Abstract
Aims
The study aims (1) to evaluate the effect of Mesorhizobium tianshanense on plant proline and polyamine levels of Lotus tenuis and its modulatory effect during plant response to short-term salt stress and (2) to compare these effects with those caused by mycorrhizal symbiosis.
Methods
Experiments consisted of a randomized factorial design of two factors: salinity (two levels, 0 and 150 mM NaCl) and symbiosis (three levels, uninoculated, Glomus intraradices, and M. tianshanense).
Results
Salinization led to increased proline levels regardless of plant organ and symbiotic status, excepting mycorrhizal L. tenuis roots. Salinity diminished the total polyamine level of control and rhizobial plants but not in mycorrhizal ones. Variations in the pattern response of the three individual polyamines (putrescine, spermidine, and spermine) differed in accordance with the symbiotic status of the plant, highlighting a divergence on proline and polyamine metabolisms between rhizobial and mycorrhizal symbiosis.
Conclusions
Spermidine and spermine contributed the most with the salt-induced root polyamine increment observed upon salinization in roots of nodulated plants, suggesting that these polyamines might mediate an adaptive role of the plant–M. tianshanense symbiosis in L. tenuis plants growing in a saline environment.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The amino acid proline is one of the compatible osmolytes that most commonly build up in the cytoplasm to prevent the osmotic imbalance resulting from the excessive accumulation of toxic ions within the vacuole (Hasegawa et al. 2000). On other hand, many authors have reported that polyamine (PA) accumulation is the immediate response to salinity in different crop plants species (Erdei et al. 1996; Chattopadhyay et al. 2002; Ghosh et al. 2011). Polyamines are aliphatic molecules of low molecular weight, absolutely required for eukaryotic cell growth. Putrescine (Put), spermidine (Spd), and spermine (Spm) are the most common PA in plants (Cohen 1998). These compounds are thought to play an important role during the interaction between plants and root symbionts (Kytöviita and Sarjala 1997; Sannazzaro et al. 2007) and in plant responses to diverse environmental stresses (Groppa and Benavides 2008). A number of reports have shed some light about the physiological role of increased PA in salinized plants. These include maintaining cellular pH and ion balance, stabilizing membranes, and functioning as nitrogen reserve to be used after stress relief by the plant (reviewed by Mansour 2000). In addition, a number of studies have revealed a correlation between PA and proline contents in plants due to a close relationship between PA and proline metabolism (Smith 1985; Aziz et al. 1998; Santa-Cruz et al. 1999; Gaspar et al. 2000; Zhao et al. 2001; Theiss et al. 2002; Tonon et al. 2004; Sotiropoulos et al. 2007; Su and Bai 2008). Most legumes have the ability to establish mutualistic symbiotic relationships with soil N-fixing bacteria (collectively known as rhizobia) and arbuscular mycorrhizal fungi. However, symbiosis effects on plant proline and PA levels under salinity has received little attention. Diouf et al. (2005) found that, in two Acacia species grown under salt stress, leaf proline accumulation was lower in co-inoculated plants than in rhizobial- or mycorrhizal-inoculated ones, suggesting that co-inoculation provides the plant with improved tolerance to salt stress. Similar results were observed in Acacia saligna (Soliman et al. 2012). On other hand, variable effects of arbuscular mycorrhizal (AM) fungi on proline levels of plants grown under salt stress have been observed. Proline accumulation was greater in mycorrhized moong plants at 12.5 and 25 mM NaCl at 40 and 62 days after sowing (Jindal et al. 1993), whereas non-AM faba bean plants accumulated much more proline than AM plants at various salinity ranges (Rabie and Almadini 2005).
Lotus tenuis (Waldst. and Kit., syn. Lotus glaber; Kirkbride 2006) is a salt-tolerant glycophyte that became the best adapted legume forage in saline soils of the Flooding Pampa (the most important cattle production region in Argentina). In addition, L. tenuis also has the ability to establish mutualistic symbiotic relationships with diazotrophic, salt-tolerant bacteria belonging to the Mesorhizobium genus (Estrella et al. 2009). L. tenuis also associates with the arbuscular mycorrhizal fungus Glomus intraradices (Schenck and Smith) (Sannazzaro et al. 2004). In 2007, Sannazzaro and colleagues provided evidence supporting the idea that the regulation of plant PA levels might be a mechanism whereby the arbuscular fungus G. intraradices increased the tolerance of a L. tenuis salt-sensitive genotype to a long-term NaCl (200 mM) exposition.
In most of the studies, which have addressed the effect of biotic or abiotic factors on growth regulator balance, measurements were performed when these factors have already caused obvious changes on plant growth. However, variations in levels of growth factors might be attributable to developmental differences instead of evidence of changes induced by the studied factors themselves (Shaul-Keinan et al. 2002).
The aims of this work were: (1) to evaluate the effect of rhizobial symbiosis on plant proline and PA levels and its modulatory effect during plant response to short-term salt-stress and (2) to compare these results with effects caused by mycorrhizal symbiosis.
In plants, several physiological parameters were found to be size-dependent. For example, in Pisum sativum L., Spd accumulation was often associated with growing tissue activity and organogenesis, whereas higher Spd/Put ratios were related to rapid growth (Perez-Amador et al. 1995). Inversely, in a study on flexibility in growth and polyamine composition of the crucifer Pringlea antiscorbutica (Hennion et al. 2006), smaller plants showed the highest Spd contents and high Spd/Put ratios were more characteristic of smaller plants. Size dependency of abscisic acid and sensitivity to auxin were also observed respectively in vascular epiphytes (Zotz et al. 2001) and barley seedlings (Liptay and Davidson 1971). Besides plant regulation, other physiological parameters were found to be size-dependent, such as photosynthetic capacity (Zotz 1997; Zotz and Ziegler 1999), stomatal regulation (Schmidt and Zotz 2001), whole-plant water relations (Schmidt and Zotz 2001), and nutrient allocation to reproduction (Zotz 2000). Therefore, to reduce eventual overlapping effects of salinity itself and those derived from differences in plant growth, we measured biochemical parameters in a phenological state where significant salt-induced morphometric changes had not become evident yet, following a similar approach to that adopted for hormonal balances by Shaul-Keinan et al. (2002).
Materials and methods
Experiments consisted of a randomized factorial design of two factors: salinity (two levels, 0 and 150 mM NaCl) and symbiosis (three levels, uninoculated, G. intraradices, and Mesorhizobium tianshanense). Five plants were grown in each Leonard jar, and there were four jars per treatment. Experiment was repeated once.
Biological material and growth conditions
Seeds of L. tenuis cv. INTA Pampa were scarified with sulfuric acid (95–98%), washed in distilled water, and sown in Petri plates containing water–agar (0.8%). Plates were incubated during 10 days in a growth chamber, with a 16/8 h photoperiod at 24°C/19°C (day/night) and 60/80 ± 5% of relative humidity. Light intensity (200 μmol m−2 s−1) was provided by daylight and Grolux fluorescent lamps (F 40 W). Seedlings were transferred to sterilized 250-ml Leonard jars (Sigma) containing a 1:1 (v/v) perlite/vermiculite mixture as substrate and half-strength (0.5×) Hoagland solution (Hoagland and Arnon 1950), provided by capillarity. Phosphorus and nitrogen contents in the nutrient solution were as follows: uninoculated (C) and mycorrhizal (M) plants: Hoagland 0.5× (15.5 ppm P=KH2PO4 0.5 mM and 210.1 ppm N (Ca(NO3)2 5 mM+KNO3 5 mM); nodulated (R) plants: Hoagland 0.5× (31 ppm P=KH2PO4 1 mM and 105.05 ppm N (Ca(NO3)2 2.5 mM+KNO3 2.5 mM). For saline treatments, these solutions were supplemented with 150 mM NaCl. Nutrient solutions were renewed every time it fell below 10% of the total Leonard jar volume.
G. intraradices (BAFC 3108) was multiplied in 1,000 ml pot cultures with soil/perlite mixture (1:3 v/v) and Sorghum halepense (L.) Pers. (=Andropogon halepensis Brot.) as host during 4 months. Fungal inoculum consisted of 1 g of root fragments with no less than 70% of their root length colonized by the fungus. For rhizobial inoculum, the M. tianshanense strain (BA151), isolated from a saline soil at the Flooding Pampa (Estrella et al. 2009), was grown in liquid TY medium (Beringer 1974) at 28°C. The efficiency of this strain as a L. tenuis growth promoter had been previously demonstrated (Estrella et al. 2009; Sannazzaro et al. 2011). Inoculum consisted of 1 ml of the bacterial suspension (DO = 109 CFU/ml), added to the crown of each seedling. The G. intraradices inoculum was incorporated during seedling transference to Leonard jars, whereas M. tianshanense was inoculated 1 day after seedlings were transplanted. Uninoculated controls received an equal amount of autoclaved inoculum. The saline stress was applied 30 days after transplanting, when at least 50% of the root was colonized by G. intraradices and plants inoculated with M. tianshanense were nodulated.
Plants were harvested 5 days after the saline treatment was initiated. Harvested plants were divided in stems, leaf, and roots. One part of the plant material was used for dry weight measurement and the rest for biochemical determinations. For morphometric determinations, one plant from each jar was used, so that each plant was a replicate. For analytical determinations, the remaining four plants in each jar were pooled, with each pool treated as one replicate. The presence of G. intraradices was determined visually by clearing and staining the roots with Trypan blue in lactophenol (Phillips and Hayman 1970). Percentage of mycorrhizal root colonization was estimated according to McGonigle et al. (1990).
Proline was estimated spectrophotometrically by the ninhydrin reaction under conditions described elsewhere (Maiale et al. 2004). Briefly, proline was extracted by boiling plant material (500 mg) in 2 ml distilled water. Then, 500 μl of Na-citrate buffer (0.2 mol/l, pH 4.6) and 2 ml of 1% ninhydrin (acetic acid/water 60:40) were added to 500 μl extract. The mixture was boiled for 1 h, extracted with 2 ml toluene, and then centrifuged. Organic phase was read at 520 nm. Proline standard (Sigma, USA) was treated in the same way as the plant extracts.
Free polyamines were estimated by analyzing dansyl-derivatives by reversed-phase high-performance liquid chromatography (HPLC) as described previously (Marcé et al. 1995). To extract free PA, pools of 300 mg of plant material were frozen in liquid N2 and homogenized. The homogenate (300 mg) was resuspended in 1 ml of perchloric acid 5% (v/v), incubated on ice during 30 min and centrifuged at 15,000×g for 15 min. Pellet was discarded and the supernatant kept at −20°C (solution C). Aliquots (200 μl) of free PA were derivatized with dansyl chloride and determined by HPLC according to Jiménez-Bremont et al. (2007). For the dansylation reaction, 200 μl of solution C (see above) was added to 10 μl of 0.1 mM heptanodiamine (internal standard, ICN) plus 200 μl saturated Na2CO3 and 400 μl dansyl chloride–acetone 1% (w/v). After 16 h at 25°C in the dark, 100 μl of proline 100% (w/v) was added to stop the reaction and the dansyl-derived PA extracted with 500 μl toluene. Then, the organic phase (400 μl) was evaporated under vacuum and resuspended in 400 μl acetonitrile. PA was separated by HPLC (ISCO 2350, ISCO Inc., Lincoln, NE) with a reverse-phase column Sephasil C18 (Amersham Pharmacia) and detected with a spectrofluorometer (Variant Fluorichrom). The solvent mix was obtained with a gradient programmer ISCO 2360, flow 1.5 ml/min as follows—0–4.5 min, acetonitrile–H2O 70:30; 4.5–9 min, acetonitrile 100; 9–15 min, acetonitrile–H2O (70:30). Peak areas were integrated, normalized to heptanodiamine, and then interpolated into a PA standard calibration curve.
Statistical analysis
Data was subjected to two-way analysis of variance using the symbiosis, salt, symbiosis–salt interaction, and error as variation sources and means compared by Duncan’s test. The Pearson's coefficient was used to attest for correlation between the increased percentages of proline and Put due to salt addition.
Results
No differences in total, shoot, and root dry weights were observed due to salt stress after 5 days of salinization (Tables 1 and 2). However, the root dry weight was influenced by a salinity × symbiosis interaction. Under the saline condition, nodulated roots were heavier than those of the corresponding control. In contrast, profound changes in proline and PA contents were observed due to the salinity × symbiosis interaction (Table 1).
Proline content
Salinization led to increased proline levels regardless of plant organ and symbiotic status, excepting roots of G. intraradices-inoculated plants (Table 3). Under the salt stress condition, symbiotic plants presented significantly lower proline contents than control ones. In the absence of NaCl, the symbiotic status did not largely affect the total proline level of plants. However, the root proline level of unsalinized plants was raised by the bacterial symbiosis, whereas both symbioses induced a reduction in the stem proline content.
Free PA content
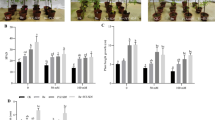
Salinity and symbiosis factors interacted on PA levels (Table 1). Salinity diminished the total PA level of the plant in control and rhizobial treatments but not in the mycorrhizal one (Fig. 1). M. tianshanense led to a 30% higher total PA content, regardless the saline condition, whereas G. intraradices reduced this level in the absence of salt.
Total, root, stem, and leaf free polyamine contents of L. tenuis plants. Nutrient solution containing 150 mM NaCl was applied to 40-day-old plants for 5 days. C: uninoculated, M: mycorrhizal, and R: rhizobial-inoculated plants, plus sign: salinized, and minus sign: unsalinized plants. Means (±SE; n = 4) with the same letter within each column are not significantly different (Duncan, P < 0.05)
Root PA level decreased due to salt addition in non-symbiotic and mycorrhizal plants (Fig. 1). Despite this diminution, mycorrhizal salt-stressed plants showed a higher root PA level than their corresponding non-mycorrhizal control. Contrarily, root PA level of nodulated plants was increased by salinity.
Salinity diminished the stem PA content in control and nodulated plants but not in mycorrhizal ones (Fig. 1). In the leaf, salinity increased the PA level in non-inoculated and mycorrhizal plants and decreased it in nodulated ones. Under the non-saline condition, mycorrhizal colonization lowered, whereas the rhizobacteria elevated stem and leaf PA contents. On other hand, root PA contents of unsalinized plants were not affected by AM inoculation, but they were slightly raised by the bacterial symbiont (Fig. 1).
Salt effect differed according to the type of polyamine and the symbiotic status. Upon salinization, total Put decreased in control and rhizobial plants, but it remained unchanged in the AM symbiosis treatment (Fig. 2). In control plants (although not in rhizobial ones), the percentage of total Put reduction correlated with that of total proline increment, induced by salt stress (r = 0.81; P < 0.0001). Conversely, total Spm was incremented by salt treatment in all cases, whereas total Spd was slightly higher in mycorrhizal and control treatments, and it did not vary in nodulated plants (Fig. 2). Under the non-saline condition, AM plants diminished total Put level and presented similar Spd and Spm levels, whereas nodulated plants had higher total Put, Spd, and Spm contents compared with uninoculated ones (Fig. 2).
Total Put, Spd, and Spm contents of L. tenuis plants. Nutrient solution containing 150 mM NaCl was applied to 40-day-old plants for 5 days. C: uninoculated, M: mycorrhizal, and R: rhizobial-inoculated plants, plus sign: salinized, and minus sign: unsalinized plants. Means (±SE; n = 4) with the same letter within each column are not significantly different (Duncan, P < 0.05)
In the root, salinity reduced the Put content in all treatments (Fig. 3), whereas symbiosis did not affect this level under the non-saline condition. Salinity significantly increased root Spd levels in nodulated plants and root Spm levels in all treatments. In contrast, root Spd was diminished or remained invariable upon salinization in control and AM plants, respectively (Fig. 3). On the other hand, AM colonization did not affect root Spd and Spm levels of unsalinized plants, whereas inoculation with M. tianshanense incremented the root Spd content.
Root Put, Spd, and Spm contents of L. tenuis plants. Nutrient solution containing 150 mM NaCl was applied to 40-day-old plants for 5 days. C: uninoculated, M: mycorrhizal, and R: rhizobial-inoculated plants, plus sign: salinized, and minus sign: unsalinized plants. Means (±SE; n = 4) with the same letter within each column are not significantly different (Duncan, P < 0.05)
In general, the patterns of PA changes due to salinity and symbiosis in stem (Fig. 4) and leaf (Fig. 5) were similar. Stem and leaf Put contents were negatively affected by salinity in control and nodulated plants. Notwithstanding this, stem and leaf Put levels in salt-treated nodulated plants were significantly higher than those observed in the corresponding uninoculated control. In AM plants, contrarily, these contents remained unaffected by salinity. Under the non-saline condition, the Put content of these organs tended to decrease and increase in mycorrhizal and nodulated plants, respectively. On other hand, salinity had no effect on the Spd level in stems (Fig. 4), but the leaf Spd level increased due to salt addition in control and AM plants (Fig. 5). In contrast, no salinity-derived change of the leaf Spd content was registered in nodulated plants. In the leaf, the pattern of Spm variations due to salinity was similar to that of Spd (Fig. 5), whereas stem Spm was raised by salt treatment, regardless of the symbiotic status (Fig. 4).
Stem Put, Spd, and Spm contents of L. tenuis plants. Nutrient solution containing 150 mM NaCl was applied to 40-day-old plants for 5 days. C: uninoculated, M: mycorrhizal, and R: rhizobial-inoculated plants, plus sign: salinized, and minus sign: unsalinized plants. Means (±SE; n = 4) with the same letter within each column are not significantly different (Duncan, P < 0.05)
Leaf Put, Spd, and Spm contents of L. tenuis plants. Nutrient solution containing 150 mM NaCl was applied to 40-day-old plants for 5 days. C: uninoculated, M: mycorrhizal, and R: rhizobial-inoculated plants, plus sign: salinized, and minus sign: unsalinized plants. Means (±SE; n = 4) with the same letter within each column are not significantly different (Duncan, P < 0.05)
In the absence of NaCl, mycorrhizal colonization did not affect stem and leaf Spd levels (Figs. 4 and 5) but reduced the leaf Spm content (Fig. 5). In contrast, all these contents were incremented by M. tianshanense (Figs. 4 and 5).
Discussion
There are a number of studies supporting the view that proline accumulation in response to salt stress is a good indicator of a higher stress perception (Tal et al. 1979; Colmer et al. 1995; Vaidyanathan et al. 2003; Maiale et al. 2004; Sannazzaro et al. 2007). On this basis, our results showing variable salt-derived increases of proline accumulation among treatments (Table 3) would indicate that L. tenuis plants experienced different degrees of salt stress according to their symbiotic status. The fact that, under the salt stress condition, symbiotic plants presented significantly lower proline contents than control ones constitutes a hint that the last were more stressed than the former, mycorrhizal plants being the less affected ones.
Salt-derived accumulations in the proline level were formerly observed upon salinization in plants of Lotus corniculatus, a species phylogenetically closely related to L. tenuis (Márquez et al. 2005). Such similarity in the proline level response to an osmotic factor between both Lotus species may be linked to their common evolution in dry and saline soil environments (Allan et al. 2004).
Inoculation with M. tianshanense led to a 75% increase in the root proline level (Table 3), in line with findings showing that this amino acid is essential for the plant–rhizobia symbiosis and inherent to the nodulation process itself (Kohl et al. 1988; Jiménez-Zurdo et al. 1997; King et al. 2000). On the contrary, no effect by AM colonization was registered on the root proline level of unsalinized plants (Table 3), in coincidence with previous observations in L. tenuis genotypes (Sannazzaro et al. 2007).
Upon salinization, total PA reductions were registered in uninoculated and nodulated plants, although not in AM ones. Such reductions were primarily due to a strong depressing effect of salinity on Put contents (Figs. 2, 3, 4, and 5). In uninoculated L. tenuis plants, salt-induced Put diminution correlated with a proline rise, in agreement with previous observations in other uninoculated plant species subjected to saline stress (Aziz et al. 1998; Tonon et al. 2004; Sotiropoulos et al. 2007; Su and Bai 2008). These results find support in the fact that salinity promotes diamine oxidase (Smith 1985; Aziz et al. 1998; Su and Bai 2008), whose catalytic activity on Put may contribute to proline accumulation (Bouchereau et al. 1999; Gaspar et al. 2000). On other hand, correlations between rises in proline and declines in Put may also be registered through conversion of glutamate (a common proline and Put precursor) to proline (Santa-Cruz et al. 1999; Gaspar et al. 2000; Zhao et al. 2001; Tonon et al. 2004). This conversion was also reported to be induced under salt stress in several plant species like cashews (da Rocha et al. 2012), Saussurea amara (Zhang et al. 2011), and tobacco (Wang et al. 2011). Interestingly, no correlation between salt-induced Put diminution and proline rise was registered in nodulated L. tenuis plants, what leads to suggest that the presence of M. tianshanense in the root may influence the interconnection of these two metabolic pathways. To date, there is very little information available about the effect of root symbionts on both proline and PA metabolism, two shared pathways, and the investment of further research efforts in this area might provide valuable information to be used for the improvement of legume tolerance to soil salinity.
Our results also showed that, in control and nodulated L. tenuis plants, salt-induced reduction of Put levels took place regardless the plant organ, whereas the stress affected Spm and Spm levels in an organ-dependent manner (e.g., Spd, Figs. 3 and 5). Organ-specific differences in the PA metabolism response to salinity have been formerly described in other plant species (Legocka and Kluk 2005; Stetsenko et al. 2009; Radyukina et al. 2009). Also, our results showing that shoots of rhizobial plants presented higher contents of free PA, compared with those of control and mycorrhizal ones, are in agreement with previous reports in alfalfa inoculated with Rhizobium meliloti and Glomus fasciculatum (Goicoechea et al. 1998) remarking a divergence between both types of symbiosis and also, the peculiarities of their PA metabolism. In this regard, the Spd augmentation observed in roots of nodulated plants is in line with the salt-induced over-expression, in a Sinorhizobium meliloti strain, of the carboxynospermidine decarboxylase, enzyme playing an important role in the Spd biosynthesis (Shamseldin et al. 2006). In turn, the Spm increase in nodulated, salt-stressed L. tenuis plants could be explained by the rise of its precursor, Spd (Fig. 3).
Besides the mechanisms mentioned in the “Introduction” by which root PA would intervene in the salt-tolerance process by plants, a relationship between polyamine catabolism mediated by amine oxidases and cell growth has been recently proposed. On one hand, it has been reported that apoplastic reactive oxygen species (ROS) promote cellular elongation in maize coleoptile (Schopfer et al. 2001) and A. thaliana root (Foreman et al. 2003). On the other hand, the enzyme polyamine oxidase (PAO) oxidizes Spd and Spm on their secondary amino groups (Federico and Angelini 1991) producing hydrogen peroxide (H2O2) among other products. In maize, higher free PA levels were observed in segments of salt-treated plants, compared with the untreated control (Rodríguez et al. 2009). In these plants, PAO activity provided the amount of ROS in the apoplast needed to sustain the elongation of leaf blades, showing a relationship between Spd and Spm, and cell growth under salinity. A similar relationship was demonstrated in soybean hypocotyls (Campestre et al. 2011). In the present work, L. tenuis plants were harvested before salt-derived differences in growth became obvious. Future research should address the occurrence of possible links between combined proline and PA metabolisms, and L. tenuis plant growth.
Finally, it has been suggested that the salt-induced metabolic alteration of homospermidine (a Spd analog) is closely related to the salt stress tolerance of fast-growing rhizobia (Fujihara 2009). All this information, along with the fact that accumulated proline may be used as a nitrogen source or as osmoticum by plants (Stewart and Larher 1980; Trotel et al. 1996), offers the notion that these polyamines might mediate an adaptive role of the plant–M. tianshanense symbiosis in L. tenuis plants growing in a saline environment. However, the mechanisms that govern Spm and Spd (or homoSpd)-mediated salt resistance in bacteria remain unclear (Fujihara 2009).
Abbreviations
- DAO:
-
Diamine oxidase
- PA:
-
Polyamines
- Put:
-
Putrescine
- Spd:
-
Spermidine
- Spm:
-
Spermine
References
Allan GJ, Francisco-Ortega J, Santos-Guerra A, Boerner E, Zimmer EA (2004) Molecular phylogenetic evidence for the geographic origin and classification of Canary Island Lotus (Fabaceae: Loteae). Mol Phylogenet Evol 32:123–138
Aziz A, Martin-Tanguy J, Larher F (1998) Stress-induced changes in polyamine and tyramine levels can regulate proline accumulation in tomato leaf discs treated with sodium chloride. Physiol Plantarum 104:195–202
Beringer JE (1974) R factor transfer in Rhizobium leguminosarum. J Gen Microbiol 84:188–198
Bouchereau A, Aziz A, Larher F, Martin-Tanguy J (1999) Polyamines and environmental challenges: recent development. Plant Sci 140:103–125
Campestre MP, Bordenave CD, Origone AC, Menéndez AB, Ruiz OA, Rodríguez AA, Maiale SJ (2011) Polyamine catabolism is involved in response to salt stress in soybean hypocotyls. J Plant Physiol 168:1234–1240
Chattopadhyay MK, Tabor CW, Tabor H (2002) Absolute requirement of spermidine for growth and cell cycle progression of fission yeast (Schizosaccharomyces pombe). P Natl Acad Sci 99:10330–10334
Cohen SS (1998) A guide to the polyamines. Oxford University Press, New York
Colmer TD, Epstein E, Dvorak J (1995) Differential solute regulation in leaf blades of various ages in salt-sensitive wheat and a salt-tolerant wheat × Lophopyrum elongatum (host) A. Löve amphiploid. Plant Physiol 108:1715–1724
da Rocha IMA, Vitorello VA, Silva JS, Ferreira-Silva SL, Viégas RA, Silva EN, Silveira JAG (2012) Exogenous ornithine is an effective precursor and the δ-ornithine amino transferase pathway contributes to proline accumulation under high N recycling in salt-stressed cashew leaves. J Plant Physiol 169:41–49
Diouf D, Duponnois R, Ba AT, Neyra M, Lesueur D (2005) Symbiosis of Acacia auriculiformis and Acacia mangium with mycorrhizal fungi and Bradyrhizobium spp. improves salt tolerance in greenhouse conditions. Aust J Plant Physiol 32:1143–1152
Erdei L, Szegletes Z, Barabas K, Pestenacz A (1996) Responses in polyamine titer under osmotic and salt stress on sorghum and maize seedlings. J Plant Physiol 147:599–603
Estrella MJ, Muñoz S, Soto MJ, Ruiz O, Sanjuan J (2009) Genetic diversity and host range of rhizobia nodulating Lotus tenuis in typical soils of the Salado River Basin (Argentina). Appl Environ Microbiol 75:1088–1098
Federico R, Angelini R (1991) Polyamine catabolism in plants. In: Slocum RD, Flores HE (eds.) Biochemistry and physiology of polyamines in plants. Boca Raton, FL,CRC Press, pp 41-56
Foreman J, Demidchik V, Bothwell JHF, Mylona P, Miedema H, Angel Torres M, Linstead P, Costa S, Brownlee C, Jones JDG, Davies JM, Dolan L (2003) Reactive oxygen species produced by NADPH oxidase regulate plant cell growth. Nature 422:442–446
Fujihara S (2009) Biogenic amines in rhizobia and legume root nodules. Microbes Environ 24:1–13
Gaspar T, Kevers C, Hausman J-F, Faivre-Rampant O, Boyer N, Dommes J, Penel C, Greppin H (2000) Integrating phytohormone metabolism and action with primary biochemical pathways. I. Interrelationships between auxins, cytokinins, ethylene and polyamines in growth and development processes. In: Greppin H, Penel C, Broughton WJ, Strasser R (eds) Integrated plant systems. University of Geneva, Switzerland, pp 163–191
Ghosh N, Adak MK, Ghosh PD, Gupta S, Sen Gupta DN, Mandal C (2011) Differential responses of two rice varieties to salt stress. Plant Biotechnol Rep 5:89–103
Goicoechea N, Szalai NG, Antolín MC, Sánchez-Díaz M, Paldi E (1998) Influence of arbuscular mycorrhizae and Rhizobium on free polyamines and proline levels in water-stressed alfalfa. J Plant Physiol 153:706–711
Groppa MD, Benavides MP (2008) Polyamines and abiotic stress: recent advances. Amino Acids 34:35–45
Hasegawa PM, Bressan RA, Zhu J-K, Bohnert HJ (2000) Plant cellular and molecular responses to high salinity. Annu Rev Plant Phys 51:463–499
Hennion F, Frenot Y, Martin-Tanguy J (2006) High flexibility in growth and polyamine composition of the crucifer Pringlea antiscorbutica in relation to environmental conditions. Physiol Plant 127:212–224
Hoagland DR, Arnon DL (1950) The water culture method for growing plants without soil. Calif Agricult Exp Stat Circ 374:1–39
Jiménez-Bremont JF, Ruiz OA, Rodríguez-Kessler M (2007) Modulation of spermidine and spermine levels in maize seedlings subjected to long-term salt stress. Plant Physiol Bioch 45:812–821
Jiménez-Zurdo JI, García-Rodríguez FM, Toro N (1997) The Rhizobium meliloti putA gene: its role in the establishment of the symbiotic interaction with alfalfa. Mol Microbiol 23:85–93
Jindal V, Atwala A, Sekhon BS, Singh R (1993) Effect of vesicular-arbuscular mycorrhizae on metabolism of moong plants under NaCl salinity. Plant Physiol Bioch 31:475–481
King ND, Hojnacki D, O'Brian MR (2000) The Bradyrhizobium japonicum proline biosynthesis gene proC is essential for symbiosis. Appl Environ Microbiol 66:5469–5471
Kirkbride JH Jr (2006) The scientific name of narrow-leaf trefoil. Crop Sci 46:2169–2170
Kohl DH, Schubert KR, Carter MB, Hagedorn CH, Shearer G (1988) Proline metabolism in N2-fixing root nodules: energy transfer and regulation of purine synthesis. P Natl Acad Sci 85:2036–2040
Kytöviita M-M, Sarjala T (1997) Effects of defoliation and symbiosis on polyamine levels in pine and birch. Mycorrhiza 7:107–111
Legocka J, Kluk A (2005) Effect of salt and osmotic stress on changes in polyamine content and arginine decarboxylase activity in Lupinus luteus seedlings. J Plant Physiol 162:662–668
Liptay A, Davidson D (1971) Coleoptile growth: variation in elongation patterns of individual coleoptiles. Ann Bot 35:91–1002
Maiale S, Sanchez D, Guirado A, Vidal A, Ruiz O (2004) Spermine accumulation under salt stress. J Plant Physiol 161:35–42
Mansour MMF (2000) Nitrogen containing compounds and adaptation of plants to salinity stress. Biol Plant 43:491–500
Marcé M, Brown DS, Capell T, Figueras X, Tiburcio AF (1995) Rapid high-performance liquid chromatographic method for the quantitation of polyamines as their dansyl derivatives: application to plant and animal tissues. J Chromatogr B Biomed Appl 666:329–335
Márquez AJ, Betti M, García-Calderón M, Pal'ove-Balang P, Díaz P, Monza J (2005) Nitrate assimilation in Lotus japonicus. J Exp Bot 56:1741–1749
McGonigle TP, Miller MH, Evans DG, Fairchild GL, Swan JA (1990) A new method which gives an objective measure of colonization of roots by vesicular-arbuscular mycorrhizal fungi. New Phytol 115:495–501
Pérez-Amador MA, Carbonell J, Granell A (1995) Expression of arginine decarboxylase is induced during early fruit development and in young tissues of Pisum sativum (L.). Plant Mol Biol 28:997–1009
Phillips JM, Hayman DS (1970) Improved procedures for clearing roots and staining parasitic and vesicular arbuscular mycorrhizal fungi for rapid assessment of infection. T Brit Mycol Soc 55:158–161
Rabie GH, Almadini AM (2005) Role of bioinoculants in development of salt-tolerance of Vicia faba plants under salinity stress. Afr J Biotechnol 4:210–220
Radyukina NL, Mapelli S, Ivanov YV, Kartashov AV, Brambilla I, Kuznetsov VV (2009) Homeostasis of polyamines and antioxidant systems in roots and leaves of Plantago major under salt stress. Russ J Plant Physl 56:323–331
Rodríguez AA, Maiale SJ, Menéndez AB, Ruiz OA (2009) Polyamine oxidase activity contributes to sustain maize leaf elongation under saline stress. J Exp Bot 60:4249–4262
Sannazzaro A, Ruiz O, Albertó E, Menéndez A (2004) Presence of different arbuscular mycorrhizal infection patterns in roots of Lotus glaber plants growing in the Salado River basin. Mycorrhiza 14:139–142
Sannazzaro AI, Echeverria M, Albertó EO, Ruiz OA, Menéndez AB (2007) Modulation of polyamine balance in Lotus glaber by salinity and arbuscular mycorrhiza. Plant Physiol Bioch 45:39–46
Sannazzaro A, Bergottini V, Paz R, Castagno L, Menéndez A, Ruiz O, Pieckenstain F, Estrella M (2011) Comparative symbiotic performance of native rhizobia of the Flooding Pampa and strains currently used for inoculating Lotus tenuis; in this region. Anton Leeuw Int J G 99:371–379
Santa-Cruz A, Acosta M, Rus A, Bolarin MC (1999) Short-term salt tolerance mechanisms in differentially salt tolerant tomato species. Plant Physiol Bioch 37:65–71
Schmidt G, Zotz G (2001) Ecophysiological consequences of differences in plant size: in situ carbon gain and water relations of the epiphytic bromeliad, Vriesea anguinolenta. Plant Cell Environ 24:101–112
Schopfer P, Plachy C, Frahry G (2001) Release of reactive oxygen intermediates (superoxide radicals, hydrogen peroxide, and hydroxyl radicals) and peroxidase in germinating radish seeds controlled by light, gibberellin and abscisic acid. Plant Physiol 125:1591–1602
Shamseldin A, Nyalwidhe J, Werner D (2006) A proteomic approach towards the analysis of salt tolerance in Rhizobium etli and Sinorhizobium meliloti strains. Curr Microbiol 52:333–339
Shaul-Keinan O, Gadkar V, Ginzberg I, Grünzweig JM, Chet I, Elad Y, Wininger S, Belausov E, Eshed Y, Atzmon N, Ben-Tal Y, Kapulnik Y (2002) Hormone concentrations in tobacco roots change during arbuscular mycorrhizal colonization with Glomus intraradices. New Phytol 154:501–507
Smith TA (1985) Polyamines. Ann Rev Plant Physio 36:117–143
Soliman ASh, Shanan NT, Massoud ON, Swelim DM (2012) Improving salinity tolerance of Acacia saligna (Labill.) plant by arbuscular mycorrhizal fungi and Rhizobium inoculation. Afr J Biotechnol 11:1259–1266
Sotiropoulos TE, Therios IN, Tsirakoglou V, Dimassi KN (2007) Response of the quince genotypes BA 29 and EMA used as pear rootstocks to boron and salinity. Int J Fruit Sci 6:93–101
Stetsenko LA, Rakitin VY, Shevyakova NI, Kuznetsov VV (2009) Organ-specific changes in the content of free and conjugated polyamines in Mesembryanthemum crystallinum plants under salinity. Russ J Plant Physl+ 56:808–813
Stewart GR, Larher F (1980) Accumulation of amino acids and related compounds in relation to environmental stress. In: Miflin BJ (ed) The biochemistry of plants, Vol 5. Academic Press, New York, pp 609–635
Su G, Bai X (2008) Contribution of putrescine degradation to proline accumulation in soybean leaves under salinity. Biol Plantarum 52:796–799
Tal M, Katz A, Heikin H, Dehan K (1979) Salt tolerande in the wild relatives of the cultivated tomato: porline accumulation in Lycopersicon esculentum Mill., L. peruvianum Mill. and Solanum pennelli Cor. treated with NaCI and polyethylene glycol. New Phytol 82:349–355
Theiss C, Bohley P, Voigt J (2002) Regulation by polyamines of ornithine decarboxylase activity and cell division in the unicellular green alga Chlamydomonas reinhardtii. Plant Physiol 128:1470–1479
Tonon G, Kevers C, Faivre-Rampant O, Graziani M, Gaspar T (2004) Effect of NaCl and mannitol iso-osmotic stresses on proline and free polyamine levels in embryogenic Fraxinus angustifolia callus. J Plant Physiol 161:701–708
Trotel P, Bouchereau A, Niogret MF, Larher F (1996) The fate of osmo-accumulated proline in leaf discs of rape (Brassica napus L.) incubated in a medium of low osmolarity. Plant Sci 118:31–45
Vaidyanathan H, Sivakumar P, Chakrabarty R, Thomas G (2003) Scavenging of reactive oxygen species in NaCl-stressed rice (Oryza sativa L.)—differential response in salt-tolerant and sensitive varieties. Plant Sci 165:1411–1418
Wang K, Liu Y, Dong K, Dong J, Kang J, Yang Q, Zhou H, Sun Y (2011) The effect of NaCl on proline metabolism in Saussurea amara seedlings. Afr J Biotechnol 10:2886–2893
Zhang J, Zhang Y, Du Y, Chen S, Tang H (2011) Dynamic metabonomic responses of tobacco (Nicotiana tabacum) plants to salt stress. J Proteome Res 10:1904–1914
Zhao F-G, Sun C, Liu Y-L (2001) Ornithine pathway in proline biosynthesis activated by salt stress in barley seedlings. Acta Bot Sin 43:36–40
Zotz G (1997) Photosynthetic capacity increases with plant size. Bot Acta 110:306–308
Zotz G (2000) Size dependence in the reproductive allocation of Dimerandra emarginata, an epiphytic orchid. Ecotropica 6:95–99
Zotz G, Ziegler H (1999) Size-related differences in carbon isotope discrimination in the epiphytic orchid, Dimerandra emarginata. Naturwissenschaften 86:39–40
Zotz G, Thomas V, Hartung W (2001) Ecophysiological consequences of differences in plant size: abscisic acid relationships in the epiphytic orchid Dimerandra emarginata. Oecologia 129:179–185
Acknowledgments
This work was supported by: Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET, Argentina), Agencia Nacional de Promoción Científica y Tecnológica (PICT 20517), UBACYT x143. M.E is CONICET scholarship holder. A.B.M. is a Universidad de Buenos Aires (UBA) and CONICET researcher, and O.A.R. and A.I.S are CONICET researchers.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Katharina Pawlowski.
Rights and permissions
About this article
Cite this article
Echeverria, M., Sannazzaro, A.I., Ruiz, O.A. et al. Modulatory effects of Mesorhizobium tianshanense and Glomus intraradices on plant proline and polyamine levels during early plant response of Lotus tenuis to salinity. Plant Soil 364, 69–79 (2013). https://doi.org/10.1007/s11104-012-1312-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-012-1312-6