Abstract
Lotus tenuis (Wadst. & Kit.) is a perennial legume widely grown for pasture in the flood-prone and salt affected Pampa region of Argentina. The physiology of salt and waterlogging tolerance in L. tenuis (four cultivars) was evaluated, and compared with Lotus corniculatus (three cultivars); the most widely cultivated Lotus species. Overall, L. tenuis cultivars accumulated less Na+ and Cl−, and more K+ in shoots than L. corniculatus cultivars, when exposed to 200 mM NaCl for 28 days in aerated or in stagnant solutions. Root porosity was higher in L. tenuis cultivars due to greater aerenchyma formation. In a NaCl dose–response experiment (0–400 mM NaCl in aerated solution), L. tenuis (cv. Chaja) accumulated half as much Cl− in its shoots than L. corniculatus (cv. San Gabriel) at all external NaCl concentrations, and about 30% less shoot Na+ in treatments above 250 mM NaCl. Ion distributions in shoots were determined for plants at 200 mM NaCl. L. tenuis (cv. Chaja) again accumulated about half as much Cl− in old leaves, young leaves and stems, compared with concentrations in L. corniculatus (cv. San Gabriel). There were not, however, significant differences between the two species for Na+ concentrations in the various shoot tissues. The higher root porosity, and maintenance of lower shoot Cl− and Na+ concentrations in L. tenuis, compared with L. corniculatus, contributes to the greater tolerance to combined salt and waterlogging stress in L. tenuis. Moreover, significant variation for tolerance to combined salinity and waterlogging stress was identified within both L. tenuis and L. corniculatus.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The area of land affected by secondary salinity (salinity caused by human activity) is steadily increasing, with recent worldwide estimates that over 70 million ha of agricultural land is affected (FAO 2005). A large proportion of saline areas also suffer from waterlogging or intermittent flooding (Smedema and Shiati 2002; Barrett-Lennard 2003). Revegetation of valley floors with perennial species in areas affected by dryland salinity should reduce recharge and the spread of salinity (George et al. 2004). Perennial pastures that produce fodder on saline soils subject to waterlogging should improve farm productivity. Lotus tenuis Wadst. & Kit. (syn. Lotus glaber Mill., Lotus tenufolius Reichb., Lotus corniculatus L. subsp. tenufolius L. P. Fourn) is a perennial pasture legume that has naturalized in the flooding Pampa region of Argentina (Kade et al. 2003). Previous studies have reported L. tenuis to be salt tolerant (Ayers 1948; Schachtman and Kelman 1991; Rogers et al. 1997a) and waterlogging tolerant (Vignolio et al. 1999; Mendoza et al. 2005; Striker et al. 2005) and have suggested this species has potential for use in saline discharge areas (Dear et al. 2003). However, the impact of combined salt and waterlogging stress on L. tenuis has not been studied and the mechanisms underlying salt tolerance in L. tenuis have not been elucidated.
Salinity impacts on plants in two main ways: osmotic stress and ion toxicity (Greenway and Munns 1980). Osmotic stress is caused by ions (mainly Na+ and Cl−) in the soil solution decreasing the availability of water to roots (Munns et al. 1995). Ion toxicity occurs when plant roots take up Na+ and/or Cl− and these ions accumulate to detrimental levels in leaves (Munns 1993). Ion imbalances and nutrient deficiency, particularly for K+ nutrition, can also occur (Maathuis and Amtmann 1999). After plants have been exposed to salinity, growth is initially reduced due to osmotic stress, but ion-specific effects will occur following longer exposure (e.g., days to weeks or more), and these later effects are more important in determining long-term tolerance to salinity (Greenway and Munns 1980; Munns and Termaat 1986; Munns et al. 1995). Ion-specific effects, such as toxicity to enzymes (Flowers et al. 1977), are minimized in plants that restrict the entry of Na+ and Cl− and also sequester into vacuoles the ions that do enter, thus regulating the concentrations in the cytoplasm (Munns 2005).
Waterlogging causes soil to become low in oxygen. This reduces ATP production in roots, with detrimental effects such as reduced growth, interference with membrane transport, and eventually death of at least a portion of the root system (Barrett-Lennard 2003). Adaptations to waterlogged soils include development of adventitious roots, formation of aerenchyma (enlarged gas spaces that reduce resistance to gas movement within the plant), barriers to radial oxygen loss in roots, and the induction of anaerobic metabolism in those tissues that experience anoxia (Armstrong 1979; Gibbs and Greenway 2003). For many plant species, when salinity and waterlogging occur together, a large increase in Na+ and/or Cl− concentrations in shoots occurs due to increased entry of these ions into oxygen-deficient roots (Barrett-Lennard 2003). Thus, waterlogging can exacerbate the effects of salinity (e.g., maize, Drew et al. 1988). Revegetation of saline and waterlogged soils requires plant species that can tolerate soil oxygen deficiency, or more accurately avoid severe hypoxia by forming aerenchyma to enhance internal oxygen transport, and regulate the transport of Na+ and Cl− to avoid ion toxicity in shoot tissues. This combination of traits does exist in the plant kingdom; for example, in salt marsh species (Drew et al. 1988).
Although L. tenuis has been reported to be both salt and waterlogging tolerant from studies of these stresses individually (references cited above), responses to the combination of the two stresses have not been determined, despite the importance of the interaction of these stresses in many field situations (Barrett-Lennard 2003). This study evaluated the interactive effects of salinity and waterlogging on Na+ and Cl− concentrations in shoots of L. tenuis and on other parameters, such as growth and root porosity. L. tenuis was compared with L. corniculatus, the most widely cultivated Lotus species. L. corniculatus is regarded as less salt (Dear et al. 2003) and flooding (Striker et al. 2005) tolerant than L. tenuis, but comparison of the responses of these two species to combined stresses of salinity and waterlogging has not previously been reported.
Materials and methods
Experiment 1: salt and waterlogging tolerance of Lotus tenuis and Lotus corniculatus cultivars
Four cultivars of L. tenuis (Chaja, Matrero, Larrañaga, Angostura) and three of L. corniculatus (San Gabriel, BL-E, BL-SE) were used to identify within- and between-species variation in responses to root-zone NaCl and stagnant treatments. BL-E and BL-SE are breeding lines provided by Dr John Ayres (NSW Department of Primary Industries, Australia). All other cultivars are commercially available and were provided by INIA, Uruguay. For simplicity, BL-E and BL-SE will also be referred to as ‘cultivars’ in this paper. Scarified seeds were washed with 0.04% (w/v) NaHClO3 and then thoroughly rinsed with deionized (DI) water. Seeds were imbibed for 3 h in aerated 0.5 mM CaSO4 solution in the dark. Imbibed seeds were placed on mesh floating on 10% aerated nutrient solution in 4.5 l pots. The full strength nutrient solution consisted of macronutrients (mM): 0.5 KH2PO4, 3.0 KNO3, 4.0 Ca (NO3)2, 1.0 MgSO4; and micronutrients (μM): 37.5 FeNa3EDTA, 23.0 H3BO3, 4.5 MnCl2, 4.0 ZnSO4, 1.5 CuSO4 and 0.05 MoO3. Seeds were kept in darkness in a 20/15°C (day/night) phytotron for 3 days. The seedlings, still on mesh, were then transferred to 25% nutrient solution and exposed to light. Seven days after imbibition seedlings were transplanted into 50% aerated nutrient solution in 4.5 l pots covered in aluminium foil. Seedlings (six per pot) were held using polystyrene foam in holes (2 cm diameter) in the pot lids. Two weeks after imbibition the solution was changed to full strength. Nutrient solutions were changed weekly and topped up with DI water as required. Average photosynthetically active radiation (PAR) in the phytotron at midday during the experimental period was 1,245 μmol m−2 s−1.
An initial harvest was taken 28 days after imbibition, and four treatments were imposed: aerated (0 mM NaCl); NaCl (aerated, 200 mM NaCl); stagnant [non-aerated and with 0.1% (w/v) agar, 0 mM NaCl] and stagnant-plus-NaCl [non-aerated and with 0.1% (w/v) agar, 200 mM NaCl]. For the plants that were harvested, roots and the stem base were rinsed in DI water and gently blotted dry. For each replicate, the six plants were bulked together and root and shoot fresh and dry masses were measured. For pots allocated to the NaCl treatments, NaCl was added daily in 50 mM increments until the final concentration of 200 mM. The next day hypoxia was imposed in those pots assigned to stagnant and stagnant-plus-NaCl treatments, by bubbling with N2 gas until the dissolved O2 concentration was <10% of air-saturated solution. One day later the nutrient solution in these pots was changed to a stagnant deoxygenated solution containing 0.1% (w/v) agar. This method simulates the decrease in dissolved O2 and increase in ethylene that occurs under waterlogged conditions (Wiengweera et al. 1997). Each treatment × species combination was represented by three replicate pots (six plants per pot) in a completely randomized block design, where blocks were designated according to phytotron position.
The final harvest was taken 56 days after imbibition (28 days after salt treatment commenced). Roots were separated from shoots and shoot fresh masses were recorded. Dead leaves were separated from the rest of the shoot and combined with dead leaves that had senesced and dropped during the treatment period. Roots were separated into main root and lateral roots and a sub-sample taken for porosity measurements. Root porosity was measured based on the method of Raskin (1983), using the equations as modified by Thomson et al. (1990). Newly formed lateral roots about 8 cm in length and closest to the crown were removed from one plant per pot for sectioning to evaluate aerenchyma. Transverse sections were cut approximately 4 cm from the root tip. Root, shoot and dead leaf samples were oven dried at 70°C for 3 days and dry masses were recorded.
Concentrations of Na+, K+ and Cl− were measured in the dried tissue samples. Dried samples were ground to a fine powder. To extract the ions, 10 ml of 0.5 M HNO3 was added to 0.1 g of ground tissue (exact amounts of acid and ground tissue were recorded) in vials and the samples were shaken for 48 h in darkness at 30°C. Diluted extracts were analyzed for Na+, K+ (Jenway PFP7 Flame Photometer) and Cl− (Buchler-Cotlove Chloridometer). Values were validated using a reference plant tissue sample with known concentrations of Na+, K+ and Cl−.
Experiment 2: NaCl dose–responses for Lotus tenuis (cv. Chaja) and Lotus corniculatus (cv. San Gabriel)
This experiment measured responses to salinity of L. tenuis (cv. Chaja) and L. corniculatus (cv. San Gabriel) over a range of NaCl concentrations. Seed germination and growth conditions were the same as in Experiment 1, except the average PAR in the phytotron at midday during the experimental period was 1,613 μmol m−2 s−1. Seven salt treatments were imposed 28 days after imbibition and NaCl was added daily in 50 mM increments until the final concentrations of: 0 (Control), 100, 150, 200, 250, 300 and 400 mM NaCl. Each treatment × species combination was represented by three replicate pots (six plants per pot) in a completely randomized design.
An initial harvest was taken when treatments were imposed (28 days after imbibition), and a final harvest was taken 28 days after NaCl treatments commenced. Roots and the stem base were rinsed in DI water and gently blotted dry. Roots were separated from shoots and shoot fresh masses recorded. Root and shoot samples were oven dried at 70°C for 3 days and dry masses recorded. Concentrations of Na+, K+ and Cl− were measured in shoot samples as described in Experiment 1.
Experiment 3: ion partitioning within shoots of Lotus tenuis (cv. Chaja) and Lotus corniculatus (cv. San Gabriel)
This experiment was done in parallel with Experiment 2; hence all growth conditions were the same as described above. Treatments of 0 and 200 mM NaCl were imposed, each for three replicate pots each of L. tenuis and L. corniculatus. Plants were harvested after 28 days treatment and roots and the stem base were rinsed in DI water and gently blotted dry. The shoots of all six plants per replicate pot were divided into the following components: the three terminal leaflets of each pentafoliate leaf on the main stem were separated from youngest to oldest, the remainder of the shoot was divided into stems and all other green leaves (OGL). Concentrations of Na+, K+ and Cl− were measured in the different shoot components after being oven dried, using the methods described in Experiment 1, except the following volumes of 0.5 M HNO3 were used for the extractions: 50–100 mg samples = 10 ml, 20–50 mg samples = 5 ml and <20 mg samples = 2.5 ml.
Statistical analyses
All statistical analyses used Genstat for Windows 8th Edition (Genstat software, VSN International, Hemel Hempstead, UK). Residuals were checked for normality and homogeneity and no transformations were necessary. ANOVA was used to identify overall significant differences and interactions between cultivars and treatments. Significant differences between treatment means were determined using Tukey’s HSD comparison, paired t-tests or orthogonal contrasts, depending on the complexity of the data set. Unless otherwise stated, the significance level was P ≤ 0.05.
Results
Within- and between-species variability in responses to salinity and waterlogging
Plant growth
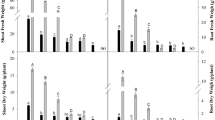
Stagnant treatment reduced the average shoot dry masses of L. tenuis cultivars by 21% and L. corniculatus cultivars by 30% relative to aerated treatment (Table 1). NaCl (200 mM) treatment reduced the shoot dry mass of both species by about 50%. Stagnant-plus-NaCl treatment reduced shoot dry mass of L. tenuis on average by 56%, compared with 67% for L. corniculatus. Shoot dry mass was not significantly different between cultivars within each treatment of aerated, stagnant or NaCl alone. In contrast, in the stagnant-plus-NaCl treatment, shoot dry mass did vary significantly amongst the cultivars of L. tenuis and L. corniculatus (Fig. 1a). For example, stagnant-plus-NaCl treatment reduced the shoot dry mass of L. tenuis cultivar Angostura by 32% less than for L. corniculatus cultivar BL-E.
Responses of shoot (a) and root (b) dry mass of Lotus tenuis and Lotus corniculatus cultivars when grown for 28 days with or without 200 mM NaCl in either aerated or stagnant nutrient solution (Experiment 1). Data are presented as shoot or root dry mass in treatments expressed as a percentage of dry mass in aerated (0 mM NaCl) control. Four cultivars of L. tenuis: Chaja (Ch), Larrañaga (Lr), Matrero (Mt), Angostura (An); and three cultivars of L. corniculatus: San Gabriel (Sg), BL-SE, BL-E; were evaluated. Four weeks after germination seedlings were exposed to one of four treatments for 28 days: aerated (0 mM NaCl), stagnant (0 mM NaCl, non-aerated), NaCl (200 mM NaCl, aerated) and stagnant-plus-NaCl (200 mM NaCl, non-aerated). Values are means of three replicate pots (with six plants per pot) ± standard errors. There were no significant differences between cultivars for NaCl and stagnant treatments, whereas there were significant differences for the stagnant with NaCl treatment based on a Tukey’s HSD test (indicated with different letters; P < 0.05)
Stagnant treatment had no significant effect on root dry mass for any of the genotypes, whereas NaCl alone and stagnant-plus-NaCl treatments caused decreases of 40–70% in root dry mass relative to aerated controls (Fig. 1b). Overall, NaCl treatment reduced the root dry mass of both L. tenuis and L. corniculatus to similar levels (55–60% of aerated), whereas in the stagnant-plus-NaCl treatment the root dry mass of L. tenuis genotypes were reduced less than for those of L. corniculatus (61% versus 52% of aerated). Genotypes within each species only showed significant differences for the stagnant-plus-NaCl treatment (Fig. 1b). Even within L. tenuis, the root dry mass varied from 28% (Larrañaga) to 60% (Angostura) of the values in aerated non-saline controls.
Root porosity
Main roots and lateral roots had similar porosities, therefore only results for main roots are presented. Both stagnant treatments increased root porosity compared to plants in aerated treatments, although the extent of this increase varied for the cultivars (Fig. 2). Overall, L. tenuis had a higher average root porosity compared with L. corniculatus for all treatments (Table 1). There was significant variability within and between species for root porosity, although the variability was greater within L. tenuis. Chaja and Angostura had similar root porosities and these were up to twofold greater than in Larrañaga and Matrero for all treatments (Fig. 2). Within L. corniculatus, the cultivar BL-SE had the highest root porosity in all treatments and this was greatest in the stagnant non-saline treatment (22 ± 1%).
Porosity of main roots of Lotus tenuis and Lotus corniculatus cultivars when grown for 28 days with or without 200 mM NaCl in either aerated or stagnant nutrient solution (Experiment 1). Four cultivars of L. tenuis: Chaja (Ch), Larrañaga (Lr), Matrero (Mt), Angostura (An); and three cultivars of L. corniculatus: San Gabriel (Sg), BL-SE, BL-E; were evaluated. Four-week-old plants were treated for 28 days with one of the following treatments: aerated (0 mM NaCl), stagnant (0 mM NaCl, non-aerated), NaCl (200 mM NaCl, aerated) and stagnant-plus-NaCl (200 mM NaCl, non-aerated). Values are means of three replicate pots (with six plants per pot) ± standard errors. Cultivars with a different letter for a particular treatment were significantly different based on a Tukey’s HSD test (P < 0.05)
The increased root porosity in stagnant treatments was likely caused by aerenchyma formation. All cultivars had aerenchyma present to varying degrees under stagnant conditions and those with greater root porosity (Fig. 2) generally had more extensive aerenchyma (not shown). In aerated solutions, the cultivars Larrañaga, Matrero (L. tenuis), San Gabriel and BL-E (L. corniculatus) had no aerenchyma present in sections taken at 4 cm behind the root tip, which was consistent with the low root porosities (<10%) measured for these cultivars when in aerated solution.
Shoot ion concentrations
The 200-mM NaCl treatment increased shoot Na+ concentrations to as high as 2,000 μmol g−1 DM (Fig. 3a). The stagnant-plus-NaCl treatment further increased shoot Na+ concentration compared with aerated NaCl treatment, and this increase ranged from 20% to over 100%. Cultivars of L. tenuis generally had lower shoot Na+ than those of L. corniculatus (Table 1). For stagnant-plus-NaCl, the average shoot Na+ concentration for L. corniculatus cultivars was 18% higher than for those of L. tenuis (Table 1). Stagnant-plus-NaCl treatment resulted in much greater between-, and within-, species variability for shoot Na+ concentrations, compared with plants in the aerated NaCl treatment. For example, within L. tenuis, shoot Na+ concentration for Chaja was almost half the concentration of Larrañaga. Na+ and Cl− concentrations in shoots of plants in non-saline aerated or stagnant treatments were low (<4 μmol g−1 DM for Na+ and <80 μmol g−1 DM for Cl−) and did not differ amongst the cultivars (data not shown).
Shoot Na+ (a) and Cl− (b) concentrations of Lotus tenuis and Lotus corniculatus cultivars when grown for 28 days with or without 200 mM NaCl in either aerated or stagnant nutrient solution (Experiment 1). Four cultivars of L. tenuis: Chaja (Ch), Larrañaga (Lr), Matrero (Mt), Angostura (An); and three cultivars of L. corniculatus: San Gabriel (Sg), BL-SE, BL-E; were evaluated. Four-week-old plants were treated for 28 days with one of the following treatments: aerated (0 mM NaCl), stagnant (0 mM NaCl, non-aerated), NaCl (200 mM NaCl, aerated) and stagnant-plus-NaCl (200 mM NaCl, non-aerated). Values are means of three replicate pots (with six plants per pot) ± standard errors. Cultivars with a different letter for a particular treatment were significantly different based on a Tukey’s HSD test (P < 0.05)
NaCl treatment increased shoot Cl− concentrations up to about 1,600 μmol g−1 DM (Fig. 3b). Stagnant-plus-NaCl treatment significantly increased shoot Cl− concentrations compared to the aerated NaCl treatment for all cultivars, and only for Chaja (L. tenuis) and BL-SE (L. corniculatus) was this increase in shoot Cl− <25%. On average, the shoot Cl− concentration of L. tenuis was about 20% less than L. corniculatus, for NaCl and stagnant-plus-NaCl treatments (Table 1). There was significant within- and between-species variability in shoot Cl− concentrations for aerated NaCl and stagnant-plus-NaCl treatments. The cultivars Chaja, Angostura (L. tenuis) and BL-SE (L. corniculatus) had much lower shoot Cl− concentrations compared with the other cultivars (Fig. 3b). Within L. tenuis, shoot Cl− concentrations ranged from 727 to 1,662 μmol g−1 DM in aerated NaCl treatment. The variability within species was even greater for stagnant-plus-NaCl treatment, with up to a threefold difference between cultivars within each species.
Shoot K+ concentrations were decreased by all treatments compared with aerated controls (Fig. 4). On average, L. tenuis and L. corniculatus had similar shoot K+ concentrations within all treatments (Table 1). For plants in aerated non-saline solution, there were no significant differences in shoot K+ concentrations between cultivars; and all averaged over 1,500 μmol g−1 DM (Fig. 4). Stagnant treatment reduced shoot K+ concentrations by more than 30% compared with aerated controls for Larrañaga, Matrero (L. tenuis) and BL-E (L. corniculatus), while for the other cultivars shoot K+ was similar to that in aerated controls. NaCl treatment reduced shoot K+ by about 50% compared with that in aerated controls for most cultivars, except for BL-SE (L. corniculatus), which was severely reduced to only one-third of that in aerated controls. In contrast to Na+ and Cl−, shoot K+ concentrations did not vary significantly between cultivars for stagnant-plus-NaCl treatment.
Shoot K+ concentrations of Lotus tenuis and Lotus corniculatus cultivars when grown for 28 days with or without 200 mM NaCl in either aerated or stagnant nutrient solution (Experiment 1). Four cultivars of L. tenuis: Chaja (Ch), Larrañaga (Lr), Matrero (Mt), Angostura (An); and three cultivars of L. corniculatus: San Gabriel (Sg), BL-SE, BL-E; were evaluated. Four-week-old plants were treated for 28 days with one of the following treatments: aerated (0 mM NaCl), stagnant (0 mM NaCl, non-aerated), NaCl (200 mM NaCl, aerated) and stagnant-plus-NaCl (200 mM NaCl, non-aerated). Values are means of three replicate pots (with six plants per pot) ± standard errors. Cultivars with a different letter for a particular treatment were significantly different based on a Tukey’s HSD test (P < 0.05). There was no significant difference between cultivars within aerated or stagnant with NaCl treatments
Concentrations of Na+, Cl− and K+ were also measured in dead leaves collected from L. tenuis and L. corniculatus cultivars during the treatment period. As expected, Na+ and Cl− concentrations in the dead leaves were higher compared with those in the whole shoots, but only slightly, and the differences between the cultivars followed the same trends as whole shoots (therefore data not shown).
In summary, the greatest variability among cultivars in shoot ion concentrations was for: Cl− in NaCl treatment and in stagnant-plus-NaCl treatment; Na+ in stagnant-plus-NaCl treatment; and K+ for plants in stagnant treatment.
Responses of Lotus tenuis (cv. Chaja) and Lotus corniculatus (cv. San Gabriel) to increasing NaCl concentrations
Based on the results of Experiment 1, L. tenuis cultivar Chaja and L. corniculatus cultivar San Gabriel were selected for a more detailed assessment of salt tolerance in a dose-response experiment, with treatments up to 400 mM NaCl. These cultivars were chosen because Chaja had lower shoot Na+ and Cl− concentrations than San Gabriel.
Plants continued to grow and produce new leaves even after 28 days at 400 mM NaCl, indicating these cultivars of both species are very salt tolerant. At 150 and 200 mM NaCl, shoot dry mass relative to plants at 0 mM NaCl was reduced significantly less for L. tenuis compared with L. corniculatus (Fig. 5a). For example, at 200 mM NaCl, shoot dry mass was reduced by about 30% for L. tenuis and 50% for L. corniculatus. However, at higher NaCl concentrations, shoot dry mass was equally reduced for both cultivars. NaCl treatment reduced root dry mass less than shoot dry mass (Fig. 5a, b). L. tenuis root dry mass was reduced by about 20% less than that of L. corniculatus at 150 and 200 mM NaCl (Fig. 5b). At 400 mM NaCl, the roots of both species were equally reduced by about 55% relative to plants at 0 mM NaCl.
Responses of shoot (a) and root (b) dry mass of Lotus tenuis (cv. Chaja) and Lotus corniculatus (cv. San Gabriel) treated for 28 days with 0–400 mM NaCl (Experiment 2). Four-week-old plants were treated with one of seven NaCl concentrations from 0 to 400 mM, for 28 days. Treatments were applied in daily increments of 50 mM NaCl until the final concentration was reached. Values are means of three replicate pots (with six plants per pot) ± standard errors. Asterisk indicates L. tenuis and L. corniculatus were significantly different based on orthogonal contrasts (P < 0.05)
Shoot concentrations of Na+, K+ and Cl− were measured in Chaja (L. tenuis) and San Gabriel (L. corniculatus) after 28 days treatment with 0–400 mM NaCl. As expected, Na+ and Cl− shoot concentrations increased and K+ decreased as the external NaCl concentration increased (Fig. 6). Shoot Cl− concentrations of L. tenuis were significantly less than for L. corniculatus at all NaCl levels except 0 mM (Fig. 6a). The difference between the two species increased at greater external NaCl concentrations, ranging from differences of 40% to over 100% from 100 to 400 mM NaCl. In contrast to Cl−, there was no significant difference in shoot Na+ concentrations between L. tenuis and L. corniculatus until the external NaCl concentration reached 300 and 400 mM (Fig. 6b). At 400 mM NaCl, shoot Na+ concentrations of L. corniculatus were 30% higher than for L. tenuis. The shoot Cl−/Na+ ratio reflects the large difference in Cl− concentration between the species. For L. tenuis, shoot Cl−/Na+ ranged from 0.4 to 0.5 for all treatments and this was significantly less than in L. corniculatus, which ranged from 0.8 to 0.9. There were no significant differences in shoot K+ concentrations between the two species (Fig. 6c) and hence the K+/Na+ ratio at all NaCl levels followed the differences in shoot Na+ concentrations.
Shoot concentrations of Cl− (a), Na+ (b) and K+ (c) for Lotus tenuis (cv. Chaja) and Lotus corniculatus (cv. San Gabriel) treated for 28 days with 0–400 mM NaCl (Experiment 2). Four-week-old plants were treated with one of seven NaCl concentrations from 0 to 400 mM, for 28 days. Treatments were applied in daily increments of 50 mM NaCl until the final concentration was reached. Values are means of three replicate pots (with six plants per pot) ± standard errors. Asterisk indicates L. tenuis and L. corniculatus were significantly different based on orthogonal contrasts (P < 0.05)
Ion partitioning in different-aged leaflets at 200 mM NaCl
Experiment 2 showed that at 200 mM NaCl, the dry masses of Chaja (L. tenuis) and San Gabriel (L. corniculatus) were both reduced to approximately half of those in aerated controls, and these two cultivars differed in shoot Cl− concentrations. Possible differences in ion partitioning were therefore evaluated in individual leaflets along the main stem. At 0 mM NaCl, there were no differences between the two species and no significant trends between different shoot components for all three ions measured (data not shown). At 200 mM NaCl, for both species, there were significant increases in Na+ and Cl− concentrations as leaflet age increased; while for K+ there was no difference between leaflets of different ages (Fig. 7). Cl− concentrations were up to twofold higher in L. corniculatus than in L. tenuis for all shoot components (Fig. 7a), consistent with the whole shoot results (Fig. 6a). In contrast to Cl−, there was no significant difference between L. tenuis and L. corniculatus for Na+ concentrations in any of the individual leaflets, stems or OGL; again consistent with the whole shoot results (Fig. 6b). Na+ concentrations increased from under 200 μmol g−1 DM in the newly emerged leaflets to over 3,500 μmol g−1 DM in the older leaflets (Fig. 7b). Interestingly, L. corniculatus had 25–85% higher K+ concentrations than L. tenuis in the individual leaflets, despite no significant difference between the species in whole shoot or stem K+ concentrations.
Concentrations of Cl− (a), Na+ (b) and K+ (c) in different-aged leaflets and other tissues for Lotus tenuis (cv. Chaja) and Lotus corniculatus (cv. San Gabriel) treated for 28 days with 200 mM NaCl (Experiment 3). Four-week-old plants were treated with 200 mM NaCl for 28 days. Ion concentrations were measured in whole shoot, stems, individual main stem leaflets (youngest L1 to oldest L14) and other green leaves. Values are means of three replicate pots (with six plants per pot) ± standard errors. Asterisk indicates L. tenuis and L. corniculatus were significantly different based on paired t-tests (P < 0.05)
Discussion
This study showed that not only is L. tenuis highly salt tolerant (still growing after 28 days at 400 mM NaCl) and tolerant of root-zone oxygen deficiency, but it is also tolerant of these stresses when combined. While there was significant variability within both L. tenuis and L. corniculatus for growth responses and traits associated with salt tolerance (regulation of shoot ion concentrations) and waterlogging tolerance (root porosity), overall L. tenuis cultivars were more tolerant.
A major finding in this study was the large differences in shoot Cl− concentrations between L. tenuis (cv. Chaja) and L. corniculatus (cv. San Gabriel). L. tenuis accumulated about half as much Cl− in its shoots compared with L. corniculatus at NaCl concentrations from 100 to 400 mM (Fig. 6a). This difference occurred in old leaflets, young leaflets and stems (Fig. 7a). These differences in Cl− were in contrast to Na+ concentrations, which only differed between the cultivars at the highest external NaCl concentrations (300 and 400 mM). While Na+ ‘exclusion’ is an important determinant of salt tolerance (Tester and Davenport 2003; Munns 2005), Cl− ‘exclusion’ from shoots is also critical for plant survival in saline conditions (Greenway and Munns 1980). Cl− ‘exclusion’ has been linked to salt tolerance in many species, particularly dicots; including Trifolium (Winter 1982; Rogers et al. 1997b), Lupinus (van Steveninck et al. 1982), Citrus (Romero-Aranda et al. 1998; Moya et al. 2003) and Glycine (Luo et al. 2005). Therefore, the low shoot concentration of Cl− in L. tenuis cultivar Chaja could be an important trait contributing to salt tolerance in this cultivar.
Although there was no difference between L. tenuis (cv. Chaja) and L. corniculatus (cv. San Gabriel) cultivars in shoot Na+ concentrations when grown at 200 mM NaCl in aerated solution; when in the stagnant-plus−200 mM NaCl, L. corniculatus had a twofold higher shoot Na+ concentration than L. tenuis (Fig. 3). Cl− concentration was also about twice as high in L. corniculatus shoots compared with L. tenuis for both aerated NaCl and stagnant-plus-NaCl treatments. These increases in shoot Na+ and Cl− concentrations might explain the sensitivity of L. corniculatus to combined salt and waterlogging stresses. Root porosity (i.e., aerenchyma formation) was lower for L. corniculatus than for L. tenuis. Lower porosity would result in less O2 being available to roots (cf. Armstrong 1979). Oxygen deficiency in L. corniculatus roots would be expected to impair respiration, leaving less energy (ATP) available for ion transport and thereby reduce Na+ and Cl− ‘exclusion’ from shoots (Barrett-Lennard 2003). A breakdown in Na+ and Cl− ‘exclusion’ caused by the combination of salinity and waterlogging has been found in other species less tolerant to waterlogging, for example wheat (Akhtar et al. 1994; Barrett-Lennard et al. 1999), maize (Drew et al. 1988), subterranean clover (Rogers and West 1993), Banksia attenuata (Carter et al. 2006) and Eucalyptus globulus (Marcar 1993).
The ability to ‘exclude’ toxic ions is likely to be correlated with salt tolerance (e.g., Tester and Davenport 2003). Measurements of Na+ and Cl− in shoots revealed that L. tenuis accumulates less Cl− than L. corniculatus at 0–400 mM NaCl and less Na+ at external NaCl concentrations above 300 mM. However, this was not consistently correlated with tolerance, as there was no significant difference between L. tenuis and L. corniculatus in relative reductions in shoot dry mass (% of 0 mM NaCl) for salt treatments >200 mM NaCl (Fig. 5). For stagnant-plus-NaCl treatment, differences in shoot Na+ and Cl− concentrations (Fig. 3) were also not always correlated with differences in dry mass between the L. tenuis and L. corniculatus cultivars (Fig. 1), although overall L. tenuis cultivars had higher dry mass (Table 1). The experiments in this study measured dry mass after 28 days of salt treatment, which may not be sufficient time for genotypic differences in growth to be significant (cf. Munns et al. 1995), particularly for perennial species. Similarly, when two cultivars of L. tenuis varying in salt tolerance were grown at 200 mM for 28 days, there was no significant difference in growth between the cultivars (Sannazzaro et al. 2006). A longer time frame is often required for differences in rates of ion accumulation to be reflected in growth differences, particularly between closely related cultivars (e.g., wheat, Munns et al. 1995).
Lotus tenuis is regarded as more salt tolerant (Dear et al. 2003) and waterlogging tolerant (Striker et al. 2005) than L. corniculatus. The present study found these reputations were generally true, but also showed large variability within both L. tenuis and L. corniculatus. Variability in salt tolerance has been previously observed in L. tenuis and L. corniculatus cultivars (Schachtman and Kelman 1991). Variability in flooding tolerance has been measured for L. tenuis (Vignolio et al. 1999), which has also been reported as more waterlogging tolerant than L. corniculatus (Striker et al. 2005). However, to our knowledge, no previous studies have compared the responses of these two species to combined salt and waterlogging stress. The present study showed that overall, L. tenuis cultivars were more tolerant than L. corniculatus cultivars to combined salt and waterlogging treatments based on dry matter production relative to aerated treatment. The higher porosity of roots, and lower Cl− and Na+ in shoots, of L. tenuis grown in stagnant-plus-NaCl treatment, is likely to contribute to its better tolerance of combined salt and waterlogging, compared with L. corniculatus. Under aerated NaCl treatment (0–400 mM), the interesting finding that L. tenuis (Chaja) accumulated half as much shoot Cl− (while Na+ was the same), compared with L. corniculatus (San Gabriel), indicates better Cl− ‘exclusion’ as an important trait for salt tolerance in Lotus species.
References
Akhtar J, Gorham J, Qureshi RH (1994) Combined effect of salinity and hypoxia in wheat (Triticum aestivum L.) and wheat-Thinopyrum amphiploids. Plant Soil 166:47–54
Armstrong W (1979) Aeration in higher plants. Adv Bot Res 7:225–332
Ayers AD (1948) Salt tolerance of birdsfoot trefoil. J Am Soc Agron 40:331–334
Barrett-Lennard E (2003) The interaction between waterlogging and salinity in higher plants: causes, consequences and implications. Plant Soil 253:35–54
Barrett-Lennard EG, Ratingen P, van Mathie MH (1999) The developing pattern of damage in wheat (Triticum aestivum L.) due to the combined stresses of salinity and hypoxia: experiments under controlled conditions suggest a methodology for plant selection. Aust J Agric Res 50:129–136
Carter JL, Colmer TD, Veneklaas EJ (2006) Variable tolerance of wetland tree species to combined salinity and waterlogging is related to regulation of ion uptake and production of organic solutes. New Phytol 169:123–134
Dear BS, Moore GA, Hughes SJ (2003) Adaptation and potential contribution of temperate perennial legumes to the southern Australian wheatbelt: a review. Aust J Exp Agric 43:1–18
Drew MC, Guenther J, Lauchli A (1988) The combined effects of salinity and root anoxia on growth and net Na+ and K+-accumulation in Zea mays grown in solution culture. Ann Bot 61:41–53
FAO (2005) Global network on integrated soil management for sustainable use of salt-affected soils. FAO Land and Plant Nutrition Management Service, Rome, Italy. http://www.fao.org/ag/agl/agll/spush
Flowers TJ, Troke PF, Yeo AR (1977) The mechanism of salt tolerance in halophytes. Annu Rev Plant Physiol 28:89–121
George RJ, Bennet D, Speed R (2004) Salinity management—the case for focusing on wheatbelt valleys. Salinity solutions conference, Bendigo, 2–5 August 2004
Gibbs J, Greenway H (2003) Mechanisms of anoxia tolerance in plants I Growth, survival and anaerobic catabolism. Funct Plant Biol 30:1–47
Greenway H, Munns R (1980) Mechanisms of salt tolerance in non-halophytes. Ann Rev Plant Physiol 31:149–190
Kade M, Pagani EA, Mendoza RE (2003) Phosphorus utilization efficiency in populations of narrow-leaf birdsfoot trefoil. Commun Soil Sci Plant 34:271–284
Luo Q, Bingjun Y, Liu Y (2005) Differential selectivity to chloride and sodium ions in seedlings of Glycine max and G. soja under NaCl stress. J Plant Physiol 162:1003–1012
Maathuis FJM, Amtmann A (1999) K+ nutrition and Na+ toxicity: the basis of cellular K+/Na+ ratios. Ann Bot 84:123–133
Marcar NE (1993) Waterlogging modifies growth, water use and ion concentrations in seedlings of salt-treated Eucalyptus camaldulensis, E. tereticornis, E. robusta and E. globulus. Aust J Plant Physiol 20:1–13
Mendoza R, Escudero V, Garcia I (2005) Plant growth, nutrient acquisition and mycorrhizal symbioses of a waterlogging tolerant legume (Lotus glaber Mill.) in a saline-sodic soil. Plant Soil 275:305–315
Moya JL, Gomez-Cadenas A, Primo-Millo E, Talon M (2003) Chloride absorption in salt-sensitive Carrizo citrange and salt-tolerant Cleopatra mandarin citrus rootstocks is linked to water use. J Exp Bot 54:825–833
Munns R (1993) Physiological processes limiting plant growth in saline soils: some dogmas and hypotheses. Plant Cell Environ 16:15–24
Munns R (2005) Genes and salt tolerance: bringing them together. New Phytol 167:645–663
Munns R, Termaat A (1986) Whole-plant responses to salinity. Aust J Plant Physiol 13:143–160
Munns R, Schachtman DP, Condon AG (1995) The significance of a two-phase growth response to salinity in wheat and barley. Aust J Plant Physiol 13:143–160
Raskin I (1983) A method for measuring leaf density, thickness and internal gas. Hortscience 18:698–699
Rogers ME, West DW (1993) The effects of rootzone salinity and hypoxia on shoot and root growth in Trifolium species. Ann Bot 72:503–509
Rogers ME, Noble CL, Pederick RJ (1997a) Identifying suitable forage legume species for saline areas. Aust J Exp Agric 37:639–645
Rogers ME, Noble CL, Halloran GM, Nicolas ME (1997b) Selecting for salt tolerance in white clover (Trifolium repens): chloride ion exclusion and its heritability. New Phytol 135:645–654
Romero-Aranda R, Moya JL, Tadeo FR, Legaz F, Primo-Millo E, Talon M (1998) Physiological and anatomical disturbances induced by chloride salts in sensitive and tolerant citrus: beneficial and detrimental effects of cations. Plant Cell Environ 21:1243–1253
Sannazzaro AI, Ruiz OA, Alberto E, Menendez AB (2006) Presence of different arbuscular mycorrhizal infection patterns in roots of Lotus glaber plants growing in the Salado River basin. Mycorrhiza 14:279–287
Schachtman DP, Kelman WM (1991) Potential for Lotus germplasm for the development of salt, aluminium and manganese tolerant pasture plants. Aust J Agric Res 42:139–149
Smedema LK, Shiati K (2002) Irrigation and salinity: a perspective review of the salinity hazards of irrigation development in the arid zone. Irrigation Drainage Syst 16:161–174
Striker GG, Insausti P, Grimoldi AA, Ploschuk EL, Vasellati V (2005) Physiological and anatomical basis of differential tolerance to soil flooding of Lotus corniculatus L. and Lotus glaber Mill. Plant Soil 276:301–311
Tester M, Davenport R (2003) Na+ tolerance and Na+ transport in higher plants. Ann Bot 91:503–527
Thomson CJ, Armstrong W, Waters I, Greenway H (1990) Aerenchyma formation and associated oxygen movement in seminal and nodal roots of wheat. Plant Cell Environ 13:395–403
Van Steveninck RFM, Van Steveninck ME, Stelzer R, Lauchli A (1982) Studies on the distribution of Na and Cl in two species of lupin (Lupinus luteus and Lupinus angustifolius) differing in salt tolerance. Physiol Plant 56:465–473
Vignolio OR, Fernandez ON, Maceira NO (1999) Flooding tolerance in five populations of Lotus glaber Mill (Syn. Lotus tenuis Waldst. et. Kit.). Aust J Agric Res 50:555–559
Wiengweera A, Greenway H, Thomson CJ (1997) The use of agar nutrient solution to simulate lack of convection in waterlogged soils. Ann Bot 80:115–123
Winter E (1982) Salt tolerance of Trifolium alexandrinum L. II. Ion balance in relation to its salt tolerance. Aust J Plant Physiol 9:227–237
Acknowledgments
NLT is on a Grains Research and Development Corporation PhD scholarship with supplementary research funding provided by the CRC for Plant-based Management of Dryland Salinity and The AW Howard Memorial Trust. Special thanks to Dr Bo Wang for comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Teakle, N.L., Real, D. & Colmer, T.D. Growth and ion relations in response to combined salinity and waterlogging in the perennial forage legumes Lotus corniculatus and Lotus tenuis . Plant Soil 289, 369–383 (2006). https://doi.org/10.1007/s11104-006-9146-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-006-9146-8