pasinetti GM (2010) Flavonoids and isoflavonoids: From plant biology to agriculture and neuroscience. Plant Physiol 154:453–457. https://doi.org/10.1104/pp.110.161430
Fauteux F, Chain F, Belzile F et al (2006) The protective role of silicon in the Arabidopsis-powdery mildew pathosystem. Proc Natl Acad Sci U S A 103:17554–17559. https://doi.org/10.1073/pnas.0606330103
Fawe A, Abou-Zaid M, Menzies JG, Bélanger RR (1998) Silicon-mediated accumulation of flavonoid phytoalexins in cucumber. Phytopathology 88:396–401. https://doi.org/10.1094/PHYTO.1998.88.5.396
Fox J, Weisberg S (2019) An R companion to applied regression
Foyer CH, Lam HM, Nguyen HT et al (2016) Neglecting legumes has compromised human health and sustainable food production. Nat Plants 2:16112. https://doi.org/10.1038/NPLANTS.2016.112
Frew A, Powell JR, Allsopp PG et al (2017) Arbuscular mycorrhizal fungi promote silicon accumulation in plant roots, reducing the impacts of root herbivory. Plant Soil 419:423–433. https://doi.org/10.1007/s11104-017-3357-z
Gano-Cohen KA, Wendlandt CE, Stokes PJ et al (2019) Interspecific conflict and the evolution of ineffective rhizobia. Ecol Lett 22:914–924. https://doi.org/10.1111/ele.13247
Hartwig UA, Maxwell CA, Joseph CM, Phillips DA (1990) Chrysoeriol and luteolin released from alfalfa seeds induce nod genes in Rhizobium meliloti. Plant Physiol 92:116–122. https://doi.org/10.1104/pp.92.1.116
Hassan S, Mathesius U (2012) The role of flavonoids in root-rhizosphere signalling: opportunities and challenges for improving plant-microbe interactions. J Exp Bot 63:3429–3444. https://doi.org/10.1093/jxb/err430
Hayat S, Hayat Q, Alyemeni MN et al (2012) Role of proline under changing environments: a review. Plant Signal Behav 7:1456–1466. https://doi.org/10.4161/psb.21949
Hernandez-Apaolaza L (2014) Can silicon partially alleviate micronutrient deficiency in plants? a review. Planta 240:447–458. https://doi.org/10.1007/s00425-014-2119-x
Hildebrandt TM, Nunes Nesi A, Araújo WL, Braun HP (2015) Amino acid catabolism in plants. Mol Plant 8:1563–1579. https://doi.org/10.1016/j.molp.2015.09.005
Hiltpold I, Demarta L, Johnson SN et al (2017) Silicon and other essential element composition in roots using X-ray fluorescence spectroscopy: a high throughput approach. Invertebr Ecol Australia's Grasslands Proc Ninth ACGIE. https://doi.org/10.6084/m9.figshare.4806532.v3
Hirsch AM (1992) Developmental biology of legume nodulation. New Phytol 122:211–237. https://doi.org/10.1111/j.1469-8137.1992.tb04227.x
Hodson MJ, White PJ, Mead A, Broadley MR (2005) Phylogenetic variation in the silicon composition of plants. Ann Bot 96:1027–1046. https://doi.org/10.1093/aob/mci255
Hothorn T, Bretz F, Westfall P et al (2021) Package ‘multcomp’. https://CRAN.R-project.org/package=multcomp
Howieson JG, Dilworth MJ (2016) Working with rhizobia. In: Murdoch University. Bytes ’n Colours, Canberra, p 314
Johnson SN, Hartley SE, Ryalls JMW et al (2017) Silicon-induced root nodulation and synthesis of essential amino acids in a legume is associated with higher herbivore abundance. Funct Ecol 31:1903–1909. https://doi.org/10.1111/1365-2435.12893
Johnson SN, Hawes C, Karley AJ (2009) Reappraising the role of plant nutrients as mediators of interactions between root- and foliar-feeding insects. Funct Ecol 23:699–706. https://doi.org/10.1111/j.1365-2435.2009.01550.x
Johnson SN, Ryalls JMW, Gherlenda AN et al (2018) Benefits from below: Silicon supplementation maintains legume productivity under predicted climate change scenarios. Front Plant Sci 9:202. https://doi.org/10.3389/fpls.2018.00202
Johnson SN, Waterman JM, Hall CR (2020) Increased insect herbivore performance under elevated CO2 is associated with lower plant defence signalling and minimal declines in nutritional quality. Sci Rep 10:14553. https://doi.org/10.1038/s41598-020-70823-3
Jones CG, Daniel Hare J, Compton SJ (1989) Measuring plant protein with the Bradford assay - 1. Evaluation and standard method. J Chem Ecol 15:979–992. https://doi.org/10.1007/BF01015193
Kapulnik Y, Joseph CM, Phillips DA (1987) Flavone limitations to root nodulation and symbiotic nitrogen fixation in alfalfa. Plant Physiol 84:1193–1196. https://doi.org/10.1104/pp.84.4.1193
Kassambara A (2018) Package “ggpubr.” https://CRAN.R-project.org/package=ggpubr
Katz O (2014) Beyond grasses: The potential benefits of studying silicon accumulation in non-grass species. Front Plant Sci 5:376. https://doi.org/10.3389/fpls.2014.00376
Kenicer G (2005) Legumes of the World. Edited by G. Lewis, B. Schrire, B. MacKinder & M. Lock. Royal Botanic Gardens, Kew. 2005. xiv + 577 pp., colour photographs & line drawings. ISBN 1 900 34780 6. £55.00 (hardback). Edinburgh J Bot 62:195–196. https://doi.org/10.1017/S0960428606190198
Klotzbücher T, Klotzbücher A, Kaiser K et al (2018) Variable silicon accumulation in plants affects terrestrial carbon cycling by controlling lignin synthesis. Glob Chang Biol 24:e183–e189. https://doi.org/10.1111/gcb.13845
Laffont C, Blanchet S, Lapierre C et al (2010) The compact root architecture1 gene regulates lignification, flavonoid production, and polar auxin transport in Medicago truncatula. Plant Physiol 153:1597–1607. https://doi.org/10.1104/pp.110.156620
Lea PJ, Sodek L, Parry MAJ et al (2007) Asparagine in plants. Ann Appl Biol 150:1–26. https://doi.org/10.1111/j.1744-7348.2006.00104.x
Leimer KR, Rice RH, Gehrke CW (1977) Complete mass spectra of the per-trimethylsilylated amino acids. J Chromatogr A 141:355–375. https://doi.org/10.1016/S0021-9673(00)93539-8
Lux A, Lukačová Z, Vaculík M et al (2020) Silicification of Root Tissues Plants 9:1–20. https://doi.org/10.3390/plants9010111
Maj D, Wielbo J, Marek-Kozaczuk M, Skorupska A (2010) Response to flavonoids as a factor influencing competitiveness and symbiotic activity of Rhizobium leguminosarum. Microbiol Res 165:50–60. https://doi.org/10.1016/j.micres.2008.06.002
Mali M, Aery NC (2009) Effect of silicon on growth, biochemical constituents, and mineral nutrition of cowpea. Commun Soil Sci Plant Anal 40:1041–1052. https://doi.org/10.1080/00103620902753590
Massey FP, Ennos AR, Hartley SE (2007) Grasses and the resource availability hypothesis: The importance of silica-based defences. J Ecol 95:414–424. https://doi.org/10.1111/j.1365-2745.2007.01223.x
Massey FP, Hartley SE (2009) Physical defences wear you down: Progressive and irreversible impacts of silica on insect herbivores. J Anim Ecol 78:281–291. https://doi.org/10.1111/j.1365-2656.2008.01472.x
Mathesius U (2001) Flavonoids induced in cells undergoing nodule organogenesis in white clover are regulators of auxin breakdown by peroxidase. J Exp Bot 52:419–426. https://doi.org/10.1093/jxb/52.suppl_1.419
Mattioli R, Costantino P, Trovato M (2009) Proline accumulation in plants: Not only stress. Plant Signal Behav 4:1016–1018. https://doi.org/10.4161/psb.4.11.9797
Maxwell CA, Hartwig UA, Joseph CM, Phillips DA (1989) A chalcone and two related flavonoids released from alfalfa roots induce nod genes of Rhizobium meliloti. Plant Physiol 91:842–847. https://doi.org/10.1104/pp.91.3.842
Miao B-H, Han X-G, Zhang W-H (2010) The ameliorative effect of silicon on soybean seedlings grown in potassium-deficient medium. Ann Bot 105:967–973. https://doi.org/10.1093/aob/mcq063
Nelwamondo A, Dakora FD (1999) Silicon promotes nodule formation and nodule function in symbiotic cowpea (Vigna unguiculata). New Phytol 142:463–467. https://doi.org/10.1046/j.1469-8137.1999.00409.x
Ng JLP, Hassan S, Truong TT et al (2015) Flavonoids and auxin transport inhibitors rescue symbiotic nodulation in the Medicago truncatula cytokinin perception mutant cre1. Plant Cell 27:2210–2226. https://doi.org/10.1105/tpc.15.00231
Peck MC, Fisher RF, Long SR (2006) Diverse flavonoids stimulate NodD1 binding to nod gene promoters in Sinorhizobium meliloti. J Bacteriol 188:5417–5427. https://doi.org/10.1128/JB.00376-06
Pueppke SG, Bolaños-Vásquez MC, Werner D et al (1998) Release of flavonoids by the soybean cultivars McCall and Peking and their perception as signals by the nitrogen-fixing symbiont Sinorhizobium fredii 1. Plant Physiol 117:599–606. https://doi.org/10.1104/pp.117.2.599
Putra R, Powell JR, Hartley SE, Johnson SN (2020) Is it time to include legumes in plant silicon research? Funct Ecol. https://doi.org/10.1111/1365-2435.13565
Putra R, Vandegeer RK, Karan S et al (2021) Silicon enrichment alters functional traits in legumes depending on plant genotype and symbiosis with nitrogen-fixing bacteria. Funct Ecol. https://doi.org/10.1111/1365-2435.13912
Quigley KM, Griffith DM, Donati GL, Anderson TM (2020) Soil nutrients and precipitation are major drivers of global patterns of grass leaf silicification. Ecology. https://doi.org/10.1002/ecy.3006
R Core Team (2021) R: A language and environment for statistical computing
Raven JA (1983) The transport and function of silicon in plants. Biol Rev 58:179–207. https://doi.org/10.1111/j.1469-185x.1983.tb00385.x
Reidinger S, Ramsey MH, Hartley SE (2012) Rapid and accurate analyses of silicon and phosphorus in plants using a portable X-ray fluorescence spectrometer. New Phytol 195:699–706. https://doi.org/10.1111/j.1469-8137.2012.04179.x
Russell A, Lenth V, Buerkner P et al (2021) Package ‘emmeans.’ https://CRAN.R-project.org/package=emmeans
Ryalls JMW, Moore BD, Riegler M et al (2015) Amino acid-mediated impacts of elevated carbon dioxide and simulated root herbivory on aphids are neutralized by increased air temperatures. J Exp Bot 66:613–623. https://doi.org/10.1093/jxb/eru439
Sachs JL, Quides KW, Wendlandt CE (2018) Legumes versus rhizobia: A model for ongoing conflict in symbiosis. New Phytol 219:1199–1206. https://doi.org/10.1111/nph.15222
Schaller J, Brackhage C, Dudel EG (2012) Silicon availability changes structural carbon ratio and phenol content of grasses. Environ Exp Bot 77:283–287. https://doi.org/10.1016/j.envexpbot.2011.12.009
Schwab CG, Broderick GA (2017) A 100-year review: protein and amino acid nutrition in dairy cows. J Dairy Sci 100:10094–10112. https://doi.org/10.3168/jds.2017-13320
Simmonds MSJ (2003) Flavonoid-insect interactions: Recent advances in our knowledge. Phytochemistry 64:21–30. https://doi.org/10.1016/S0031-9422(03)00293-0
Steiner F, Zuffo AM, Bush A, da Santos DMS (2018) Silicate fertilization potentiates the nodule formation and symbiotic nitrogen fixation in soybean. Pesqui Agropecuária Trop 48:212–221. https://doi.org/10.1590/1983-40632018v4851472
Terpolilli J, Hill Y, Tian R et al (2013) Genome sequence of Ensifer meliloti strain WSM1022; a highly effective microsymbiont of the model legume Medicago truncatula A17. Stand Genomic Sci 9:315–324. https://doi.org/10.4056/sigs.4608286
Terpolilli JJ, O’Hara GW, Tiwari RP et al (2008) The model legume Medicago truncatula A17 is poorly matched for N2-fixation with the sequenced microsymbiont Sinorhizobium meliloti 1021. New Phytol 179:62–66. https://doi.org/10.1111/j.1469-8137.2008.02464.x
Vessey JK (1994) Measurement of nitrogenase activity in legume root nodules: In defense of the acetylene reduction assay. Plant Soil 158:151–162. https://doi.org/10.1007/BF00009490
Vu QV, Marwick B, Hester J, Held M (2011) Package ‘ggbiplot’. https://CRAN.R-project.org/package=ggbiplot
Wang M, Gao L, Dong S et al (2017) Role of silicon on plant–pathogen interactions. Front Plant Sci 8:701. https://doi.org/10.3389/fpls.2017.00701
Wasson AP, Pellerone FI, Mathesius U (2006) Silencing the flavonoid pathway in Medicago truncatula inhibits root nodule formation and prevents auxin transport regulation by rhizobia. Plant Cell 18:1617–1629. https://doi.org/10.1105/tpc.105.038232
Westhoek A, Clark LJ, Culbert M et al (2021) Conditional sanctioning in a legume-Rhizobium mutualism. Proc Natl Acad Sci U S A 118:e2025760118. https://doi.org/10.1073/pnas.2025760118
Wickham H, Hester J, Chang W (2021) Package ‘devtools’. https://CRAN.R-project.org/package=devtools
Young N, Debellé F, Oldroyd G (2011) The Medicago genome provides insight into the evolution of rhizobial symbioses. Nature 480:520–524. https://doi.org/10.1038/nature10625.The
Zeier J (2013) New insights into the regulation of plant immunity by amino acid metabolic pathways. Plant, Cell Environ 36:2085–2103. https://doi.org/10.1111/pce.12122
Acknowledgements
We thank Tamara Weyman, Burhan Amiji, Premchand Maisnam, Fikadu Biru and Tarikul Islam for their generous assistance during the plant harvest. We also thank Dr. Jason Ng (the Australian National University) and Dr. Pushpinder Matta for their technical help with the mass spectrometry analysis for flavonoids and foliar C/N analysis, respectively, and the Joint Mass Spectrometry Facility at the Australian National University. R.P. is the holder of a scholarship as part of an Australian Research Council Future Fellowship (FT170100342) awarded to S.N.J., the Australian Steel Mill Services (ASMS) and the University of York in the United Kingdom.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Contributions
R.P., J.R.P., S.E.H., and S.N.J. constructed the idea and design of the experiment; R.P. conducted the experiment; U.M. assisted with flavonoid analysis; J.M.W. and D.W. ran the amino acids and soluble protein analyses; R.P. analysed the data with some statistical advice from J.R.P. and led the writing of the manuscript. J.M.W., U.M., D.W., J.R.P., S.E.H. and S.N.J. critically contributed to manuscript drafts. Lastly, all authors gave final approval for publication.
Corresponding author
Ethics declarations
Conflict of interest
None of the authors has conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Responsible Editor: Martin J. Hodson.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Putra, R., Waterman, J.M., Mathesius, U. et al. Benefits of silicon-enhanced root nodulation in a model legume are contingent upon rhizobial efficacy. Plant Soil 477, 201–217 (2022). https://doi.org/10.1007/s11104-022-05358-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-022-05358-9