Abstract
Aims
Cover crop species selection for soybean (Glycine max) production under no-tillage (NT) management may affect soil organic C sequestration by altering the quantity and quality of C inputs, thereby affecting cropping system sustainability. If so, the underlying mechanisms for such regulation are still unclear.
Methods
We assessed changes in soil C and N fractions at 0-0.1 m depth and soil C stock at 0-0.6 m depth during the last three years of dry-season cover cropping in a soybean production system managed with NT for 9 years on a Rhodic Hapludox in Sao Paulo, Brazil. Dry-season management treatments were repeated yearly in a split-plot scheme. Main plots during the fall-winter were (1) ruzigrass (Urochloa ruziziensis), (2) grain sorghum (Sorghum bicolor), and (3) the intercropping of ruzigrass and sorghum. Subplots during spring prior to planting soybean were (a) pearl millet (Pennisetum glaucum), (b) sunn hemp (Crotalaria juncea), and (c) forage sorghum (Sorghum bicolor).
Results
Soil C and N fractions were affected according to crop residue characteristics of the rotations. Higher soil C stocks in 2012 and 2015 (7 % an average) were observed at 0.2–0.4 m depth by ruzigrass compared to sorghum. High crop residue input with ruzigrass in the fall-winter sequestered 0.61 Mg C ha− 1 yr− 1 at 0-0.1 m soil depth compared with lower C sequestration using grain sorghum (0.29 Mg C ha− 1 yr− 1).
Conclusions
The quantity and quality of crop residues impact its retention on soil surface controlling the dynamics of soil C and N fractions and can be considered relevant for soil C sequestration. These aspects could contribute to the mitigation of atmospheric CO2 in crop production systems.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Soils hold the largest C pool in terrestrial ecosystems, and therefore changes in soil organic carbon (SOC) can impact atmospheric carbon dioxide (CO2) concentration (Stockmann et al. 2013). Historically, converting forested areas into agricultural land reduced SOC content (Deng et al. 2016). However, recent conservation management of agricultural soils has promoted SOC sequestration and mitigation of CO2 emissions to the atmosphere (Tang et al. 2019). Importantly, soil management controls whether soil acts as a sink or source of C to the atmosphere (Farina et al. 2017; Minasny et al. 2017).
Soil disturbance following a monoculture crop with seasonal fallow periods, and lack of crop rotations with insufficient soil cover violate the key features of conservation agriculture (Derpsch et al. 2014), often resulting in SOC depletion, and especially in the tropics (Lal 2002). Alternatively, the practices of no-till (NT) management to maintain soil cover and using cover crops along with diverse crop rotations are strategies to increase SOC sequestration in cropping systems (Rodríguez Martín et al. 2019). Soil organic C sequestration in agricultural systems is regulated by the type of cropping systems (Luo et al. 2010), which are important to optimize for the sustainability of agricultural production (Rigon and Calonego 2020). Additionally, changes in SOC depend on soil type and climate (Hoyle et al. 2013), soil texture, and mineralogy (Bayer et al. 2006b), and type and quantity of crop residues (Xu et al. 2017). However, how SOC responds to different crop residues under a variety of crop management systems remains unclear (Zhao et al. 2018; Kong et al. 2019).
Most SOC sequestration studies have focused on differing soil management (e.g. conventional tillage and no-till). Fewer studies have been conducted on differences between crop rotations with specific attention to crop residue quality. Crop residue quality may play a significant role in controlling SOC accumulation in NT cropping systems, particularly under tropical conditions (Raphael et al. 2016; Rigon et al. 2020). According to these authors cumulative crop residue production and N content influence the accumulation of SOC and its fractions. Faster SOC turnover in tropical soils due to high temperature and precipitation leads to rapid decomposition of crop residues and potentially limited SOC accumulation (Bolliger et al. 2006; Powlson et al. 2016).
Grasses have high biomass production potential and their residues may provide recalcitrant compounds, such as lignin, that slow decomposition leading to greater and longer lasting surface residue cover of the soil surface (Pimentel et al. 2015; Akhtar et al. 2018). Lignin is chemically connected to cellulose and hemicellulose in the cellulosic fiber walls, providing strength and rigidity to plant structures (Thevenot et al. 2010). In addition, lignin has been considered an important contributor to stable SOC pools owing to innate biochemical recalcitrance (Stevenson 1994; Kögel-Knabner 2002). With legumes as cover crop, crop residues have enhanced N concentration and this may alter the soil microbial community to increase SOC sequestration in tropical soils (Bayer et al. 2016; Justes 2017). The beneficial effect of cover crops in the whole cropping system will depend on how they are managed (Büchi et al. 2018). Therefore, a new challenge is to develop and improve crop rotation schemes with high organic C inputs that maximize the benefits of NT as a strategy to promote SOC sequestration and soil quality (Vieira et al. 2009).
Limited knowledge exists on the role of crop residue type and quality as drivers for SOC sequestration, specifically in tropical cropping systems. The objective of this study was to assess C inputs and characteristics of crop rotations affecting soil C and N fractions and SOC sequestration under NT. We hypothesized that (i) greater residue quality from legumes and lower residue quality from grasses in crop rotations will impact residue retention and soil C and N fractions influencing SOC sequestration; and (ii) greater C and N inputs of different crop rotations will increase SOC sequestration and C retention efficiency. This information is needed to understand how to design cropping systems for enhancing long-term SOC dynamics and develop reasonable management strategies for SOC sequestration that work for farmers and the environment.
Materials and methods
Study site
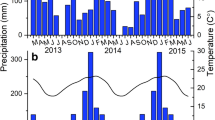
The study was established in 2006 on a Rhodic Hapludox (Soil Survey Staff 2014) in Botucatu, Sao Paulo, Brazil (22°49’S, 48°25’ W). The climate is mesothermal with a well-defined dry season from May to September and mean annual rainfall of 1450 mm. Monthly temperature and rainfall between 2012 and 2015 and the average from 2006 to 2011 are shown in Fig. 1.
Experimental design and crop rotation management
The experiment was laid out as a split-plot arrangement in four randomized blocks. Main plots were crop species in the fall-winter, planted around the first half of April. Split plots were type of spring crops, sown around the second half of September. Soybean (Glycine max (L.) Merrill) was sown across the entire experiment as the cash crop in summer in Nov/Dec. The factorial arrangement of three main plots and three sub plots resulted in nine soybean cropping system treatments (Table 1), maintained consistently from 2006 to 2015.
Main plots were grain sorghum (Sorghum bicolor), ruzigrass (Urochloa ruziziensis), and intercropping of sorghum and ruzigrass planted in 5 m x 30 m plots. Sorghum was sown at 11 kg ha− 1 with row spacing of 0.34 m. Ruzigrass was sown at 22 kg ha− 1 with row spacing of 0.17 m. For the intercropped treatment, the same spacing and seed rates were used as for sole crops. Ruzigrass seeds were placed in the fertilizer box of the seeder and distributed in the same row as sorghum. In Aug/Sep sorghum plots were harvested, and afterwards crops were chemically desiccated with glyphosate. In both cases, crop residues were left on the soil surface.
Sub plots in the spring were pearl millet (Pennisetum glaucum), sunn hemp (Crotalaria juncea), and forage sorghum (Sorghum bicolor) planted in 5 m x 10 m areas. Pearl millet, sunn hemp, and forage sorghum were sown at 25, 30, and 15 kg ha− 1, respectively, with row spacing of 0.17 m in second half of September. In spring at pre-flowering stage (Nov/Dec), cover crops were chemically desiccated with glyphosate, and residues were left on the soil surface. Soybean was sown soon thereafter. No fertilizers were used in winter and spring seasons.
Soybean was sown in the three years of this phase of the project on 5 Dec 2012, 13 Nov 2013, and 2 Dec 2014 at 400,000 seeds ha− 1 with row spacing of 0.45 m. Since the beginning of the experiment, soybean was the only fertilized crop, receiving 50 kg K2O ha− 1 and 50 kg P2O5 ha− 1 each year as potassium chloride and triple superphosphate, respectively. Crop management was the same in each year from 2006 to 2015.
Crop residue
Two crop residue samples (0.25 m2 each) were randomly collected from each plot at the end of each of the three growing periods during each of the three years; totaling 9 crop seasons as follows: fall-winter 2012, 2013 and 2014, spring crops 2012, 2013 and 2014, and soybean 2012/2013, 2013/2014, and 2014/2015. Crop residue samples were dried at 55°C for 48 h and weighed. Samples were ground, homogenized, and analyzed for C and N contents with an elemental analyzer (LECO-TruSpec® CHNS). Sub-samples from all plots in fall-winter 2014 and spring crops 2014 were analyzed for cellulose, hemicellulose, and lignin (Silva and Queiroz 2002).
Soil
Soil was sampled with a push probe after winter crops in 2012 and after soybean harvest in 2015 by compositing three soil cores (5 cm diameter) from each experimental unit at soil depths of 0-0.1, 0.1–0.2, 0.2–0.4, and 0.4–0.6 m. Subsamples collected in 2012 were analyzed for chemical (Raij et al. 2001) and physical and granulometric properties (Danielson and Sutherland 1986) (Table 2). For SOC analysis, soil samples were air-dried, ball-milled, and analyzed with an elemental analyzer (LECO-TruSpec® CHNS). Subsamples collected in 2015 at 0-0.1-m depth were analyzed for soil C and N fractions (Franzluebbers and Stuedemann 2008). Briefly, soil microbial biomass C was determined with chloroform fumigation-incubation without subtraction of a control. The flush of CO2 following rewetting of dried soil (3 d) and cumulative C and N mineralization during 24 d of incubation were determined with aerobic incubation of soil at 50 % water-filled pore space and 25°C. Duplicate 50-g soil samples in 60 mL glass jars were wetted and placed in a 1-L canning jar along with a vial containing 10 mL of 1 mol L− 1 NaOH to trap CO2 and a vial of water to maintain humidity. Alkali traps were replaced at 3 and 10 d of incubation and CO2–C determined by titration with 1 mol L− 1 HCl with vigorous stirring in the presence of BaCl2 (which precipitated to form BaCO3) to a phenolphthalein endpoint. At 10 d, one of the subsamples was removed and fumigated with CHCl3 under vacuum for 1 d, vapors removed, placed into a separate canning jar along with vials of alkali and water, and incubated at 25°C for 10 d. Potential C mineralization was calculated from the cumulative evolution of CO2 during 24 d of incubation. Basal soil respiration was assumed from the linear rate of C mineralization during the 10- to 24-d period. Mineralizable N was determined from the difference in inorganic N concentration between 0 and 24 d of incubation. Inorganic N (NH4–N + NO2–N + NO3–N) was determined from the filtered extract of a 10-g subsample of dried (55°C for 3 d) and sieved (≤ 2 mm) soil that was shaken with 20 mL of 2 mol L− 1 KCl for 30 min using salicylate-nitroprusside and hydrazine autoanalyzer.
Another sampling for undisturbed soil samples was conducted in 2012 and 2015. Trenches approximately 0.4 m wide, 0.8 m long and 0.6 m deep were opened in each plot and samples were taken at the center in each soil depths (0-0.1, 0.1–0.2, 0.2–0.4, and 0.4–0.6 m) using a volumetric single ring (5.0 cm high x 4.8 cm wide) (Blake 1965) to calculate C stock of the soil profile (Veldkamp 1994) according to Eq. 1:
where SOC is soil organic carbon (g kg− 1), Bd is soil bulk density (Mg m− 3), and sl is soil layer (cm). Calculations of C sequestration, C sequestration rate, and C retained from crop residues (CRCR) were based on de Moraes Sá et al. (2015) and Yadav et al. (2019) according to Equations 2, 3, and 4, respectively.
where: years is (2015–2012) and cumulative C crop residue input is the crop residue yield (Mg ha− 1) during 9 seasons starting from the winter crop in 2012 to soybean in 2014/2015.
Data analysis
Homogeneity and normality of data were tested prior to analysis of variance (p < 0.05). Differences among means were compared by the t-test (LSD, p < 0.05).
Results
Crop residue
Across nine growing periods from the fall-winter season of 2012 to the summer soybean season of 2014/2015, cumulative crop residue inputs were 34.5 Mg ha− 1, while C and N contents were, respectively, 15.2 Mg C ha− 1 and 527 kg N ha− 1 when averaged across cropping treatments (Table 3). The fall-winter and spring crop treatments impacted (p < 0.05) cumulative crop residue mass and C and N inputs, but there was no interaction between fall-winter and spring crop treatments.
Crop residue and C inputs from ruzigrass were, on average, 9–10 % greater (p < 0.05) than from other fall-winter crops (grain sorghum intercropped with ruzigrass or grain sorghum only). Difference in crop residue N input from ruzigrass was even greater (30–50 %) compared to grain sorghum and sorghum intercropped with ruzigrass.
Among spring crops, sunn hemp had 30–40 % greater (p < 0.05) N input than from pearl millet and forage sorghum. In addition, crop residue mass and C input by sunn hemp were 9 % greater (p < 0.05) than from forage sorghum, but not differing from those of pearl millet.
Crop residues from ruzigrass during fall-winter and from sunn hemp during spring had unique fiber compositions (Table 3). Besides greater N inputs, both crop residues had greater lignin content (p < 0.05) and lower hemicellulose and cellulose contents (p < 0.05) relative to other species in their respective cropping seasons.
Soil
Soil C and N fractions at 0-0.1 m in 2015
Soil C and N fractions were affected only by fall-winter crop treatments and not by spring crop treatments nor the interaction between fall-winter and spring crop treatments (Table 4). Soil-test biological activity (STBA), net N mineralization (NMIN), particulate organic N (PON), and the fraction of SOC that was mineralizable during 24 days of incubation (CMIN) were ~ 20 % greater (p < 0.05) when cropped with ruzigrass alone than when intercropped with grain sorghum.
Soil C stock and C sequestration rate
Across crop rotation treatments, C stock increased ~ 6 % at 0-0.1-m soil depth between 2012 and 2015 [(p < 0.05), 22.7 and 24.1 Mg C ha− 1, respectively]. However, in deeper soil layers, C stocks were more similar during this time period (Table 5).
Among crop rotations, C stock was only different between ruzigrass alone and intercropping at 0.2-0.4-m soil depth in both years analyzed [(p < 0.05), Table 5]. This distinction of ruzigrass alone compared to intercropping also occurred in the total C stock of the soil profile in 2015 (0-0.6 m soil depth). However, it is important to highlight that the increment from 2012 to 2015 tended to be greater (p = 0.24, data not shown) with sorghum alone (2.1 Mg C ha− 1) than from ruzigrass alone and intercropped (1.4 and 0.5 Mg C ha− 1).
Fall-winter crop treatments also impacted C sequestration rate, although this was limited to the 0-0.1-m soil depth (Table 6). Soil C sequestration rate ranged from 0.29 to 0.61 t C ha− 1 yr− 1 in sorghum only and ruzigrass only treatments, respectively. At deeper depths, soil C sequestration was not statistically different from zero.
As a fraction of total C input from crop residue production, ruzigrass in fall-winter retained 0.112 kg kg− 1, and was greater (p < 0.05) than from grain sorghum, which was only 0.060 kg kg− 1. As observed for C stocks and C sequestration, there was no influence of spring species on C retention from crop residues.
Discussion
Crop residue inputs
Average annual crop residue input was 11.5 Mg ha-1 (5 Mg C ha-1). Crop residue C input was considered greater than the minimum of 4 Mg C ha-1 to maintain soil C stocks in soybean cropping systems with NT in tropical regions (Bayer et al. 2006a; Vieira et al. 2009). In weathered soils with dry winter, soil cover is recognized as an important part of soil conservation (Jantalia et al. 2007). Hence, crop residue inputs are needed to compensate for SOC depletion from fast residue decomposition with high temperature and abundant moisture in the rainy season (Lal 2002). In general, grass species are preferred to produce abundant biomass that persists on the soil surface (Teixeira et al. 2014; Rigon et al. 2018), which ruzigrass can produce in fall-winter. Although intercropping with ruzigrass has become more widely used to increase biomass production (Crusciol et al. 2015), the low crop residue production in the intercropped treatment may have been due to the absence of N fertilization in our experiment, which would intensify the competition with similar physiological crop types (Vidal and Merotto Jr 2010). In addition, under limited water supply in the winter, the competition between crops can be intensified (Zegada-Lizarazu et al. 2006). Alternatively, the intermediate production values from the intercropped treatment compared to both sole crops suggest a simple dilution of the ruzigrass impact with grain sorghum.
Greater accumulated N inputs from ruzigrass can be explained by remarkably vigorous root growth with fast turnover and rhizodeposition (Cavalcante et al. 2019) that can capture cycled N from decomposition of organic matter. A recent study found that ruzigrass can increase the nitrification potential of soil N and the abundance of N-fixing microorganisms (Rocha et al. 2020). In addition, N from biological nitrogen fixation (BNF) may be as high as 20 % in the tissues of ruzigrass (Reis et al. 2001), since it was discovered that Urochloa spp are able to obtain N contributions from BNF (Boddey and Victoria 1986). Further studies are suggested to assess the contribution from BNF of forages in cropping systems. Urochloa species are also recognized as efficient nutrient cyclers in integrated cropping systems (Garcia et al. 2008). Sunn hemp stands out among legumes for its high potential BNF (Chikowo et al. 2004). Input of N represented an average increase of 70 kg N ha-1 year-1, a large portion of which may have been from BNF. This characteristic should be considered as an effective substitute for application of synthetic fertilizers (Kaye and Quemada 2017), providing greater sustainability for tropical cropping systems. These results are even more relevant in our experiment due to the absence of N fertilization since its establishment in 2006.
In general, the quality of crop residues is related to its composition, such as N content, C:N ratio, and lignin content (Chen et al. 2014). Usually, grass species have greater lignin levels than legumes (del Río et al. 2007). High lignin content from sunn hemp (Table 2) was similar to that observed by Palm et al. (2001). These authors classified sunn hemp as a residue with high quality based on N content, lignin, and polyphenolic content. Talbot and Treseder (2012) suggested that lignin decomposes during all stages of crop residue mineralization, not only after other structural compounds decompose. Considering the relatively low C:N ratio of ruzigrass (C:N = 24.7) and sunn hemp (C:N = 24.9), we can infer high lability, despite the high lignin contents. Cotrufo et al. (2013) proposed that labile residue compounds could help stabilize SOC in soils with high C stabilization capacity, such as in this soil with high Fe and Al oxides. In addition, the effect of litter quality on stable SOC is most easily observed at moderate saturation deficits (Castellano et al. 2015). With these considerations in mind, crop rotations that diversify residue input characteristics, such as with high lignin and C:N ratio, could better maintain soil cover during the dry winter period when managed with NT. In contrast, during the rainy season of spring it may be more important to have crop species with low C:N ratio and rapid N mineralization to provide nutrients for the developing crops. Our results suggest that residue quality during crop rotations with NT might need to be matched to environmental conditions varying among seasons in tropical environments.
Soil
Soil C and N fractions
Soil microorganisms produce enzymes that decompose crop residues and soil organic matter according to substrate quality to meet their demands for food (Wang et al. 2015). For this reason, availability of high-quality crop residues with easily decomposable C stimulates soil mineralization rates (Shahbaz et al. 2017). In addition to large crop residue C inputs, intrinsic characteristics of ruzigrass residues can help explain greater soil-test biological activity and cumulative C mineralization. According to Cotrufo et al. (2015, 2013) and Lehmann and Kleber (2015), easily decomposable residues contribute significantly to SOC formation. Ruzigrass cropping in fall-winter also resulted in greater particulate organic N and net N mineralization (Table 3). Although sunn hemp is recognized for its high-quality residues and inputs from BNF, it did not significantly impact particulate organic N [(p = 0.11) Table 3]. It is important to highlight that particulate organic N values were the same when cropped in the spring with sunn hemp as in the fall-winter with ruzigrass (0.17 mg kg-1). It may be possible that the quantity of sunn hemp residues (Table 2) were not sufficient to impact particulate organic N (Table 4). This explanation may be the same as to why particulate organic C was not affected by cropping treatments. Soil organic C and total N were also not affected by cropping treatments in earlier years of this experiment (Raphael et al. 2016). More active fractions of C and N may have been more sensitive to change than this passive fraction of particulate organic C and N, which are generally composed of surface residues and roots (Franzluebbers et al. 1999) that provide a source of energy and nutrients for the community of decomposing microorganisms (Baldock and Skjemstad 2000). Our results suggest that both quantity and quality of crop residues are important for affecting soil C and N changes under NT.
Soil C stock and C sequestration rate
Crop residue retention on the soil surface across the years of this experiment (2006–2015), helped to explain the greatest soil C stocks nearest the soil surface (i.e. 0-0.1-m depth) in 2015 (24.2 ± 1.01 Mg C ha− 1). Although crop residue inputs appeared to have been sufficient to maintain and increase soil C stocks at the soil surface, there was no differentiation of effects by fall-winter and spring cropping treatments.
The capacity of soil to sequester C over time may be reduced (West and Six 2007), due possibly to C saturation (Six et al. 2002). Soil organic C addition by roots has been considered a strategy to increase SOC stock in deeper soil profile (Lorenz and Lal 2005; Thorup-Kristensen et al. 2020), and our results provided some evidence of this with greater SOC stock at 0.2–0.4 m depth with ruzigrass in both years. Urochloa has abundant root mass that can easily reach 1 m soil depth (Quattrocchi 2006). In addition, rhizodeposition of organic C from exudates (Cavalcante et al. 2019) can contribute to SOC stock change in the profile (Mancin et al. 2013). Hence, we expected an influence of crop rotations on C sequestration in deeper soil, but this did not occur. There was a trend for C sequestration in soil profile assessed (0-0.6 m depth, p = 0.24) by fall winter crops, as well as in the spring by grasses, which may have provided root biomass influence. Limited knowledge exists on the role of cropping systems as drivers for SOC accumulation in deeper soil, specifically in weathered soils. Recent studies with crop rotations under NT observed influence on soil C and N fractions only at the soil surface (Neto et al. 2020; Rigon et al. 2020) Raphael et al. (2016) found that soil under ruzigrass had the greatest degree of soil organic matter humification in previous years of this experiment in the topsoil. The inputs of aboveground biomass regulated soil C sequestration at the soil surface. Soil C changes deeper in the profile will likely be influenced by deep-penetrating roots, but this possibility will need more time to assess in this experiment in the tropics.
Carbon saturation in clayey soils may be less prevalent than in sandy soils (Jagadamma and Lal 2010; Feng et al. 2013). Clay fractions of soil often have greater capacity to sequester SOC, and this effect may have played a role in why SOC stock was greater with depth, corresponding to greater clay content in this soil depth (Table 2).
Cropping system intensification is expected to have greater rates of soil C sequestration (Robertson et al. 2018), due to greater production of crop residue C that can either keep constant or increase C retention rate (Li et al. 2016; Kumar and Nath 2019). If C input from the three years of this phase of the experiment were extrapolated across the nine years total, then average C sequestration rate of 0.48 Mg C ha− 1 yr− 1 would represent average mitigation of 1.7 Mg CO2 ha− 1 yr− 1. Similar soil C sequestration rates have been observed in other tropical soils (Bayer et al. 2000, 2006a; de Moraes Sá et al. 2001; Moraes Sá et al. 2015). Annual soil C sequestration rate of 0.5 Mg C ha− 1 yr− 1 was estimated for conversion of conventional tilled cropland to NT in tropical environments (Cerri et al. 2004). Annual SOC sequestration rate by cover crops has been estimated at 0.3 Mg C ha− 1 yr− 1 across a variety of soil types, climate, and management systems (Poeplau and Don 2015). Besides C inputs from crop residue, soil C stabilization is essential to guarantee an increase in soil C sequestration rate.
In highly weathered soils, Fe and Al oxides provide additional protection of SOC to decomposition through organo-mineral interactions (physical-chemical protection) (Six et al. 2002). Cropping intensification can affect nutrient cycling (Hazra et al. 2019). Abundant soil cover and minimal soil disturbance form the principles of conservation agriculture, making it possible to restore SOC (Lal 2015). Cropping intensification changes the quantity and quality of crop residues, thereby impacting SOC turnover (Liu et al. 2014; Novelli et al. 2017), through soil aggregation and dynamics of soil C and N fractions (Rigon et al. 2020).
It is widely accepted that litter quality can affect soil organic matter stabilization (Castellano et al. 2015). However, the role of crop residue quality in the formation of soil organic matter is not completely understood, and labile C substrates appear to impact soil C sequestration (Cotrufo et al. 2013), thereby promoting more stable and humidified soil organic matter fractions (Samahadthaiy et al. 2010; Cyle et al. 2016; Kumar and Nath 2019). A wide range of soil C sequestration rates may be a result of differing quality of crop residues that affect the amount of C sequestered in agricultural soils (Christopher and Lal 2007). Crop residue quality may have been why C sequestration rate was low with sorghum (0.29 Mg C ha− 1 yr− 1) and high with ruzigrass (0.61 Mg C ha− 1 yr− 1) and sunn hemp (0.54 Mg C ha− 1 yr− 1). Cellulose is considered more labile and is usually decomposed faster than lignin (Chen et al. 2018). In the long-term, soil C stability is highly dependent on the chemical nature of the substrate, such as lignin contributing more to the pool of humic substances (Torres et al. 2014). Thus, some aspects of our original hypotheses were confirmed based on quantity of crop residue inputs and quality of their intrinsic characteristics. It was clear that high C input to soil and use of NT were important management characteristics to affect change in SOC, as observed earlier (Rigon et al. 2020).
Carbon retention from crop residues
The fraction of C input from crop residues that is retained as SOC represents an important index to indicate efficiency of C conservation in soil (Bhattacharyya et al. 2012; Yadav et al. 2016). Variation of this fraction from 6 to 11 % in our study was similar to that of Cotrufo et al. (2013). Higher quality residues sequester a greater fraction of C input, especially in soils with strong mineral stabilization as in tropical soils (Cotrufo et al. 2015). Similar values of efficiency for C retention have been observed in other studies in temperate (Han et al. 2018) and tropical environments (Hok et al. 2015), and specific to Brazilian soils (de Moraes Sá et al. 2015). According to many of these reports, crop-specific characteristics have a direct impact on soil C inputs and their retention as SOC.
Differences in crop residue C retention can be explained by the contrasting decomposition characteristics of residues during the dry winter season. Ruzigrass had both greatest production and high lignin content, confirming its efficiency in C retention. Substrate utilization efficiency by microorganisms may also have an impact on the decomposition process of crop residues (Cotrufo et al. 2015). These results indicate that biochemical recalcitrance may be an important mechanism of soil organic matter stabilization. This would reinforce the hypothesis that intrinsic characteristics of crop residues could be a strategy to enhance soil C sequestration under NT. Thevenot et al. (2010), in a review, suggest that the accumulation and stabilization of organic matter can be derived from lignin in plant residues. Stabilization could occur in the clay fraction, but the mechanisms remain unclear, suggesting the necessity to pursue this hypothesis further. Therefore, additional studies should be undertaken to explore how crop residue quantity and quality can be manipulated to change the soil C dynamic.
Conclusions
In general, soil C and N fractions were impacted according to crop residue quality and quantity in the crop rotation. Ruzigrass in the dry season of fall-winter increased SOC stock in the soil profile, and combined with greater crop residue supply and N and lignin contents, led to greater efficiency of SOC conversion and greatest soil organic C sequestration rate at 0-0.1-m soil depth. It is important to note that under an intensive cropping rotation i.e. three crops annually, changing SOC stock deeper in the profile will require further investigation over time.
Maintaining protective soil residue cover during the dry winter and providing biologically based C and N inputs to the main crop through decomposition in the wet spring may be a sustainable cropping strategy under NT in tropical conditions. Hence, appropriate crop species choice based on residue characteristics in the rotation play an important role to achieve soil C retention and sequestration. Our results suggest that the quantity and quality of crop residue deposited at the soil surface in intensive cropping systems under NT can be important factors for SOC retention and sequestration, thereby mitigating against rising atmospheric CO2 concentration.
Data availability
Data generated or analysed during this study are included in this published article.
References
Akhtar K, Wang W, Ren G et al (2018) Changes in soil enzymes, soil properties, and maize crop productivity under wheat straw mulching in Guanzhong, China. Soil Tillage Res 182:94–102. https://doi.org/10.1016/j.still.2018.05.007
Baldock JA, Skjemstad JO (2000) Role of the soil matrix and minerals in protecting natural organic materials against biological attack. Org Geochem 31:697–710. https://doi.org/10.1016/S0146-6380(00)00049-8
Bayer C, Mielniczuk J, Amado TJC et al (2000) Organic matter storage in a sandy clay loam acrisol affected by tillage and cropping systems in southern Brazil. Soil Tillage Res 54:101–109. https://doi.org/10.1016/S0167-1987(00)00090-8
Bayer C, Lovato T, Dieckow J et al (2006a) A method for estimating coefficients of soil organic matter dynamics based on long-term experiments. Soil Tillage Res 91:217–226. https://doi.org/10.1016/j.still.2005.12.006
Bayer C, Martin-Neto L, Mielniczuk J et al (2006b) Carbon sequestration in two Brazilian Cerrado soils under no-till. Soil Tillage Res 86:237–245. https://doi.org/10.1016/j.still.2005.02.023
Bayer C, Gomes J, Zanatta JA et al (2016) Mitigating greenhouse gas emissions from a subtropical Ultisol by using long-term no-tillage in combination with legume cover crops. Soil Tillage Res 161:86–94. https://doi.org/10.1016/j.still.2016.03.011
Bhattacharyya P, Roy KS, Neogi S et al (2012) Effects of rice straw and nitrogen fertilization on greenhouse gas emissions and carbon storage in tropical flooded soil planted with rice. Soil Tillage Res 124:119–130. https://doi.org/10.1016/j.still.2012.05.015
Blake GR (1965) Bulk density. Methods of soil analysis. Wiley, Hoboken, pp 374–390
Boddey RM, Victoria RL (1986) Estimation of biological nitrogen fixation associated with Brachiaria and Paspalum grasses using15N labelled organic matter and fertilizer. Plant Soil 90:265–292. https://doi.org/10.1007/BF02277403
Bolliger A, Magid J, Amado JCT et al (2006) Taking stock of the Brazilian “Zero-Till revolution”: a review of landmark research and farmers’ practice. Adv Agron 91:47–110. https://doi.org/10.1016/S0065-2113(06)91002-5
Büchi L, Wendling M, Amossé C et al (2018) Importance of cover crops in alleviating negative effects of reduced soil tillage and promoting soil fertility in a winter wheat cropping system. Agric Ecosyst Environ 256:92–104. https://doi.org/10.1016/j.agee.2018.01.005
Castellano MJ, Mueller KE, Olk DC et al (2015) Integrating plant litter quality, soil organic matter stabilization, and the carbon saturation concept. Glob Chang Biol 21:3200–3209. https://doi.org/10.1111/gcb.12982
Cavalcante DM, de Castro MF, Chaves MTL et al (2019) Effects of rehabilitation strategies on soil aggregation, C and N distribution and carbon management index in coffee cultivation in mined soil. Ecol Indic 107:105668. https://doi.org/10.1016/j.ecolind.2019.105668
Cerri CEP, Paustian K, Bernoux M et al (2004) Modeling changes in soil organic matter in Amazon forest to pasture conversion with the Century model. Glob Chang Biol 10:815–832. https://doi.org/10.1111/j.1529-8817.2003.00759.x
Chen B, Liu E, Tian Q et al (2014) Soil nitrogen dynamics and crop residues. A review. Agron Sustain Dev 34:429–442. https://doi.org/10.1007/s13593-014-0207-8
Chen X, Hu Y, Feng S et al (2018) Lignin and cellulose dynamics with straw incorporation in two contrasting cropping soils /704/47/4113 /704/158/47 /38/77 article. Sci Rep 8:1–10. https://doi.org/10.1038/s41598-018-20134-5
Chikowo R, Mapfumo P, Nyamugafata P, Giller KE (2004) Mineral N dynamics, leaching and nitrous oxide losses under maize following two-year improved fallows on a sandy loam soil in Zimbabwe. Plant Soil 259:315–330. https://doi.org/10.1023/B:PLSO.0000020977.28048.fd
Christopher SF, Lal R (2007) Nitrogen management affects carbon sequestration in North American cropland soils. CRC Crit Rev Plant Sci 26:45–64. https://doi.org/10.1080/07352680601174830
Cotrufo MF, Wallenstein MD, Boot CM et al (2013) The Microbial Efficiency-Matrix Stabilization (MEMS) framework integrates plant litter decomposition with soil organic matter stabilization: Do labile plant inputs form stable soil organic matter? Glob Chang Biol 19:988–995. https://doi.org/10.1111/gcb.12113
Cotrufo MF, Soong JL, Horton AJ et al (2015) Formation of soil organic matter via biochemical and physical pathways of litter mass loss. Nat Geosci 8:776–779. https://doi.org/10.1038/ngeo2520
Crusciol CAC, Nascente AS, Borghi E et al (2015) Improving soil fertility and crop yield in a tropical region with palisadegrass cover crops. Agron J 107:2271–2280. https://doi.org/10.2134/agronj14.0603
Cyle KT, Hill N, Young K et al (2016) Substrate quality influences organic matter accumulation in the soil silt and clay fraction. Soil Biol Biochem 103:138–148. https://doi.org/10.1016/j.soilbio.2016.08.014
Danielson RE, Sutherland PL (1986) Soil sampling and methods of analysis. Methods of soil analysis. Physical and mineralogical method, 2nd edn. Madison, Soil Science Society of America, Wisconsin, pp 443–460
de Moraes Sá JC, Cerri CC, Dick W et al (2001) Chronosequence in a Brazilian Oxisol. Water 65:1486–1499
de Moraes Sá JC, Séguy L, Tivet F et al (2015) Carbon depletion by plowing and its restoration by no-till cropping systems in oxisols of subtropical and tropical agro-ecoregions in Brazil. L Degrad Dev 26:531–543. https://doi.org/10.1002/ldr.2218
del Río JC, Gutiérrez A, Rodríguez IM, Ibarra D, Martínez AT (2007) Composition of non-woody plant lignins and cinnamic acids by Py-GC/MS, Py/TMAH and FT-IR. J Anal Appl Pyrol 79:39–46
Deng L, Zhu G-y, Tang Z-s, Shangguan Z-p (2016) Global patterns of the effects of land-use changes on soil carbon stocks. Glob Ecol Conserv 5:127–138. https://doi.org/10.1016/j.gecco.2015.12.004
Derpsch R, Franzluebbers AJ, Duiker SW et al (2014) Why do we need to standardize no-tillage research? Soil Tillage Res 137:16–22. https://doi.org/10.1016/j.still.2013.10.002
Farina R, Marchetti A, Francaviglia R et al (2017) Modeling regional soil C stocks and CO2emissions under Mediterranean cropping systems and soil types. Agric Ecosyst Environ. https://doi.org/10.1016/j.agee.2016.08.015
Feng W, Plante AF, Six J (2013) Improving estimates of maximal organic carbon stabilization by fine soil particles. Biogeochemistry 112:81–93. https://doi.org/10.1007/s10533-011-9679-7
Franzluebbers AJ, Stuedemann JA (2008) Early response of soil organic fractions to tillage and integrated crop-livestock production. Soil Sci Soc Am J 72:613–625. https://doi.org/10.2136/sssaj2007.0121
Franzluebbers AJ, Langdale GW, Schomberg HH (1999) Soil carbon, nitrogen, and aggregation in response to type and frequency of tillage. Soil Sci Soc Am J 63:349–355
Garcia RA, Crusciol CAC, Calonego JC, Rosolem CA (2008) Potassium cycling in a corn-brachiaria cropping system. Eur J Agron 28:579–585
Han X, Xu C, Dungait JAJ et al (2018) Straw incorporation increases crop yield and soil organic carbon sequestration but varies under different natural conditions and farming practices in China: A system analysis. Biogeosciences 15:1933–1946. https://doi.org/10.5194/bg-15-1933-2018
Hazra KK, Nath CP, Singh U et al (2019) Diversification of maize-wheat cropping system with legumes and integrated nutrient management increases soil aggregation and carbon sequestration. Geoderma 353:308–319. https://doi.org/10.1016/j.geoderma.2019.06.039
Hok L, de Moraes Sá JC, Boulakia S et al (2015) Short-term conservation agriculture and biomass-C input impacts on soil C dynamics in a savanna ecosystem in Cambodia. Agric Ecosyst Environ 214:54–67. https://doi.org/10.1016/j.agee.2015.08.013
Hoyle FC, D’Antuono M, Overheu T, Murphy DV (2013) Capacity for increasing soil organic carbon stocks in dryland agricultural systems. Soil Res 51:657–667. https://doi.org/10.1071/SR12373
Jagadamma S, Lal R (2010) Distribution of organic carbon in physical fractions of soils as affected by agricultural management. Biol Fertil Soils 46:543–554. https://doi.org/10.1007/s00374-010-0459-7
Jantalia CP, Resck DVS, Alves BJR et al (2007) Tillage effect on C stocks of a clayey Oxisol under a soybean-based crop rotation in the Brazilian Cerrado region. Soil Tillage Res 95:97–109. https://doi.org/10.1016/j.still.2006.11.005
Justes E (2017) Cover Crops for Sustainable Farming. Springer, Netherlands. https://doi.org/10.1007/978-94-024-0986-4
Kaye JP, Quemada M (2017) Using cover crops to mitigate and adapt to climate change. A review. Agron Sustain Dev 37. https://doi.org/10.1007/s13593-016-0410-x
Kögel-Knabner I (2002) The macromolecular organic composition of plant and microbial residues as inputs to soil organic matter: Fourteen years on. Soil Biol Biochem 105:A3–A8. https://doi.org/10.1016/j.soilbio.2016.08.011
Kong D, Liu N, Wang W et al (2019) Soil respiration from fields under three crop rotation treatments and three straw retention treatments. PLoS One 14:1–21. https://doi.org/10.1371/journal.pone.0219253
Kumar N, Nath CP (2019) Impact of zero-till residue management and crop diversification with legumes on soil aggregation and carbon sequestration. Soil Tillage Res 189:158–167. https://doi.org/10.1016/j.still.2019.02.001
Lal R (2002) The potential of soils of the tropics to sequester carbon and mitigate the greenhouse effect. Adv Agron 76:1–30. https://doi.org/10.1016/s0065-2113(02)76002-1
Lal R (2015) Sequestering carbon and increasing productivity by conservation agriculture. J Soil Water Conserv 70:55A-62A. https://doi.org/10.2489/jswc.70.3.55A
Lehmann J, Kleber M (2015) The contentious nature of soil organic matter. Nature 528:60–68. https://doi.org/10.1038/nature16069
Li S, Li Y, Li X et al (2016) Effect of straw management on carbon sequestration and grain production in a maize-wheat cropping system in Anthrosol of the Guanzhong Plain. Soil Tillage Res 157:43–51. https://doi.org/10.1016/j.still.2015.11.002
Liu C, Lu M, Cui J et al (2014) Effects of straw carbon input on carbon dynamics in agricultural soils: A meta-analysis. Glob Chang Biol 20:1366–1381. https://doi.org/10.1111/gcb.12517
Lorenz K, Lal R (2005) The depth distribution of soil organic carbon in relation to land use and management and the potential of carbon sequestration in subsoil horizons. Adv Agron 88:35–66. https://doi.org/10.1016/S0065-2113(05)88002-2
Luo Z, Wang E, Sun OJ (2010) Can no-tillage stimulate carbon sequestration in agricultural soils? A meta-analysis of paired experiments. Agric Ecosyst Environ 139:224–231. https://doi.org/10.1016/j.agee.2010.08.006
Mancin CR, De Souza LCF, Vitorino ACT et al (2013) Matéria orgânica em um Latossolo Vermelho distroférrico sob sistema plantio direto consolidado. Semin Agrar 34:635–648. https://doi.org/10.5433/1679-0359.2013v34n2p635
Minasny B, Malone BP, McBratney AB et al (2017) Soil carbon 4 per mille. Geoderma 292:59–86. https://doi.org/10.1016/j.geoderma.2017.01.002
Neto JF, Franzluebbers AJ, Crusciol CAC et al (2020) Soil carbon and nitrogen fractions and physical attributes affected by soil acidity amendments under no-till on Oxisol in Brazil. Geoderma Reg 24:e00347. https://doi.org/10.1016/j.geodrs.2020.e00347
Novelli LE, Caviglia OP, Piñeiro G (2017) Increased cropping intensity improves crop residue inputs to the soil and aggregate-associated soil organic carbon stocks. Soil Tillage Res 165:128–136. https://doi.org/10.1016/j.still.2016.08.008
Palm CA, Gachengo CN, Delve RJ et al (2001) Organic inputs for soil fertility management in tropical agroecosystems: Application of an organic resource database. Agric Ecosyst Environ 83:27–42. https://doi.org/10.1016/S0167-8809(00)00267-X
Pimentel LG, Weiler DA, Pedroso GM, Bayer C (2015) Soil N2O emissions following cover-crop residues application under two soil moisture conditions. J Plant Nutr Soil Sci 178:631–640. https://doi.org/10.1002/jpln.201400392
Poeplau C, Don A (2015) Carbon sequestration in agricultural soils via cultivation of cover crops - A meta-analysis. Agric Ecosyst Environ 200:33–41. https://doi.org/10.1016/j.agee.2014.10.024
Powlson DS, Stirling CM, Thierfelder C et al (2016) Does conservation agriculture deliver climate change mitigation through soil carbon sequestration in tropical agro-ecosystems? Agric Ecosyst Environ 220:164–174. https://doi.org/10.1016/j.agee.2016.01.005
Quattrocchi U (2006) World dictionary of grasses: Common names, scientific names, epynyms, synonyms, and etymology. CRC Press, Boca Raton
Raij BV, Andrade JC, Cantarella H, Quaggio JA (2001) Chemical analysis for evaluation of the fertility of tropical soils. Instituto Agronômico, Campinas
Raphael JPA, Calonego JC, Milori DMBP, Rosolem CA (2016) Soil organic matter in crop rotations under no-till. Soil Tillage Res 155:45–53. https://doi.org/10.1016/j.still.2015.07.020
Reis VM, Dos Reis Fábio BJ, Quesada DM et al (2001) Biological nitrogen fixation associated with tropical pasture grasses. Aust J Plant Physiol 28:837–844. https://doi.org/10.1071/pp01079
Rigon JPG, Calonego JC (2020) Soil carbon fluxes and balances of crop rotations under long-term no-till. Carbon Balance Manag 15:19. https://doi.org/10.1186/s13021-020-00154-3
Rigon JPG, Calonego JC, Rosolem CA et al (2018) Cover crop rotations in no-till system: Short-term CO2 emissions and soybean yield. Sci Agric 75:18–26. https://doi.org/10.1590/1678-992x-2016-0286
Rigon JPG, Franzluebbers AJ, Calonego JC (2020) Soil aggregation and potential carbon and nitrogen mineralization with cover crops under tropical no-till. J Soil Water Conserv 75:601–609. https://doi.org/10.2489/jswc.2020.00188
Robertson AD, Zhang Y, Sherrod LA, Rosenzweig ST, Ma L, Ahuja L, Schipanski ME (2018) Climate Change Impacts on Yields and Soil Carbon in Row Crop Dryland Agriculture. J Environ Qual 47:684–694
Rocha KF, Kuramae EE, Borges BMF et al (2020) Microbial N-cycling gene abundance is affected by cover crop specie and development stage in an integrated cropping system. Arch Microbiol 202:2005–2012. https://doi.org/10.1007/s00203-020-01910-2
Rodríguez Martín JAy, Álvaro-Fuentes J, Gabriel JL et al (2019) Soil organic carbon stock on the Majorca Island: Temporal change in agricultural soil over the last 10 years. Catena 181:104087. https://doi.org/10.1016/j.catena.2019.104087
Samahadthaiy P, Vityakon P, Saenjan P (2010) Effects of different quality plant residues on soil carbon accumulation and aggregate formation in atropical sandy soil in northeast Thailand as revealed by a 10-year field experiment. Land Degrad Dev 21:463–473. https://doi.org/10.1002/ldr.982
Shahbaz M, Kuzyakov Y, Sanaullah M et al (2017) Microbial decomposition of soil organic matter is mediated by quality and quantity of crop residues: mechanisms and thresholds. Biol Fertil Soils 53:287–301. https://doi.org/10.1007/s00374-016-1174-9
Silva DJ, Queiroz AC (2002) Food analysis: chemical and biological methods, 3rd edn. UFV, Viçosa
Six J, Conant RT, Paul EA, Paustian K (2002) Stabilization of organic matter by soil minerals: Implications for C-saturation of soils. Plant Soil 241:155–176
Soil Survey Staff (2014) Keys to soil taxonomy, 12th edn. USDA-Natural Resources Conservation Service, Washington, DC
Stevenson FJ (1994) Humus chemistry: genesis, composition, reactions, 2nd edn. Wiley, New York
Stockmann U, Adams MA, Crawford JW et al (2013) The knowns, known unknowns and unknowns of sequestration of soil organic carbon. Agric Ecosyst Environ 164:80–99. https://doi.org/10.1016/j.agee.2012.10.001
Talbot JM, Treseder KK (2012) Interactions among lignin, cellulose, and nitrogen drive litter chemistry-decay relationships. Ecology 93:345–354. https://doi.org/10.1890/11-0843.1
Tang H, Liu Y, Li X et al (2019) Carbon sequestration of cropland and paddy soils in China: potential, driving factors, and mechanisms. Greenh Gases Sci Technol 885:872–885. https://doi.org/10.1002/ghg.1901
Teixeira RA, Soares TG, Fernandes AR, Braz AM de S (2014) Grasses and legumes as cover crop in no-tillage system in northeastern Pará Brazil. Acta Amaz 44:411–418. https://doi.org/10.1590/1809-4392201305364
Thevenot M, Dignac MF, Rumpel C (2010) Fate of lignins in soils: A review. Soil Biol Biochem 42:1200–1211. https://doi.org/10.1016/j.soilbio.2010.03.017
Thorup-Kristensen K, Halberg N, Nicolaisen M et al (2020) Digging deeper for agricultural resources, the value of deep rooting. Trends Plant Sci 25:406–417. https://doi.org/10.1016/j.tplants.2019.12.007
Torres IF, Bastida F, Hernández T et al (2014) The role of lignin and cellulose in the carbon-cycling of degraded soils under semiarid climate and their relation to microbial biomass. Soil Biol Biochem 75:152–160. https://doi.org/10.1016/j.soilbio.2014.04.007
Veldkamp E (1994) Organic carbon turnover in three tropical soils under pasture after deforestation. Soil Sci Soc Am J 58:175–180
Vidal R, Merotto A Jr (2010) Inicialismo. In: VIDAL, R. (Ed). Interação negativa entre plantas: inicialismo, alelopatia e competição. Porto Alegre: Evangraf, 2010. p. 33–49
Vieira FCB, Bayer C, Zanatta JA et al (2009) Building up organic matter in a subtropical paleudult under legume cover-crop-based rotations. Soil Sci Soc Am J 73:1699–1706. https://doi.org/10.2136/sssaj2008.0241
Wang H, Boutton TW, Xu W et al (2015) Quality of fresh organic matter affects priming of soil organic matter and substrate utilization patterns of microbes. Sci Rep 5:1–13. https://doi.org/10.1038/srep10102
West TO, Six J (2007) Considering the influence of sequestration duration and carbon saturation on estimates of soil carbon capacity. Clim Change 80:25–41. https://doi.org/10.1007/s10584-006-9173-8
Xu Y, Chen Z, Fontaine S et al (2017) Dominant effects of organic carbon chemistry on decomposition dynamics of crop residues in a Mollisol. Soil Biol Biochem 115:221–232. https://doi.org/10.1016/j.soilbio.2017.08.029
Yadav GS, Babu S, Das A et al (2016) Effect of organic and inorganic fertilizers and rice straw on carbon sequestration and soil fertility under a rice–rice cropping pattern. Carbon Manag 10:619–629. https://doi.org/10.1080/17583004.2018.1545518
Yadav GS, Babu S, Das A et al (2019) Soil carbon dynamics and productivity of rice–rice system under conservation tillage in submerged and unsubmerged ecologies of Eastern Indian Himalaya. Carbon Manag 10:51–62. https://doi.org/10.1080/17583004.2018.1545518
Zegada-Lizarazu W, Izumi Y, Iijima M (2006) Water competition of intercropped pearl millet with cowpea under drought and soil compaction stresses. Plant Prod Sci 9:123–132. https://doi.org/10.1626/pps.9.123
Zhao Y, Zhang J, Müller C, Cai Z (2018) Temporal variations of crop residue effects on soil N transformation depend on soil properties as well as residue qualities. Biol Fertil Soils 54:659–669. https://doi.org/10.1007/s00374-018-1291-8
Funding
This work was supported by the São Paulo Research Foundation - Fapesp [grant numbers 13/23359-4, 15/23389-6, and 17/23029-5].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest/Competing interests
We have no conflicts of interest to disclose.
Additional information
Responsible Editor: Zucong Cai.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rigon, J.P.G., Calonego, J.C., Capuani, S. et al. Soil organic C affected by dry‐season management of no‐till soybean crop rotations in the tropics. Plant Soil 462, 577–590 (2021). https://doi.org/10.1007/s11104-021-04878-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-021-04878-0