Abstract
Background and aims
Land cover change (LCC) from natural forest (NF) to plantations (PF) has occurred worldwide over the past several decades. However, the different LCC effects on soil aggregate C and N turnover in various climatic zones remain uncertain.
Methods
Soil samples were taken from both NF and PF at five sites along an approximately 4200 km north-south transect in eastern China. We measured soil aggregate C and N concentrations, and δ13C and δ15N.
Results
The soil aggregate distribution is similar between NF and PF, except that the mass proportion of microaggregate is lower in NF. The impacts of LCC on soil C and N concentrations, and δ13C and δ15N vary among five climate zones. The changes in soil aggregate C and N concentrations and δ15N induced by LCC show nonlinear relationships with climatic factors (i.e., MAT and MAP), and there is a linear relationship between soil aggregate Δδ13C (calculated by subtracting PF from NF) and MAT and MAP. The soil aggregate C and N concentrations, and δ13C and δ15N show clear trends along the climatic transect. In addition, the impacts of LCC are more obvious in topsoil than in subsoil.
Conclusion
Our findings highlight that the impacts of LCC on soil C and N concentrations are related to climatic factors. Specifically, that the increased decomposition of soil C in PF than NF can be compensated by higher C inputs with increasing MAT and MAP.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Forest soils store a large portion of terrestrial carbon (C) (Dixon et al. 1994) but simultaneously may release large sums of C when they undergo land use or environmental changes (Guillaume et al. 2015; Houghton and Nassikas 2017). To fulfill societal needs for valuable timber and other economic forest products, natural forests (NFs) have been widely converted to intensively managed plantations (PFs) during the past several decades (Lin et al. 2018; Llorente et al. 2010; Straaten et al. 2015; Yang et al. 2019). There is substantial evidence showing significant differences in the quantity and quality of C input through litterfall or root exudation between NF and PF (Hertel et al. 2009; Lewis et al. 2016) and on management practices including tillage, fertilization, thinning, drainage, and harvesting (Jandl et al. 2007; Li et al. 2014). It is very important to know how land cover change (LCC) from NF to PF alters soil C and N cycles (Chen et al. 2016; Straaten et al. 2015). Thus, the influences of LCC on soil C and N have been widely investigated during the past two decades (Chen et al. 2004; Li et al. 2005; Lin et al. 2018; Straaten et al. 2015). Currently, however, there is still uncertainty regarding positive, negative or negligible effects on soil C and N at a large scale under various climatic conditions (Lewis et al. 2016).
Soil aggregates are the basic units of soil structure, and have an important role in controlling soil C and N stocks and stabilization (Tisdall and Oades 1982; Six and Paustian 2014; Zhu et al. 2017). Moreover, soil organic matter fractions can serve as early indicators for the change in soil C and N stocks induced by LCC (He et al. 2008; Leifeld and Kögel-Knabner 2005). A hierarchy model was proposed by Tisdall and Oades (1982), who stated that mineral particles are bound together to form microaggregates by various cements, including persistent organic materials, crystalline oxides and highly disordered aluminosilicates. These microaggregates, in turn, build macroaggregates due to transient and temporal agents such as polysaccharides, roots, and fungal hyphae (Spohn and Giani 2011). Due to the various aggregate binding agents, the stability of soil aggregates and the allocated C and N in different-size aggregates have different sensitivities to land use and cover change (Tisdall and Oades 1982; Chen et al. 2017). Land cover change from natural forests to intensively managed plantations has been well known to strongly affect soil aggregation and C and N pools (Ashagrie et al. 2005; Chen et al. 2017; Goma-Tchimbakala 2009; He et al. 2008). However, fewer studies have focused on the efficiency and mechanism of aggregate C and N turnover associated with the input of plant residues and output of microbial mineralization.
Isotopic abundances of 13C and 15N in soils can change as a result of fractionation processes occurring during soil C and N cycles (Bol et al. 1999; Gerschlauer et al. 2019; Guillaume et al. 2015), thereby providing new information on soil C and N turnover controlled by biotic and abiotic factors (Garten et al. 2000; Ma et al. 2012; Ngaba et al. 2019; Wang et al. 2018). Generally, high litter inputs can lead to low values of δ13C and δ15N in soils, whereas enhanced soil organic matter (SOM) mineralization can enrich soil δ13C and δ15N (Garten et al. 2000; Natelhoffer and Fry 1988). Land use and cover change can alter C and N source inputs, SOM decomposition rates, soil erosion, and N cycle processes e.g., mineralization, nitrification and gas N loss (Chen et al. 2016; Guillaume et al. 2015; Hertel et al. 2009; Ross et al. 1999). Therefore, it has been suggested that the conversion of natural forests to plantations can lead to the changes in 13C and 15N in plant and soils at the stand scale (Deng et al. 2016; Llorente et al. 2010; Guillaume et al. 2015; Ngaba et al. 2019; Zeller et al. 2007). To our knowledge, however, no studies have systematically examined the isotopic signatures of 13C and 15N in soil aggregates following the conversion of natural forests to managed plantations along a national-scale climate zone.
China is currently the country most affected by LCC from natural forests to intensively managed plantations in the world (Lin et al. 2018; Yang et al. 2009). The destruction of primary forests is regarded as a main factor causing significant loss of soil C and N pools (Lewis et al. 2016; Wang et al. 2016). Thus, this study focuses on soil aggregate C and N concentrations, and 13C and 15N abundances at the surface (0–10 cm) and subsurface (10–20 cm) layers in the NF and adjacent PF across five climate zones along an approximately 4200 km transect from northern to southern China. We aim to evaluate whether the impacts of LCC on soil aggregate C and N differ among climate zones and between topsoil and subsoil, and to determine the relationship between the changes in soil aggregate C and N concentrations, and δ13C and δ15N with climatic factors, i.e., mean annual temperature (MAT) and mean annual precipitation (MAP). This study can provide new information on the effects of LCC under the background of global climate change. Based on the aforementioned studies, we hypothesized that (1) soil aggregate stability and C and N allocated in aggregates would decline in plantations because of intensive management disturbances, but (2) soil aggregate δ13C and δ15N would be enriched in plantations due to increasing SOM mineralization relative to adjacent natural forests. We also hypothesized that (3) the impacts of LCC on soil aggregate C and N would vary among climate zones, and the declines of soil aggregate C and N concentrations and the enrichments of aggregate δ13C and δ15N would increase with increasing MAT and MAP.
Materials and methods
Study site and sampling
The study sites are located in the eastern forest of China (18.44–52.92°N, 108.01–122.79°E) and have elevations of 275 to 800 m. In August 2017, soil samples were collected at five sites along a north-south transect in eastern China, including Mohe (MH), Qingyuan (QY), Huitong (HT), Dinghushan (DH), and Jianfengling (JF) (Fig. S1). At each site, natural forests (NFs) and paired adjacent plantations (PFs) converted from native vegetation were selected to ensure comparable stand conditions. The characteristics of climatic conditions, soil types and vegetation compositions in each stand are shown in Table 1. Three plots of 20 m × 20 m were set up in each stand and soils were taken at surface (0–10 cm) and subsurface (10–20 cm) layers, packed in zip locked polythene bags and taken back to the laboratory immediately for analysis of soil aggregate distribution, soil C and N concentrations, and isotopic abundances of 13C and 15N.
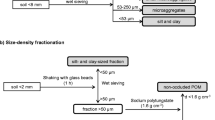
Soil aggregate fractionation
The aggregate size distribution and stability are determined using a wet sieving protocol (Kemper and Rosenau 1986). In summary, soils are air-dried for 2 weeks, and a sieve of 8 mm is used to remove roots, coarse plant residues and any stones >8 mm. Then, 40 g air-dried soil is used for aggregate size fractionation by wet sieving and is placed on a set of six nested sieves of 5, 2, 1, 0.5, 0.25 and 0.053 mm. The sieves are shaken vertically for 30 min at a rate of 35 strokes min−1 in a water bucket. The soil aggregate fractions retained in each sieve were carefully washed off the sieve into a preweighed aluminous container, oven-dried at 105 °C and weighed. A portion of soil aggregate <0.053 mm retained in water was also collected and oven dried at 105 °C for determination of soil C, N and isotopic abundances of 13C and 15N, but the mass was calculated from the total soil mass minus the dry mass of the other six aggregates. Each fraction was agitated for 18 h with 5% sodium hexametaphosphate (1:3 soil: liquid ratio) to correct the presence of coarse fragments and sands.
Soil C and N concentrations and natural abundance of 13C and 15N analysis
Soil C and N concentrations and stable isotopic abundances of 13C and 15N were measured using an isotope ratio mass spectrometer (IRMS) (IsoPrime 100, Isoprime Ltd., UK) connected to a CN elemental analyzer (Vario MICRO cube, Elementar, Germany). Carbon and nitrogen stable isotope abundances are calculated as δ13C and δ15N (‰) using the following formula:
where Rsample is the13C:12C or 15N:14N ratio in the samples and Rstandard is the 13C:12C or 15N:14N ratio in the standard. The Pee Dee Belemnite (PDB) and atmospheric N2 (δ15N = 0.0‰) are used as the standards for carbon and nitrogen, respectively. Internal standards (L-histidine, glycine, D-glutamic and acetanilide) are used to check the precision of isotopic compositions. In general, the analytical precisions for δ13C and δ15N are better than 0.2‰.
Statistical analysis
All statistical analyses were conducted using R software (version 3.5.1). Analysis of variance (ANOVA) was performed to test the effects of climate zone, land use, soil depth and soil aggregate size on the mass proportion of soil aggregate size fractions, C and N concentrations, and δ13C and δ15N values. Comparisons among different climate zones were tested using Tukey’s honest significant difference at p < 0.05. All data were subjected to the assumptions of normality and homogeneity of variance before conducting the ANOVA procedure. Regression analysis is performed to fit the relationships between climatic factors (i.e., MAT and MAP) and the changes in soil aggregate C and N and δ13C and δ15N calculated from NF minus PF, and among soil aggregate C and N and δ13C and δ15N values in natural forests and plantations.
Results
Soil aggregate mass proportion and mean weight diameter
Climate zones have significant influences on the mass proportion of each aggregate fraction (Table 2; Fig. 1). However, forest type and soil depth do not differ in the mass proportion of each fraction (Table 2), except that the averaged mass proportion of 0.053–0.25 mm microaggregate across five sampling sites and two depths is significantly higher in PF (21.4%) than in NF (17.4%). There is a forest type × climate zone interaction on the mass proportion of 0.5–1 mm aggregate size (Table 2, p = 0.03). Mean weight diameters (MWD) are not different between NF and PF but vary among climate zones with a significantly lower MWD in the south subtropical (SST) zone (Table 2, Fig. S2).
Mass proportion of each soil aggregate fraction in natural forests and plantations among five climate zones along a north-south transect in eastern China. NF, natural forest; PF, plantation forest. CT, cold temperate zone; MT, mid-temperate zone; MST, mid-subtropical zone; SST, south subtropical zone; TRO, marginal tropical zone
Soil aggregate C and N concentration
In general, soil C and N concentrations (g kg−1 aggregate) are almost uniformly distributed across all 7 aggregate fractions (Fig. 2a, b), except for a lower C concentration in the <0.053 mm fraction than in the 1–2 mm fraction (p = 0.03). The impacts of LCC on soil aggregate C and N concentrations vary among climate zones (Table 3). The averaged C concentration over aggregate sizes and depth is higher in PF than NF in the cold temperate (CT) zone (p < 0.001), but there are lower aggregate C concentrations in PF in both mid-temperate (MT) (p = 0.02) and tropical (TRO) zones (p = 0.03) compared to NF (Fig. 3a). The soil aggregate N concentration decreases from NF to PF in the MT (p = 0.02), SST (p < 0.001) and TRO (p = 0.03) zones, but PF has a higher aggregate N concentration than NF in the CT zone (Fig. 3b).
Distribution of soil (a) C and (b) N concentrations (g kg−1 aggregate) and (c) δ13C and (d) δ15N among aggregate size fractions under natural forests and plantations. The different lowercase letters indicate significant differences among aggregate size fractions. NF, natural forest; PF, plantation forest
Impacts of land cover change from natural forests to plantations on soil aggregate (a) C and (b) N concentrations and (c) δ13C and (d) δ15N among the five climate zones. The different lowercase letters on bars indicate significant differences among climate zones under natural forests and the different capital letters on bars indicate significant differences among climate zones under plantations. ***, ** and * indicate significant differences between natural forest and plantation in each climate zone at the level of p < 0.001, p < 0.01 and p < 0.05, and ns indicates a non-significant difference, respectively. NF, natural forest; PF, plantation forest. CT, cold temperate zone; MT, mid-temperate zone; MST, mid-subtropical zone; SST, south subtropical zone; TRO, marginal tropical zone
Climate has a strong influence on soil aggregate C and N concentrations (Table 3) but differs between NF and PF. In plantations, soil aggregate C decreases from the CT zone to the mid-subtropical (MST) zone but then slightly increases in the SST and TRO zones. However, NF has the highest aggregate C in the MT zone (Fig. 3a). The soil aggregate N concentrations in both NF and PF all increase from the CT to MT zones but then decline in the MST, SST and TRO zones (Fig. 3b). Furthermore, the changes in soil aggregate C concentration (ΔC) and N concentration (ΔN) calculated by subtracting PF from NF show nonlinear relationships with mean annual temperature (MAT) and precipitation (MAP) (Fig. 4a-d). In addition, although soil aggregate C and N all decrease with increasing soil depth in both NF and PF (Table 3; Fig. 2a, b), the declines of aggregate C and N concentrations are larger in NF (14.38 C g kg−1 and 0.85 N g kg−1) than PF (9.67 C g kg−1 and 0.48 N g kg−1).
Soil aggregate δ13C and δ15N content
Soil δ13C does not differ across all aggregate fractions (Table 3; Fig. 2c), but soil δ15N is significantly higher in the <0.053 mm aggregate size than in the other fractions (Fig. 2d). Soil aggregate δ13C and δ15N are significantly affected by forest type, soil depth and climate zone, and there are significant interactions between forest type and site location, and between forest type and soil depth (Table 3). The mean values of δ13C over aggregate sizes and soil depth are enriched after natural forests are converted to plantations, except in the CT zone (Fig. 3c). Similarly, PF has higher values of soil aggregate δ15N than NF in the CT, MT and TRO zones, but there are no differences in soil aggregate δ15N between NF and PF in either the MST or SST zones (Fig. 3d).
Mean values of soil δ13C over seven aggregate fractions in NF and PF show similar trends along climate zones and increase from the CT zone to MST zones but then decline to the TRO zone (Fig. 3c). Furthermore, soil aggregate Δδ13C has negative relationships with increasing MAT and MAP (Fig. 4e, f). However, the trend of soil aggregate δ15N along climate zones differs between NF and PF. The soil aggregate δ15N in the NF increases from the CT to MST zones and then declines to the TRO zone (Fig. 3d). For PF, however, the soil aggregate δ15N values in the CT, MT, MST and TRO zones are not different but are significantly higher than those in the SST zone. The soil aggregate Δδ15N at both soil depths shows nonlinear relationships with MAP (Fig. 4h), but there is no clear relationship between MAT and the different soil aggregate δ15N at 10–20 cm (Fig. 4g). Moreover, there are forest type × soil depth interactions on both δ13C and δ15N in soil aggregates (Table 3). Soil δ13C are significantly enriched along the soil depth in NF (p < 0.001), but there is no difference in soil δ13C between the two soil layers in PF (p = 0.08, Fig. 2c). Compared to the surface layer (0–10 cm), soil aggregate δ15N is enriched by 28% and 14% in NF and PF, respectively, at the 10–20 cm depths (Fig. 2d).
Relationships among soil C and N concentrations and δ13C and δ15N
The statistical analyses show a positive correlation between soil C and N concentration in both NF (r = 0.91, p < 0.001) and PF (r = 0.78, p < 0.001) (Fig. 5a). Moreover, there is a significantly positive correlation between soil aggregate δ13C and δ15N in NF (r = 0.71, p < 0.001) but not in PF (r = 0.19, p = 0.12) (Fig. 5b). In NF, soil δ13C and δ15N are negatively correlated with aggregate C and N concentrations, respectively (Fig. 5c, d). However, soil δ15N is not correlated with soil N concentration (Fig. 4d), and there is a weak negative correlation between soil aggregate δ13C and C concentration in PF (Fig. 5c).
Relationships of (a) soil aggregate C and N concentrations, (b) soil aggregate δ13C and δ15N, (c) soil aggregate C concentration and δ13C, and (d) soil aggregate N concentration and δ15N. Each point presents the mean value of three replication plots (n = 3). NF, natural forests; PF, plantations. The solid line indicates the regression of natural forests, and the dotted line indicates the regression of plantations
Discussion
Soil aggregate size distribution, and aggregate C and N concentrations as affected by LCC
In this study, we found an increase in microaggregates (0.053–0.25 mm) and silt + clay (<0.053 mm) but a decreasing mass proportion of macroaggregates (> 0.25 mm) in the intensively managed plantations relative to adjacent natural forests. These findings supported our first hypothesis that LCC from natural forests to plantations could decrease the stability of soil aggregates, and agreed with a previous study by Yang et al. (2009) who also found that converting old-growth native forests to Castanopsis Kawakamii and Cunninghamia lanceolata Lamb. plantations decreased the mass proportion of macroaggregates in southern China. Compared to natural forests, intensively managed plantations and croplands are usually exposed to various anthropogenic disturbances, e.g., site preparation, tree planting and furrow fertilization, which can disintegrate large aggregates into smaller aggregates and resulting in an increase in the proportion of small aggregates in soils (Abrishamkesh et al. 2011; Richards et al. 2009). Consequently, the excluded disturbances related to land use and cover change have been widely suggested to be prone to physical restoration of aggregate stability (Blanco-Canqui and Lal 2004; Pohl et al. 2012; Six et al. 1999). In addition, soil aggregation is mediated by complex factors, such as abiotic (clay minerals, carbonates, exchangeable cations), biotic (plant roots, soil fauna and microorganisms), and environmental (soil temperature and moisture) factors (Bronick and Lal 2005; Pohl et al. 2012; Tisdall and Oades 1982). In the present study, the changes in soil aggregate size distribution might be related to the reduction in SOC in plantations (Chen et al. 2004; Lin et al. 2018), and furthermore, the higher plant diversity, vegetation cover and root density in natural forests were likely beneficial for forming and stabilizing macroaggregates in soils (Le Bissonnais et al. 2018; Pohl et al. 2012).
The changes in C and N in soil aggregates can usually be used as early indicators of soil C and N changes (Leifeld and Kögel-Knabner 2005) and provide us with the mechanisms of soil C and N pool stabilization induced by LCC (Gelaw et al. 2015; Six et al. 2000; Zhu et al. 2017). We observed that the averaged soil C concentration across aggregate fractions declined significantly in the plantations in the MT and TRO zones compared with the natural forests. Similarly, several previous studies also observed a net loss of soil C and N after land conversion from natural forest to plantations (He et al. 2008; Lyu et al. 2017; Richards et al. 2009). This result is suggested to be related to the reduction of C inputs into soils and the increasing organic matter decomposition caused by the disturbances of site preparation and burning before plantation establishment (Chen et al. 2016; Deng et al. 2016; Goma-Tchimbakala 2009). Moreover, soil aggregation is an important mechanism for the stabilization of soil organic matter, and in less disturbed systems, the formation of macroaggregates can stabilize and protect soil organic C (Gelaw et al. 2015; Li et al. 2005; Six et al. 2000; Zhu et al. 2017). However, the soil aggregate C concentration was higher in PF than in NF in the cold temperate zone, and no differences in the soil aggregate C concentration were observed in the MST and SST zones after LCC. Our results indicated that land conversion from NF to PF had a strong influence on soil C and N dynamics, but the impacts varied among climate zones with positive, negative, or even no influence. Such large spatial-scale site-to-site variations in SOC were also observed by Lewis et al. (2016), who found that SOC was significantly higher in the pine plantation at two of the eight sites, was higher in the native vegetation at two sites and did not differ at four sites. They explained that the variance among sites was due to the different plantation species, the different environmental drivers (e.g., rainfall) and the higher degree of variability between sites (e.g., different plantation age, site preparation and management methods, soil types, and native vegetation types).
It is not surprising to us that soil aggregate C and N concentrations decreased with soil depth in both NF and PF. However, there were forest type × soil depth interactions on soil aggregate C and N concentrations, and the vertical differences in soil aggregate C and N between the two layers decreased in plantations compared with natural forests. This result indicated that land conversion altered the vertical distribution of soil aggregate C and N. Compared to subsoil, topsoil C and N are usually more sensitive to land cover and land use change (Gaudinski et al. 2000; Ngaba et al. 2019) because of enhanced topsoil erosion after natural forest conversion to plantations (Guillaume et al. 2015). In addition, the smaller differences in aggregate C and N concentrations along soil depth in plantations could be related to intensive management practices, such as digging, scarification, or stump lifting, resulting in mixing topsoil into the subsurface layer (Mobley et al. 2015).
Soil aggregate δ13C and δ15N as affected by LCC
Stable isotopes of 13C and 15N in soil aggregate fractions are an excellent integrative and potentially powerful tool for understanding the mechanisms of soil C and N changes after land cover change (John et al. 2005; Llorente et al. 2010). According to the concept of aggregate hierarchy, microaggregates are bound together into macroaggregates by transient binding agents and temporary binding agents (Tisdall and Oades 1982; Six et al. 2000). The consequences of this aggregate hierarchy are an increase in C concentration but a depletion of δ13C with increasing aggregate-size fraction (John et al. 2005). However, our data did not support the aggregate hierarchy model. Although a lower C in the <0.053 mm fraction was observed in the present study, we did not observe significant differences in soil δ13C with increasing aggregate-size fraction. Similar to the results in our study, John et al. (2005) also found that soil δ13C was uniformly distributed across soil aggregate fractions in forest soil. Some recent studies suggested that the formation of fine mineral-stabilized organic matter derived recently from litter plays an important role in SOM formation and stability (Cotrufo et al. 2015), which might explain the lower values of δ13C and δ15N because of the new plant C inputs in microaggregate fractions.
The mean values of δ13C and δ15N across soil aggregates were generally higher in the plantations than in natural forests. This result is consistent with our second hypothesis and with another previous study by Zeller et al. (2007) that found that soil under Norway spruce displayed a lower δ13C value than soil under a natural stand. In our study, there were significantly negative relationships of soil δ13C and C concentrations, and δ15N and N concentrations in NF, which was supported by other previous studies (Evans and Belnap 1999; Garten et al. 2000). This suggests that the enriched soil δ13C values in the plantations might be related to increased SOM decomposition due to site disturbances (Chen et al. 2016; Deng et al. 2016; Natelhoffer and Fry 1988). Moreover, the rate of litter input and the litter’s isotope abundance can also influence the isotopic abundances of soil C and N (Natelhoffer and Fry 1988). Usually, the litter decomposition rate is slower in plantations due to the lower litter quality compared with natural forests (Li et al. 2005), which results in fewer inputs of depleted litter C in plantations. One of our previous studies observed that the isotopic abundances of 13C and 15N in forest floor litter in NF were generally lower than those in PF (Ngaba et al. 2019) and which furthermore led to the higher values of soil δ13C and δ15N in plantations.
In addition, our results showed that there are increasing trends of δ13C and δ15N with soil depth in natural forests. The explanations of soil δ13C along soil depth are generally grouped into three categories: mixing, selective decomposition and preservation of compounds enriched in δ13C, and kinetic fractionation during humification (Bird et al. 2002a; Diochon and Kellman 2008; Garten et al. 2000). However, these depth trends of soil δ13C were not observed in plantations, and the magnitude of the 15N shift from the surface to depth was less in PF than in NF. The declined gradients of soil aggregate δ13C and δ15N in plantations might be related to the mechanisms of topsoil erosion, mechanical mixing between soil layers, and increasing C mineralization in topsoil with disturbances compared to intact natural forests (Diochon and Kellman 2008; Eshetu and Högberg 2000; Guillaume et al. 2015).
Changes in soil aggregate distribution, C and N concentrations, and δ13C and δ15N among climate zones
For our third and last hypothesis, we found that climate zone had significant impacts on the mass proportion of each soil aggregate fraction, the averaged C and N concentrations, and isotopic abundances of 13C and 15N across aggregates. Though the trends of soil aggregate C and N concentrations, and aggregate δ13C and δ15N along the climate zones were slightly different between NF and PF, we found that the soil C and N concentrations generally increased from the cold temperate zone to the mid-temperate zone, declined to the subtropical zone, and then again exhibited a slight increase in the tropical zone. In the present study, the lower C and N concentrations in NF in the cold temperate zone compared to the mid-temperate zone might be related to the short-term restoration of the natural forests at the site where one serious fire disaster occurred in 1987. Usually, climate factors can influence soil C and N dynamics through biotic processes of forest productivity and decomposition of SOM (Post et al. 1985). In our study, however, the pattern of forest biomass productivity cannot explain the trends of soil C and N along the north-south transect in eastern China, considering that net ecosystem productivity decreased with increasing latitude (Yu et al. 2013; Wen and He 2016). Many previous studies have reported that the decomposition of SOC increases with increasing MAT, thereby causing soil C and N loss and enrichment of soil isotopic abundances of 13C and 15N (Bird et al. 2002b; Garten et al. 2000; Homann et al. 2007; Högberg 1997). However, the relationships of soil C and N turnover with MAP are more complex and usually show an inflection point (Jobbágy and Jackson 2000; Wang et al. 2018). The decomposition rate of soil organic matter can decline when MAP is very high and anaerobic conditions dominate (Wang et al. 2018). In addition, a decline in water use efficiency at wetter sites can promote the depletion of heavy C and N isotopes in soils (Austin and Vitousek 1998; Peri et al. 2012).
It is interesting that our data show the changes in soil C and N concentrations, and δ13C and δ15N induced by LCC are related to MAT and MAP. In the plantations soil C becomes more enriched in 13C with increased MAT and MAP, suggesting greater decomposition in the plantations compared to the natural forests with increased MAT and MAP (Fig. 4e, f). And yet, soil C pools converged to the same value between the natural forests and plantations at the highest MAT and MAP (Fig. 4a, b), suggesting that C inputs in plantations kept up with higher decomposition rates at these sites. Although there are many previous studies reporting positive, negative or no impacts of land cover change from natural forests to plantations on soil C and N pools in various climate zones (Chen et al. 2004; Guillaume et al. 2015; Li et al. 2014; Lin et al. 2018; Lewis et al. 2016; Yang et al. 2019), there is still little research comparing the different effects of LCC along climatic factors. Liao et al. (2012) reported larger declines in soil C in the temperate zone than in the tropical zone after land cover change from natural forests to plantations. In their review on the changes in SOC induced by LCC worldwide, Guo and Gifford (2002) found that the release of soil C was different among areas with precipitation <1000 mm, 1000–1500 mm and > 1500 mm. Several mechanisms might explain the varied impacts of LCC on soil C and N. First, the soil temperature sensitivity (Q10) of soil C and N was different between natural forests and plantations (Yang et al. 2017), thereby resulting in the varied response of soil C and N between natural forests and plantations with increasing MAT. Second, soil C accumulation and 13C abundance are affected by the quantity and quality of plant C sources (Natelhoffer and Fry 1988; Hatton et al. 2015). The differential rates of productivity and vegetation composition between natural forests and plantations varied among climate zones (Brown et al. 2020; Cai et al. 2019), which might also contribute to the differences in soil C and N changes induced by LCC among climate zones with increasing MAT and MAP.
Conclusion
Our results indicated that the impacts of LCC on soil aggregate C and N, and δ13C and δ15N varied among climate zones with increasing, decreasing or no influence on soil aggregate C and N because of the site-to-site variances of the linked differences in climatic conditions, soil texture and dominant vegetation. Moreover, our results implied that the changes in soil C and N concentrations, and isotopic abundances of 13C and 15N are related to climatic factors and that at large spatial scales, natural environmental factors (e.g., MAT and MAP) had a greater influence on soil C and N dynamics compared to the anthropogenic disturbance of natural forests converting to managed plantations. Considering the limits of our study, in the future, more studies are needed to rule out the relative contribution of climatic factors on soil C and N dynamics compared to other factors, such as vegetation production and composition, soil texture, and plantation age.
References
Abrishamkesh S, Gorji M, Asadi H (2011) Long-term effects on land use on soil aggregate stability. Int Agrophys 25:103–108
Ashagrie Y, Zech W, Guggenberger G (2005) Transformation of a Podocarpus falcatus dominated natural forest into a monoculture Eucalyptus globulus plantation at Munesa, Ethiopia: soil organic C, N and S dynamics in primary particle and aggregate-size fractions. Agric Ecosyst Environ 106:89–98. https://doi.org/10.1016/j.agee.2004.07.015
Austin AT, Vitousek PM (1998) Nutrient dynamics on a precipitation gradient in Hawai’i. Oecologia 113:519–529. https://doi.org/10.1007/s004420050405
Bird MI, Santruckova H, Arneth A, Grigoriev S, Gleixner G, Kalaschnikov YN, Lloyd J, Schulze ED (2002a) Soil carbon inventories and carbon-13 on a latitude transect in Siberia. Tellus B 54(5):631–641. https://doi.org/10.3402/tellusb.v54i5.16699
Bird MI, Santruckova H, Lloyd J, Lawson E (2002b) The isotopic composition of soil organic carbon on a north-south transect in western Canada. Eur J Soil Sci 53:393–403. https://doi.org/10.1007/s11104-012-1304-6
Blanco-Canqui H, Lal R (2004) Mechanisms of carbon sequestration in soil aggregates. Crit Rev Plant Sci 23:481–504. https://doi.org/10.1080/07352680490886842
Bol RA, Harkness DD, Huang Y, Howard DM (1999) The influence of soil processes on carbon isotope distribution and turnover in the British uplands. Eur J Soil Sci 50(1):41–51. https://doi.org/10.1046/j.1365-2389.1999.00222.x
Bronick CJ, Lal R (2005) Soil structure and management: a review. Geoderma 124:3–22. https://doi.org/10.1016/j.geoderma.2004.03.005
Brown HCA, Berninger FA, Larjavaara M, Appiah M (2020) Above-ground carbon stocks and timber value of old timber plantations, secondary and primary forests in southern Ghana. For Eco Manag 472:118236. https://doi.org/10.1016/j.foreco.2020.118236
Cai H, Li F, Jin G (2019) Fine root biomass, production and turnover rates in plantations versus natural forests: effects of stand characteristics and soil properties. Plant Soil 436:463–474. https://doi.org/10.1007/s11104-019-03948-8
Chen CR, Xu ZH, Mathers NJ (2004) Soil carbon pools in adjacent natural and plantations forests of subtropical Australia. Soil Sci Soc Am J 68:282–291. https://doi.org/10.2136/sssaj2004.2820
Chen G, Yang Y, Yang Z, Xie J, Guo J, Gao R, Yin Y, Robinson D (2016) Accelerated soil carbon turnover under tree plantations limits soil carbon storage. Sci Rep 6:19693. https://doi.org/10.1038/srep19693
Chen C, Liu W, Jiang X, Wu J (2017) Effects of rubber-based agroforestry systems on soil aggregation and associated soil organic carbon: implications for land use. Geoderma 299:13–24. https://doi.org/10.1016/j.geoderma.2017.03.021
Cotrufo MF, Soong JL, Horton AJ, Campbell EE (2015) Formation of soil organic matter via biochemical and physical pathways of litter mass loss. Nat Geosci 8:776–779
Deng L, Wang KB, Tang ZS, Shangguan ZP (2016) Soil organic carbon dynamics following natural vegetation restoration: evidence from stable carbon isotopes (δ13C). Agric Ecosyst Environ 221:235–244. https://doi.org/10.1016/j.agee.2016.01.048
Diochon A, Kellman L (2008) Natural abundance measurements of 13C indicate increased deep soil carbon mineralization after forest disturbance. Geophys Res Lett 35(14). https://doi.org/10.1029/2008GL034795
Dixon RK, Brown S, Houghton RA, Solomon AM, Trexler MC, Wisniewski J (1994) Carbon pools and flux of global forest ecosystems. Science 263:185–190. https://doi.org/10.1126/science.263.5144.185
Eshetu Z, Högberg P (2000) Effects of land use on 15N natural abundance of soils in Ethiopian highlands. Plant Soil 222(1–2):109–117. https://doi.org/10.1023/A:1004777301260
Evans RD, Belnap J (1999) Long-term consequences of disturbance on nitrogen dynamics in an arid ecosystem. Ecology 80(1):150–160. https://doi.org/10.1890/0012-9658(1999)080[0150:LTCODO]2.0.CO;2
Garten JCT, Cooper LW, Post WM III, Hanson PJ (2000) Climate controls on forest soil C isotope ratios in the southern Appalachian Mountains. Ecology 81(4):1108–1119. https://doi.org/10.1890/0012-9658(2000)081[1108:CCOFSC]2.0.CO;2
Gaudinski JB, Trumbore SE, Davidson EA, Zheng S (2000) Soil carbon cycling in a temperate forest: radiocarbon-based estimates of residence times, sequestration rates and partitioning of fluxes. Biogeochemistry 51:33–69. https://doi.org/10.1023/A:1006301010014
Gelaw AM, Singh BR, Lal R (2015) Organic carbon and nitrogen associated with soil aggregates and particle sizes under different land uses in Tigray, northern Ethiopia. Land Degrad Dev 26:690–700. https://doi.org/10.1002/ldr.2261
Gerschlauer F, Saiz G, Costa DS, Kleyer M, Dannenmann M, Kiese R (2019) Stable carbon and nitrogen isotopic composition of leaves, litter, and soils of various ecosystems along an elevational and land-use gradient at Mount Kilimanjaro, Tanzania. Biogeosciences 16:409–424. https://doi.org/10.5194/bg-16-409-2019
Goma-Tchimbakala J (2009) Carbon and nitrogen storage in soil aggregates from different Terminali superba age plantations and natural forest in Kouilou, Congo. Int J Soil Sci. https://doi.org/10.3923/ijss.2009.104.113
Guillaume T, Damris M, Kuzyakov Y (2015) Losses of soil carbon by converting tropical forest to plantations: erosion and decomposition estimated by δ13C. Glob Chang Biol 21(9):3548–3560. https://doi.org/10.1111/gcb.12907
Guo LB, Gifford RM (2002) Soil carbon stocks and land use change: a meta analysis. Glob Chang Biol 8:345–360. https://doi.org/10.1046/j.1354-1013.2002.00486.x
Hatton PJ, Castanha C, Torn MS, Bird JA (2015) Litter type control on soil C and N stabilization dynamics in a temperate forest. Glob Chang Biol 21:1358–1367. https://doi.org/10.1111/gcb.12786
He Y, Xu Z, Chen C, Burton J, Ma Q, Ge Y, Xu J (2008) Using light fraction and macroaggregate associated organic matters as early indicators for management-induced changes in soil chemical and biological properties in adjacent native and plantation forests of subtropical Australia. Geoderma 147(3–4):116–125. https://doi.org/10.1016/j.geoderma.2008.08.002
Hertel D, Harteveld MA, Leuschner C (2009) Conversion of a tropical forest into agroforest alters the fine root-related carbon flux to the soil. Soil Biol Biochem 41(3):481–490. https://doi.org/10.1016/j.soilbio.2008.11.020
Högberg P (1997) 15N natural abundance in soil–plant systems. New Phytol 137(2):179–203
Homann PS, Kapchinske JS, Boyce A (2007) Relations of mineral-soil C and N to climate and texture: regional differences within the conterminous USA. Biogeochemistry 85(3):303–316. https://doi.org/10.1007/s10533-007-9139-6
Houghton RA, Nassikas AA (2017) Global and regional fluxes of carbon from land use and land cover change 1850–2015. Glob Biogeochem Cycles 31:456–472. https://doi.org/10.1002/2016GB005546
Jandl R, Lindner M, Vesterdal L, Bauwens B, Baritz R, Hagedorn F, Johnson DW, Minkkinen K, Byrne KA (2007) How strongly can forest management influence soil carbon sequestration? Geoderma 137(3–4):253–268. https://doi.org/10.1016/j.geoderma.2006.09.003
Jobbágy EG, Jackson RB (2000) The vertical distribution of soil organic carbon and its relation to climate and vegetation. Ecol Appl 10(2):423–436. https://doi.org/10.1890/1051-0761(2000)010[0423:TVDOSO]2.0.CO;2
John B, Yamashita T, Ludwig B, Flessa H (2005) Storage of organic carbon in aggregate and density fractions of silty soils under different types of land use. Geoderma 128:63–79. https://doi.org/10.1016/j.geoderma.2004.12.013
Kemper WD, Rosenau RC (1986) Aggregate stability and size distribution. In: Klute A (ed) Methods of soil analysis, Part 1: physical and mineralogical methods, 2nd edn. ASA and Soil Science Society of America, Madison, Wisconsin, pp 425–442
Le Bissonnais Y, Prieto I, Roumet C, Nespoulous J, Metayer J, Huon S, Villatoro M, Stokes A (2018) Soil aggregate stability in Mediterranean and tropical agro-ecosystems: effect of plant roots and soil characteristics. Plant Soil 424(1–2):303–317. https://doi.org/10.1007/s11104-017-3423-6
Leifeld J, Kögel-Knabner I (2005) Soil organic matter fractions as early indicators for carbon stock changes under different land-use? Geodema 124:143–155. https://doi.org/10.1016/j.geoderma.2004.04.009
Lewis T, Simth TE, Hogg B, Swift S, Verstraten L, Bryant P, Wehr BJ, Tindale N, Menzies NW, Dala RC (2016) Conversion of sub-tropical native vegetation to introduced conifer forest: impacts on below-ground and above-ground carbon pools. For Ecol Manag 370:65–75. https://doi.org/10.1016/j.foreco.2016.03.058
Li Y, Xu M, Zou X et al (2005) Comparing soil organic carbon dynamics in plantation and secondary forest in wet tropics in Puerto Rico. Glob Chang Biol 11:239–248. https://doi.org/10.1111/j.1365-2486.2005.00896.x
Li YF, Zhang JJ, Chang SX, Jiang PK, Zhou GM, Shen ZM, Wu JS, Lin L, Wang ZS, Shen MC (2014) Converting native shrub forests to Chinese chestnut plantations and subsequent intensive management affected soil C and N pools. For Eco Manag 312:160–169. https://doi.org/10.1016/j.foreco.2013.10.008
Liao C, Luo Y, Fang C, Chen J, Li B (2012) The effects of plantation practice on soil properties based on the comparison between natural and planted forests: a meta-analysis. Glob Ecol Biogeogr 21:318–327. https://doi.org/10.1111/j.1466-8238.2011.00690.x
Lin Z, Li Y, Tang C, Luo Y, Fu W, Cai X, Li Y, Yue T, Jiang P, Hu S, Chang SX (2018) Converting natural evergreen broadleaf forests to intensively managed moso bamboo plantations affects the pool size and stability of soil organic carbon and enzyme activities. Biol Fertil Soils 54(4):467–480. https://doi.org/10.1007/s00374-018-1275-8
Llorente M, Glaser B, Turrión (2010) Anthropogenic disturbance of natural forest vegetation on calcareous soils alters soil organic matter composition and natural abundance of 13C and 15N in density fractions. Eur J For Res 129:1143–1153. https://doi.org/10.1007/s10342-010-0402-3
Lyu M, Xie J, Ukonmaanaho L, Jiang M, Li Y, Chen Y, Yang Z, Zhou Y, Lin W, Yang Y (2017) Land use change exerts a strong impact on deep soil C stabilization in subtropical forests. J Soils Sediments 17:2305–2317. https://doi.org/10.1007/s11368-016-1428-z
Ma JY, Sun W, Liu XN, Chen FH (2012) Variation in the stable carbon and nitrogen isotope composition of plants and soil along a precipitation gradient in northern China. PLoS One 7:e51894. https://doi.org/10.1371/journal.pone.0051894
Mobley ML, Lajtha K, Kramer MG, Bacon AR, Heine PR, Richter DD (2015) Surficial gains and subsoil losses of soil carbon and nitrogen during secondary forest development. Glob Chang Biol 21:986–996. https://doi.org/10.1111/gcb.12715
Natelhoffer KJ, Fry B (1988) Controls on natural nitrogen-15 and carbon-13 abundances in forest soil organic matter. Soil Sci Soc Am J 52:1633–1640. https://doi.org/10.2136/sssaj1988.03615995005200060024x
Ngaba MJY, Hu YL, Bol R, Ma XQ, Jin SF, Mgelwa AS (2019) Effects of land use change from natural forest to plantation on C, N and natural abundance of 13C and 15N along a climate gradient in eastern China. Sci Rep 9(1):1–12. https://doi.org/10.1038/s41598-019-52959-z
Peri PL, Ladd B, Pepper DA, Bonser SP, Laffan SW, Amelung W (2012) Carbon (δ13C) and nitrogen (δ15N) stable isotope composition in plant and soil in southern Patagonia's native forests. Glob Chang Biol 18:311–321. https://doi.org/10.1111/j.1365-2486.2011.02494.x
Pohl M, Graf F, Buttler A, Rixen C (2012) The relationship between plant species richness and soil aggregate stability can depend on disturbance. Plant Soil 355:87–102. https://doi.org/10.1007/s11104-011-1083-5
Post WM, Pastor J, Zinke PJ, Stangenberger AG (1985) Global patterns of soil nitrogen storage. Nature 317. https://doi.org/10.1038/317613a0
Richards AE, Dalal RC, Schmidt S (2009) Carbon storage n a ferrosol under subtropical rainforest tree plantations, and pasture in linked to soil aggregation. Aus J Soil Res 47:341–350
Ross DJ, Tate KR, Scott NA, Feltham CW (1999) Land-use change: effects on soil carbon, nitrogen and phosphorus pools and fluxes in three adjacent ecosystems. Soil Biol Biochem 31(6):803–813. https://doi.org/10.1016/S0038-0717(98)00180-1
Six J, Paustian K (2014) Aggregate-associated soil organic matter as an ecosystem property and a measurement tool. Soil Biol Biochem 68:A4–A9. https://doi.org/10.1016/j.soilbio.2013.06.014
Six J, Elliott ET, Paustian K (1999) Aggregate and soil organic matter dynamics under conventional and no-tillage systems. Soil Sci Soc Am J 63(5):1350–1358. https://doi.org/10.2136/sssaj1999.6351350x
Six J, Elliott ET, Paustian K (2000) Soil macroaggregate turnover and microaggregate formation : a mechanism for C sequestration under no-tillage agriculture. Soil Biol Biochem 32(14):2099–2103. https://doi.org/10.1016/S0038-0717(00)00179-6
Spohn M, Giani L (2011) Impacts of land use change on soil aggregation and aggregate stabilizing compounds as dependent on time. Soil Biol Biochem 43:1081–1088. https://doi.org/10.1016/j.soilbio.2011.01.029
Straaten O, Corre MD, Wolf K, Tchienkoua M, Cuellar E, Matthews RB, Veldkamp E (2015) Conversion of lowland tropical forests to tree cash crop plantations loses up to one-half of stored soil organic carbon. PNAS 112:9956–9960. https://doi.org/10.1073/pnas.1504628112/-/DCSupplemental
Tisdall JM, Oades JM (1982) Organic-matter and water-stable aggregates in soils. J Soil Sci 33:141–163. https://doi.org/10.1111/j.1365-2389.1982.tb01755.x
Wang K, Deng L, Ren Z, Shi W, Chen Y, Shang-Guan Z (2016) Dynamics of ecosystem carbon stocks during vegetation restoration on the loess plateau of China. J Arid Land 8(2):207–220. https://doi.org/10.1007/s40333-015-0091-3
Wang C, Houlton BZ, Liu D, Hou J, Cheng W, Bai E (2018) Stable isotope constraints on global soil organic carbon turnover. Biogeosciences 15:987–995. https://doi.org/10.5194/bg-15-987-2018
Wen D, He N (2016) Forest carbon storage along the north-south transect of eastern China: spatial patterns, allocation, and influencing factors. Ecol Indic 61:960–967. https://doi.org/10.1016/j.ecolind.2015.10.054
Yang Y, Guo J, Chen G, Ying Y, Gao R, Lin C (2009) Effects of forest conversion on soil labile organic carbon fractions and aggregate stability in subtropical China. Plant Soil 323:153–162. https://doi.org/10.1007/s11104-009-9921-4
Yang K, He R, Yang W, Li Z, Zhuang L, Wu F, Tan B, Liu Y, Zhang L, Tu L, Xu Z (2017) Temperature response of soil carbon decomposition depends strongly on forest management practice and soil layer on the eastern Tibetan Plateau. Sci Rep 7:4777. https://doi.org/10.1038/s41598-017-05141-2
Yang Z, Chen S, Liu X, Xiong D, Xu C, Arthur MA, McCulley RL, Shi S, Yang Y (2019) Loss of soil organic carbon following natural forest conversion to Chinese fir plantation. For Ecol Manag 449:117476. https://doi.org/10.1016/j.foreco.2019.117476
Yu G, Zhu X, Fu Y et al (2013) Spatial patterns and climate drivers of carbon fluxes in terrestrial ecosystems of China. Glob Chang Biol 19:798–810. https://doi.org/10.1111/gcb.12079
Zeller B, Brechet C, Maurice JP, Le Tacon F (2007) 13C and 15N isotopic fractionation in trees, soils and fungi in a natural forest stand and a Norway spruce plantation. Ann For Sci 64(4):419–429. https://doi.org/10.1051/forest:2007019
Zhu GY, Shangguan ZP, Deng L (2017) Soil aggregate stability and aggregate-associated carbon and nitrogen between natural restoration grassland and Chinese red pine plantation on the loess plateau. Catena 149:253–260. https://doi.org/10.1016/j.catena.2016.10.004
Acknowledgments
This research was supported by the National Natural Science Foundation of China (No. U1805243), State Key Laboratory of Soil and Sustainable Agriculture and China Scholarship Council (CSC) University Scholarship Program. We thank Liu Jun, Wang Yu-Zhe, Qu Ru-Ping and Qiu Qing-Yan, and Ge Zhi-Qiang, Xu Wen-Bin, Huang Zhi-Jing and Wei Qiang for sampling in field and laboratory analysis. We are also grateful to Karanja Joe for the constructive suggestions on this paper.
Author information
Authors and Affiliations
Contributions
M.N. elaborated in sampling, and obtained and analyzed data; M.N. and R.B. wrote the first draft of the paper. Y.H. designed study concept. R.B. and Y.H. critically revised the final paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Feike A. Dijkstra.
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOC 400 kb)
Rights and permissions
About this article
Cite this article
Ngaba, M.J.Y., Bol, R. & Hu, YL. Stable isotopic signatures of carbon and nitrogen in soil aggregates following the conversion of natural forests to managed plantations in eastern China. Plant Soil 459, 371–385 (2021). https://doi.org/10.1007/s11104-020-04754-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-020-04754-3