Abstract
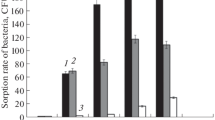
The effect of initial inoculum density of the antagonistic bacterial strains Pseudomonas fluorescens B5 and Pseudomonas corrugata 2140 (103 to 108 CFU per seed pellet) on sugar beet seedling colonisation, in situ bioluminescence and antagonistic activity towards Pythium ultimum was investigated. Populations of the bacteria colonising sugar beet root systems approached an apparent carrying capacity of 105 to 106 CFU per plant after 12 d growth, irrespective of inoculum density. This meant an up to 320-fold population increase at low inoculum densities and a decrease at high densities. Population densities of both bacteria and their corresponding in situ bioluminescence (resulting from luciferase enzyme expression from the inserted luxAB genes) reached highest levels in the hypocotyl region and in the upper root region 0–20 mm below seed level (104–106 CFU/cm section, 101–103 RLU/cm section) and decreased with root depth. In situ bioluminescence, which indicates physiological activity, was measurable at lowest antagonist initial inoculum density (103 CFU per seed pellet) and did not increase significantly with increasing inoculum density. Bioluminescence was also significantly correlated with population density. For Pseudomonas fluorescens B5, the total population size per plant and downward colonisation of the root (below 40 mm depth) increased significantly with antagonist inoculum density applied to the seeds. For Pseudomonas corrugata 2140, no significant influence of initial inoculum density on root colonisation was observable. Survival and dry weight of sugar beet seedlings in Pythium infested soil increased significantly with increasing inoculum density of Pseudomonas fluorescens B5, whereas for Pseudomonas corrugata 2140, initial densities of 104 to 106 CFU per seed resulted in maximal survival of plants.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- CFU:
-
colony forming units
- RLU:
-
relative luminescence units
References
Bahme J B and Schroth M N 1987 Spatial temporal colonization pattern of a rhizobacterium on underground organs of potato. Phytopathology 77, 1093–1100.
Bandoni R J and Koske R E 1974 Monolayers and microbial dispersal. Science 183, 1079–1080.
Beauchamp C J, Kloepper J W and Lemke P A 1993 Luminometric analysis of plant root colonization by bioluminescent pseudomonads. Can. J. Microbiol. 39, 434–441.
Berger F, Hong Li, White D, Fraser R and Leifert C 1996 Effect of pathogen inoculum, antagonist density and plant species on biological control of Phytophtora and Pythium damping-off in high-humidity fogging glasshouses. Phytopathology 86, 428–433.
Bowen G D and Rovira A D 1976 Microbial colonization of plant roots. Ann. Rev. Phytopathol. 14, 121–144.
Bull C T, Weller D M and Thomashow L S 1991 Relationship between root colonization and suppression of Gaeumannomyces graminis var. triticii by Pseudomonas fluorescens strain 2–79. Phytopathology 81, 954–958.
Dowling D N and O’Gara F 1994 Metabolites of Pseudomonas involved in biocontrol of plant disease. Trends Biotechnol. 12, 133–141.
Faiz S M A and Weatherly P E 1982 Root contraction in transpiring plants. New Phytol. 92, 333–343.
Frey-Klett P, Churin J-L, Pierrat J-C, Garbaye J 1999 Dose effect in the dual inoculum of an ectomycorrhizal fungus and a mycorrhiza helper bacterium in two forest nurseries. Soil Biol. Biochem. 31, 1555–1562.
Fukui R, Schroth M N, Hendson M and Hancock J G 1994a Interaction between strains in sugar beet spermospheres and their relationship to pericarp colonization by Pythium ultimum in soil. Phytopathology 84, 1322–1330.
Fukui R, Schroth M N, Hendson M, Hancock J G and Firestone M K 1994b Growth Patterns and metabolic activity of pseudomonads in sugar beet spermospheres: relationship to pericarp colonization by Pythium ultimum. Phytopathology 84, 1331–1338.
Fukui R, Poinar E I, Bauer P H, Schroth M N, Hendson M, Wang X-L and Hancock J G 1994c Spatial colonization patterns and interactions of bacteria on inoculated sugar beet seed. Phytopathology 84, 1338–1345.
Grant F A, Prosser J I, Killham K and Glover L A 1992 Luminescence based detection of Erwinia carotovora subsp. carotovora in soil. Soil Biol. Biochem. 24, 961–967.
Heupel M 1992 Biologische Bekämpfung bodenbürtiger Wurzelbranderreger der Zuckerrübe (Beta vulgaris L. ssp. vulgaris var. altissima DOELL) durch den Einsatz mikrobieller Antagonisten. Ph.D. thesis. University of Göttingen.
Howie W J, Cook R J and Weller D M 1987 Effects of soil matric potential and cell motility on wheat root colonization by fluorescent Pseudomonas suppressive on take all. Phytopathology 77, 286–292.
Huck M G, Klepper B and Taylor, H M 1970 Diurnal variations in root diameter. Plant Physiol. 45, 529–530.
Johnson K B 1994 Dose response relationships and inundative biological control. Phytopathology 84, 780–784.
Katarýan B T and Torgashova G G 1976 Spectrum and activity of the herbicidal effect of 2,4-Diacetylphloroglucinol. Dokl. Acad. Nauk. Arm. SSR 63, 109–112.
Killham K S and Yeomans C V 2001 Rhizosphere carbon flow measurement and implications: from isotopes to reporter genes. Plant Soil 232, 91–96.
Knox, O G G 2000 Exploiting nitrate respiration to optimise antagonistic control of root disease in soil. Thesis submitted for the degree of Doctor of Philosophy, University of Aberdeen.
Larkin R P and Fravel D R 1999 Mechanisms of action and dose response relationships governing biological control of Fusarium wilt of tomato by nonpathogenic Fusarium spp. Phytopathology 89, 1152–1161.
Liddell C M and Parke J L 1989 Enhanced colonization of pea tap-roots by a fluorescent pseudomonad biocontrol agent by water infiltration into soil. Phytopathology 79, 1327–1332.
Loper J E, Haack C A and Schroth M N 1985 Population dynamics of soil pseudomonads in the Rhizosphere of potato (Solanum tuberosum L.) Appl. Env. Microbiol. 49, 416–422.
De Lorenzo V, Herrero M, Jakubzik U and Timmis K N 1990 Mini-Tn5 transposon derivatives for insertion mutagenesis, promoter probing, and chromosomal insertion of cloned DNA in gram-negative eubacteria. J. Bacteriol. 172, 6568–6572.
Martin F N and Loper J E 1999 Soilborne diseases caused by Pythium spp. Ecology, epidemiology and prospects for biological control. Crit. Rev. Plant Sci. 18, 111–181.
Maurhofer M, Kell C, Schnider U, Volsard C, Haas D and Defago G 1992 Influence of enhanced antibiotic production in Pseudomonas fluorescens strain CHA0 on its disease suppressive capacity. Phytopathology 82, 190–195.
Meikle A, Killham K, Prosser J I and Glover L A 1992 Luminescence measurement of population activity of genetically modified Pseudomonas fluorescens in the soil. FEMS Microbiol. Lett. 99, 217–220.
Montesinos E and Bonaterra A 1996 Dose-Response Models in biological control of plant pathogens: an empirical verification. Phytopathology 86, 464–472.
Osburn R M and Schroth M N 1984 Dynamics of Pythium ultimum infection and damping-off of sugar beets and biocontrol by a strain of Pseudomonas putida. (abstract) Phytopathology 74, 835.
Parke J l, Moen R, Rovira A D and Bowen G D 1986 Soil water flow affects the rhizoshere distribution of a seed borne biocontrol agent, Pseudomonas fluorescens. Soil Biol. Biochem. 18, 583–588.
Raaijmakers J M, Bonsall R F and Weller D M 1999 Effect of population density of Pseudomonas fluorescens on production of 2,4-diacetylphloroglucinol in the rhizosphere of wheat. Phytopathology 89, 470–475.
Raaijmakers J M, Leeman M, van Oerschot M M P, van der Sluis I, Schippers B and Bakker, P A H M 1995 Dose response relationships in biological control of Fusarium wilt of radish by Pseudomonas ssp. Phytopathology 85, 1075–1081.
Rattray E A S, Prosser J I, Glover L A and Killham K 1995 Characterization of rhizosphere colonization by luminescent Enterobacter cloacae at the population and single cell levels. Appl. Env. Microbiol. 61, 2950–2957.
Ramos C, Molina L, Mølbak L, Ramos J L and Molin S 2000 A bioluminescent derivative of Pseudomonas putida KT2440 for deliberate release into the environment. FEMS Microbiol. Ecol. 34, 91–102.
Russell C N 1996 Lux gene reporting of the interaction between Gaeumannomyces graminis and an antagonistic pseudomonad in the wheat rhizosphere. Thesis submitted for the degree of Doctor of Philosophy, University of Aberdeen.
Ryder M H and Borrett M A 1991 Root colonization by non-fluorescent pseudomonads used for the control of wheat take-all. In Plant Growth-Promoting Rhizobacteria-Progress and Prospects. Eds. C Keel, B Koller and G Défago. pp. 302–307. International Organization for Biological and Integrated Control of Noxious Animals and Plants IOBC/WPRS Bulletin XIV/8.
Ryder, M H and Rovira A D 1993 Biological control of take-all of glasshouse-grown wheat using strains of Pseudomonas corrugata isolated from wheat field soil. Soil Biol. Biochem. 25, 311–320.
Shaw J J, Dane F, Geiger D and Kloepper J W 1992 Use of bioluminescence for the detection of genetically engineered microorganisms released into the environment. Appl. Env. Microbiol. 58, 267–273.
Sørensen J, Jensen L E and Nybroe O 2001 Soil and rhizosphere as habitats for Pseudomonas inoculants: new knowledge on distribution, activity and physiological state derived from micro-scale and single-cell studies. Plant Soil 232, 97–108.
Suslow T V and Schroth M N 1982 Rhizobacteria on sugar beets: Effects of seed application and root colonization on yield. Phytopathology 72, 199–206.
Trevors J T, van Elsas J D, van Overbeek, L S and Starodub M E 1990 Transport of a genetically engineered Pseudomonas fluorescens strain through a soil microcosm. Appl. Env. Microbiol. 56, 401–408.
White D, Leifert C, Ryder M H and Killham K 1996 Lux gene technology- A strategy to optimize biological control of soil borne diseases. New Phytol. 133, 173–181.
de Weger L A, Dunbar P, Mahaffee W F, Lugtenberg B J J and Sayler G S 1991 Use of bioluminescence markers to detect Pseudomonas spp. in the rhizosphere. Appl. Env. Microbiol. 57, 3641–3644.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schmidt, C.S., Agostini, F., Leifert, C. et al. Influence of inoculum density of the antagonistic bacteria Pseudomonas fluorescens and Pseudomonas corrugata on sugar beet seedling colonisation and suppression of Pythium damping off. Plant Soil 265, 111–122 (2004). https://doi.org/10.1007/s11104-005-8943-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-005-8943-9