Abstract
Background and aims
The inoculation of cereal crops with plant growth-promoting bacteria (PGPB) is a potential strategy to improve fertilizer-N acquisition by crops in soils with low capacity to supply N. A study was conducted to assess the impact of three inoculants on grain yield, protein content, and urea-15 N recovery in maize (Zea mays L.) under Cerrado soil and climate conditions.
Methods
The main treatments included inoculants containing (i) Azospirillum brasilense strain Sp245, (ii) A. brasilense strains AbV5 + AbV6, (iii) Herbaspirillum seropedicae strain ZAE94, and (iv) a non-inoculated control. The subtreatments were (i) urea-N fertilization (100 kg N ha−1) at 30 days after sowing and (ii) no N addition at the stage. To determine fertilizer-N recovery, 15N–labelled urea was applied in microplots.
Results
Inoculants carrying A. brasilense improved urea-15 N acquisition efficiency in maize and also improved grain yield compared to the non-inoculated control, while urea-N fertilization enhanced grain quality by providing higher protein content.
Conclusion
Our results suggest that the inoculation of maize grains with PGPB represents a strategy to improve fertilizer-N recovery and maize yield in Cerrado soil with a low capacity to supply N.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Approximately 70 million Mg of maize (Zea mays L.) were produced in Brazil in 2016 from a cropped area of 15.9 Mha and this amount represents more than one-third of the total grain production in the country (CONAB 2016). However, maize is commonly grown in low-fertility soils and thus requires high fertilizer-N inputs. For example, at the beginning of this decade, maize crops consumed 25% of the total commercial fertilizer-N used in Brazil (Heffer 2013). A significant amount of this fertilizer-N is used on soils with poor fertility with low levels of potentially mineralizable N, such as sandy Oxisols in the Cerrado region (Alves et al. 2016). The use of biological techniques to improve fertilizer-N use efficiency can represent a sustainable alternative for cereal-growing in these tropical areas. It is also important to consider the frequent occurrence of dry spells in the Cerrado region (Martins et al. 2015), which are commonly associated to inefficient N use by plants (Hu and Schmidhalter 2005) since reduction in the transpiration rate induced by drought stress reduces the crop N uptake by lowering the transportation of this nutrient from roots to shoots (Tanguilig et al. 1987). Therefore, the inoculation of cereal crops with PGPB is a potential strategy for improving N acquisition from fertilizers and reducing environmental risks associated with fertilization (Owen et al. 2015; Herrera et al. 2016).
The fact that the use of bacterial inoculants in agriculture has a tremendous positive impact on world food production and the preservation of environmental resources is indisputable (e.g., Alves et al. 2003). Specifically, the benefits of diazotrophic PGPB on grasses have been studied over the last six decades (Döbereiner 1953, 1959), enabling the development of specific inoculants that have been commercially applied worldwide in the most important cereals, including maize (Zea mays L.), rice (Oryza sativa L.), and wheat (Triticum aestivum L.) (Cassan et al. 2015). However, even in countries that have engaged in pioneering PGPB research, such as Brazil, the large-scale application of bacterial inoculants to grasses in agricultural areas is incipient and remains challenging. It is estimated that less than 10% of the areas cropped with grasses in Brazil use bacterial inoculants, which represents only 7% of the total amount of bacterial inoculants commercialized in Brazil in 2015; more than 90% of the inoculants are applied to soybean (Urquiaga et al. 2015).
Many in-depth studies have been conducted to improve knowledge on the bacteria-plant association, employing techniques involving genomics, proteomics, and metabolomics (Mukherjee et al. 2016; Spaepen and Vanderleyden 2015) as well as co-inoculation (Yegorenkova et al. 2016) and the industrial production of inoculants (Bashan and Bashan 2015). However, there is still a lack of information regarding the magnitude and exact mechanisms involved in the positive effects of bacterial inoculants on N nutrition in plants under field conditions, including the role played by PGPB in the recovery of N derived from fertilizer. To this end, isotope techniques can generate reliable results regarding the potential for biological techniques to improve fertilizer use efficiency. The use of isotope techniques are particularly important in studies of plant N nutrition, taking into account the complexity of its biogeochemical cycle having three main sources: biological N2 fixation, soil organic matter mineralization, and fertilizer-N. In addition to the known role of diazotrophic bacteria on biological N2 fixation in grasses (Urquiaga et al. 2012; Alves et al. 2015), these microorganisms indirectly affect plants by stimulating growth through the production of regulators, such as auxins, as demonstrated in previous studies (e.g., Spaepen and Vanderleyden 2015). The impact of this hypothesized positive effect of diazotrophic bacteria on N uptake through growth promotion (Spaepen and Vanderleyden 2011; Hungria et al. 2016) can be measured through isotope-aided balance studies, which may contribute to demonstrating an important additional benefit of inoculant application to cereals under field conditions (improvement of fertilizer-N recovery) and provide further scientific support for the large-scale application of bacterial inoculants to these crops, especially maize.
Among the diazotrophic bacteria that may have a growth-promoting effect, Azospirillum brasilense is a rhizobacteria that has been used in the field inoculation of grasses with great success (Ferreira et al. 2013; Wisniewski-Dye et al. 2013; Hungria et al. 2016). Other diazotrophic bacteria have also been investigated for possible use as inoculants for grasses such as Herbaspirillum seropedicae, which has been associated with maize and other grasses (Baldani et al. 2000; Monteiro et al. 2012; Alves et al. 2015). Selecting PGPB strains adapted for a specific soil condition can enhance the success of large-scale inoculant applications and thus improve grain yield.
Specifically, field studies examining the role of PGPB in the improvement of fertilizer-N recovery under Cerrado region conditions are especially important for agriculture in Brazil, considering that this is the most significant region in that country legally apt for agriculture expansion (Coelho 2011). However, there is a paucity of research regarding the role of PGPB in maize production in this important agricultural region. Therefore, the objective of this study was to assess the impact of inoculation with A. brasilense (strains Sp245 and AbV5 + AbV6) and H. seropedicae (strain ZAE94) on grain yield, protein content, and urea-N recovery by maize in a Cerrado Oxisol with a low capacity to supply N.
Materials and methods
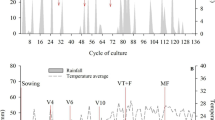
The experiment was conducted in the 2012–2013 summer cropping season in Luis Eduardo Magalhães, in western Bahia State, Brazil. The experimental area is located in the tropical megathermal zone, or Köppen’s Aw (a tropical climate with a dry winter and an average temperature in the coldest month >18 °C) and is located within the Cerrado biome (12°06’S, 46°02’W and an altitude of 837 m). Rainfall during the cropping season (July 2012–June 2013) was 1187 mm with an annual distribution that peaks between October and March and a severe dry season from April to September. The cumulative precipitation from maize sowing to harvest was 895 mm with dry spells that are typical of the Cerrado region, such as that observed from 17 to 30 days after maize sowing (Fig. 1).
The soil in the experimental area was a sandy Oxisol (Xanthic Hapludox), based on U.S. Soil Taxonomy (Soil Survey Staff 2010). Physical, chemical and mineralogical soil properties are shown in Table 1. Cerrado Oxisols are characterized by a relatively high content of iron and aluminum oxides in the clay fraction with low Ruxton ratio (SiO2:Al2O3) values, that indicate an intense weathering process (Table 1). This area has been used for commercial production of maize for twenty years, i.e., since the Cerrado vegetation was replaced by agriculture. No-tillage cultivation had been in use for several years before the field experiment, but conventional tillage (ploughing and disking) was reinstated in the 2010/2011 crop season.
A three-way hybrid of maize (Z. mays L. ATL300) was mechanically sown on November 27, 2012, with a row spacing of 0.50 m and 3.3 plants m−1. NPK 4–14–8 was applied as a banded starter fertilizer (18 kg N ha−1) below and to the side of the seed position. Potassium (125 kg K ha−1) was broadcast 15 days after sowing as potassium chloride. The treatments were applied in a split-plot arrangement in a randomized complete block design with 4 replicates. The main treatments were three inoculants applied to the maize grains just before sowing operation and a non-inoculated control. The inoculants were (i) A. brasilense strain Sp245, (ii) A. brasilense strains AbV5 + AbV6, and (iii) H. seropedicae strain ZAE94. The Herbaspirillum inoculant was prepared in sterile peat at a concentration of 108 cells g−1 of substrate and applied at a rate of 25 g kg−1 of seeds. The two Azospirillum inoculants were prepared in liquid formulation at a concentration of 5 × 108 cells mL−1 and applied at a rate of 5 mL kg−1 of seeds. To increase adhesion of the three inoculants to the seeds, a 100 g L−1 sucrose solution was added at a rate of 5 mL kg−1 seeds. The sucrose solution was mixed with 2 kg of maize seeds in a plastic bag and the inoculant was carefully mixed and allowed to air-dry in the shade for 30 min.
The treatments (seed inoculants) were applied in plots consisting of 5 maize rows, and each row was 20.0 m long for a total area of 50 m2. These main plots were further split into two 10.0-m long subplots (25 m2) that received the sub-treatments: in one urea was broadcast on the soil surface 30 days after sowing and in other no N was added at this stage. Urea was broadcast 27 days after sowing at a rate of 100 kg N ha−1. The two outer rows and 1.5 m at the ends of each row were considered the borders of each subplot. To determine the recovery of urea-N by maize, 15N–labelled urea (1.0 atom% 15N in excess) was applied in a microplot within each fertilized subplot. One microplot was established in an 3.0-m2 area (1.0 m of width × 3.0 m of length) in the center of each fertilized subplot (each replicate). At harvest, 169 days after sowing, the 5 maize plants occupying the middle 1.5 m of the central row of the 15N–labelled microplot were collected and their grain was separated and weighed. The remaining material (straw) was also weighed and finely chopped and subsamples of the grain and straw were dried in a forced-air oven at 65 °C for 48 h, reweighed, and ground (Wiley mill, 2 mm sieve). The dry plant material was then powder-milled and analyzed for total N and 15N (Arnold and Schepers 2004), following the procedures described by Ramos et al. (2001). The remainder of the subplot was also harvested and weighted to obtain a more precise estimate of maize yield.
The percentage of N in the plant derived from the 15N–labelled fertilizer (%Ndff) was calculated as described by the IAEA (2001) as follows:
With %Ndff value, the N content in plant material (%N) and the dry matter (DM) yield in kg ha−1, it was possible to calculate the amount of N in the plants derived from fertilizer as follows:
Therefore, the urea-N recovery by plants as a percentage (%N recovery) was calculated using to the following equation:
The grain moisture content was adjusted to 130 g kg−1 before the statistical analyses and the protein content in the grains was obtained by multiplying the grain N content by 5.68 (Sriperm et al. 2011). The data of yield, N uptake, protein content, and 15N recovery data were submitted to analysis of variance (ANOVA), and the means were compared using Tukey’s HSD test (α = 0.05). ANOVA was performed after determining the normality of errors (Shapiro-Wilk’s test) and the homogeneity of variance (Bartlett’s test) of the data. Statistical analyses were performed using R software (R Development Core Team 2014).
Results
A significant inoculation effect was observed in both the maize straw and grain yields of maize (Tables 2 and 3). Both inoculants carrying A. brasilense improved grain yield compared to the H. seropedicae inoculant and to the non-inoculated control (Table 3). Independent of the strain and urea-N fertilization, the net increase in grain yield resulting from inoculation with A. brasilense, that is, the difference between the yield of the non-inoculated control and that of inoculated treatments, was generally higher than 1000 kg ha−1 of grain. The improvement in grain yield associated with A. brasilense was very similar for strains Sp245 and AbV5 + AbV6 (Table 3) and, with or without the application of urea-N, the increases of grain yield attributed to inoculation with these strains resulted in a greater than 30% increase in grain yield (Table 3). On the other hand, the inoculant carrying H. seropedicae did not have a significant effect on maize yield (straw or grains) (Table 3). The improvement in grain yield attributed to A. brasilense inoculation compared to the non-inoculated control (>1000 kg ha−1) was even greater than the improvement induced by urea-N fertilization compared to the unfertilized treatments (475 to 876 kg ha−1).
The effects of the inoculants on straw and grain yield were similar (Table 3). Inoculants with A. brasilense (both strains Sp245 and AbV5 + AbV6) increased straw yield relative to the non-inoculated control, with or without urea-N fertilization (Table 3). Similarly to the grain yield results, inoculation with A. brasilense (both strains) increased straw yield more than urea-N fertilization. From the results presented in Table 3, it is possible to calculate the maize harvest index, namely, the weight of the maize grains as a percentage of the sum of the straw and grain weights, which ranged from 41% to 47% in present study.
The parameter in this study indicating the quality of maize grains was the protein content, which was affected by urea-N fertilization without being significantly influenced by inoculation (Tables 2 and 4). On average, the urea-N fertilization increased the grain protein content by 20% compared to the unfertilized treatments (Table 4) and although the effect of the inoculants on grain protein content was not statistically significant (Table 2), there was a trend of higher protein content in grain of inoculated maize (84 to 85 g kg−1), compared to the non-inoculated control (80 g kg−1).
The uptake of N in grains and straw was affected by both inoculation and urea-N fertilization (Tables 2 and 5) and the effects of inoculants on the uptake of N in grains was similar to the effects on grain yield (Tables 3 and 5). The N uptake by grain of maize inoculated with A. brasilense amounted to 61–63 kg N ha−1, which was higher than with H. seropedicae (50 kg N ha−1) and in the non-inoculated control (43 kg N ha−1) (Table 5). The N uptake by the straw of the unfertilized maize was not influenced by inoculation, but inoculation with strains of A. brasilense had a significant positive effect on the N uptake by the straw of the maize fertilized with urea. In general, as observed for grain yield, the total N uptake (straw + grains) was affected more by inoculation than urea-N fertilization. Compared to the non-inoculated control, the A. brasilense Sp245 and AbV5 + AbV6 strains increased, on average, the total N uptake by maize by 51% and 59%, respectively, while urea-N fertilization the increased total N uptake by 41%, on average, compared to the unfertilized treatments (Table 5).
The results of 15N in the grains and straw indicated that the percentage of N in the plant derived from the labelled urea (%Ndff) was not significantly affected by the inoculants, and there was even a trend toward higher values in the inoculated treatments (Tables 2 and 6). Considering the values of %Ndff as well as the %N and the yield of grain and straw, it was possible to obtain the recovery percentage of urea-N by maize and its distribution in the two components (Fig. 2). It is important to note that the inoculants had a significant effect on the urea-N recovery by maize and on its distribution in the grain and straw. The recovery of urea-N by maize shoots (grains + straw) was 58% following inoculation with A. brasilense strains AbV5 + AbV6, 50% with A. brasilense strain Sp245, and only 34% in the non-inoculated maize (Fig. 2). Inoculation with H. seropedicae strain ZAE94 did not significantly increase urea-N recovery by maize plants (Fig. 2), and the urea-N recovery in grains was very similar in the two A. brasilense treatments, i.e., 36% of the applied urea-N, which was markedly higher than the 23% recovery of urea-N in the grains of non-inoculated maize (Fig. 2). Conversely, considering the impact of inoculants on the recovery of urea-N by maize straw, there were no significant differences among the A. brasilense strain Sp245 (14%), H. seropedicae strain ZAE94 (13%), and the non-inoculated maize (11%). However, the inoculation of maize with A. brasilense strains AbV5 + AbV6 doubled the urea-N recovery in straw compared to the 11% observed in the non-inoculated control (Fig. 2). Overall, an increase in grain yield was closely associated with higher urea-N recovery by maize inoculated with A. brasilense compared to non-inoculated maize (Table 3 and Fig. 2).
Recovery of urea-N in maize in response to inoculants. Urea was applied at a rate of 100 kg N ha−1 and its isotope enrichment was 1.0 atom% 15N in excess. Among inoculants, values followed by same letters are not significantly different based on Tukey’s test (α = 0.05). Underlined letters indicate comparision of total fertilizer-N recovery (straw + grains) among inoculants. Error bars represent the standard error of the mean (n = 4)
Discussion
The results of the present study, it was shown that inoculants with PGPB had a significant positive impact on N plant nutrition and on maize grain yield, and the impact was even higher than that of urea-N fertilization, which had more pronounced effects on grain protein content. These results showed that the inoculants quantitatively improved maize production, whereas the qualitative aspect maize production, as measured by grain protein content, was affected more by urea-N fertilization. Our results indicated that inoculants carrying A. brasilense markedly improved nutrient acquisition efficiency, as revealed by a significantly higher recovery of 15N derived from urea (+71%), which resulted in higher grain yield (Fig. 2 and Table 3). This result indicates that PGPB can be used to improve the conversion of an expensive agricultural input (fertilizer-N) into the final product (maize grains), thus mitigating economic and environmental problems caused by the inefficient use of synthetic fertilizer-N by maize. The PGPB-induced increase in maize grain yield in our study is consistent with results obtained by Hungria et al. (2010), who performed experiments in other regions of Brazil with inoculants carrying A. brasilense strains AbV5 + AbV6. In a sandy Oxisol (732 g kg−1 of sand) from Ponta Grossa in Parana State, these authors found that an inoculant carrying those A. brasilense strains increased grain yields by a range from 591 to 787 kg ha−1.
Although the exact mechanisms underlying the PGPB effect on N acquisition by maize was not evaluated in the present study, it is very likely that the improvement in urea-15 N recovery by maize inoculated with A. brasilense is associated with its well-known ability to produce plant growth regulators (Tien et al. 1979; Cohen et al. 2009; Spaepen and Vanderleyden 2015). Specifically, it was previously demonstrated that the strains Sp245 and AbV5 of A. brasilense, which were tested in the present study, have the ability to produce indoles (Dobbelaere et al. 1999; Ona et al. 2005; Ferreira et al. 2015), which can be one of the most important factors affecting plant growth. For example, this putative mechanism of growth promotion might have improved the ability of the plants to more efficiently explore the soil, as indicated in previous studies using A. brasilense (Cohen et al. 2015; Ambrosini et al. 2016), but it has also been well documented also that PGPB can help plants tolerate drought stress (Nadeem et al. 2015; Ngumbi and Kloepper 2016). Therefore, it is important to consider that the impact of PGPB on the nutrient use efficiency, including N from urea as revealed by 15N–recovery (Fig. 2), cannot be separated from the role of these bacteria in the possible alleviation of drought stress (Hu and Schmidhalter 2005). Casanovas et al. (2002) showed that drought stress tolerance in maize seedlings was improved by inoculating seeds with A. brasilense Sp245, which is one of the strains tested in the present study. This mechanism can contribute to the effect of A. brasilense on the fertilizer-N acquisition (Fig. 2), since the reduction in the transpiration rate of the plant caused by drought stress reduces N uptake by lowering the transportation of this nutrient from the roots to the shoots (Tanguilig et al. 1987). For example, this might have been particularly important during the dry spells that occurred two weeks before and one week after urea-N sidedressing, which corresponded to the maize growth stages V3 to V7 (Fig. 1). Dry spells, which normally occur from one to three weeks during the peak rainy season, are a characteristic of the Cerrado biome, where they are called “veranico” (Kornelius et al. 1979). In general, the role of PGPB in the alleviation of drought stress as well other abiotic stressors, such as low nutrient levels (Ferreira et al. 2013), is gaining attention as a strategy to increase food production (Ngumbi and Kloepper 2016). Therefore, the use of PGPB can play a special role in cereal crops cultivated in soils commonly subjected to abiotic stresses, such as the highly weathered Cerrado soils with low capacity to supply N and frequent of dry spells.
Factors explaining the different responses of maize to inoculants carrying different PGPB (strains of A. brasilense compared to H. seropedicae) include the ability of these bacteria to fix N2 from the atmosphere (Alves et al. 2015; Ambrosini et al. 2016) and produce plant regulators (Radwan et al. 2004) as well as the bacteria-genotype interaction (Garcia de Salamone et al. 1996; Alexandre 2015; Pereg et al. 2016; Scharf et al. 2016). In addition, previous studies have indicated that Azospirillum develop better in soils with lower contents of organic matter (Fallik et al. 1988) and clay (Ferreira et al. 2013) and these two factors might have maximized the effects of the A. brasilense inoculants in the sandy Oxisol of the present study (Table 4 and Fig. 2), which has low clay (~10%) and soil organic matter contents, as indicated by its low C and N contents (Table 1). A possible explanation for the lack of an effect by H. seropedicae ZAE94 on maize yield in the present study is a bacteria strain-maize genotype interaction that suppressed the effect of inoculation. Many studies in the literature suggest that the positive effect of PGPB is dependent on plant genotype (Garcia de Salamone et al. 1996; Baldani et al. 2000; Monteiro et al. 2012), and it is important to consider that, although inoculation of maize with H. seropedicae ZAE94 did not have a positive effect on the yield and N nutrition of maize in the present study (Tables 3 and 5), this bacterium has previously been shown to have positive effects. For instance, Alves et al. (2015) found that this PGPB played a significant role in the improvement of BNF in maize. In the present study, we attempted to quantify BNF using the 15N natural abundance technique, but the poor 15N signature of the soil available N (δ15N < 5‰), as measured by an analysis of non-N2-fixing reference plants, did not permit an accurate and precise estimate with this methodology. Nonetheless, the analysis of unfertilized maize showed a tendency toward a decrease in the abundance of 15N in maize with inoculation, but the effect was not statistically significant (data not shown). The δ15N values were 5.3 ± 0.8‰ in the non-inoculated control, 4.2 ± 0.4‰ in maize inoculated with A. brasilense strain Sp245, 3.5 ± 0.1‰ with A. brasilense strain AbV5 + AbV6 and 3.5 ± 0.4‰ with H. seropedicae ZAE94. The use of other techniques to quantify of N2 fixation, such as isotope dilution (Boddey et al. 1995; IAEA 2001), would be necessary reliably estimate the N in maize derived from BNF in the type of the soil in the present study.
Additional studies that consider roles of the other agronomic factors in the inoculation success (e.g., genotype, supplying of irrigation) and a deeper mechanistic understanding of the role of PGPB in alleviating the abiotic stresses commonly found in Cerrado are needed. Such studies may help support a recommendation for the large-scale agronomic use of inoculants in the production of maize and other non-leguminous crops in the Cerrado region, which is one of the most important regions in the world that are apt for agricultural expansion.
Conclusions
The results of the present study indicated that the inoculation of maize seeds with PGPB represents a strategy for improving fertilizer-N recovery and maize yield in a highly weathered Cerrado soil with low capacity to supply N. The grain quality, measured by protein content, was affected more by urea-N fertilization, whereas the quantitative parameter of grain production (grain yield) was more affected by inoculation with PGPB. The Azospirillum-induced increase in maize grain yield was higher than the urea-N fertilization effect. The inoculation of maize with PGPB to improve urea-N use efficiency can be considered an economically and environmentally sound strategy for cereal production, as urea is the most commonly used N-fertilizer, especially in tropical soils with very low contents of this nutrient, such as weathered Oxisols in the Cerrado region.
Abbreviations
- DM:
-
Dry mass
- Ndff:
-
N in the plant derived from 15N–labelled fertilizer
- PGPB:
-
Plant growth-promoting bacteria
References
Alexandre G (2015) Chemotaxis in Azospirillum. In: Cassán FD, Okon Y, Creus CM (eds) Handbook for Azospirillum. Springer, Basel, pp 101–114. doi:10.1007/978-3-319-06542-7
Alves BJR, Boddey RM, Urquiaga S (2003) The success of BNF in soybean in Brazil. Plant Soil 252:1–9. doi:10.1023/A:1024191913296
Alves GC, Videira SS, Urquiaga S, Reis VM (2015) Differential plant growth promotion and nitrogen fixation in two genotypes of maize by several Herbaspirillum inoculants. Plant Soil 387:307–321. doi:10.1007/s11104-014-2295-2
Alves BJR, Martins MR, Boddey RM, Jantalia CP, Urquiaga S (2016) Importância da fixação biológica de nitrogênio para a sustentabilidade agrícola no Cerrado. In: Flores RA, Cunha PP (eds) Práticas de manejo do solo para adequada nutrição de plantas no Cerrado, 1st edn. Gráfica UFG, Goiânia, pp 220–259
Ambrosini A, Souza R, Passaglia LMP (2016) Ecological role of bacterial inoculants and their potential impact on soil microbial diversity. Plant Soil 400:193–207. doi:10.1007/s11104-015-2727-7
Arnold SL, Schepers JS (2004) A simple roller-mill grinding procedure for plant and soil samples. Commun Soil Sci Plant Anal 35:537–545. doi:10.1081/CSS-120029730
Baldani VLD, Baldani JI, Döbereiner J (2000) Inoculation of rice plants with endophytic diazotrophs Herbaspirillum seropedicae and Burkholderia spp. Biol Fertil Soil 30:485–491. doi:10.1007/s003740050027
Bashan Y, de-Bashan LE (2015) Inoculant preparation and formulations for Azospirillum spp. In: Cassán FD et al. (eds) Handbook for Azospirillum. Springer, Switzerland, pp 469–485. doi:10.1007/978-3-319-06542-7_26
Boddey RM, Oliveira OC, Alves BJR, Urquiaga S (1995) Field application of the 15 N isotope dilution technique for the reliable quantification of plant-associated biological nitrogen fixation. Nutr Cycl Agroecosyst 42:77–87. doi:10.1007/978-94-009-1706-4_7
Casanovas EM, Barassi CA, Sueldo RJ (2002) Azospirillum inoculation mitigates water stress effects in maize seedlings. Cereal Res Commun 30:343–350
Cassan FD, Okon Y, Creus CM (2015) Handbook for Azospirillum. Springer, Basel. doi:10.1007/978-3-319-06542-7
Coelho FB (2011) O Cerrado e a integração nacional, 63ª Reunião Anual da SBPC SBPC. Goiânia, GO, Brazil
Cohen AC, Travaglia CN, Bottini R, Piccoli PN (2009) Participation of abscisic acid and gibberellins produced by endophytic Azospirillum in the alleviation of drought effects in maize. Botany 87:455–462. doi:10.1139/B09-023
Cohen A, Bottini R, Pontin M, Berli F, Moreno D, Boccanlandro H, Travaglia C, Picocoli P (2015) Azospirillum brasilense ameliorates the response of Arabidopsis thaliana to drought mainly via enhancement of ABA levels. Physiol Plant 153:79–90. doi:10.1111/ppl.12221
CONAB (2016) Acompanhamento da safra brasileira 2015/2016: Sétimo levantamento <www.conab.gov.br>. Accessed 20 June 2016
R Development Core Team (2014) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. <http://www.R-project.org>
Dobbelaere S, Croonenborghs A, Thys A, Broek AV, Vanderleyden J (1999) Phytostimulatory effect of Azospirillum brasilense wild type and mutant strains altered in IAA production on wheat. Plant Soil 212:153–162. doi:10.1023/A:1004658000815
Döbereiner J (1953) Azotobacter em solos ácidos. Bol Inst Ecol Exp Agr 11:1–36
Döbereiner J (1959) Influência da cana-de-açúcar na população de Beijerinckia do solo. Rev Bras Biol 19:251–258
EMBRAPA – Empresa Brasileira de Pesquisa Agropecuária (1997) Manual de métodos de análise de solos, 2nd edn. Centro Nacional de Pesquisa de Solos, Rio de Janeiro
Fallik E, Okon Y, Fischer M (1988) Growth response of maize roots to Azospirillum inoculation: effect of soil organic matter content, number of rhizosphere bacteria and timing of inoculation. Soil Biol Biochem 20:45–49. doi:10.1016/0038-0717(88)90125-3
Ferreira AS, Pires RR, Rabelo PG, Oliveira RC, Luz JMQ, Brito CH (2013) Implications of Azospirillum brasilense inoculation and nutrient addition on maize in soils of the Brazilian Cerrado under greenhouse and field conditions. Appl Soil Ecol 72:103–108. doi:10.1016/j.apsoil.2013.05.020
Ferreira EPB, Castro AP, Martin-Didonet CCG, Fageria NK (2015) Agronomical performance of upland rice cultivars inoculated with Azospirillum brasilense depends on the plant genotype. Commun Soil Sci Plant Anal 46:1751–1762. doi:10.1080/00103624.2015.1043456
Garcia de Salamone IE, Döbereiner J, Urquiaga S, Boddey RM (1996) Biological nitrogen fixation in Azospirillum strain-maize genotype associations as evaluated by the 15N isotope dilution technique. Biol Fertil Soil 23:249–256. doi:10.1007/BF00335952
Heffer P (2013) Assessment of fertilizer use by crop at the global level. International Fertilizer Industry Association, Paris
Herrera JM, Rubio G, Levy L, Delgado JA, Lucho-Constantino CA, Islas-Valdez S, Pellet D (2016) Emerging and established technologies to increase nitrogen use efficiency of cereals. Agronomy 6:25. doi:10.3390/agronomy6020025
Hu Y, Schmidhalter U (2005) Drought and salinity: a comparison of their effects on mineral nutrition of plants. J Plant Nutr Soil Sci 168:541–549. doi:10.1002/jpln.200420516
Hungria M, Campo RJ, Souza EM, Pedrosa FO (2010) Inoculation with selected strains of Azospirillum brasilense and A. lipoferum improves yields of maize and wheat in Brazil. Plant Soil 331:413–425. doi:10.1007/s11104-009-0262-0
Hungria M, Nogueira MA, Araujo RS (2016) Inoculation of Brachiaria spp with the plant growth-promoting bacterium Azospirillum brasilense: an environment-friendly component in the reclamation of degraded pastures in the tropics. Agric Ecosyst Environ 221:125–131. doi:10.1016/j.agee.2016.01.024
IAEA - International Atomic Energy Agency (2001) Training course series No 14. Use of isotope and radiation methods in soil and water management and crop nutrition. IAEA, Vienna
Kornelius E, Saueressig MG, Goedert WJ (1979) Pasture establishment and management in the Cerrado of Brazil. In: Sanchez PA, Tergas LE (eds) Pasture production in acid soils of the tropics. CIAT, Cali, pp 147–166
Martins MR, Jantalia CP, Polidoro JC, Batista JN, Alves BJR, Boddey RM, Urquiaga S (2015) Nitrous oxide and ammonia emissions from N fertilization of maize crop under no-till in a Cerrado soil. Soil Till Res 151:75–81. doi:10.1016/j.still.2015.03.004
Monteiro RA, Balsanelli E, Wassem R, Morin AM, Brusamarello-Santos LCC, Schmidt MA, Tadra-Sfeir MZ, Pankievicz VCS, Cruz LM, Chubatsu LS, Pedrosa FO, Souza EM (2012) Herbaspirillum-plant interactions: microscopical, histological and molecular aspects. Plant Soil 356:175–196. doi:10.1007/s11104-02-1125-7
Mukherjee T, Kumar D, Burriss N, Xie Z, Alexandre G (2016) Azospirillum brasilense chemotaxis depends on two signaling pathways regulating distinct motility parameters. J Bacteriol 198:1764–1772. doi:10.1128/JB.00020–16
Nadeem SM, Naveed M, Ahmad M, Zahir ZA (2015) Rhizosphere bacteria for crop production and improvement of stress tolerance: mechanisms of action, applications, and future prospects. In: Arora N (ed) Plant microbes symbiosis: applied facets. Springer, Berlin, pp 1–36. doi:10.1007/978-81-322-2068-8_1
Ngumbi E, Kloepper J (2016) Bacterial-mediated drought tolerance: current and future prospects. Appl Soil Ecol 105:109–125. doi:10.1016/j.apsoil.2016.04.009
Ona O, Van Impe J, Prinsen E, Vanderleyden J (2005) Growth and indole-3-acetic acid biosynthesis of Azospirillum brasilense Sp245 is environmentally controlled. FEMS Microbiol Lett 246:125–132. doi:10.1016/j.femsle.2005.03.048
Owen D, Williams AP, Griffith GW, Withers PJA (2015) Use of commercial bio-inoculants to increase agricultural production through improved phosphrous acquisition. Appl Soil Ecol 86:41–54. doi:10.1016/j.apsoil.2014.09.012
Pereg L, de-Bashan LE, Bashan Y (2016) Assessment of affinity and specificity of Azospirillum for plants. Plant Soil 399:389–414. doi:10.1007/s11104-015-2778-9
Radwan TSD, Mohamed ZK, Reis VM (2004) Effect of inoculation with Azospirillum and Herbaspirillum on production of indolic compounds and growth of wheat and rice seedlings. Pesq Agropec Bras 39:987–994. doi:10.1590/S0100-204X2004001000006
Ramos MG, Villatoro MAA, Urquiaga S, Alves BJR, Boddey RM (2001) Quantification of the contribution of biological nitrogen fixation to tropical green manure crops and the residual benefit to a subsequent maize crop using 15N-isotope techniques. J Biotechnol 91:105–115. doi:10.1016/S0168-1656(01)00335-2
Scharf BE, Hynes MF, Alexandre GM (2016) Chemotaxis signaling systems in model beneficial plant–bacteria associations. Plant Mol Biol 90:549–559. doi:10.1007/s11103-016-0432-4
Soil Survey Staff (2010) Keys to soil taxonomy, 11th edn. USDA NRCS, Washington, DC
Spaepen S, Vanderleyden J (2011) Auxin and plant-microbe interactions. In: Mark E, Weijers D, Leyser O, Ljung K (eds) Cold Spring Harbor Perspectives in Biololgy. Cold Spring Harbor Laboratory Press. doi:10.1101/cshperspect.a001438
Spaepen S, Vanderleyden J (2015) Auxin signaling in Azospirillum brasilense: a proteome analysis. In: Bruijn FJ (ed) Biological Nitrogen Fixation. John Wiley & Sons Inc, Hoboken. doi:10.1002/9781119053095.ch91
Sriperm N, Pesti GM, Tillman PB (2011) Evaluation of the fixed nitrogen-to-protein (N:P) conversion factor (6.25) versus ingredient specific N:P conversion factors in feedstuffs. J Sci Food Agric 91:1182–1186. doi:10.1002/jsfa.4292
Tanguilig VC, Yambao EB, O’Toole JC, Dedatta SK (1987) Water-stress effects on leaf elongation, leaf water potential, transpiration, and nutrient-uptake of rice, maize, and soybean. Plant Soil 103:155–168. doi:10.1007/BF02370385
Tien TM, Gaskins MH, Hubbell DH (1979) Plant growth substances produced by Azospirillum brasilense and their effect on the growth of pearl millet (Pennisetum americanum L.). Appl Environ Microbiol 37:1016–1024
Urquiaga S, Xavier RP, de Morais RF, Batista RB, Schultz N, Leite JM (2012) Maia e Sá J, Barbosa KP, de Resende AS, Alves BJR, Boddey RM (2011) evidence from field nitrogen balance and 15N natural abundance data for the contribution of biological N2 fixation to Brazilian sugarcane varieties. Plant Soil 356:5–21. doi:10.1007/s11104-011-1016-3
Urquiaga S, Alves BJR, Reis VM, Jantalia CP, Xavier GR, Zilli JE, Martins MR, Baldani JI, Boddey RM (2015) Balanço de N na produção de grãos e cana-de-açúcar na agricultura brasileira: Impacto da fixação biológica de N2. In: Book of Abstracts, Fertbio. Araxa, MG, Brazil
Wisniewski-Dye F, Drogue B, Borland S, Prigent-Combaret C (2013) Azospirillum-plant interaction: from root colonisation to plant growth promotion. In: Gonzalez MBR, González-López J (eds) Beneficial plant-microbial interactions: ecology and applications. CRC Press, Boca Raton, pp 237–269. doi:10.1201/b15251-12
Yegorenkova IV, Tregubova KV, Burygin GL, Matora LY, Ignatov VV (2016) Assessing the efficacy of co-inoculation of wheat seedlings with the associative bacteria Paenibacillus polymyxa 1465 and Azospirillum brasilense Sp245. Can J Microbiol 62:279–285. doi:10.1139/cjm-2015-0647
Acknowledgements
We acknowledge the Coordination for the Improvement of Higher Education Personnel (CAPES-EMBRAPA agreement) and the Foundation for the Support of Research in the State of Rio de Janeiro (FAPERJ) for the post-doctoral scholarship awarded to M. R. Martins and the research grant “FAPERJ - Cientista do Nosso Estado” awarded to B.J.R. Alves, S. Urquiaga, V. M. Reis, and R. M. Boddey. We also thank the Brazilian National Council for Scientific and Technological Development (CNPq) for the fellowship awarded to the same authors. We gratefully acknowledge Altiberto M. Baêta and Renato Moutinho da Rocha for their assistance with the laboratory analyses, Henrique A. Silva from for his assistance with the field experiment and Josimar N. Batista for the precipitation data.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Stéphane Compant .
Rights and permissions
About this article
Cite this article
Martins, M.R., Jantalia, C.P., Reis, V.M. et al. Impact of plant growth-promoting bacteria on grain yield, protein content, and urea-15 N recovery by maize in a Cerrado Oxisol. Plant Soil 422, 239–250 (2018). https://doi.org/10.1007/s11104-017-3193-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-017-3193-1