Abstract
The use of growth-promoting and potentially nitrogen-fixing (N) bacteria in corn is a viable alternative to increase grain yield, reduce costs and environmental risks. However, the role of Azospirillum brasilense in the process of N utilization by plants needs to be better understood for its efficient use. Thus, the objective of this study was to evaluate the responses of inoculation with A. brasilense in corn cultivated in succession to cover crops associated with doses of mineral N. Two experiments were carried out in Augusto Pestana and Santa Maria (Rio Grande do Sul, Brazil), growing maize in succession to winter crops (black oats and forage radish), under doses of mineral N in top dressing (0, 75, 150 and 225 kg of N ha− 1), associated with A. brasilense inoculation. It was found that corn inoculation under black oat responded better to N rates applied to the thousand seed weight (TSW), grain yield (GY) (Augusto Pestana) and relative chlorophyll content (RCC) (Santa Maria). This was not evidenced when on forage radish residue, which quadratically adjusted the variables regarding N doses. In Santa Maria, the yield components were influenced by the interaction between the predecessor crop and the N dose, with a linear response to N doses when under black oat, obtaining isolated positive effects of inoculation for TSW and GY. It was concluded that the effect of inoculation of corn with A. brasilense is affected by residue from previous crops and N dose, with greater N demand in succession to black oat.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Corn (Zea mays L.) is the main cereal grown in the world, being used in agriculture, food and pharmaceutical industries and as energy source for ethanol production (Hafiz et al. 2015; USDA 2021). Currently, it is estimated that world production is approximately 1,12 billion tons. Brazil is the third largest producer in the world (115,602 million tons) with an area of over 20 million hectares (CONAB 2022). New areas of cultivation are limited geographically and by the competition for space established by summer crops.
Due to the dry matter production of corn, the need for nitrogen (N) is high (Setiyono et al. 2010; Morris et al. 2018), which implies a high demand for mineral N (16,4 to 22,3 kg of N/t of grains). Due to leaching, volatilization and surface runoff processes, the efficiency of mineral N application has become limited with values close to 50% under the best conditions (Szulc et al. 2020). As a result, crop production costs and water contamination can be high (Lorensini et al. 2012). Thus, options that can reduce the use of N by plants, maintain or increase grain productivity, are of paramount importance to the crop production chain.
The relationship between N need and fertilization depends on the predecessor crop, soil organic matter content and expected yield (CQFS-RS/SC 2016). The different crops that preceded maize are capable of immobilizing N (Qin et al. 2021), exerting a negative allelopathic effect on the rhizosphere (Koehler-Cole et al. 2020), presenting symptoms of nutritional deficiency and reduced crop growth. Thus, changes in the plant transcriptome are evidenced, with modification of the root system architecture, chlorophyll and amino acid content (Bang et al. 2021). As a consequence, there will be a reduction in photosynthesis and production of photoassimilates, which are essential for increasing grain and dry matter yields (Fu et al. 2020). Genetic improvement provides maize cultivars with high yield potential, however the environment and management (especially N) are capable of restricting these high yields (Skonieski et al. 2019).
The use of growth-promoting rhizobacteria, with the capacity to biologically fix atmospheric N (BNF), produce phytohormones and increase the root system, are alternatives in reducing costs and increasing productivity (Backer et al. 2018; Breedt et al. 2017). Azospirillum brasilense, when associated with grasses, can provide N to the crop via BNF, act as a hormone promoter, improve the utilization of nitrogen fertilizers, reduce the doses of mineral N applied, and minimize the environmental impact of the use of mineral N (Hungria et al. 2010; Shin et al. 2016; Souza et al. 2019). This has resulted in corn yields close to 10 t ha− 1 with a 30 to 40% reduction in nitrogen fertilization, due to the higher foliar nitrogen content, number of grains per ear and root development (Oliveira et al. 2017; Scott et al. 2020; Artyszak and Gozdowski 2020).
The use of cover crops in the winter period cycles nutrients, improves soil physicochemical properties and biological activities, reduces soil temperature, maintains moisture and suppresses weeds in the no-tillage system (Silva et al. 2017; Pellin et al. 2019; Vincent-Caboud et al. 2019). In corn production systems, black oat (Avena strigosa Schreb.) and radish (Raphanus sativus L.) are species widely used in the off-season due to their rapid growth for soil cover and low seed cost for implementation. However, these species differ significantly in terms of Carbon:Nitrogen (C:N) ratio and residue decomposition rate, with a direct effect on the release of nutrients, mainly N, with oat residues and forage radish showing an average C:N ratio of 42 and 29, respectively (Acosta et al. 2014). Nutrient immobilization was obtained in the first thirty days after black oat desiccation, with a gradual productivity reduction of the corn crop after long periods of cultivation without nitrogen fertilization, due to the high input of dry matter poor in N and C/N ratio above 30, resulting in nutritional deficiency in the crop (Piva et al. 2021). On the other hand, forage radish has a higher rate of degradation and release of nutrients (Michelon et al. 2019).
In this sense, the amount and origin of the dry matter of the cover crops change the rhizosphere and the microbiological relationships between the soil and the cultivated plants (Vukicevich et al. 2016; Fanin et al. 2016). Thus, the inoculation of corn with A. brasilense under black oat or radish straw can cause different responses to the doses of N applied. The objective of this study was to evaluate the responses of inoculation with A. brasilense in the corn crop cultivated in succession to cover crops and under doses of mineral N.
2 Materials and methods
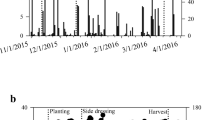
Two field experiments were carried out in the state of Rio Grande do Sul, Brazil. Experiment I was carried out in the municipality of Augusto Pestana (28° 26’ 30” S; 54° 0’58” W and altitude of 400 m), in soil classified as typical dystroferric red Latosol (EMBRAPA 2013), exhibiting the following soil chemical characteristics in the 0–10 cm depth layer: pH: 5.9; P: 13.4 mg dm− 3; K: 141 mg dm− 3; Ca: 5.4 cmolc dm− 3; Mg: 1.6 cmolc dm− 3; Cu: 7.3 mg dm− 3; Zn: 2.0 mg dm− 3; Mn: 6.7 mg dm− 3; S: 19.8 mg dm− 3. Experiment II was carried out in Santa Maria (29º 43’04” S; 53º 44’ 01” W and altitude of 116 m), in soil of the area classified as sandy (Arênico) Dystrophic Red Argisol (EMBRAPA 2013), with the following chemical characteristics quantified in the layer of 0–10 cm: pH: 6.7; P: 21.4 mg dm− 3; K: 54 mg dm− 3; Ca: 7.7 cmolc dm− 3; Mg: 3.0 cmolc dm− 3. The meteorological conditions of precipitation and average temperature during the growing season were adequate in experiment I (Fig. 1 A). However, there was a lower accumulated volume of rainfall in experiment II, with lower water availability in the flowering period of the crop (Fig. 1B).
Both experiments were carried out in a randomized block design with four replications. The treatments were distributed in a trifactorial (2 × 2 × 4), evaluating two predecessor crops (forage radish and black oat), use or not of inoculant based on A. brasilense and four doses of mineral N applied in topdressing (0, 75, 150 and 225 kg of N ha− 1). The experimental units consisted of six rows of five meters in length, spaced at 0.45 m, totaling 13.5 m2. The evaluations were carried out in the three central rows, using four meters in length in each plot, with a useful area of 5.4 m2.
The cover crops in both locations were sown in the second half of June 2017, with a seeder-fertilizer consisting of double discs and rows spaced at 17 cm, regulated to distribute 400 seeds m− 2 of black oat and 300 seeds m− 2 of forage radish. The base fertilization was carried out at sowing with the application of 350 kg ha− 1 of the formulated NPK (05-20-20), adding 120 kg ha− 1 of urea in top dressing applied to plants with four leaves. The desiccation of cover crops in experiment I took place in the second half of September, with a tank mixture of glyphosate + 2.4-D amine, applied at doses of 720 and 1005 g a.i. ha− 1, respectively. In experiment II, desiccation was also carried out in the second half of September with glyphosate + atrazine, applied in a tank mixture at a dose of 1080 g a.i. and 1750 g a.i. ha− 1, respectively. The herbicide paraquat at a dose of 400 g a.i. ha− 1 was applied at both sites two days before corn sowing.
The sowing of simple hybrid corn AG 9025, super early with VT PRO3 technology was carried out on nov/01/2017 (Experiment I) and out/27/2017 (Experiment II), at a density of 3.5 plants per linear meter and spacing 0.45 m between rows. Inoculation with A. brasilense (strains AbV5 and AbV6) was carried out via seed treatment, at a dose of 5 mL kg− 1 of seed, at the time of sowing. In experiment I, the base fertilization consisted of 366 kg ha− 1 of triple superphosphate and 120 kg ha− 1 of potassium chloride. In experiment II, 150 kg ha-¹ of triple superphosphate and 150 kg ha-¹ of potassium chloride were used. Topdressing nitrogen was applied at phenological stages V4, V6 and V10, according to the phenological scale of Ritchie et al. (1993), with application of one third of the dose established for each treatment (0, 75, 150 and 225 kg ha− 1) in the defined phenological stages.
The effects of the treatments were evaluated by counting the final plant stand (FPS), root dry mass (RDM), number of grains per row (NGR), number of rows per ear (NRE), relative chlorophyll content (RCC), thousand seed weight (TSW) and grain yield (GY). The plant stand was determined by directly counting the number of plants, with results expressed in plants per meter square (plants m− 2). The root dry mass was determined by collecting three plants per plot, which were washed in running water and subjected to drying in an oven at 60ºC until constant mass, with weighing and values expressed in g plant− 1. Additionally, the relative chlorophyll content was determined in three leaves of three different plants per plot, measured two days after each nitrogen application, with the aid of the Clorofilog Falker equipment. The yield components, number of rows per ear and number of grains per row were obtained from 10 ears randomly collected from each plot, after harvest, by directly counting the components. Grain yield (kg ha− 1) was determined from the harvest of 5.4 m² of the plot, with samples subjected to threshing, cleaning and weighing, with values adjusted to 13% humidity. Additionally, four samples of 100 grains were used to measure the thousand seed weight, with average values expressed in grams and corrected for 13% humidity.
Data were submitted to analysis of variance using the F test (p < 0.05) and the means of treatments were compared using the Scott-Knott test (p < 0.05), with the aid of the Sisvar® software (Ferreira 2014). The data were submitted to regression analysis for the N dose factor and bar graph for the cover crop and inoculation factors, using Sigma Plot 14.0 software.
3 Results and discussion
Analysis of variance for experiment I indicated a significant effect [previous crop (C) x inoculation (I) x N dose] for the variables TSW and GY, followed by double interaction (C x N) for NRE and (I x N) for the RDM. In addition, there was a double interaction (C x I) for the RCC and an isolated effect of N doses for the FPS. For experiment II, there was a significant interaction between the factors (C x I x N) only for the RCC. However, the double interaction (C x N) occurred for RDM, NGR, TSW and GY. Followed by the isolated effect of the inoculation for the variables TSW and GY.
FPS was higher in experiment I when the N dose was 143 kg ha− 1 (Fig. 2 A), resulting in 5.95 plants m− 2, which was 11% higher than the control dose. For experiment II, the isolated effect of the cover crop indicated that forage radish promoted an increase of 8% in FPS compared to black oat, obtaining 6.7 plants m− 2 (Fig. 2B). Nitrogen fertilizer doses can vary depending on the hybrid and history of the area, which converge to a satisfactory FPS, with an increase in biomass and productivity due to higher FPS (Basir et al. 2016; Piazzoli et al. 2021). Additionally, cover crops can help in the process of accumulating organic carbon in the soil and increasing nutrient cycling, improving soil chemical characteristics and increasing N availability (Santos et al. 2014). Therefore, the sowing of the corn crop under forage radish promoted the highest FPS values, which may be associated with the rapid release of macronutrients from the tissue and the straw being in a more advanced decomposition process. Due to the lower lignin content, lignin:N and C:N ratio, promote the stimulation of decomposing microorganisms (Oliveira et al. 2016; Rutan et al. 2022). However, due to the higher C:N ratio found in black oats (23:1), the decomposition and release of nutrients became slower, due to the temporary immobilization of N by microorganisms (Martins et al. 2014), which possibly led to lower FPS. However, the main effect on FPS is probably associated with the physical phenomenon of straw in the soil and the quality of the sowing equipment, which can generate variability in the plant stand as a function of the ability to cut biomass and uniform seed deposition.
For RDM, in experiment I, there was a significant increase in the presence of A. brasilense associated with 150 kg ha− 1 of N (Fig. 3 A), obtaining a value of 85 g pl− 1, with an increase of 13% in relation to non-inoculated plants at the same dose of N. Additionally, for the non-inoculated treatment, the dose of N required to reach the maximum accumulation of RDM was 125 kg ha− 1, with 76 g pl− 1, with reduction for higher doses. In experiment II, the highest RDM means were obtained when sown under forage radish and inoculated, with an average increase of 56% and 36% compared to inoculated black oat and uninoculated radish, respectively (Fig. 3B). Additionally, for the interaction of C x N, it was verified that corn sowing on forage radish (Fig. 3 C) presented maximum technical efficiency for RDM with 139 kg ha− 1, producing roots of 50 g pl− 1. However, on black oat there was no response of the variable, regardless of the dose of N applied, with average root production of 20,08 g pl− 1, a value generally much lower than when on the forage radish. The increase in RDM when inoculated is related to a greater biosynthesis of indole-3-acetic acid (auxins) (Spaepen et al. 2014; Pii et al. 2019; Cassán et al. 2020), which stimulates the production of root hairs, as well as increased root mass and length (Heydari et al. 2018; Xiong et al. 2020). With this, there is a greater root interception and, consequently, greater response of the growing shoot/productivity plant. It was observed that root growth was maximized when the predecessor crop was forage radish (Fig. 3B). This is due to the pivotal roots of this crop acting as soil decompactors, in addition to promoting high production of biomass and N in the tissues, obtaining higher RDM, total length and corn grain yield (Rutan and Steinke 2019; Zhang et al. 2022).
The negative relationship of black oat on corn RDM (Fig. 3B C) was probably due to a lower nutrient release rate, due to the higher C:N ratio compared to forage radish, altering straw decomposition (Balota et al. 2014). Consequently, there is a limitation of N in the initial stages of corn, since the BNF performed by the bacterium can supply only 5 to 18% of the N necessary for plant growth (Cassán et al. 2020). This corroborates the biotic immobilization of N in the body of decomposer microorganisms from residues with higher C:N ratio (Cao et al. 2020) (Fig. 3B C). In addition, inoculation promoted maize plants with higher RDM at higher doses of N (dose of 150 kg ha− 1, Fig. 3 A). This may be related to the increase in root mass promoted by the bacterium, allowing greater use of the applied N (Barbosa et al. 2022), thereby improving the productivity components (Galindo et al. 2020). Better responses were obtained for RDM at doses between 100 and 168 kg ha− 1 (Ordónez et al. 2021), similar to the data found in this research.
The NRE presented significance in experiment I when the plants were submitted to increasing doses of N on black oat residues, obtaining 15.13 rows per ear at the highest dose (Fig. 4 A). However, when corn was sowed on forage radish, no response was evidenced for N doses, with mean values of 14.7 rows per ear (Fig. 4 A). There was no response for the NRE variable regarding treatments in experiment II. On the other hand, the significance for NGR was verified only in experiment II, with nitrogen fertilization response when the crop was sown on black oat, obtaining the highest value of 30 grains row− 1, at a dose of 179 kg ha− 1 of N (Fig. 4B). This was not observed when the crop was sown on forage radish, with average values of 24.6 grains row− 1.
Although the definition of some agronomic parameters is genetically established, management practices can modify their expression in the field. Therefore, the greater response of nitrogen fertilization in plots containing black oat residues (Fig. 4 A and 4B), is a consequence of the high immobilization of N, which can lead to a decline of up to 25% on corn grain yield when without N application (Lourente et al. 2007). On the other hand, cover crops such as legumes or radish can provide high amounts of N to maize plants, with a direct influence on the productivity components of the crop, and there may be no response to increasing doses of N (Valicheski et al. 2020). This is due to the rapid mineralization of nutrients in the straw, resulting in an adequate supply of N in periods of greater demand in the corn crop (Aita et al. 2001).
Relative chlorophyll content under nitrogen doses (A) and interaction for the factors cover crop x inoculation (B) in experiment I. Interaction cover crop x inoculation x nitrogen in experiment II (C). NI: Non-inoculated and IN: Inoculated. *Uppercase compare cover crops and lowercase the inoculation, by Scott-Knott test (p < 0,05)
The RCC values in experiment I showed significance for the N dose and C x I interaction (Fig. 5 A and 5B). There was a quadratic adjustment according to the doses of N applied, with maximum value at the dose of 135 kg ha− 1 (Fig. 5 A). Inoculation with A. brasilense in corn sown on black oat promoted a 6% increase in RCC, compared to the control, which was not observed on forage radish (Fig. 5B). Considering these results, the RCC response to the increase in N dose may be linked to the essentiality of the nutrient in the constitution of the chlorophyll molecule, amino acids and proteins (HIREL et al. 2005), which justifies the correlation between the availability of the nutrient and the largest composite construction of the photosynthetic apparatus. Thus, there is an input of approximately 30% of foliar N for the construction of thylakoid membrane components such as chlorophylls (Ghannoum et al. 2005; Bassi et al. 2018), which may be associated with the best results in the presence of the bacteria on black oat due to greater root absorption capacity and BNF. This is mainly due to the immobilization of N by oat residues when not inoculated.
In experiment II, it was found that the triple interaction C x I x N was significant for RCC. Treatments containing black oat and A. brasilense showed an increasing linear model, obtaining a RCC of 62 at the highest dose of N, with an increase of 17% compared to the absence of N in top dressing. However, when sowing was carried out on forage radish and the presence of A. brasilense, there was no RCC response regardless of the applied N dose (average of 55). When inoculation was not used, a quadratic model for sowing on black oat was verified, with maximum technical efficiency around 156 kg ha− 1 of N. On the other hand, when sowing was carried out on forage radish, an increasing linear model was observed for the variable with RCC of 55 at the highest dose of N (225 kg ha− 1). In general, it was found that the highest RCC values were obtained on black oat residues with Azospirillum brasilense. This may be related to inoculation in the source-sink relationship of the plant, with greater activity of enzymes linked to carbohydrate metabolism, increase in total biomass and crop productivity. This will stimulate greater synthesis of chlorophyll, which is one of the main components of the photosynthetic apparatus (Mekureyaw et al. 2022). In addition, it presents a direct response to nitrogen fertilization under the increase of leaf area and RCC, increasing up to 13% in corn plants inoculated and submitted to 300 kg ha− 1 N (Muller et al. 2016; Galindo et al. 2019).
Considering TSW and GY, a significant C x I x N interaction was observed in experiment I. There was an increasing linear behavior for both variables when the crop was inoculated with A. brasilense and sowing was carried out on black oat, obtaining 434 g (TSW) and 10.775 kg ha− 1 (GY) are calculated at the highest dose of N (Fig. 6 A and 6 C). However, when there was no inoculation, a quadratic model was evidenced for TSW and GY, with maximum technical efficiency close to 220 kg ha− 1 and 263 kg ha− 1, reaching values of 440 g TSW and 10,986 kg ha− 1 GY. The inoculation of corn with A. brasilense on forage radish showed a quadratic adjustment, with maximum technical efficiency obtained at the dose of 174 kg of N ha− 1 TSW and 196 kg of N ha− 1 GY, obtaining respectively 430 g and 10.774 kg ha− 1 (Fig. 6 A and 6 C). However, without inoculation, there was an increasing linear model TSW and second order PG, with values of 430 g and 10.883 kg ha− 1, at doses of 225 and 216 kg of N ha− 1, respectively.
In experiment II, inoculation with A. brasilense increased TSW and GY by 5% and 9%, respectively (Fig. 6E F). In addition, a double interaction (C x N) was observed for both analyzed variables (Fig. 6B and D). It was found that sowing on black oat and the application of 150 kg ha− 1 of N promoted the highest values in TSW (382 g). However, this did not occur for forage radish, which showed a linear increase for TSW with increasing N dose (Fig. 6B). Additionally, it was found that GY was influenced by the type of cover crop and dose applied. The sowing carried out on black oat promoted an increase of 1.873 kg ha− 1 or 22% compared to forage radish at the highest nitrogen applied rate (Fig. 6D). However, in the absence of nitrogen fertilization, corn sowing on black oat reduced TSW and GY by 5% and 14%, respectively. Thus, indicating dependence on the input of nitrogen fertilizers in this system. Additionally, it appears that the productive potential of this area was lower, due to the lower volume and irregularity of rainfall during the crop cycle, mainly in the phenological stage of bolting-flowering (Fig. 1B).
Several authors have reported increased corn productivity ranging from 8.7 to 30% by inoculation with A. brasilense. This is due to greater plant development, increased number of grains per ear and grain mass (Hungria et al. 2010; Oliveira et al. 2018; Galindo et al. 2019; Fernandes et al. 2020). This corroborates the data obtained in experiment II for the effect of bacteria on TSW and GY (Fig. 6E F). Recently, research has shown that plant inoculation can be associated with high doses of N (above 100 kg ha− 1), providing greater absorption and efficiency of use of this nutrient, with an economic return of 7% in relation to the condition without A. brasilense (Galindo et al. 2020; Caires et al. 2021). In experiment I, it was verified that the productivity of treatments inoculated on black oat and forage radish showed maximum technical efficiency under high nitrogen doses 225 kg ha− 1 and 196 kg ha− 1, respectively (Fig. 6B). In this sense, although the BNF performed by the bacterium can partially contribute to the N demand, it can be paralyzed at certain times by the deleterious action of oxygen on the nitrogenase enzyme. This increases the energy expenditure for atmospheric N2 fixation by up to 2.5 times (Pankievicz et al. 2019), which requires the application of nitrogen fertilizers to meet the demand for N. However, it should be noted that the sowing of corn after a grass species presents an increase in N demand to overcome the temporary immobilization of the nutrient, around 30 kg ha− 1 in the present study. This corroborates the results reported by Valicheski et al. (2020), who found that corn yields above 10 t ha− 1 under black oat, required higher doses of N.
Considering the inoculation responses and N doses on black oat, the results reinforce the role of cover crops in the efficiency of seed inoculation with diazotrophic bacteria. Thus, cover crops, especially grasses, are one of the factors with the greatest influence on soil microbial activity, having a direct effect on the number of colony-forming units and microbial respiration (C-CO2) (Kim et al. 2020). Therefore, the immobilization of N in the initial period of decomposition of black oats (47% less NO3− in the soil solution up to 50 days after desiccation) (Drost et al. 2020) may favor the process of root-bacteria association. This is because the greater presence of N available in the system, affects the efficiency of BNF (Saturno et al. 2017).
Cover crops influence soil chemical and physical characteristics, with increased levels of N, P, K, Ca and Mg, reduced N leaching, weed suppression, reduced compaction and biopore construction (Wendling et al. 2016; Cottney et al. 2020; Ogilvie et al. 2021), often increasing grain yield. Due to the higher C:N ratio of black oat, the management of this crop can determine the success of the crop, since the lack of synchrony between the degradation of residues and the release of nutrients from the straw reduces the grain yield between 4 and 13% (Abdalla et al. 2019; Rutan and Steinke 2019), which justifies the linear response of nitrogen fertilization on black oat in both experiments. Thus, the management of nitrogen fertilization in systems that integrate cover plants with a higher C:N ratio require more N to meet the nutritional demand of the plant, especially in the initial phenological stages (Rutan and Steinke 2019; Piva et al. 2021).
4 Conclusion
Inoculation of the corn crop with A. brasilense promotes positive effects on most of the phenological parameters that affect corn productivity, as well as having an influence on the root mass, regardless of the predecessor crop. Thus, inoculation with A. brasilense increased corn yield by between 2 and 9% on average.
Regarding the response to N, greater effects were observed when on black oat residues, due to the immobilization of N by the straw, with better yields obtained at the highest rates of N evaluated, but with a more expressive response up to 150 kg ha− 1. Regarding forage radish residues, there was a lower response to fertilization with N due to the rapid mineralization of this residue in the soil, positively influencing the supply of N to corn cultivated in succession.
References
Abdalla M, Hastings A, Cheng K et al (2019) A critical review of the impacts of cover crops on nitrogen leaching, net greenhouse gas balance and crop productivity. Glob Change Biol 25:2530–2543. https://doi.org/10.1111/gcb.14644
Acosta JAA, Amado TJC, Silva LS et al (2014) Decomposição da fitomassa de plantas de cobertura e liberação de nitrogênio em função da quantidade de resíduos aportada ao solo sob sistema plantio direto. Ciênc Rural 44:801–809. https://doi.org/10.1590/S0103-84782014005000002
Aita C, Basso CJ, Ceretta CA et al (2001) Plantas de cobertura de solo como fonte de nitrogênio ao milho. Rev Bras Ciênc Solo 25:157–165. https://doi.org/10.1590/S0100-06832001000100017
Artyszak A, Gozdowski D (2020) Is it possible to replace part of the mineral nitrogen dose in maize for grain by using growth activators and plant growth-promoting rhizobacteria? Agronomy, 10: 1–10. https://doi.org/10.3390/agronomy10111647
Backer R, Rokem JS, Ilangumaran G (2018) Plant growth-promoting rhizobacteria: context, mechanisms of action, and roadmap to commercialization of biostimulants for sustainable agriculture. Front Plant Sci 9:1–17. https://doi.org/10.3389/fpls.2018.01473
Balota EL, Calegari A, Nakatani AS et al (2014) Benefits of winter cover crops and no-tillage for microbial parameters in a Brazilian Oxisol: A long-term study. Agric Ecosyst and Environ 97:31–40. https://doi.org/10.1016/j.agee.2014.07.010
Bang TC, Husted S, Laursen KH et al (2021) The molecular–physiological functions of mineral macronutrients and their consequences for deficiency symptoms in plants. New Phytol 229:2446–2469. https://doi.org/10.1111/nph.17074
Barbosa JZ, Roberto LA, Hungria M et al (2022) Meta-analysis of maize responses to Azospirillum brasilense inoculation in Brazil: benefits and lessons to improve inoculation efficiency. Appl Soil Ecology 170:1–10. https://doi.org/10.1016/j.apsoil.2021.104276
Basir A, Jan MT, Arif M et al (2016) Response of tillage, nitrogen and stubble management on phenology and crop establishment of wheat. Int J Agric Biol 18:1–8. https://doi.org/10.17957/IJAB/14.0030
Bassi D, Menossi M, Mattiello L et al (2018) Nitrogen supply influences photosynthesis establishment along the sugarcane leaf. Sci Rep 8:1–13. https://doi.org/10.1038/s41598-018-20653-1
Breedt G, Labuschagne N, Coutinho TA (2017) Seed treatment with selected plant growth-promoting rhizobacteria increases maize yield in the field. Ann Appl Biol 171:229–236. https://doi.org/10.1111/aab.12366
Caires EF, Bini AR, Barão LFC et al (2021) Seed inoculation with Azospirillum brasilense and nitrogen fertilization for cereal crops under a continuous no-till system. Agron J 113:560–576. https://doi.org/10.1002/agj2.20488
Cao Y, Zhao F, Zhang Z et al (2020) Biotic and abiotic nitrogen immobilization in soil incorporated with crop residue. Soil & Tillage Res 202:1–6. https://doi.org/10.1016/j.still.2020.104664
Cassán F, Coniglio A, López G et al (2020) Everything you must know about Azospirillum and its impact on agriculture and beyond. Biol Fertil Soils 56:461–479. https://doi.org/10.1007/s00374-020-01463-y
Conab (2022) Acompanhamento da safra brasileira – grãos. https://www.conab.gov.br/info-agro/safras/graos/boletim-da-safra-de-graos, accessed 19 April 2022
Cottney P, Black L, White E et al (2020) The correct cover crop species integrated with slurry can increase biomass, quality and nitrogen cycling to positively affect yields in a subsequent spring barley rotation. Agronomy 10:1–27. https://doi.org/10.3390/agronomy10111760
Comissão de química e fertilidade do solo - CQFS-RS/SC (2016) Manual de calagem e adubação para os Estados do Rio Grande do Sul e de Santa Catarina. 11a ed. Porto Alegre: Sociedade Brasileira de Ciência do Solo - Núcleo Regional Sul
Drost SM, Rutgers M, Wouterse M et al (2020) Decomposition of mixtures of cover crop residues increases microbial functional diversity. Geoderma 361:1–11. https://doi.org/10.1016/j.geoderma.2019.114060
Embrapa (2013) Sistema brasileiro de classificação de solos, 3 edn. [Embrapa], Brasília, p 353
Fanin N, Fromin N, Bertrand I (2016) Functional breadth and home-field advantage generate functional differences among soil microbial decomposers. Ecology 97:1023–1037. https://doi.org/10.1890/15-1263.1
Fernandes VB, Mingotte FLC, Morello OF et al (2020) Agronomic performance of maize under Azospirillum brasilense inoculation and top-dressing nitrogen fertilization. Rev Bras Milho Sorgo 19:1–15. https://doi.org/10.18512/rbms2020v19e1185
Ferreira DF (2014) Sisvar: a Guide for its Bootstrap procedures in multiple comparisons. Ciênc Agrotec 38:109–112. https://doi.org/10.1590/S1413-70542014000200001
Fu W, Wang Y, Ye Y et al (2020) Grain yields and nitrogen use efficiencies in different types of stay-green maize in response to nitrogen fertilizer. Plants 9:1–16. https://doi.org/10.3390/plants9040474
Galindo FS, Buzetti S, Rodrigues WL et al (2020) Inoculation of Azospirillum brasilense associated with silicon as a liming source to improve nitrogen fertilization in wheat crops. Sci Rep 10:1–18. https://doi.org/10.1038/s41598-020-63095-4
Galindo FS, Teixeira-Filho MCM, Buzetti S et al (2019) Maize yield response to nitrogen rates and sources associated with Azospirillum brasilense. Agron J 111:1985–1997. https://doi.org/10.2134/agronj2018.07.0481
Ghannoum O, Evans JR, Chow WS et al (2005) Faster rubisco is the key to superior nitrogen-use efficiency in NADP-Malic enzyme relative to NAD-Malic enzyme C4 grasses. Plant Physiol 137:638–650. https://doi.org/10.1104/pp.104.054759
Hafiz S, Djima A, Adolphe A et al (2015) Biodiversity of local varieties of corn cultivation among farmers in Benin. J Agric Crop Res 3:85–99
Heydari MM, Brook RM, Jones DL (2018) The role of phosphorus sources on root diameter, root length and root dry matter of barley (Hordeum vulgare L.). J Plant Nutr 42:1–15. https://doi.org/10.1080/01904167.2018.1509996
Hirel B, Andrieu B, Valadier MH et al (2005) Physiology of maize II: Identification of physiological markers representative of the nitrogen status of maize (Zea mays) leaves during grain filling. Physiol Plant 124:178–188. https://doi.org/10.1111/j.1399-3054.2005.00511.x
Hungria M, Campo RJ, Souza EM et al (2010) Inoculation with selected strains of Azospirillum brasilense and A. lipoferum improves yields of maize and wheat in Brazil. Plant Soil 331:413–425. https://doi.org/10.1007/s11104-009-0262-0
Kim N, Zabaloy MC, Guan K et al (2020) Do cover crops benefit soil microbiome? A meta-analysis of current research. Soil Biol Biochem 142:1–14. https://doi.org/10.1016/j.soilbio.2019.107701
Koehler-cole K, Everhart SE, Gu Y et al (2020) Is allelopathy from winter cover crops affecting row crops? Agric Environ Lett 5:1–6. https://doi.org/10.1002/ael2.20015
Lorensini F, Ceretta CA, Girotto E et al (2012) Lixiviação e volatilização de nitrogênio em um Argissolo cultivado com videira submetida à adubação nitrogenada. Ciênc Rural 42:1173–1179
Lourente ERP, Ontocelli R, Souza LCF et al (2007) Culturas antecessoras, doses e fontes de nitrogênio nos componentes de produção do milho. Acta Sci Agron 29:55–61. https://doi.org/10.4025/actasciagron.v29i1.66
Martins RP, Comin JJ, Gatiboni LC et al (2014) Mineralização do nitrogênio de plantas de cobertura, solteiras e consorciadas, depositadas sobre um solo com histórico de cultivo de cebola. Rev Ceres 1:587–596. https://doi.org/10.1590/0034-737X201461040020
Mekureyaw MF, Beierholm AE, Nybroe O et al (2022) Inoculation of tomato (Solanum lycopersicum) roots with growth promoting pseudomonas strains induces distinct local and systemic metabolic biosignatures. Physiol Mol Plant Pathol 117:1–10. https://doi.org/10.1016/j.pmpp.2021.101757
Michelon CJ, Junges E, Casali CA et al (2019) Atributos do solo e produtividade do milho cultivado em sucessão a plantas de cobertura de inverno. Rev Ciênc Agrovet 18:230–239. https://doi.org/10.5965/223811711812019230
Morris TF, Murrel S, Beegle DB et al (2018) Strengths and limitations of nitrogen rate recommendations for corn and opportunities for improvement. Agron J 110:1–37. https://doi.org/10.2134/agronj2017.02.0112
Muller TM, Sandini IE, Rodrigues JD et al (2016) Combination of inoculation methods of Azospirilum brasilense with broadcasting of nitrogen fertilizer increases corn yield. Ciênc Rural 46:210–215. https://doi.org/10.1590/0103-8478cr20131283
Ogilvie CM, Ashiq W, Vasava HB et al (2021) Quantifying root-soil interactions in cover crop systems: A review. Agriculture 11:1–15. https://doi.org/10.3390/agriculture11030218
Oliveira ALM, Santos OJAP, Marcelino PRF et al (2017) Maize Inoculation with Azospirillum brasilense Ab-V5 cells enriched with exopolysaccharides and polyhydroxybutyrate results in high productivity under low n fertilizer input. Front Microbiol 8:1–18. https://doi.org/10.3389/fmicb.2017.01873
Oliveira FC, Netto MS, Araujo LS et al(2016) Corn development and production in function of sources of nitrogen fertilizers and doses. Rev Caatinga 29: 812–821, 2016. https://doi.org/10.1590/1983-21252016v29n405rc
Oliveira RP, Lima SF, Alvarez RCF et al (2018) Azospirillum brasilense inoculation and management of fertilizer nitrogen in maize. Braz J Agric 93:347–361
Ordónez RA, Castellano MJ, Danalatos GN et al (2021) Insufficient and excessive N fertilizer input reduces maize root mass across soil types. F Crops Res 267:1–13. https://doi.org/10.1016/j.fcr.2021.108142
Pankievicz VCS, Irving TB, Maia LGS et al (2019) Are we there yet? The long walk towards the development of efficient symbiotic associations between nitrogen-fixing bacteria and non-legumious crops. BMC Biol 17:1–17. https://doi.org/10.1186/s12915-019-0710-0
Pellin DMP, Souza FR, Wendland SB et al (2019) Soil coverage plants preceding maize culture submitted to doses of nitrogen fertilization. Braz J Agric 94:153–165
Piazzoli D, Sapucay MJLC, Prando AM et al (2021) Plant density and nitrogen topdressing of high-altitude main-season corn. Semina: Ciênc Agrár 42:2651–2668. https://doi.org/10.5433/1679-0359.2021v42n5p2651
Pii Y, Aldrighetti A, Valentinuzzi F et al (2019) Azospirillum brasilense inoculation counteracts the induction of nitrate uptake in maize plants. J Exp Bot 70:1313–1324. https://doi.org/10.1093/jxb/ery433
Piva JT, Bratti F, Locatelli JL et al (2021) Use of winter cover crops improves maize productivity under reduced nitrogen fertilization: a long-term study. Bragantia 80:1–11. https://doi.org/10.1590/1678-4499.20200165
Qin Z, Guan K, Zhou W et al (2021) Assessing the impacts of cover crops on maize and soybean yield in the U.S. Midwestern agroecosystems. F Crops Res 273:108404. https://doi.org/10.1016/j.fcr.2021.108264
Ritchie SW, Hanway JJ, Benson GO (1993) How a corn plant develops. Iowa State University of Science and Technology Cooperative Extension Service, Ames
Rutan J, Steinke K (2019) Corn nitrogen management following daikon radish and forage oat cover crops. Soil Sci Soc Am J 83:181–189. https://doi.org/10.2136/sssaj2018.07.0269
Rutan J, Rosenzweig N, Steink K (2022) Soil bacterial community composition following cover crops and corn nitrogen management. Soil Sci Soc Am J 86:51–64. https://doi.org/10.1002/saj2.20336
Santos IL, Caixeta CF, Souza AATC et al (2014) Cover plants and mineral nitrogen: effects on organic matter fractions in an oxisol under no tillage in the cerrado. Rev Bras Ciênc Solo 38:1874–1881. https://doi.org/10.1590/S0100-06832014000600022
Saturno DF, Cerezini P, Silva PM et al (2017) Mineral nitrogen impairs the biological nitrogen fixation in soybean of determinate and indeterminate growth types. J Plant Nutr 40:1690–1701. https://doi.org/10.1080/01904167.2017.1310890
Scott S, Housh A, Powell G et al (2020) Crop yield, ferritin and Fe (II) boosted by Azospirillum brasilense (HM053) in Corn. Agronomy 10:1–12. https://doi.org/10.3390/agronomy10030394
Setiyono TD, Walters DT, Cassman KG et al (2010) Estimating maize nutrient uptake requirements. F Crops Res 118:158–168. https://doi.org/10.1016/j.fcr.2010.05.006
Shin W, Islam R, Benson A et al (2016) Role of diazotrophic bacteria in biological nitrogen fixation and plant growth improvement. Korean J Soil Sci Fert 49:17–29. https://doi.org/10.7745/KJSSF.2016.49.1.017
Silva MP, Arf O, Sá ME et al (2017) Plantas de cobertura e qualidade química e física de Latossolo Vermelho distrófico sob plantio direto. Rev Bras Ciênc Agrár 12:60–67. https://doi.org/10.5039/agraria.v12i1a5424
Skonieski FR, Viégas J, Martin TN et al (2019) Effect of nitrogen topdressing fertilization and inoculation of seeds with Azospirillum brasilense on corn yield and agronomic characteristics. Agronomy 9:1–11. https://doi.org/10.3390/agronomy9120812
Souza EM, Galindo FS, Teixeira filho MCM et al (2019) Does the nitrogen application associated with Azospirillum brasilense inoculation influence corn nutrition and yield? Rev Bras Eng Agríc Ambient 23:53–59. https://doi.org/10.1590/1807-1929/agriambi.v23n1p53-59
Spaepen S, Bossuyt S, Engelen K et al (2014) Phenotypical and molecular responses of Arabidopsis thaliana roots as a result of inoculation with the auxin-producing bacterium Azospirillum brasilense. New Phytol 201:850–861. https://doi.org/10.1111/nph.12590
Szulc P, Barlóg P, Ambrozy-Deregowska K et al (2020) In-soil application of NP mineral fertilizer as a method of improving nitrogen yielding efficiency. Agronomy 10:1–13. https://doi.org/10.3390/agronomy10101488
Usda (2021) https://www.fas.usda.gov/data/world-agricultural-production. Accessed 29 july 2021
Valicheski RR, Cruz SJS, Eger JG et al (2020) Cover crops and nitrogen fertilization - Nutritional aspects and agronomic performance of maize grown in sucession. Rev Bras Milho Sorgo 19:1–16. https://doi.org/10.18512/1980-6477/rbms2020.v19.e1095
Vincent-caboud L, Casagrande M, David C et al (2019) Using mulch from cover crops to facilitate organic no-till soybean and maize production. A review. Agron Sustain Dev 45:1–15. https://doi.org/10.1007/s13593-019-0590-2
Vukicevich E, Lowery T, Bowen P et al (2016) Cover crops to increase soil microbial diversity and mitigate decline in perennial agriculture. A review. Agron Sustain Dev 36:1–14. https://doi.org/10.1007/s13593-016-0385-7
Wendling M, Buchi L, Amossé C et al (2016) Influence of root and leaf traits on the uptake of nutrients in cover crops. Plant Soil 409:419–434. https://doi.org/10.1007/s11104-016-2974-2
Xiong P, Zhang Z, Hallett PD et al (2020) Variable responses of maize root architecture in elite cultivars due to soil compaction and moisture. Plant Soil 455:79–91. https://doi.org/10.1007/s11104-020-04673-3
Zhang Z, Yan L, Wang Y et al (2022) Bio-tillage improves soil physical properties and maize growth in a compacted vertisol by cover crops. Soil Soc Am J 1–14. https://doi.org/10.1002/saj2.20368
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lima, C.S., Ceolin, C., Muller, D. et al. Inoculation with Azospirillum brasilense in corn cultivated on cover crops and nitrogen doses. Symbiosis 87, 237–247 (2022). https://doi.org/10.1007/s13199-022-00870-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13199-022-00870-z