Abstract
Background and aims
Planted forests, established on non-forest lands, play an important role in enhancing terrestrial carbon (C) sequestration. Understanding the changes in soil C, nutrients and stoichiometry in planted forests is important for forest management.
Methods
We conducted a global meta-analysis of changes in C, nitrogen (N), phosphorus (P) and sulfur (S) and their stoichiometry in mineral soils of planted forest across broad climatic zones from 139 papers.
Results
Soil C and N are slightly decreased after forestation on grassland, moderately increased after forestation on cropland, and substantially increased after forestation on barren land. Forestation does not affect total soil P, but the available P is significantly depleted after the forestation of grassland and cropland with N-fixers. Changes in soil nutrients (N, P and S) and shifts in stoichiometry (ratios of C:N, C:P and N:P) are significantly related to soil C dynamics (p < 0.05). Soil C sequestration is the lowest in the boreal zone, and greater under plantation with N-fixing species than under non-fixing species.
Conclusion
Changes in soil C and nutrients after forestation mainly differ to prior land use. Compared with forestation of grassland, forestation of barren land is a more effective approach to enhancing C sequestration.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Planted forests, established on non-forest lands by afforestation or reforestation, are one type of practice that may have an important effect on terrestrial carbon (C) cycle (Pan et al. 2011). Large-scale forestation (including afforestation and reforestation) can sequester substantial amount of CO2 from the atmosphere and then assimilate the C into living biomass and dead organic matter in soils (Silver et al. 2000; Persson et al. 2013), providing an opportunity to offset C emission through land-use change. Better assessments of soil C change in planted forest can reduce the considerable uncertainty in estimating C sequestration and emissions in terrestrial ecosystems, and provide a scientific basis for predicting changes in soil fertility for use by policy-makers in forest management (Johnson and Curtis 2001; Li et al. 2012a). However, the magnitude and direction of soil C changes after forestation varies greatly. For instance, soil C change after forestation of cropland has been found to be inconsistent across geographic and/or climatic zones, ranging from close to zero in the United States (US) (Nave et al. 2013) to +20 % in the top 10 cm in Northern Europe (Bárcena et al. 2014), +50 % in the tropical zone (Don et al. 2011) and +68.6 % in the top 10 cm in China (Song et al. 2014). Soil organic C (SOC) content serves as a proxy for soil organic matter (SOM) with a conversion factor of 2 (Pribyl 2010), and the SOM is a major contributor of plant nutrients and soil fertility (Whitbread 1995). Therefore, quantifying soil C change in planted forests is essential for understanding the global C cycle and the evolution of forest soil fertility.
Nitrogen (N), phosphorus (P) and sulfur (S) are important macronutrients for plant growth and soil C sequestration, and they vary with stand development and global environmental change (Bradford et al. 2008; Oulehle et al. 2011; Cleveland et al. 2013). Altered N, P and S content have strong impacts on C dynamics in forest ecosystems (Oulehle et al. 2011; Goll et al. 2012). The regulatory effects of N, P and S on soil C sequestration in forest ecosystems can be largely explained by three factors. Firstly, soil N and P availability is crucial in controlling net primary productivity (NPP) and C allocation in forest ecosystems (Cleveland et al. 2013; Fernandez-Martinez et al. 2014). Limited amounts of available N and P in soils may constrain the rate of input of organic matter from NPP (Goll et al. 2012; Cleveland et al. 2013). Available S is a less critical limiting factor for plant productivity (Mitchell et al. 1992), but it plays a major role in the structure of proteins and functioning of enzymes in plant metabolism and by its interactions with other elements (Droux 2004). Secondly, soil nutrient availability regulates the decomposition rate of soil C by altering the structure and metabolism of the soil microbial community (Moscatelli et al. 2005; Strickland et al. 2010). Soil nutrient availability also determines the output rate of soil C by regulation of labile C (e.g., dissolved organic matter) released from the litter and mineral layer (Cleveland and Townsend 2006; Borken et al. 2011) and controls the rates of the net humification efficiency for soil organic matter (Kirkby et al. 2013; Kirkby et al. 2014). Thirdly, shifts in the C:nutrient stoichiometry of soil and microbial biomass could regulate the soil C sequestration process by altering the structure of soil microbial communities and the biogeochemical cycle (Griffiths et al. 2012; Sistla and Schimel 2012). For instance, the soil C:N ratio is closely related to the N mineralization rates of soil organic material and C allocation in forest ecosystems (Alberti et al. 2014).
In previous studies, soil C change after afforestation of non-forested land has been evaluated according to different categories of intrinsic edaphic factors (e.g., mineralogy and texture) (Laganière et al. 2010; Don et al. 2011), soil depths (Shi et al. 2013), plant genera (e.g., Pinus and Eucalyptus) (Berthrong et al. 2009), climatic conditions and anthropogenic management (Laganière et al. 2010; Poeplau et al. 2011). These pioneering studies offered a basic understanding of soil C change in forested land and potential determinants at large scales, but several points remain to be further investigated. First, previous studies mainly focused on changes in C and N in forested agricultural soils (i.e. grassland, pasture and cropland) (Laganière et al. 2010; Li et al. 2012a), while the response of soil P, S and stoichiometry, and the effects of forestation on barren land, defined as the land with sparse vegetation cover and eroded soils, have not been thoroughly evaluted. Second, the potential impacts of changes in nutrient dynamics and soil stoichiometry on soil C sequestration in planted forests at large scales are still unknown. Third, the observations included in previous global meta-analysis were mainly located in Europe, the Americas and Oceania (Berthrong et al. 2009; Laganière et al. 2010), while observations in Asia and Africa were rarely included. These shortcomings point to knowledge gaps in our understanding of soil C sequestration, nutrient cycles and stoichiometric variations in planted forest ecosystems.
In this study, we conducted a global meta-analysis of the changes in soil C, N, P, S and their stoichiometry in soils of planted forests spanning the global range of climate conditions, prior land use, plantation age, and tree species. The main objectives of this study were to (1) quantify the changes in C, N, P, S concentrations and their stoichiometry under different categories of explanatory factors across broad climatic zones, and (2) investigate the potential determinants of soil C-sequestration in planted forests at global scales.
Materials and methods
Data collection
Data on C, N, P and S concentrations in soil and microbial biomass following afforestation and reforestation at global scale were collected from studies published between 1993 and 2015 in both English and Chinese (including journal articles and dissertations). In order to reduce publication bias, literature was selected according to the following 4 criteria (Shi and Han 2014): (1) the concentrations of SOC and nutrient of non-forest land (reference) and forested plots must have been assessed; (2) the same stratified method for soil sampling must have been employed for forested and reference plots, and samples must be collected to a depth of at least 10 cm; (3) plantation age and dominant species of forested plots must have been reported; and (4) when the chronosequence approach was employed, the soil type must be consistent and stand age must be no less than 3 years. We considered changes in the concentration of soil C and nutrients in soils of planted forests established on non-forest land under natural conditions. Thus, we excluded data from sampling sites that were fertilized and/or harvested during forest development. Numerical values were extracted from digital figures using GetData Graph Digitizer (Version 2.22, Russian Federation).
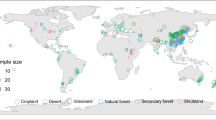
The profile of forest soil mainly consists of the O horizon (forest floor) and mineral horizon, particularly for temperate and boreal forests (Thuille and Schulze 2006). Few properties of the forest floor were monitored in the selected publications. Thus, we focused on changes in properties in the mineral horizon. As soil pH plays a critical role in nutrient availability (Lucas and Davis 1961), we also extracted data on soil pH at forested and reference plots. Finally, a total of 16 soil properties, including soil pH, total organic C concentration (SOC, g kg-1), total N concentration (TN, g kg-1), total P concentration (TP, mg kg-1), total S concentration (TS, mg kg-1), easily available N concentration (AN, sum of ammonia and nitrate, mg kg-1), easily available P concentration (AP, mg kg-1), sulfate concentration (SO 2 −4 , mg kg-1), soil microbial C concentration (mg kg-1), soil microbial N concentration (mg kg-1), soil microbial P concentration (mg kg-1), and the C:N:P:S stoichiometry (mass ratios of soil C:N, C:P, C:S, N:P and C:N of microbial biomass, unitless) in the topsoil depth (within 30 cm depth) were compiled. Because soil element (C, N and P) concentrations are highly influenced by sampling depth (Jackson et al. 2000), values from shallow sampling depths (<10 cm) were excluded. If an element concentration in the topsoil was measured at different sampling depths, a depth-weighted mean was calculated. The fertilization by atmospheric N deposition can increase C stock in forest biomass and the upper mineral soils (de Vries et al. 2009; Pregitzer et al. 2008). However, we did not consider the influences of N deposition on soil C sequestration after forestation due to the lack of annual N deposition rate during the stand development at sampling sites. Then, a final dataset on the response of soil properties to afforestation and reforestation was compiled, including detailed information on site location, plantation age, climate factors (including mean annual precipitation (MAP, mm) and mean annual temperature (MAT, °C), experimental design, soil sampling depths and repeated sampling from 139 published studies from 1993 to 2015 (Fig. 1; Table S1).
Data classification
Both afforestation and reforestation refer to establishment of forest on non-forest land in most definitions (Lund 1999; IPCC 2000). The distinction between afforestation and reforestation is determined by whether the land supported trees during the past X (e.g., 30, 50) years (reforestation) or not (afforestation) from the perspective of land-use and land-cover change (IPCC 2000; Verchot et al. 2007). Nevertheless, the definition of reforestation has different implications for different countries and organizations (Lund 1999; IPCC 2000). For example, from a forestry management perspective, the definition of reforestation includes regeneration after harvesting (IPCC 2000), however, it does not reflect a change of land-use designation. Thus, we defined reforestation activities as the conversion of non-forest land to planted forest in this study. Since the SOC stock after deforestation decreased rapidly and became steady after 10 years (Poeplau et al. 2011), we assumed that changes in the soil properties at the reforested sites were quite similar to the afforested sites. This is also supported by a global meta-analysis (Liao et al. 2012). Therefore, we used the term “forestation” to include both afforestation and reforestation. According to prior land use, the type of planted forest was categorized as forest established on grassland (FG), forest established on cropland (FC) and forest established on barren land (FB).
Tree species, particularly the N-fixing properties of species, has been found to determine the direction and magnitude of changes in soil C and other nutrients after forestation (Resh et al. 2002). Tree species of the genus Eucalyptus are widely planted on non-forested land, and their impacts on soil nutrients have been widely investigated (Berthrong et al. 2012; Eclesia et al. 2012). Thus, the tree species were categorized as Eucalyptus, broadleaf trees excluding Eucalyptus and N-fixing species, coniferous trees, mixed forests including both broadleaf and coniferous trees, N-fixing species and others (like bamboo species) in plantations (Marin-Spiotta and Sharma 2013).
Generally, the default time period for soil C stock changes after land-use change is 20 years (IPCC 2006; Don et al. 2011). Furthermore, the mean site age where most soil properties were measured is close to 20 years (Table 1). Thus, plantation age was divided into < 20 years (younger plantation, mean, 10 years; range, 1–19 years) and ≥ 20 years (older plantation, mean, 37 years; range, 20–200 years). Climatic zone has a significant influence on soil C and N change after forests have been established on non-forested land (Laganière et al. 2010; Li et al. 2012a). In order to compare the response of soil C and nutrients to forestation spanning the global range of climate conditions, we adopted the climatic zone classification in the IPCC Good Practice Guidance for Land Use, Land-Use Change and Forestry (LULUCF) (IPCC 2006). The IPCC classification distinguishes 8 climatic zones according to temperature and moisture gradients (IPCC 2006). However, soil sampling sites were unevenly distributed across the climatic zones (e.g., sampling sites located in the tropical moist zone, tropical dry zone and cool temperate zone with dry climate accounting for 2, 4 and 31 % of total sampling sites, respectively). This uneven distribution may bias the results. In order to compare our estimates with other studies, we classified the climatic zones into 4 types across a mean annual temperature gradient: tropical (MAT > 18 °C), warm temperate (10 °C < MAT < 18 °C), cool temperate (4 °C < MAT < 10 °C) and boreal zones (MAT < 4 °C) (IPCC 2006; Laganière et al. 2010).
Data analysis
Main calculations
The soil element stock in a fixed layer i were calculated using equation 1,
where C i is soil element stock (Mg ha−1) and S i is soil element concentration (g kg−1) for i sampling layer, respectively; γ i is the bulk density (g cm−3) and h i is thickness (cm) of soil layer (Yang et al. 2007).
Soil bulk density estimates are critical for calculations of C i , but many studies did not report it. We used a logarithmic linear function γ = -0.115 × ln(SOC) + 1.17, which was established with the reported values of soil bulk density (γ) and SOC concentration from published literatures, to estimate missing soil bulk density. More details about this empirical function are available in Shi et al. (2013). In order to compare our estimates with previous studies, only the SOC and TN stocks were calculated.
In our preliminary data set, soil sample depth varied among the studies between 10 and 30 cm. In order to make comparable estimates of SOC and TN pool change to the same sample depth, the SOC and TN stock changes with irregular sample depths (h) were adjusted to those of the top 20 cm of the sample using methodology adopted by Yang et al. (2011) and Li et al. (2012a) (equations 2 and 3).
where Y is the cumulative proportion of the SOC (or TN) stock from the soil surface to depth h (cm);βis the relative rate of decrease in the soil C (or TN) stock with soil depth (0.9786 for SOC and 0.9831 for TN, respectively) (Jobbágy and Jackson 2000; Jobbágy and Jackson 2001); C20 is the expected soil C (or TN) stock adjusted to the top 20 cm soil layer at a specific site; h is the original soil depth available in individual studies (cm); C h is measured SOC (or TN) stock at sample depth h (cm) at a specific site.
Then the absolute change in SOC (or TN) stock in the top 20 cm depth (ΔC j , Mg ha-1) for j site was calculated with equation (3).
C ej is the SOC (or TN) stock of afforested sites (Mg ha−1), C cj is the SOC (or TN) stock of the j observation for control sites (Mg ha−1).
Meta analysis
The soil properties in the upper mineral soil under forestation were analyzed in the meta-analysis. The natural logarithm of the response ratio (R +) was employed to determine the effect size (Hedges et al. 1999).
where X e and X c are the averages of a specific variable in forested and control sites, respectively. The values of effect size which were outside 3 standard deviations of the mean of each soil property were considered as outliers and discarded according to Pauta criterion. To include as many studies as possible, an unweighted meta-analysis was used (Adams et al. 1997). The mean effect size (lnR +) was calculated, and 95 % confidence intervals (CI) were generated using a bootstrapping procedure (4, 999 iterations) on Meta-win software (Rosenberg et al. 2000). We calculated the mean effect size for each category (type of previous land use, climatic zone, plantation age and plant species), and tested the significance of differences among the various levels using a random model. For each categorical group, total heterogeneity (Q t ) was calculated as the sum of within group heterogeneity (Q w ) and between group heterogeneity (Q b ) (Hedges et al. 1999). If the Q b is significant according to the Q-statistic, the response ratios are considered to be significantly different among the various levels (Liu and Greaver 2009).
The percentage of each response variable (y) was estimated using Formula (5) (Liao et al. 2012).
If the 95 % confidence interval (CI) of y did not overlap zero, then the percentage difference in soil properties change is considered to be significantly affected by forestation (Liao et al. 2012).
To explore the potential determinants of SOC changes in forested land, statistical analysis was performed in two steps. First, we ran linear regressions between the effect size of SOC [Ln(R soc )] and the effect size of other soil properties to test the effects of internal factors (including changes in soil pH, nutrients and stoichiometry) on soil C dynamics using SAS REG procedures (Release 9.1, SAS Institute Inc., Cary, NC, USA, 2004). Post hoc power analysis, a well-established statistical tool with which to overcome errors that arise from significant changes in heterogeneous soils due to insufficient replication (Wang et al. 2011), was performed with each linear regression model using SAS POWER procedures (α = 0.05). The post hoc power analysis uses the obtained sample size and effect size to detect the probability of getting a significant linear slope between effect size of SOC concentration and other soil properties at a given p < 0.05 of the data. Secondly, a general linear model (GLM) was run to test the effects of external factors (prior land-use, tree species, climatic zones as categorical variables, MAP, MAT and plantation age as continuous variables) on the effect size of soil C and nutrients that strongly correlated with soil C. Only significant effects were included in the final model (p < 0.05). The GLM analysis was also performed to determine absolute changes in soil C and TN stock, respectively.
Results
General Changes in soil pH, C, N, P, and S and stoichiometric ratios
The changes in soil properties after forestation in individual studies varied from negative to positive (Table 1). Significant values of the Q b test indicated that prior land use was the most important factor influencing changes in the investigated variables after forestation (Table 1). The meta-analysis showed that mean soil pH was decreased by 0.34 units for FC and 0.33 units for FG (p < 0.05), respectively (Table 2). The mean SOC stock was changed by +26.8 % in FC soils (p < 0.05), by +50.6 % in FB soils (p < 0.05), but no change in FG soils (–2.6 %, p > 0.05). TN stock was significantly changed by -12.6 % for FG, +16 % for FC, and +37.8 % for FB (p < 0.05). The changes in SOC and TN concentrations are quite similar to their corresponding changes in stocks (Table 2). TP concentration was not changed under any plantation type (p > 0.05). TS concentration changed significantly by -15.6 % for FG (p < 0.05).
Regarding soil nutrient availabilities, inorganic N concentration was increased by +48.1 % in FB soils (p < 0.05). P availability was significantly changed by -23.6 % in FC soils (p < 0.05), by +22.2 % in FB soils (p < 0.05), but not changed in FB soils (p > 0.05). Sulfate was increased by +67.5 % in FG soils (p < 0.05). The limited available data showed that there was a significant change in microbial biomass C: -25.4 % for FG (p < 0.05), +32.1 % for FC (p < 0.05), and +160.4 % for FB (p < 0.05).
Regarding soil stoichiometry, the change in C:N ratio ranged from +7.7 % in FC soils to +9.7 % in FG soils and +15 % in FB soils (p < 0.05). A significant increase in C:P ratios occurred in FC (+52.3 %) and FB (+58.5 %) soils (p < 0.05). The limited available data shows that C:S and N:P ratios were not changed in FG and FC soils (p > 0.05), but the N:P ratio increased significantly by +38.3 % in FB soils (p < 0.05). The C:N ratio of microbial biomass increased significantly by +50.4 % in FG soils and by +47.2 % in FB soils (p < 0.05), but was not significantly changed in FC soils (p > 0.05) (Table 2).
Changes in soil pH, C, N, P, and S and stoichiometric ratios under sub-categorical variables
The range of changes in some soil properties following forestation varied under each level of categorical variable (Figs. 2, 3 and 4; Tables S2–S4). For FG, differences in changes in soil TP, TS and sulfate between older and younger plantations were significant (p < 0.05) (Fig. 2a). For FC, the increases in soil C, TN, AN, C:N ratio were larger in older forests than in younger stands (Fig. 2b) (p < 0.05). For FB, except for soil C:N ratio, changes in other soil indices between older and younger plantations were irun nsignificant (p > 0.05) (Fig. 2c). The responses of investigated variables across climatic zones varied depending on prior land use (Fig. 3; Table S3). Change in SOC stock after forestation on grassland was -11 % in the boreal zone in FG soils (p < 0.05) (Fig. 3a). There was a higher level of SOC accumulation in FC soils in the tropical zone (+50.7 %) than in the boreal zone (+15.1 %) (p < 0.05) (Fig. 3b). In FB soils, levels of SOC accumulation in the boreal zone were significantly lower than in other climatic zones (p < 0.05) (Fig. 3c). In FC soils, increases in SOC and N availability in plantations with N-fixing species were significantly greater than in plantations with broadleaf trees (p < 0.05) (Fig. 4b). Soil P availability in plantations with N-fixing species was significantly decreased by -34.1 % for FG and -31.9 % for FC (p < 0.05) (Fig. 4; Table S4).
Percentage difference in soil properties after forestation on (a) grassland or pasture (FG), (b) cropland (FC) and (c) barren land (FB) at different stand ages. The bars show the mean and 95 % CI (Table S2) and the values next to the bars indicate the sample size
Percentage difference in soil properties after forestation on (a) grassland or pasture (FG), (b) cropland (FC) and (c) barren land (FB) of different climate types. The bars show the mean and 95 % CI (Table S3) and the values next to the bars indicate the sample size
Percentage difference in soil properties after forestation on (a) grassland or pasture (FG), (b) cropland (FC) and (c) barren land (FB) for different tree species. Percentage difference under mixed forests and bamboo species were not included in this figure due to limited observations. The bars show the mean and 95 % CI (Table S4) and the values next to the bars indicate the sample size
Potential determinants controlling soil C sequestration after forestation
Soil C change following forestation was strongly related to changes in soil TN, AN, C:N ratios in soil and microbial mass, C:P ratio, N:P ratio and the C and N in soil microbial biomass with a high power value (Power > 0.80) (p < 0.01), but was not related to changes in soil pH, available P or sulfate (p > 0.05) (Fig. 5).
Relationships among effect size of the main soil properties and SOC concentration in planted forest soils. The abbreviations pH (a), TN (b), TP (c), AN (d), AP (e), TS (f), Sulfate-S (f), C:N (g), C:P (h), N:P (i), Mc (j), Mn (k), Mc:Mn (l) are soil acidity, total soil N concentration, total soil P concentration, soil inorganic N concentration, soil inorganic P concentration, soil sulfur concentration, soil sulfate concentration, soil C/N ratio, soil C/P ratio, soil N/P ratio, soil microbial C concentration, soil microbial N concentration, C/N ratio in soil microbial biomass, respectively. Solid lines are fit by an equation with p < 0.05, and dashed lines denote the equation with p > 0.05. R2 is the coefficient of determination for linear regression models, and power is the probability of getting a significant linear slope at p < 0.05 for the correlation analysis. The least significant number (LSN) indicates the number of observations required to produce a specific p value < 0.05 given that the data have the same form
Consistent with the Q b test in meta-analysis, prior land use is the most important predictor of absolute and relative changes in soil C and TN stocks in forested land (Table 3). The values of parameters in the general linear model show that across climatic zones, forested soils in the boreal zone have the lowest ability to sequester C. The relative changes in soil C and TN stocks under plantation with N-fixing species are both greater than plantations with non-N fixing species (Table ). Temperature has a positive effect on the absolute and relative changes in soil TN stock (p < 0.01). In contrast, precipitation has a general negative effect on the relative changes in soil C and TN stocks in forested ecosystems (p < 0.01) (Table 3). The precipitation also has a negative effect on the relative changes in AN concentration and soil microbial C concentration that strongly correlated with soil C in forested ecosystems (p < 0.01) (Table S5). The absolute and relative changes in Soil C and TN stocks increased with plantation age (p < 0.01) (Table 3).
Discussion
Response of soil C to forestation
The response of soil C to forestation in the light of land-use history preceding forestation has been described in previous studies (Laganière et al. 2010; Li et al. 2012a; Bárcena et al. 2014). The mean SOC stock change in the current study was -2.6 % after forestation on grassland, which is comparable to global average of -6.7 % estimated by Berthrong et al. (2009) and -8 % in northern Europe (Bárcena et al. 2014), but significantly lower than the +17.5 % estimated after conversion of grassland to secondary forest in the tropical region (Don et al. 2011) and +3 to +10 % in a previous global meta-analysis (Laganière et al. 2010). For forestation of cropland, the mean SOC stock change in the current study was +26.8 %, which is significantly higher than the estimates of no change in the US (Nave et al. 2013), and is close to +20 % estimated for northern Europe (Bárcena et al. 2014) and +18 to +26 % in a previous global meta-analysis (Guo and Gifford 2002; Laganière et al. 2010), but much lower than the +68.6 % estimated for the top 10 cm in China (Song et al. 2014). For forestation of barren land, the mean SOC stock change was +50.6 %, which is comparable to the +50 % estimated for northern Europe (Bárcena et al. 2014), and within the range of +37 % for natural barren land to +173 % for barren land after industrial activities in the US (Nave et al. 2013). According to the above comparisons, the estimates of soil C change following forestation in our study are slightly higher (for FC) or lower (for FG) than previous global estimates.
The differences between the estimates of SOC change in our study and the values in previous global meta-analysis could be mainly explained by two factors. First, the inclusion of more data sources and data sources from more varied areas. The data sources in previous global meta-analysis were mainly located in Europe, the Americas and Oceania, and rarely included observations from developing countries in Asia and Africa (Berthrong et al. 2009; Laganière et al. 2010). Our data set was compiled by integrating the observations collected in previous global meta-analysis and the latest reported observations, particularly in Asia and Africa. Second, a more detailed categorical variable of data classification was used. We have added the observations in forested barren land which with sparse vegetation cover and eroded soils, and analyzed the changes in soil C and nutrients under detailed categorical variables of climatic zones and tree species. In contrast, the prior land use was classified as grassland/pasture and cropland in previous global meta-analysis (Berthrong et al. 2009; Laganière et al. 2010; Shi et al. 2013).
Determinants of soil C sequestration in forested land
Nutrients
The soil C sequestration process is coupled with nutrient cycles in terrestrial ecosystems (Goll et al. 2012; Cleveland et al. 2013). To sequester C into soil organic matter requires sufficient amounts of N, P and S (Kirkby et al. 2013; Kirkby et al. 2014), as is shown for planted forest ecosystems by a linear relationship between soil C change and soil nutrient (TN, TP, TS and AN) changes (Fig. 5b, c, f and d respectively). The content of soil microbial biomass is regarded as a powerful indicator of soil fertility and biological quality (Jenkinson et al. 1990; Li et al. 2012b). Soil C change is also strongly and positively correlated with the C and N concentrations in microbial biomass (Fig. 5j and k respectively), indicating soil fertility is the critical internal factor determining soil C sequestration after forestation (Fernandez-Martinez et al. 2014).
Prior land use
The general linear model showed that the prior land use is the most important predictor of changes in soil C and nutrients after forestation (Table 3). The parameter values of prior land use showed that soil C sequestration potential is significantly higher in soils of forested barren land and cropland than in soils of forested grassland (Table 3). Concentrations of soil C, TN and microbial biomass C were significantly increased after forestation of barren land, but reduced or no change after forestation of grassland (Table 2). The different responses of soil C to forestation with prior land use could be explained by two factors:
First, the soils in barren land prior to forestation generally have lower organic carbon level than grassland (Fig. 6). The initial C stock in mineral soils was both negatively correlated with the relative and absolute SOC accumulation in forested land (Shi et al. 2013; Deng et al. 2014), which could be explained if the high initial SOC is due to large amounts of still recognizable plant remains in the soil that are easily decomposed due to the soil disturbances in process of site preparation, tree planting, etc. (Jandl et al. 2007).
Secondly, the barren land that was forested was mainly located in fragile areas like the Loess Plateau in China and subtropical regions with relatively poor vegetation coverage, which are prone to lose soil C and nutrients through soil erosion by water and wind (Fu et al. 2010). When the forest was established on barren land, the vegetation cover increased rapidly and soil erosion was reduced (Tang et al. 2010). The restoration of barren land can also enhance the rates of nutrient mineralization in infertile areas (Davis 1998) and increase soil C stability by forming macro-aggregates through mycorrhizal associations with plant roots and other soil microbes (Six et al. 2006). In contrast, conversion of grassland to forestland will reduce organic matter input during the early years (Don et al. 2009), and increase C decomposition of recognizable plant remains (generally high in grasslands) during site preparation resulting in an over reduction in total soil C. Therefore, we suggest that forestation of barren land is a more effective approach to enhancing C sequestration and soil fertility compared with forestation of grassland.
Climatic zones
Climatic zone has a significant influence on soil C changes in forested land (Tables 1 and 3). The values of parameters in the general linear model for both absolute and relative changes in SOC indicated that across climatic zones, mineral soils in the boreal zone after forestation have the lowest ability to sequester C. The lower C sequestration ability in the boreal zone could be explained by lower C input after forestation and the higher soil C stock before forestation. We suggest that the forested soil receive less organic matter input in the boreal zone because trees grow slowly under cold conditions (Laganière et al. 2010). The initial C stock in mineral soils in the boreal zone, particularly for FG and FB, is relatively higher than in other climatic zones (Fig. 6). The higher initial soil C stock will increase the risk of soil C loss after planted forest establishment due to the soil disturbance (Jandl et al. 2007). Additionaly, we found a positive effect of temperature on soil N accumulation, indicating a lower N accumulation in forested soil in the boreal zone than in the temperate and tropical zones in this study (Table 3). Thus, the inefficient N accumulation in cold environments could also be associated with lower soil C sequestration ability in the boreal zone, because of the strong relationship between soil C and N in forested land (Li et al. 2012a).
Precipitation
Precipitation has a generally negative effect on relative changes in concentration of C in soil and microbial biomass, total N and labile N in forested ecosystems (Table 3 and Table S5), indicating the relatively greater ability to accumulate C and nutrients in drier sites than in wetter sites. Generally, losses of soil N can be attributed to uptake by plants, surface water runoff, and leaching from topsoil to deeper soils (Davis 1998; Lorenz and Lal 2010). The negative impacts of increasing precipitation on soil C and N accumulation in forested soils might be attributed to increasing soil erosion from surface runoff, leaching into deep soils, and denitrification of N in high-precipitation regions (Kirschbaum et al. 2008). A negative impact of increasing precipitation on relative change in SOC was also found in a meta-analysis of afforestation in the US (Nave et al. 2013) and in extensive field investigations of soil C and N change after afforestation under Eucalyptus planted on grassland in South America (Berthrong et al. 2012; Eclesia et al. 2012). It should be noted that this negative impact of increasing precipitation only affected relative soil C and N change (lnR), rather than absolute change (e.g. Mg ha-1). If the changes in soil C and N are expressed in an absolute terms, precipitation might have a positive effect on soil C and N accumulation due to greater plant productivity and fine soil texture in wetter regions (Deng et al. 2014).
N-fixing species
As in previous studies (Resh et al. 2002; Binkley 2005), we found that capacities to accumulate C and N in plantations with N-fixing species were relative higher than in pure plantations without N-fixing species (Table 3; Fig. 4). N-fixing species can substantially increase the amount of available N in the soil via biological N-fixation, which can moderate soil N limitation for plant growth and reduce microbial respiration rates (Resh et al. 2002; Fisk et al. 2015). However, there was a significant depletion of available P in soils after forestation on grassland and cropland with N-fixing species (Fig. 4a and b), which has not been reported in previous studies (Resh et al. 2002; Shi et al. 2015). Plantations with N-fixing species require high P for nodule development and growth as well as nitrogenase activity (Binkley et al. 2000; DeLuca et al. 2002; Vitousek et al. 2002), and they can increase P cycling through elevated soil phosphatase activity (Zou et al. 1995; Mitchell and Ruess 2009). It has been demonstrated that the amount of N fixed by N-fixing species is controlled by levels of P supply under elevated atmospheric CO2 concentration (Edwards et al. 2006; Lam et al. 2012). Inorganic P availability can ultimately determine soil C sequestration by directly controlling N availability in soils under N-fixing species and plant biomass (Augusto et al. 2013). Decreased available P under stands with N fixing species could also result from available P being converted into SOM once the N limitation in stands without N fixers was reduced by including N fixers. Thus, we propose that decreased P availability might be a key limiting factor for further C sequestration after forestation with N-fixing species.
Limitations and uncertainties
First, the forest floor (O horizon) is a crucial part of the integrated soil profile (Thuille and Schulze 2006), playing an important role linking the soil nutrient biogeochemical cycle in forest ecosystems. Whether the C and nutrients contained in the forest floor are included or not determines the direction and magnitude of changes in C and nutrients in the whole soil profile of forested ecosystems (Richter et al. 1999). However, most studies did not include changes in C and nutrients on the forest floor after forestation, so relevant data were not available for this meta-analysis. Omitting of C sequestration in forest floor may underestimate the real soil C sequestration in forested land (Richter et al. 1999; Shi et al. 2013), particularly for the older plantation in the boreal zone and cool temperate zone if the soil C change was quantified using absolute terms (Mg C ha-1 or Mg C ha-1 yr-1). The C sequestered in forest floor contributed to 34 % of the total soil C sequestration of forested cropland in a global data synthesis (Shi et al. 2013). Therefore, more efforts are needed to quantify the C sequestered in forest floor and increase our knowledge about the role of interactions between the forest floor and the adjacent mineral soil layer in controlling C and nutrient cycles in planted forests.
Second, using both absolute and relative changes in soil C have advantages and shortages in quantifying the soil C change in forested land. In terms of absolute change, soil sampling depths are as important as soil C concentration in calculating soil C stock (equations 1 and 3) but they are inconsistent among different authors. In order to make comparable estimates, the data of absolute changes in soil C stock with irregular sample depths need to be transformed into ones with the same sample depths. The SOC stock changes were transformed to those of the top 20 cm of the sample using the methodology that commonly used in previous meta- analysis (equations 2 and 3) (Yang et al. 2011; Li et al. 2012a; Deng et al. 2014). However, it should be noted that this transformation would introduce errors and uncertainties in the estimates of SOC stock changes. The empirical equations used in the methodology for different depth transformation are about how soil C stock distributes with varied depth at large scales (Jobbágy and Jackson 2000; Jobbágy and Jackson 2001). To estimate the soil C stock at depth in forest lands is one thing, but to predict the soil C stock change in forested lands with varied depths is another. In other words, these empirical equations might be abused in estimating soil C stock change in the soil profile and/or in deep layers, assuming that the soil C change in the forested land decreases asymptotically with the increasing sample depths using a fixed coefficient β(equation 2). However, this assumption is never validated by the field observations. Observations from a nearly 50 years reforested plot with loblolly pine in the subtropical southeastern US showed that C concentration was significantly increased in surface soils (0–15 cm), with no change in sub-surface soils (15–35 cm), but significantly decreased in subsoils (35–60 cm) (Mobley et al. 2015). Accordingly, it is better to assess the soil C change in whole-profile in forested land rather than that in the upper mineral soils when using the absolute change terms.
In terms of relative change, it is less seriously affected by the soil sample depths but lacking consideration of C sequestration in forest floor (no reference available) and the pre-forestation soil C content. This limitation would be proved by the results from the linear regression analysis between the pair-data of relative and absolute SOC stock change in each climatic zone (Fig. 7). The absolute SOC stock changes are all strongly correlated to the corresponding relative changes, but the slopes are significantly differed to climatic zones [e.g., slopes for boreal zone is 41.5, which is significantly higher than 18.6 for tropical zone and 20.7 for cool temperate zone (p <0.05)] (Fig. 7). It is obviously that the amounts of soil C sequestration across climatic zones are not same when the relative changes of soil C are equal. The greater slope for boreal zone is mainly due to the higher initial soil C stock before forestation (Fig. 6). Thus, we deduced that the relative change terms might be a precise index to quantify soil C change in a climatic zone scale, rather than a robust index to quantify the ability of C-sequestration in forested land at global scales due to no consideration of pre-forestation soil C.
Third, the geographic distribution of sampling sites was uneven, which may increase the uncertainties of our estimates to predict soil C dynamics and soil fertility change in some regions. Although we have used more observations than previous studies, sampling sites in Africa and in the boreal zone (e.g. Russia and Canada) were much more rare than observations in other regions. Furthermore, changes in S have rarely been investigated in planted forest soils, which limits our ability to quantify changes in soil S and the C:S ratio after forestation.
Conclusions
The prior land use is the most important predictor of changes in soil C and nutrients after forestation. Forest established on barren land has a larger capacity to accumulate C and nutrients in soils than forest established on grassland. Soil C dynamics in forested soils are affected by internal factors (i.e. soil N, P, S and the C:N ratios of soil and microbial biomass) and by external factors (i.e. climatic zone, tree species and precipitation). The C sequestration ability of mineral soils is the lowest in the boreal zone and greater under N-fixing plantations than under non-fixing species. However, the significant reduction of P availability after the forestation of grassland and cropland with N-fixers may further limit soil C sequestration. Across the precipitation gradients, the relative changes in soil C and N are negatively correlated with increasing precipitation. We recommend that future research should add more measurements of C sequestered in forest floor, investigate the C change in whole soil profile and collect more the data from Africa as well as boreal zone in order to reduce the uncertainty of the estimates in this study.
References
Adams DC, Gurevitch J, Rosenberg MS (1997) Resampling tests for meta-analysis of ecological data. Ecology 78:1277–1283
Alberti G, Vicca S, Inglima I, Belelli-Marchesini L, Genesio L, Miglietta F, Marjanovic H, Martinez C, Matteucci G, D’Andrea E, Peressotti A, Petrella F, Rodeghiero M, Cotrufo MF (2014) Soil C:N stoichiometry controls carbon sink partitioning between above-ground tree biomass and soil organic matter in high fertility forests. Iforest-Biogeosci Forest 8:195–206. doi:10.3832/ifor1196-008
Augusto L, Delerue F, Gallet‐Budynek A, Achat DL (2013) Global assessment of limitation to symbiotic nitrogen fixation by phosphorus availability in terrestrial ecosystems using a meta‐analysis approach. Global Biogeochem Cycles 27:804–815
Bárcena TG, Kiær LP, Vesterdal L, Stefánsdóttir H, Gundersen P, Sigurdsson B (2014) Soil carbon stock change following afforestation in Northern Europe: a meta‐analysis. Glob Change Biol 20:2393–2405
Berthrong ST, Jobbágy EG, Jackson RB (2009) A global meta-analysis of soil exchangeable cations, pH, carbon, and nitrogen with afforestation. Ecol Appl 19:2228–2241
Berthrong ST, Pineiro G, Jobbagy EG, Jackson RB (2012) Soil C and N changes with afforestation of grasslands across gradients of precipitation and plantation age. Ecol Appl 22:76–86
Binkley D (2005) How nitrogen-fixing trees change soil carbon. In: Binkley D, Menyailo O (eds) Tree species effects on soils: implications for global change. Springer, Dordrecht, pp 155–164
Binkley D, Giardina C, Bashkin MA (2000) Soil phosphorus pools and supply under the influence of Eucalyptus saligna and nitrogen-fixing Albizia facaltaria. Forest Ecol Manag 128:241–247
Borken W, Ahrens B, Schulz C, Zimmermann L (2011) Site‐to‐site variability and temporal trends of DOC concentrations and fluxes in temperate forest soils. Glob Change Biol 17:2428–2443
Bradford M, Fierer N, Reynolds J (2008) Soil carbon stocks in experimental mesocosms are dependent on the rate of labile carbon, nitrogen and phosphorus inputs to soils. Funct Ecol 22:964–974
Cleveland CC, Townsend AR (2006) Nutrient additions to a tropical rain forest drive substantial soil carbon dioxide losses to the atmosphere. Proc Natl Acad Sci U S A 103:10316–10321
Cleveland CC, Houlton BZ, Smith WK, Marklein AR, Reed SC, Parton W, Del Grosso SJ, Running SW (2013) Patterns of new versus recycled primary production in the terrestrial biosphere. Proc Natl Acad Sci U S A 110:12733–12737
Davis M (1998) Soil impacts of afforestation in the high country. N Z For 42:34–38
de Vries W, Solberg S, Dobbertin M, Sterba H, Laubhann D, Van Oijen M, Evans C, Gundersen P, Kros J, Wamelink G (2009) The impact of nitrogen deposition on carbon sequestration by European forests and heathlands. Forest Ecol Manag 258:1814–1823
DeLuca TH, Zackrisson O, Nilsson M-C, Sellstedt A (2002) Quantifying nitrogen-fixation in feather moss carpets of boreal forests. Nature 419:917–920
Deng L, Gb L, Shangguan Z (2014) Land‐use conversion and changing soil carbon stocks in China’s ‘Grain‐for‐Green’Program: a synthesis. Glob Change Biol 20:3544–3556
Don A, Rebmann C, Kolle O, Scherer-Lorenzen M, Schulze E-D (2009) Impact of afforestation-associated management changes on the carbon balance of grassland. Glob Change Biol 15:1990–2002. doi:10.1111/j.1365-2486.2009.01873.x
Don A, Schumacher J, Freibauer A (2011) Impact of tropical land-use change on soil organic carbon stocks—a meta-analysis. Glob Change Biol 17:1658–1670. doi:10.1111/j.1365-2486.2010.02336.x
Droux M (2004) Sulfur assimilation and the role of sulfur in plant metabolism: a survey. Photosyn Res 79:331–348. doi:10.1023/b:pres.0000017196.95499.11
Eclesia RP, Jobbagy EG, Jackson RB, Biganzoli F, Pineiro G (2012) Shifts in soil organic carbon for plantation and pasture establishment in native forests and grasslands of South America. Glob Change Biol 18:3237–3251. doi:10.1111/j.1365-2486.2012.02761.x
Edwards EJ, McCaffery S, Evans JR (2006) Phosphorus availability and elevated CO2 affect biological nitrogen fixation and nutrient fluxes in a clover‐dominated sward. New Phytol 169:157–167
Fernandez-Martinez M, Vicca S, Janssens IA, Sardans J, Luyssaert S, Campioli M, Chapin FS, Ciais P, Malhi Y, Obersteiner M, Papale D, Piao SL, Reichstein M, Roda F, Penuelas J (2014) Nutrient availability as the key regulator of global forest carbon balance. Nat Clim Change 4:471–476. doi:10.1038/nclimate2177
Fisk M, Santangelo S, Minick K (2015) Carbon mineralization is promoted by phosphorus and reduced by nitrogen addition in the organic horizon of northern hardwood forests. Soil Biol Biochem 81:212–218
Fu X, Shao M, Wei X, Horton R (2010) Soil organic carbon and total nitrogen as affected by vegetation types in Northern Loess Plateau of China. Geoderma 155:31–35. doi:10.1016/j.geoderma.2009.11.020
Goll DS, Brovkin V, Parida BR, Reick CH, Kattge J, Reich PB, van Bodegom PM, Niinemets U (2012) Nutrient limitation reduces land carbon uptake in simulations with a model of combined carbon, nitrogen and phosphorus cycling. Biogeosciences 9:3547–3569. doi:10.5194/bg-9-3547-2012
Griffiths B, Spilles A, Bonkowski M (2012) C:N:P stoichiometry and nutrient limitation of the soil microbial biomass in a grazed grassland site under experimental P limitation or excess. Ecol Process 1:1–11. doi:10.1186/2192-1709-1-6
Guo L, Gifford R (2002) Soil carbon stocks and land use change: a meta analysis. Glob Change Biol 8:345–360
Hedges LV, Gurevitch J, Curtis PS (1999) The meta-analysis of response ratios in experimental ecology. Ecology 80:1150–1156
IPCC (2000) Land use, land-use change and forestry. In: Robert T. Watson, Bert Bolin, N H Ravindranath, David J Verardo and David J Dokken (eds) Cambridge University, Press, Cambridge, UK, The Edinburgh Building Shaftesbury Road, Cambridge, England. http://www.ipcc.ch/ipccreports/sres/land_use/index.php?idp=124
IPCC (2006) In: Gómez DR, Watterson JD, Americano BB, Ha C, Marland G, Matsika E, Namayanga LN, Osman-Elasha B, Saka J, Treanton K (eds) IPCC guidelines for national greenhouse gas inventories. Institute for Global Environmental Strategies, Kamiyamaguchi Hayama, http://www.ipcc-nggip.iges.or.jp/public/2006gl/
Jackson R, Schenk H, Jobbagy E, Canadell J, Colello G, Dickinson R, Field C, Friedlingstein P, Heimann M, Hibbard K (2000) Belowground consequences of vegetation change and their treatment in models. Ecol Appl 10:470–483
Jandl R, Lindner M, Vesterdal L, Bauwens B, Baritz R, Hagedorn F, Johnson DW, Minkkinen K, Byrne KA (2007) How strongly can forest management influence soil carbon sequestration? Geoderma 137:253–268. doi:10.1016/j.geoderma.2006.09.003
Jenkinson D, Andrew S, Lynch J, Goss M, Tinker P (1990) The turnover of organic carbon and nitrogen in soil [and discussion]. Philos Trans R Soc Lond B Biol Sci 329:361–368
Jobbágy EG, Jackson RB (2000) The vertical distribution of soil organic carbon and its relation to climate and vegetation. Ecol Appl 10:423–436
Jobbágy EG, Jackson RB (2001) The distribution of soil nutrients with depth: global patterns and the imprint of plants. Biogeochemistry 53:51–77
Johnson DW, Curtis PS (2001) Effects of forest management on soil C and N storage: meta analysis. Forest Ecol Manag 140:227–238
Kirkby CA, Richardson AE, Wade LJ, BattenB GD, Blanchard C, Kirkegaard JA (2013) Carbon-nutrient stoichiometry to increase soil carbon sequestration. Soil Biol Biochem 60:77–86. doi:10.1016/j.soilbio.2013.01.011
Kirkby CA, Richardson AE, Wade LJ, Passioura JB, Batten GD, Blanchard C, Kirkegaard JA (2014) Nutrient availability limits carbon sequestration in arable soils. Soil Biol Biochem 68:402–409. doi:10.1016/j.soilbio.2013.09.032
Kirschbaum MU, Guo LB, Gifford RM (2008) Why does rainfall affect the trend in soil carbon after converting pastures to forests?: a possible explanation based on nitrogen dynamics. Forest Ecol Manag 255:2990–3000. doi:10.1016/j.foreco.2008.02.005
Laganière J, Angers DA, Paré D (2010) Carbon accumulation in agricultural soils after afforestation: a meta‐analysis. Glob Change Biol 16:439–453. doi:10.1111/j.1365-2486.2009.01930.x
Lam SK, Chen DL, Norton R, Armstrong R, Mosier AR (2012) Nitrogen dynamics in grain crop and legume pasture systems under elevated atmospheric carbon dioxide concentration: a meta-analysis. Glob Change Biol 18:2853–2859
Li DJ, Niu SL, Luo YQ (2012a) Global patterns of the dynamics of soil carbon and nitrogen stocks following afforestation: a meta-analysis. New Phytol 195:172–181. doi:10.1111/j.1469-8137.2012.04150.x
Li Y, Wu J, Liu S, Shen J, Huang D, Su Y, Wei W, Syers JK (2012b) Is the C:N:P stoichiometry in soil and soil microbial biomass related to the landscape and land use in southern subtropical China? Global Biogeochem Cycles 26:B4002. doi:10.1029/2012gb004399
Liao CZ, Luo YQ, Fang CM, Chen JK, Li B (2012) The effects of plantation practice on soil properties based on the comparison between natural and planted forests: a meta-analysis. Global Ecol Biogeogr 21:318–327. doi:10.1111/j.1466-8238.2011.00690.x
Liu L, Greaver TL (2009) A review of nitrogen enrichment effects on three biogenic GHGs: the CO2 sink may be largely offset by stimulated N2O and CH4 emission. Ecol Lett 12:1103–1117
Lorenz K, Lal R (2010) Nutrient and water limitations on carbon sequestration in forests. In: Lorenz K (ed) Carbon sequestration in forest ecosystems. Springer. pp 207–239
Lucas R, Davis JF (1961) Relationships between pH values of organic soils and availabilities of 12 plant nutrients. Soil Sci 92:177–182
Lund HG (1999) Aforest’by any other name. Environ Sci Policy 2:125–133
Marin-Spiotta E, Sharma S (2013) Carbon storage in successional and plantation forest soils: a tropical analysis. Global Ecol Biogeogr 22:105–117. doi:10.1111/j.1466-8238.2012.00788.x
Mitchell JS, Ruess RW (2009) N2 fixing alder (Alnus viridis spp. fruticosa) effects on soil properties across a secondary successional chronosequence in interior Alaska. Biogeochemistry 95:215–229. doi:10.1007/s10533-009-9332-x
Mitchell MJ, David MB, Harrison RB (1992) Sulphur dynamics of forest ecosystems. In: Howarth RW, Stewart JWB, Ivabov MV (eds) Sulphur cycling on the continents. Wiley, Chichester, pp 215–254
Mobley ML, Lajtha K, Kramer MG, Bacon AR, Heine PR, Richter DD (2015) Surficial gains and subsoil losses of soil carbon and nitrogen during secondary forest development. Glob Change Biol 21:986–996
Moscatelli M, Lagomarsino A, Marinari S, De Angelis P, Grego S (2005) Soil microbial indices as bioindicators of environmental changes in a poplar plantation. Ecol Indic 5:171–179
Nave LE, Swanston CW, Mishra U, Nadelhoffer KJ (2013) Afforestation effects on soil carbon storage in the United States: a synthesis. Soil Sci Soc Am J 77:1035–1047. doi:10.2136/sssaj2012.0236
Oulehle F, Evans CD, Hofmeister J, Krejci R, Tahovska K, Persson T, Cudlin P, Hruska J (2011) Major changes in forest carbon and nitrogen cycling caused by declining sulphur deposition. Glob Change Biol 17:3115–3129. doi:10.1111/j.1365-2486.2011.02468.x
Pan Y, Birdsey RA, Fang J, Houghton R, Kauppi PE, Kurz WA, Phillips OL, Shvidenko A, Lewis SL, Canadell JG (2011) A large and persistent carbon sink in the world’s forests. Science 333:988–993. doi:10.1126/science.1201609
Persson M, Moberg J, Ostwald M, Xu J (2013) The Chinese grain for green programme: assessing the carbon sequestered via land reform. J Environ Manag 126:142–146. doi:10.1016/j.jenvman.2013.02.045
Poeplau C, Don A, Vesterdal L, Leifeld J, Van Wesemael BAS, Schumacher J, Gensior A (2011) Temporal dynamics of soil organic carbon after land-use change in the temperate zone—carbon response functions as a model approach. Glob Change Biol 17:2415–2427. doi:10.1111/j.1365-2486.2011.02408.x
Pregitzer KS, Burton AJ, Zak DR, Talhelm AF (2008) Simulated chronic nitrogen deposition increases carbon storage in Northern Temperate forests. Glob Change Biol 14:142–153
Pribyl DW (2010) A critical review of the conventional SOC to SOM conversion factor. Geoderma 156:75–83
Resh SC, Binkley D, Parrotta JA (2002) Greater soil carbon sequestration under nitrogen-fixing trees compared with species. Ecosystems 5:217–231. doi:10.1007/s10021-001-0067-3
Richter DD, Markewitz D, Trumbore SE, Wells CG (1999) Rapid accumulation and turnover of soil carbon in a re-establishing forest. Nature 400:56–58. doi:10.1038/21867
Rosenberg MS, Adams DC, Gurevitch J (2000) Metawin: statistical software for meta-analysis, version 2.0. Sinauer Associates, Sunderland
Shi S, Han P (2014) Estimating the soil carbon sequestration potential of China’s Grain for Green Project. Global Biogeochem Cycles 28:1279–1294
Shi SW, Zhang W, Zhang P, Yu YQ, Ding F (2013) A synthesis of change in deep soil organic carbon stores with afforestation of agricultural soils. Forest Ecol Manag 296:53–63. doi:10.1016/j.foreco.2013.01.026
Shi SW, Han PF, Zhang P et al. (2015) The impact of afforestation on soil organic carbon sequestration on the Qinghai Plateau, China. PLoS ONE 10. doi: 10.1371/journal.pone.0116591
Silver W, Ostertag R, Lugo A (2000) The potential for carbon sequestration through reforestation of abandoned tropical agricultural and pasture lands. Restor Ecol 8:394–407
Sistla SA, Schimel JP (2012) Stoichiometric flexibility as a regulator of carbon and nutrient cycling in terrestrial ecosystems under change. New Phytol 196:68–78. doi:10.1111/j.1469-8137.2012.04234.x
Six J, Frey SD, Thiet RK, Batten KM (2006) Bacterial and fungal contributions to carbon sequestration in agroecosystems. Soil Sci Soc Am J 70:555–569. doi:10.2136/sssaj2004.0347
Song X, Peng C, Zhou G, Jiang H, Wang W (2014) Chinese Grain for Green Program led to highly increased soil organic carbon levels: a meta-analysis. Sci Rep 4:4460. doi:10.1038/srep04460
Strickland MS, Callaham MA, Davies CA, Lauber CL, Ramirez K, Richter DD, Fierer N, Bradford MA (2010) Rates of in situ carbon mineralization in relation to land-use, microbial community and edaphic characteristics. Soil Biol Biochem 42:260–269. doi:10.1016/j.soilbio.2009.10.026
Tang X, Liu S, Liu J, Zhou G (2010) Effects of vegetation restoration and slope positions on soil aggregation and soil carbon accumulation on heavily eroded tropical land of Southern China. J Soils Sediments 10:505–513
Thuille A, Schulze E-D (2006) Carbon dynamics in successional and afforested spruce stands in Thuringia and the Alps. Glob Change Biol 12:325–342. doi:10.1111/j.1365-2486.2005.01078.x
Verchot LV, Zomer R, Van Straaten O, Muys B (2007) Implications of country-level decisions on the specification of crown cover in the definition of forests for land area eligible for afforestation and reforestation activities in the CDM. Clim Change 81:415–430. doi:10.1007/s10584-006-9111-9
Vitousek PM, Cassman K, Cleveland C, Crews T, Field CB, Grimm NB, Howarth RW, Marino R, Martinelli L, Rastetter EB (2002) Towards an ecological understanding of biological nitrogen fixation. Biogeochemistry 57:1–45
Wang WJ, Qiu L, Zu YG, Su DX, An J, Wang HY, Zheng GY, Sun W, Chen XQ (2011) Changes in soil organic carbon, nitrogen, pH and bulk density with the development of larch (Larix gmelinii) plantations in China. Glob Change Biol 17:2657–2676. doi:10.1111/j.1365-2486.2011.02447.x
Whitbread A (1995) Soil organic matter: its fractionation and role in soil structure. In: Lefroy RDB, Blair GJ, Craswell ET (eds) Soil organic matter management for sustainable agriculture’, vol 56, ACIAR Proceedings., pp 124–130
Yang Y, Mohammat A, Feng J, Zhou R, Fang J (2007) Storage, patterns and environmental controls of soil organic carbon in China. Biogeochemistry 84:131–141. doi:10.1007/s10533-007-9109-z
Yang Y, Luo Y, Finzi AC (2011) Carbon and nitrogen dynamics during forest stand development: a global synthesis. New Phytol 190:977–989
Zou X, Binkley D, Caldwell BA (1995) Effects of dinitrogen-fixing trees on phosphorus biogeochemical cycling in contrasting forests. Soil Sci Soc Am J 59:1452–1458
Acknowledgments
This research was sponsored by the National Key Fundamental Research Program of China (2013CB956602), the International S & T Cooperation Program of China (2013DFA32190), National Science Foundation of China (41401277), and the Natural Sciences and Engineering Research Council of Canada (NSERC) Discover Grant. We also thank the MERITE Postdoc Scholarship program for foreign students from Quebec of Canada to S. Shi. Many thanks to two anonymous reviewers who provided constructive suggestions for improving this manuscript as well as Andreas Wilkes in Values for Development Ltd for his assistance with English language and grammatical editing of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Ingrid Koegel-Knabner.
Rights and permissions
About this article
Cite this article
Shi, S., Peng, C., Wang, M. et al. A global meta-analysis of changes in soil carbon, nitrogen, phosphorus and sulfur, and stoichiometric shifts after forestation. Plant Soil 407, 323–340 (2016). https://doi.org/10.1007/s11104-016-2889-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-016-2889-y