Abstract
To investigate the possible mechanisms of glutathione reductase (GR) in protecting against oxidative stress, we obtained transgenic tobacco (Nicotiana tabacum) plants with 30–70% decreased GR activity by using a gene encoding tobacco chloroplastic GR for the RNAi construct. We investigated the responses of wild type and transgenic plants to oxidative stress induced by application of methyl viologen in vivo. Analyses of CO2 assimilation, maximal efficiency of photosystem II photochemistry, leaf bleaching, and oxidative damage to lipids demonstrated that transgenic plants exhibited enhanced sensitivity to oxidative stress. Under oxidative stress, there was a greater decrease in reduced to oxidized glutathione ratio but a greater increase in reduced glutathione in transgenic plants than in wild type plants. In addition, transgenic plants showed a greater decrease in reduced ascorbate and reduced to oxidized ascorbate ratio than wild type plants. However, there were neither differences in the levels of NADP and NADPH and in the total foliar activities of monodehydroascorbate reductase and dehydroascorbate reductase between wild type and transgenic plant. MV treatment induced an increase in the activities of GR, ascorbate peroxidase, superoxide dismutase, and catalase. Furthermore, accumulation of H2O2 in chloroplasts was observed in transgenic plants but not in wild type plants. Our results suggest that capacity for regeneration of glutathione by GR plays an important role in protecting against oxidative stress by maintaining ascorbate pool and ascorbate redox state.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Glutathione is the major low molecular weight thiol in all living organisms. In plants, glutathione is present in millimolar concentrations in leaves and is a major pool of non-protein sulphur (Foyer and Halliwell 1976; Kunert and Foyer 1993). Numerous studies have shown that glutathione has a number of important physiological functions in plants (see reviews May et al. 1998; Noctor et al. 1998, 2002; Maughan and Foyer 2006). Glutathione is an important sulphur sink, a predominant transported form of reduced sulphur, and a regulator of sulphur assimilation (Hell 1997). Glutathione has an ability to detoxify xenobiotics, such as herbicides, by conjugation reactions catalyzed by glutathione-S-transferases (Marrs 1996). Glutathione also plays an important role in the protection of plants against oxidative stress (Alscher 1989; Noctor et al. 1998). In addition, glutathione is involved in the regulation of gene expression, cell signalling, cell cycle, plant development, and cell death (Wingate et al. 1988; May et al. 1998; Noctor et al. 1998; Sanchez-Fernández et al. 1997; Gomez et al. 2004; Maughan and Foyer 2006).
Glutathione exists with two different forms, i.e. the reduced form (γ-Glu-Cys-Gly, GSH) and the oxidized form (glutathione disulphide, GSSG), in all living organisms. Glutathione is maintained almost exclusively in the reduced form in plants and the proportion of glutathione in the reduced form is normally greater than 0.9 under non-stress conditions (Noctor et al. 1998). The physiological functions of glutathione have been mainly attributed to its reduced form in plants (Alscher 1989). Thus, it is necessary that high proportion of glutathione in the reduced form should be maintained in plants (e.g. reviews, Noctor et al. 1998; May et al. 1998; Maughan and Foyer 2006). While acting as a major antioxidant in plant cells, GSH is oxidized to the disulphide form GSSG. However, plants have evolved the mechanisms to maintain a high proportion of the reduced glutathione. GSH can be synthesized in both the chloroplast and the cytosol of plant leaves. In addition, the reduction of GSSG to GSH can be catalyzed by glutathione reductase (GR, EC 1.6.4.2) with the accompanying oxidation of NADPH (Noctor and Foyer 1998; Asada 1999).
GR is localized mainly in the chloroplast in which it represents about 80% of total GR activities in leaf tissues, but is also found in the cytosol, glyoxysomes and peroxisomes (Edwards et al. 1990; Jiménez et al. 1997). GR is involved in the ascorbate-glutathine cycle. In this cycle, efficient recycling of glutathione is ensured by GR, which in turn makes re-reduction of ascorbate efficiently (Foyer and Halliwell 1976; Nakano and Asada 1980). Ascorbate as the major substrate for reductive detoxification of H2O2 in plants must therefore be continuously regenerated from its oxidized forms. Thus, in the ascorbate-glutathine cycle, glutathione acts as a recycled intermediate in the reduction of H2O2 using electrons derived ultimately from H2O (Foyer 1997), suggesting that GR plays an important role in the protection of plants against oxidative stress.
Indeed, many studies have demonstrated that GR plays an important role in protecting plants against oxidative stress. It has been observed that stress-tolerant plants tend to have high activities of GR (Amsellem et al. 1993; Kocsy et al. 1996, 2000; Mittova et al. 2003). Moreover, it has been shown that enhanced chloroplastic GR activity in transgenic plants results in increased protection against oxidative stress (Aono et al. 1993; Broadbent et al. 1995; Foyer et al. 1995; Pilon-Smit et al. 2000). In addition, it has been reported that a decrease in GR activity in tobacco plants leads to an increased sensitivity to oxidative stress (Aono et al 1995). However, it is still unclear how GR plays an important role in protecting plants against oxidative stress.
The main objective of this study was to investigate the possible mechanisms of GR in determining tolerance to oxidative stress. To this end, we generated transgenic tobacco plants with 30–70% decreased cellular GR activities by RNAi using a cDNA fragment of tobacco chloroplastic GR gene. We assessed how GR plays an important role in protecting plants against oxidative stress. We specifically investigated the effects of decreased total GR activity on photosynthesis, the ascorbate-glutathione cycle, and the content of reactive oxygen species H2O2 in tobacco plants under oxidative stress conditions induced by the application of methyl viologen (MV).
Materials and methods
Vectors and plant transformation
The partial coding region for tobacco chloroplastic glutathione reductase gene (X76293, gi: 431954) was cloned into pKANNIBAL vector between the XbaI-ClaI sites in sense orientation and the KpnI-EcoRI sites in antisense orientation (Wesley et al. 2001). The primers used were: 5′-TTC GAA TTC TTC TCA CCC ACT CCC GTA-3′, 5′-CAC GGT ACC TTG TCA TTT TCA CTC CCA CT-3′ (igr antisense primers) and 5′-GAT TCT AGA TTC TCA CCC ACT CCC GTA-3′, 5′-GGC GAT CGA TTT GTC ATT TTC ACT CCC ACT-3′ (igr sense primers). Construct made in pKANNIBAL was subcloned as Notl fragment into pART27, then introduced into Agrobacterium tumefaciens strain LBA4404 by tri-parental mating (An 1987). Nicotiana tabaccum (Wisconsin 38) were transformed by the standard Agrobacterium-mediated transformation (Horsh et al. 1985). Regenerated plants were transplanted into sterilized soil, grown in a greenhouse at 27/20°C (day/night), with maximum PPFD of 1000 μmol m−2 s−1, and a photoperiod of 12/12 h light/dark.
Plant growth conditions and methyl viologen (MV) treatments
Four independent lines of transgenic tobacco plants (i2, i21, i28, and i42) were used in the present study. The seeds of these transgenic were allowed to germinate on agar in the presence of 50 μg l−1 kanamycin. After growth for two weeks, plants were transferred to vermiculite which was soaked with Hoagland solution containing with 0 and 20 μM MV. The transplanted plants were then grown for two weeks in a growth chamber at 25 ± 1°C with PPFD of 100 μmol m−2 s−1, a relative humidity of 75–80%, and a photoperiod of 12/12 h light/dark. All the measurements on physiological and biochemical parameters were carried out on the youngest fully expanded leaves.
RT-PCR
Total RNA was isolated from 4-week-old plants using the Trizol reagent (Invitrogen Carlsbad, CA, USA). Two μg of total RNA from each sample was used to synthesize first-strand cDNA in a 20 μl total volume (SuperScript Pre-amplification System, Promega, Madsion, WI, USA). RT-PCR were performed using the igr sense primers, and amplified DNA product were collected and analyzed following different numbers (20 cycles, 30 cycles) of amplification cycles. The tobacco actin was amplified using primers made in our laboratory to quantify the relative amounts of cDNA. Identical quantities of reverse-transcribed first strand cDNAs were used as the templates for test PCR.
Antiserum preparation
The nucleotide sequence encoding the tobacco chloroplastic GR (X76293, gi:431954) was amplified by PCR using the following primers: sense, TCAAAGCTTGCTACATCTCTGAGCACACCA and antisense ACACTCGAGTCAAACTCCAGCTGCAGCTTT. The resulting DNA of GR was subcloned as a HindIII-XhoI fragment into the plasmid pET28a to obtain an N-terminal His affinity tag fusion and the resulting plasmid was transformed into E. coli BL21 (DE3) strain. The BL21 cells were harvested after adding 0.4 mM isopropylthio-β-D-galactoside for 5 h and resuspended in 500 mM NaCl and 20 mM NaH2PO4, pH 7.8. After incubation for 30 min at 4°C in the presence of lysozyme (1 mg/ml), the bacterial lysate was sonicated 30 times for 10 s each. The overexpressed His-GR fusion protein in inclusion bodies was centrifuged at 3000g for 30 min, and the pellet was solubilized in 500 mM NaCl, 6 M urea, and 20 mM NaH2PO4, pH 7.8. The His-GR fusion protein was purified on a Ni-NTA agarose resin matrix, and the purified His-GR protein was used for rabbit immunization to raise antiserum.
Immunological analysis on 2D SDS-PAGE
The protein samples isolated directly from both leaves and chloroplasts for 2D SDS-PAGE analysis were extracted and pelleted by TCA-acetone method. For the total leaf protein samples, 100 mg tobacco leaves were homogenized in 1 ml 10% TCA-acetone solution, centrifuged at 8,000g for 5 min, and then the pellet was dried with vacuum pump. For the samples isolated directly from isolated chloroplasts, the chloroplasts were broken and pelleted by adding 4 times volume cold acetone and then the pellet was further dried with vacuum pump. The protein samples in dried pellet from leaves and chloroplasts were solubilized for 1 h in solubilization buffer (20 mM Tris-base pH 8.3, 7 M urea, 2 M thiourea, 30 mM DTT, 4% (w/v) 3-([Cholamidopropyl]dimethylammonio)-1-propane-sulphonate (CHAPS), 0.002% (w/v) bromophenol blue, 0.5% (v/v) IPG buffer, 1% (v/v) protease inhibitor (Amersham Bioscience, New York, USA).
For 2D SDS-PAGE, protein samples were diluted to 0.5 mg/ml and loaded onto strip reswell tray (Amersham Bioscience). Immobiline DryStrip (pH 3–10, 7 cm) was overlayed upside down in the reswell tray and covered with Drystrip Cover Fluid (Amersham Bioscience). After 12 h reswelling, the IEF was run at following settings: 100 V for 1 h, 500 V for 1 h, 1000 V for 1h, 8000 V to 62 kVh. The IPG strips, after IEF, were equilibrated by the Amersham Bioscience protocol and then loaded onto 5% stacking gel and 12% resolving gel. The SDS-PAGE was run on 10 mA per gel using Mini-Protean III electrophoresis unit (Bio-Rad Laboratories, Inc.).
After 2D SDS-PAGE separation, the proteins were electrotransferred onto a PVDF membrane (Amersham Biosciences, Piscataway, USA) and were detected with tobacco chloroplastic GR-antiserum made in our laboratory. Immunoblots were developed with anti-rabbit IgG (Sigma) and subjected to enhanced chemiluminescence detection (SuperSignal® West Pico Trial Kit; Pierce, Rockford, IL, USA), according to the manufacturer’s protocol.
Immunological analysis on 1D SDS-PAGE
The total leaf protein samples were extracted and pelleted by TCA-acetone method as described in immunological analysis on 2D SDS-PAGE. The protein samples were separated using 12% (w/v) acrylamide gels with 6 M urea (Laemmli 1970). The immunological analysis was carried out as described above.
Dephosphorylation
Dephosphorylation was carried out according to Edwards et al. (1990). Total leaf protein extracts were dephosphorylated by incubation with E. coli alkaline phosphatase at a concentration of 1 unit/10 μg protein, in 50 mM Tris pH 8.0, 0.1 mM ethylenediaminetetraacetic acid (EDTA), at 37°C for 24 h.
Isolation of intact chloroplasts
Intact chloroplasts were isolated by centrifugation on a Percoll density gradient according to the method described by Yoshimura et al. (2004). The percentage of intact chloroplasts was determined by measuring ferricyanide photoreduction before and after osmotic shock, and preparations used in the experiments were between 85 and 95% intact. The purity of chloroplasts was checked using the enzymes catalase, cytochrome-c oxidase, and phosphoenolpyruvate carboxylase (PEPCase) as markers for peroxidosome, mitochondria and cytosol, respectively (Robinson and Walker 1979; Edwards et al. 1990).
Analysis of gas exchange
Measurements of net photosynthetic gas exchange were made on a fully expanded attached leave of tobacco seedlings using an open system (Ciras-1, PP systems, UK). The photosynthetic rate was made at a CO2 concentration of 360 μl l−1 and at a temperature 25°C with a relative humidity 80% and growth light (100 μmol m−2 s−1).
Measurements of chlorophyll fluorescence
Chlorophyll fluorescence was measured with a PAM-2000 chlorophyll fluorescence system under atmospheric conditions (Heinz Walz, Effieltrich, Germany) as described previously (Yang et al. 2005). After a dark adaptation period of 30 min, minimum fluorescence (Fo) was determined by a weak red light. Maximum fluorescence of dark-adapted state (Fm) was measured during a subsequent saturating light pulse of with light (8000 μmol m−2 s−1 for 0.8 s). The measurements were performed on the attached leaves of tobacco seedlings. The maximal efficiency of PSII photochemistry was determined as the ratio of variable to maximal chlorophyll fluorescence (Fv/Fm) (Krause and Weis 1991).
Enzyme measurements
GR activity was determined by following the rate of GSH formation at 412 nm (González et al. 1998). Ascorbate peroxidase (APX, EC1.11.1.11) activity was determined by monitoring the decrease in absorbance at 290 nm due to ascorbate oxidation (González et al. 1998). Dehydroascorbate reductase (DHAR, EC1.8.5.1) activity was determined by the increase in absorbance at 295 nm due to DHA reduction (Knörzer et al. 1996). Monodehydroascorbate reductase (MDHAR, EC1.6.5.4) activity was measured by monitoring the decrease in absorbance at 340 nm due to the oxidation of NADPH (Arrigoni et al. 1981). Superoxide dismutase (SOD, EC1.15.1.1) activity was estimated by its ability to inhibit photoreduction of nitroblue tetrazolium (Beyer and Fridovich 1987). Catalase (CAT, EC1.11.1.6) activity was assayed by directly measuring the rate of decomposition of H2O2 at 240 nm as described by Aebi (1984).
Measurements of the metabolite pool size of the ascorbate-glutathione cycle
1 g leaf discs were frozen in liquid N2 and were homogenized in 5 ml of 10% TCA (w/v) containing 1.5 mM EDTA, and centrifuged for 15 min at 14,000g. The supernatant was used for glutathione and ascorbate analyses. The TCA extract was neutralized (diluted 1:50 with 5% Na2HPO4, pH 7.5). Total glutathione was measured using the enzymatic recycling assays. Changes in absorbance of the reaction mixture were followed at 412 nm. Oxidized glutathione was measured by first incubating 1.5 ml of the 1:50 diluted acid extract with 60 μl of 2-vinylpyridine for 1 h at 25°C. After the incubation, the measurements were carried out using the same assay. Reduced glutathione was determined as the difference between total glutathione and GSSG. All values are reported as GSH equivalents (Anderson et al. 1992). For ascorbate measurement, 200 μl TCA extract were mixed with 5 μl 5 N NaOH. 100 μl of this mixture was added potassium phosphate buffer and reaction buffer to estimate reduced ascorbate (ASC) content by the absorbance at 525 nm. Another 100 μl of the mixture was used for total amount of reduced and oxidized ascorbic acid. It was determined by reducing oxidized ascorbate (DHA) to ASC (in a potassium phosphate buffer containing DTT) before measuring. The content of DHA was determined as the difference between these two assays. The amounts of ascorbate were calculated from a standard curve of pure L-ascorbic acid (Sigma) that underwent the same treatment (González et al. 1998). The contents of NADPH and NADP were determined by enzyme cycling techniques as described by Maciejewska and Kacperska (1987).
Cytochemical detection of H2O2
H2O2 was visualized at the subcellular level using CeCl3 for localization according to Bestwick et al. (1997). Electron-dense CeCl3 deposits are formed in the presence of H2O2 and are visible by transmission electron microscopy. Leaf pieces (approximately 1–3 mm2) were excised and were incubated in freshly prepared 5 mM CeCl3 in 50 mM 3-(N-morpholino)propanesulfonic acid (MOPS) at pH 7.2 for 1 h. Leaf pieces were then fixed with 1.25% (v/v) glutaraldehyde and 1.25% (v/v) paraformaldehide in 50 mM sodium cacodylate buffer (pH 7.2) for 1 h. After fixation, tissues were washed twice in the same buffer for 10 min and post-fixed for 45 min in 1% (v/v) osmium tetroxide, and dehydrated in a graded ethanol series (30–100%, v/v) and embedded Eponaraldite (Agar Aids, Bishop’s Stortford, UK). After 12 h in pure resin, followed by a change of fresh resin for 4 h, the samples were polymerized at 60°C for 48 h. Ultrathin sections (70 nm thick) were obtained with a Leica ultramicrotome, stained with uranyl acetate and lead citrate. Electron micrographs were obtained with a JEM-1230 TEM (JEOL) at an accelerating voltage of 80 kV.
Measurements of H2O2
Total leaf H2O2 was determined according to the method as described by Veljovic-Jovanovic et al. (2002) and Queval et al. (2008). Tobacco leaves were ground to a fine powder in liquid nitrogen and the powder extracted in 1 M HClO4 and 5% insoluble PVP. The homogenate was centrifuged at 12,000g for 10 min and the supernatant was neutralized with 5 M K2CO3 to pH 5.6 in the presence of 50 μl 0.3 M phosphate buffer (pH 5.6). The homogenate was centrifuged at 12,000g for 1 min to remove KClO4. The sample was incubated prior to assay for 10 min with 1 U ascorbate oxidase (Sigma, Watford, UK) to oxidize ascorbate to eliminate the interference of ascorbate. The reaction mixture consisted of 0.1 M phosphate buffer (pH 6.5); 3.3 mM 3-(dimethylamino) benzoic acid; 0.07 mM 3-methyl-2-benzothiazoline hydazone and 50 ng horseradish peroxidase (Sigma, Watford, UK). The reaction was initiated by addition of an aliquot (50 or 100 μl) of the sample. The absorbance change at 590 nm was monitored at 25°C. To eliminate the interference of lipid peroxides, parallel aliquot for each sample was incubated for 10 min with 1 U catalase (Sigma, Watford, UK) to catalyze H2O2, the content of lipid peroxides was then determined using the same procedure as described above and their content was subtracted from each sample. It should be pointed out that for each assay, H2O2 contents in the extract were quantified by reference to an internal standard (3 nmol H2O2, added to the reaction mixture on completion of the absorbance change due to the sample).
Determination of oxidative damage to lipids
Oxidative damage to lipids was determined by measuring the content of malondialdehyde (MDA), prepared in 10% trichloroacetic acid containing 0.65% 2-thiobarbituric acid (TBA) and heated at 95°C for 25 min, as described by Hodges et al. (1999). MDA content was calculated by correcting for compounds other than MDA which absorb at 532 nm by subtracting the absorbance at 532 nm of a solution containing plant extract incubated without TBA from an identical solution containing TBA.
Measurements of chlorophyll and protein
Chlorophyll content was determined according to Arnon (1949). Protein content was determined by the dye-binding assay according to Bradford (1976).
Results
Generation of transgenic tobacco plants with decreased GR activity
In this study, we generated transgenic tobacco plants with the RNAi construct of tobacco chloroplastic GR. Fifty independent transformed lines were obtained and some of them exhibited foliar GR activities 30–70% of wild type plants. Four transgenic lines (i2, i21, i28, and i42) that had decreased GR activities were chosen to obtain progeny (T1 generation) by self-fertilization. Wild type plants had 0.028 ± 0.002 U mg−1 protein (n = 3) of GR activity. The GR activity in transgenic i2 plants was 0.019 ± 0.002 U mg−1 protein (n = 3) which was about 70% of GR activity in wild type plants. The activities of GR in transgenic i21, i28, and i42 plants were 0.0085 ± 0.0001, 0.0079 ± 0.0001, and 0.0083 ± 0.0001 U mg−1 protein (n = 3), respectively, which were about 30% of GR activity in wild type plants.
RT-PCR analysis shows that the mRNA levels of GR in transgenic plants were suppressed as compared to that in wild type plants. The mRNA levels of GR in transgenic i21, i28 and i42 plants were suppressed much greater than those in transgenic i2 plants (Fig. 1a).
a RT-PCR analyses in wild type and transgenic tobacco plants. The mRNA was isolated from wild type and transgenic tobacco plants and used for antiscript. RT-PCR was performed using the igr sense primers, and amplified DNA product were collected and analyzed following different numbers (20 cycles, 30 cycles) of amplification cycles. The tobacco actin was amplified as internal standard. b Detection of GR protein in wild type and transgenic tobacco plants. Total leaf proteins (50 μg for each sample) isolated directly from the leaves of wild type and transgenic tobacco plants were subjected to SDS-PAGE followed by western blot analysis with a rabbit antiserum against tobacco chloroplastic GR. The molecular weight of the major band protein was approximately 55 kDa. wt, wild type tobacco plants; i2, i21, i28, i42, four independent transgenic tobacco plants
Western blotting analysis on 1D SDS-PAGE was carried out using the antiserum of tobacco chloroplastic GR raised in our laboratory (Fig. 1b). Three protein bands with different molecular weights were recognized by the antiserum. The molecular weight of the major protein was approximately 55 kDa. The molecular weights of the other two minor proteins were about 50 and 47 kDa, respectively. There was a decrease in the level of the major protein in transgenic plants as compared with that in wild type plants and such a decrease was much greater in transgenic i21, i28 and i42 plants than in transgenic i2 plants. However, it seems that there was no significant change in the levels of the two minor proteins between wild type and transgenic plants.
Western blotting analysis on 2D SDS-PAGE has been used to analyze the multiple forms of GR in pea plants (Edwards et al. 1990). When analyzed on the total leaf protein extracts in wild type plants, there were ten forms of GR enzyme recognized by antiserum (Fig. 2a). The isoelectric point (pI) of these forms ranged from 4.5 to 6.2. The major forms of GR in tobacco plants were the spots 3, 4, and 5. The molecular weights of forms 1–5 were approximately 55 kDa. The molecular weights of forms 6–10 were lower and ranged about 47–50 kDa. To investigate whether the multiple forms of GR observed in this study were associated with protein glutathionylation and other thiol-disulphide exchange reactions, the total leaf protein samples were incubated with 1 mM dithiothreitol (DTT) which reverses glutathionylation and thiol-disulphide exchange (Gallogly and Mieyal 2007; Huang et al. 2008) and then the DTT-treated total leaf protein samples were subjected to 2D electrophoresis and subsequent immunoblot analysis of GR. We observed that there were no changes in the patterns of the multiple forms of GR after the total leaf protein samples were treated with DDT (data not shown), suggesting that the multiple forms of GR were not due to protein glutathionylation and other thiol-disulphide exchange reactions. Phosphorylation is one of the most common types of post-translational modifications. According to the method described by Edwards et al. (1990), we investigated the effects of dephosphorylation on the patterns of the forms of GR following 2D electrophoresis. We observed that dephosphorylation had no effects on the pattern of the forms of GR (data not shown), indicating that the multiple forms of GR were not due to phosphorylation.
2D gel electrophoresis and subsequent immunoblot analysis of GR. a Total leaf proteins (100 μg loaded for each sample) isolated directly from leaves of wild type tobacco plants. b Total chloroplastic proteins (50 μg loaded for each sample) isolated directly from chloroplasts which were isolated from leaves of wild type tobacco plants. c Total leaf proteins (100 μg loaded for each sample) isolated directly from leaves of transgenic i28 tobacco plants
It has been shown that the major forms of GR are localized in the chloroplast (Aono et al. 1993; Edwards et al. 1990). In order to investigate whether the major forms 3, 4, and 5 exist in the chloroplast or not, we further examined the forms of GR in the chloroplast. We thus isolated intact chloroplasts from the leaves of wild type plants. Intactness of the chloroplast isolated in this study was between 85 and 95%. The purity of intact chloroplasts was assessed using the enzymes catalase, cytochrome-c oxidase, phosphoenolpyruvate carboxylase (PEPCase) as the markers for peroxidosome, mitochondria and cytosol, respectively. No activities of catalase, cytochrome-c oxidase, and PEPcase were detected in the chloroplast (data not shown), suggesting that there was no other organelle contamination in the chloroplast isolated in this study. We observed that there were three types of forms of GR in the chloroplast (Fig. 2b). According to the pIs and molecular weights of these forms, these three GR forms were assigned as the forms 3, 4 and 5. These results indicate that the major forms 3, 4, of GR exist in the chloroplast in tobacco plants.
To investigate how the forms of GR were affected in transgenic plants, we further examined the changes in the forms of GR in i28 transgenic plants (Fig. 2c). As compared to the forms of GR in wild type plants, the major forms 4 and 5 were not detectable and the major form 3 was suppressed significantly in transgenic i28 plants. In terms of other minor forms of GR, the forms 6, 8 and 10 were not detectable but the levels of the forms 9 and 7 increased. There was a decrease in the level of the form 2 and no significant change in the level of the form 1 in transgenic i28 plants. Similar results were also observed in transgenic i2, i21, and i42 plants (data not shown). Since only two minor bands were observed in 1D gels and there was no significant change in the levels of the two minor proteins between wild type and transgenic plants, and also the levels of the forms 6, 8 and 10 decreased in transgenic i28 plants, it is difficult to observe an increase in the levels of the forms 7 and 9 in 1D gels as shown in Fig. 1b.
Effects of MV on leaf bleaching and oxidative damage to lipids
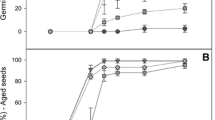
We observed that there were no significant differences in growth between wild type and transgenic plants under normal growth conditions. We examined the effects of decreased GR activity on the sensitivity of wild type and transgenic plants to oxidative stress. The herbicide MV is referred to as a source of oxidative stress and the damage caused by MV treatment under illumination is known as MV-mediated photooxidative damage. MV exerts its herbicidal activity by interfering with the intracellular electron transfer systems in plants. This disruption leads to the formation of reactive oxygen species (ROS) such as superoxide anion, H2O2 (Dodge 1994). Therefore, MV is often used to induce oxidative stress and assay the tolerance of plants to oxidative stress in plants (Aono et al. 1995; Broadbent et al. 1995; Yoshimura et al. 2004; Xiong et al. 2007; Lim et al. 2008). The seedlings of wild type and transgenic plants were treated with 20 μM MV in this study. We observed that after 8 days of the MV treatment, leaves started to show slight bleaching in transgenic i21, i28 and i42 plants. After about 14 days of MV treatment, the bleaching of leaves became to be evident in transgenic i21, i28 and i42 plants but not in wild type plants. Thus, the leaf samples at the 14th day after the MV treatment were taken for various physiological and biochemical analyses. Figure 3 shows the details on the effects of MV on leaf bleaching in wild type and transgenic plants. After treated with 20 μM MV for two weeks, we observed no obvious symptoms of leaf bleaching in wild type and transgenic i2 plants. However, leaf bleaching was evidently visible in transgenic i21, i28, and i42 plants (Fig. 3a). In order to further characterize leaf bleaching, we examined the changes in the contents of leaf chlorophyll in both wild type and transgenic plants (Fig. 3b). There were no significant differences in the content of chlorophyll in both wild type and transgenic plants under normal conditions. After treated with 20 μM MV, there was no significant decrease in the content of chlorophyll in wild type plants. However, transgenic i2 plants showed about a 10% decrease in the content of chlorophyll. Such a decrease was greater in transgenic i21, i28, and i42 plants that had about a 25% decrease in the content of chlorophyll.
a Effects of methyl viologen treatment on leaf bleaching in wild type and transgenic tobacco plants. b Effects of methyl viologen treatment on the content of leaf chlorophyll in wild type and transgenic tobacco plants. wt, wild type tobacco plants; i2, i21, i28, i42, four independent transgenic tobacco plants. The values are mean ± SD of three independent experiments
In order to investigate the effects of MV on oxidative damage to lipids in wild type and transgenic plants, we examined the changes in the contents of MDA in both wild type and transgenic plants (Fig. 4). There were no differences in the content of MDA between wild type and transgenic plants under normal conditions. After treated with 20 μM MV for two weeks, there was no significant change in the content of MDA in wild type plants. However, the content of MDA was significantly increased in transgenic plants and such an increase was much greater in transgenic i21, i28 and i42 plants than in transgenic i2 plants.
Effects of MV on photosynthetic function
Figure 5 shows the effects of MV on CO2 assimilation and PSII function in seedlings of wild type and transgenic plants. There were no significant differences in CO2 assimilation rate between wild type and transgenic plants under normal growth conditions. After treated with 20 μM MV for two weeks, there was no significant decrease in CO2 assimilation rate in wild type plants. However, there was a significant decrease in CO2 assimilation rate in transgenic plants. There was about a 40% decrease in CO2 assimilation rate in transgenic i2 plants. The decrease in CO2 assimilation rate was much greater in transgenic i21, i28 and i42 plants than in transgenic i2 plants. There was about 80% decrease in CO2 assimilation rate in transgenic i21, 28, and i42 plants (Fig. 5a). We further examined the effects of MV on the function of PSII by measuring chlorophyll fluorescence. PSII activity is expressed by the fluorescence parameter, the ratio of maximal variable fluorescence to maximal fluorescence, Fv/Fm, which is the maximal efficiency of PSII photochemistry (Krause and Weis 1991). Figure 5b shows that there were no significant differences in Fv/Fm between wild type and transgenic plants under normal conditions. After treated with 20 μM MV for two weeks, wild type plants showed no significant decrease in Fv/Fm. There was a slight in Fv/Fm in transgenic i2 plants but a significant decrease in Fv/Fm in transgenic i21, i28 and i42 plants. These results suggest that the sensitivity of photosynthetic function to oxidative stress was significantly increased in transgenic plants.
a Effects of methyl viologen treatment on CO2 assimilation rate in wild type and transgenic tobacco plants. b Effects of methyl viologen on the maximal efficiency of photosystem II photochemistry (Fv/Fm) in wild type and transgenic tobacco plants. wt, wild type tobacco plants; i2, i21, i28, i42, four independent transgenic tobacco plants. The values are mean ± SD of three independent experiments
Effects of MV on the production of H2O2
Since glutathione acts as a recycled intermediate in the reduction of H2O2 using electrons derived ultimately from H2O and GR ensures an efficient recycling of glutathione (Foyer 1997), we investigated the effects of MV on the production of H2O2 in the leaves of wild type and transgenic plants (Fig. 6). There were no significant differences in the levels of H2O2 between wild type and transgenic plants under normal conditions. After treated with 20 μM MV for two weeks, there was an increase in the levels of H2O2 in both wild type plants and transgenic plants. Moreover, such an increase was much greater in transgenic plants, in particular for i21, i28, and i42 plants, than in wild type plants.
To investigate the subcellular localization of H2O2 accumulation in the leaves of wild type and transgenic plants after treated with 20 μM MV, a cytochemical technique with CeCl3 that reacts with H2O2 to produce electron-dense deposits of cerium perhydroxides was used (Bestwick et al. 1997). Since MV treatment resulted in an obvious bleaching of the leaves in transgenic plants, the green part of the leaf in transgenic plants was taken for the analysis of H2O2 accumulation. Under normal conditions, CeCl3 deposits, indicative of the accumulation of H2O2, were not observed in the mesophyll cells in both wild type and transgenic plants (Fig. 7a, c). After treated with 20 μM MV for two weeks, H2O2 accumulation was consistently visible only in the cell walls of mesophyll cells in wild type plants (Fig. 7b). However, H2O2 accumulation was consistently visible not only in the cell walls of mesophyll cells but also in the stroma of the chloroplast in transgenic plants (Fig. 7d). In addition, we observed consistently that the chloroplast in transgenic i28 plants contained less grana stacks in the thylakoid membranes than that in wild type plants after treated with 20 μM MV for two weeks.
Cytochemical localization of H2O2 accumulation in the mesophyll cells of wild type and transgenic i28 plants under normal conditions and after treated with 20 μM methyl viologen. a Wild type tobacco plants in the absence of methyl viologen. b Wild type tobacco plants treated with 20 μM methyl viologen. c Transgenic i28 tobacco plants in the absence of methyl viologen. d Transgenic i28 tobacco plants treated with 20 μM methyl viologen. It should be noted that the green part of the leaf in transgenic i28 plants was taken for the analysis of H2O2 accumulation. Three independent experiments were carried out with similar results. Arrows indicate CeCl3 precipitates. cw, cell wall; ch, chloroplast. Bar = 1 μm
Effects of MV on activities of enzymes
GR is involved in the ascorbate-glutathine cycle in which the efficient recycling of glutathione is ensured by GR (see reviews Asada 1999). The other enzymes involved in this cycle include: APX, DHAR, and MDHAR. We thus investigated the effects of MV on the total foliar activities of these enzymes in wild type and transgenic plants (Fig. 8). Under normal conditions, there was a significant decrease in GR activity in transgenic plants as compared to wild type plants. After treated with MV for two weeks, there was a significant increase in the GR activity in both wild type and transgenic plants and such an increase was greater in transgenic plants than in wild type plants (Fig. 8a). There was no significant difference in the activity of APX between wild type plants and transgenic plants either under normal conditions or under MV treatment conditions. However, MV induced an increase in the activity of APX in both wild type and transgenic plants (Fig. 8b). There were no significant differences in the activities of DHAR and MDHAR between wild type and transgenic plants either under normal conditions or under MV treatment conditions. And, MV treatment did not induce significant changes in the activities of DHAR and MDHAR (Fig. 8c, d).
Effects of methyl viologen treatment on the activities of GR (a), APX (b), MDHAR (c), and DHAR (d) in leaves of wild type and transgenic tobacco plants. wt, wild type tobacco plants; i2, i21, i28, i42, four independent transgenic tobacco plants. The values are mean ± SD of three independent experiments
We further examined the effects of MV on total foliar activities of SOD and CAT in wild type and transgenic plants (Fig. 9). There were no significant differences in the activities of SOD and CAT between wild type and transgenic plants either under normal conditions or under MV treatment conditions. However, MV treatment induced an increase in the activities of SOD and CAT in both wild type and transgenic plants.
Effects of MV on the metabolite pool size of the ascorbate-glutathione cycle
Figure 10 shows the effects of MV on the pools of glutathione and ascorbate in wild type and transgenic plants. Under normal conditions, there were no significant differences in the levels of GSH and GSSG and the ratio of GSH/GSSG between wild type and transgenic plants. Under MV treatment conditions, no significant change in the level of GSH was observed but there was an increase in the level of GSSG in wild type plants. There was a significant increase in the levels of both GSH and GSSG in transgenic plants. A decrease in the ratios of GSH/GSSG in both wild type and transgenic plants was observed and such a decrease was much greater in transgenic plants than in wild type plants.
Effects of methyl viologen treatment on GSH (a), GSSG (b), the GSH/GSSG ratio (c), ASC (d), DHA (e), and the ASC/DHA ratio in leaves of wild type and transgenic tobacco plants. wt, wild type tobacco plants; i2, i21, i28, i42, four independent transgenic tobacco plants. The values are mean ± SD of three independent experiments
Under normal conditions, there were no significant differences in the levels of ASC and DHA and in the ratios of ASC/DHA between wild type and transgenic plants. MV treatment resulted in no significant changes in the level of ASC in wild type plants but a significant decrease in the levels of ASC in transgenic plants. In addition, MV treatment induced an increase in the levels of DHA in both wild type and transgenic plants but there were no significant differences in the levels of DHA between wild type and transgenic plants. Furthermore, MV treatment led to a decrease in the ratios of ASC/DHA in both wild type and transgenic plants and such a decrease was much greater in transgenic plants than in wild type plants.
We also examined the effects of MV on the levels of NADPH and NADP and the NADPH/NADP ratios in wild type and transgenic plants. We observed that there were no significant differences in the levels of NADPH and NADP and the NADPH/NADP ratios between wild type and transgenic plants under normal conditions and there were also neither changes in the levels of NADPH and NADP and the NADPH/NADP ratios in wild type and transgenic plants after treated with 20 μM MV (data not shown).
Relationship between glutathione redox state, ascorbate redox state and H2O2 accumulation
Figure 11 shows the relationship between the ratio of GSH/GSSG and accumulation of H2O2 as well as the relationship between the ratio of ASC/DHA and accumulation of H2O2 in wild type and transgenic plants. The ratios of GSH/GSSG and DHA/ASC decreased rapidly as the content of H2O2 increased from 200 to 400 nmol g−1 FW. Low H2O2 contents (less than 200 nmol g−1 FW) seem to allow variability in the ratios of GSH/GSSG and DHA/ASC. It seems that H2O2 content at around 200 nmol g−1 FW might be a threshold, where the further accumulation of H2O2 results in leaf bleaching. Since the ratios of GSH/GSSG and DHA/ASC are often used as indicators of glutathione redox state and ascorbate redox state, respectively, the results from Fig. 11 suggest that the decrease in glutathione and ascorbate redox states had a significant effect on accumulation of H2O2.
Discussion
GR plays an important role in response to oxidative stress in plants (see reviews, Foyer et al. 1997; May et al. 1998; Noctor et al. 1998, 2002; Maughan and Foyer 2006). Indeed, several studies have demonstrated that transgenic plants with overexpression of GR in the chloroplast have been to provide additional protection against oxidative stress (Aono et al. 1993; Broadbent et al. 1995; Foyer et al 1995). However, the physiological basis for this enhanced tolerance to oxidative stress is still unknown. In this study, we obtained the transgenic tobacco plants with 30–70% decrease in the activity of GR by using a gene (X76293, gi: 431954) encoding tobacco chloroplastic GR for the RNAi construct. Thus, it makes possible for us to investigate how GR regulates the responses to oxidative stress in transgenic tobacco plants.
In this study, our results show that an antiserum raised against GR recognized ten forms of GR in a total leaf protein extract separated by 2D electrophoresis in tobacco plants, three of which are major forms and localized in the chloroplast (Fig. 2a, b). Similar results have been reported previously that GR is localized mainly in the chloroplast (Edwards et al. 1990; Foyer et al. 1991). The occurrence of multiple GR forms is in agreement with the report showing eight isoforms in pea leaves by 2D electrophoresis (Edwards et al. 1990). Multiple isoforms of GR have also been reported in the leaves of tobacco and red spruce (Foyer et al. 1991; Hausladen and Alscher 1994). Since DTT had no effects on the patterns of the multiple forms of GR, our results suggest that the multiple forms of GR might be not due to glutathionylation and other thiol-disulphide exchange reactions. In addition, we also observed that phosphorylation had no effects on the patterns of the multiple forms of GR following 2D electrophoresis, indicating that phosphorylation was not responsible for multiple forms of GR. In addition, Edwards et al. (1990) have shown that the multiple forms of GR in pea leaves were not to due to glycosylation and phosphorylation by following 2D electrophoresis. Therefore, according to the results in this study and in Edwards et al. (1990), the multiple forms of GR in the leaves of tobacco plants might be not due to glutathionylation, phosphorylation and glycosylation.
It is still not clear how the multiple isoforms of GR arise in tobacco plants. To date, two GR genes in pea and tobacco, GOR1 and GOR2, have been characterized (Creissen et al. 1992; Creissen and Mullineaux 1995; Stevens et al. 1997, 2000). GOR1 encodes a protein co-targeted into chloroplasts and mitochondria (Creissen et al. 1992, 1995) and GOR2 encodes a protein which is extraplastidial and most likely to be located in the cytosol (Stevens et al. 2000). It seems that the numbers of genes encoding GR in tobacco contrast sharply with the number of isoforms of GR enzyme (Foyer et al. 1991). The product of GOR1 has been shown to be co-targeted to both chloroplasts and mitochondria simultaneously in pea, suggesting that a single mRNA can generate a protein with two subcellular locations (Creissen et al. 1995). Possibly, a similar situation exists in tobacco plants. In addition, it has been suggested that there may still be more GR genes in tobacco, for example encoding the cytosolic isoform(s) and the presence of multiple isoforms of GR in tobacco may be explained in part by the expression of a small gene family (Creissen and Mullineaux 1995). Furthermore, multiple forms of GR may result from translation of different mRNAs derived from the same gene by intron skipping during the splicing of nascent GR mRNAs as observed in tobacco GR cDNAs (Creissen and Mullineaux 1995). It is also possible that there may be post-translational control over isoform synthesis of GR encoded by GOR1 or GOR2 (Edwards et al. 1994) although some post-translational modifications have been excluded.
MV is referred to as a source of oxidative stress and the damage caused by MV treatment under illumination is known as MV-mediated photooxidative damage (Dodge 1994; Aono et al. 1995). Visible injury of leaf discs by MV treatment is often used to assay the tolerance of plants to oxidative stress (Foyer et al. 1991; Aono et al. 1993, 1995; Broadbent et al. 1995; Yoshimura et al. 2004). MV treatments in previous studies were normally performed on leaf discs in vitro. In this study, we examined the responses of wild type and transgenic plants in response to MV treatments in vivo. We observed that there was a visible leaf bleaching in transgenic i21, i28, and i42 plants after MV treatment for two weeks (Fig. 3). We also observed a significant decrease in CO2 assimilation and PSII activity as well as a significant accumulation of MDA and H2O2 in transgenic plants (Figs. 4, 5, 6). Therefore, our results clearly demonstrate that a decrease in GR activity leads to increased sensitivity to oxidative stress.
It has been reported that GR affects not only on the glutathione redox state (expressed by the GSH/GSSG ratio) but also on the glutathione pool size. Foyer et al. (1995) have shown that overexpression of GR in the chloroplast in poplar trees resulted in an increase in both foliar GSH and the GSH/GSSG ratio. In contrast, Broadbent et al. (1995) have demonstrated that elevated levels of GR activity in the chloroplast in transgenic tobacco plants led to only an increase in the glutathione pool but had no effects on the GSH/GSSG ratio. However, both poplar and tobacco with overexpression of GR exhibited a significant increase in the tolerance to oxidative stress (Broadbent et al. 1995; Foyer et al. 1995). It is not clear whether such increased tolerance to oxidative stress in transgenic plants with overexpression of GR was a result of an increase in the glutathione pool size or/and the GSH/GSSG ratio. In the present study, we observed that there was an increase in foliar GSH content and total glutathione pool in transgenic tobacco plants after MV treatment, although their GR activity was significantly inhibited. At the same time, the GSH/GSSG ratio decreased significantly in transgenic plants (Fig. 10), indicating that the decrease in GR activity results in an inhibition of regeneration of glutathione. Therefore, our results suggest that the capacity for regeneration of glutathione determined by GR appears to play an important role in protection against oxidative stress.
To reveal how the capacity for regeneration of glutathione plays an important role in protecting against oxidative stress, we compared the changes in accumulation of H2O2 in wild type and transgenic plants after MV treatment. We observed that there was significant accumulation of H2O2 in the leaves of transgenic plants after MV treatments (Fig. 6). Moreover, the accumulation of H2O2 in the leaves of wild type and transgenic plants increased rapidly as the ratio of GSH/GSSG decreased from around 15 to about 4 (Fig. 11). Our results further showed that significant accumulation of H2O2 was observed in the chloroplast in transgenic plants but not in the chloroplast in wild type plants under MV treatment conditions (Fig. 7). These results suggest that the decrease in the GSH/GSSG ratio resulting from inhibited GR activity may result in accumulation of H2O2, which caused oxidative damage to photosynthetic function (Fig. 4).
One of the functions of the ascorbate-glutathione cycle is to detoxify H2O2 and GR, together with the other enzymes APX, DHAR, and MDHAR, is involved in this cycle (May et al. 1998; Noctor et al. 1998). Thus, we investigated if accumulation of H2O2 was associated with the changes in the activities of these enzymes in transgenic plants under oxidative stress. We observed that there were no significant differences in the total leaf activities of APX, DHAR and MDHAR between wild type and transgenic plants either under normal conditions or after MV treatment (Fig. 8). In addition, there were also no significant differences in the total activities of SOD and CAT between wild type and transgenic plants either under normal conditions or under oxidative stress (Fig. 9). These results seem to suggest that accumulation of H2O2 in transgenic plants after MV treatment may be not associated with the changes in the activities of the other enzymes involved in the ascorbate-glutathione cycle.
We further investigated if accumulation of H2O2 was associated with the changes in the metabolite pool size of the ascorbate-glutathione cycle. Our results show that there was an increase in foliar GSH and the glutathione pool size in transgenic plants under oxidative stress (Fig. 10). In addition, we observed that there were no significant differences in foliar NADPH and NADP contents between wild type and transgenic plants under normal conditions and there were also no significant changes in both wild type and transgenic plants after MV treatments. However, a greater decease in foliar ASC was observed in transgenic plants than in wild type plants after MV treatment (Fig. 10). In plants, the major substrate for reductive detoxification of H2O2 by APX is ASC (Noctor et al. 1998; Asada 1999). The results in this study suggest that the decrease in foliar ASC may be a factor which limited a detoxification of H2O2, thus resulting in accumulation of H2O2 in transgenic plants under oxidative stress.
Foliar ASC showed a greater decrease and the ascorbate pool became more oxidized in transgenic plants than in wild type plants as a result of oxidative stress (Fig. 10), suggesting that the capacity for reduction of the glutathione pool by GR plays an important role in maintaining the ascorbate pool and the ascorbate redox state. Ascorbate-glutathione cycle is one of connections between the ascorbate pool and the glutathione pool. In this pathway, GSH and NADPH are the substrates of DHAR and MDHAR, respectively, for ascorbate regeneration (Noctor et al. 1998). However, transgenic plants in this study showed increased GSH and no changes in NADPH content, and the activities of DHAR and MDHAR as compared to wild type plants under oxidative stress. Thus, it seems that the decrease in foliar ASC and the ascorbate redox state in transgenic plants under oxidative stress is not due to the changes in the substrates and the enzymes involved in ascorbate regeneration, but most likely associated with the decrease in the glutathione redox state caused by decreased GR activity. It has been suggested that changes in the glutathione redox state may be a factor in sensing oxidative stress and plays a role in the response to oxidative stress (Foyer et al. 1997; Foyer and Noctor 2005). According to the results in this study, we tentatively propose that the decrease in the glutathione redox state by decreased GR activity under oxidative stress may induce a failure in redox sensing, which results in a decrease in the ASC and the ascorbate redox state that in turn leads to an accumulation of reactive oxygen species, thus causing oxidative damage. However, how the decrease in GR activity results in the decrease in ASC content and the ascorbate redox state under oxidative stress remains to be studied further.
In the present study, wild type and transgenic plants were treated with 20 μM MV for two weeks to induce oxidative stress. Such a MV treatment resulted in oxidative damage as indicated by leaf bleaching, lipids damage and a decrease in photosynthetic function in particular in transgenic plants (Figs. 3, 4, 5). On the other hand, such a long stress period is likely to cause signalling effects that allow the onset of acclimation processes. It has been suggested that H2O2 generated in the chloroplast during high light stress can act as both a local and systemic signal to elicit defensive or acclimatory responses (Karpinski et al. 1997, 1999, 2003; Mullineaux and Karpinski 2002; Fryer et al. 2003; Chang et al. 2004; Mullineaux et al. 2006). Pretreated with H2O2 can trigger protective functions that cause plants to acclimate to a range of stress conditions (Karpinski et al. 1999). In this study, we observed that MV treatment resulted in an increase in the content of H2O2 in the leaves in wild type and transgenic plants (Fig. 6) and accumulation of H2O2 occurred in the cell walls in MV-treated wild type and transgenic plants (Fig. 7), suggesting that there is an apparent signalling effect in MV-treated wild type and transgenic plants. Indeed, we observed that there was an increase in total foliar activities of GR, APX, SOD, and CAT as well as in the levels of reduced glutathione in MV-treated plants as compared to those that were grown in the absence of MV (Figs. 8, 9, 10). APXs are key enzymes in the antioxidant network and catalyze the reduction of H2O2 to water using ascorbate as the electron donor (Asada 1999). It has been shown that chloroplastic APXs are prone to inactivation by ROS whereas the levels of cytoplasmic APXs are known to increase significantly in response to high light stress (Asada 1999; Karpinski et al. 1997, 1999). Our results show that there was accumulation of H2O2 in the chloroplast in MV-treated transgenic plants (Fig. 7). However, we still observed that MV treatment resulted in an increase in total foliar APX activity in MV-treated plants as compared to those that were grown in the absence of MV (Fig. 9b). Thus, it is reasonable to speculate that MV-induced increase in total foliar activity of APX observed in this study may be associated with an increase in the activities of cytoplasmic APXs. It has been shown that the expression of the gene APX2, encoding a cytosoloc ascorbate peroxidase, is observed within 15 min after the onset of high light stress and associated with accumulation of H2O2 in bundle sheath cells (Karpinski et al. 1997, 1999; Fryer et al. 2003). This regulation of APX2 has been suggested to play an important role in protecting the sites of primary photosynthesis from ROS and, at the same time, stimulating ROS accumulation to signal responses to high light stress (Fryer et al. 2003). Thus, the results in this study suggest that MV-induced oxidative stress causes signalling effects that allow the onset of acclimation processes.
In conclusion, we obtained transgenic tobacco plants with significant decreased GR activity in order to investigate the possible roles of GR in protection against oxidative stress. Under oxidative stress, there was a greater decrease in the GSH/GSSG ratio but a greater increase in foliar GSH in transgenic plants than in wild type plants. On the other hand, there were greater decreases in foliar ASC and the ascorbate redox state in transgenic plants than in wild type plants. The decrease in foliar ASC and the ascorbate redox state would limit a detoxification of H2O2, thus resulting in an accumulation of H2O2, which caused oxidative damage in transgenic plants under oxidative stress conditions. The results in this study suggest that the capacity for regeneration of glutathione by GR plays an important role in protecting against oxidative stress by maintaining ascorbate pool and ascorbate redox state. The results in this study also suggest that MV-induced oxidative stress causes signalling effects that allow the onset of acclimation processes.
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126. doi:10.1016/S0076-6879(84)05016-3
Alscher RG (1989) Biosynthesis and antioxidant function of glutathione in plants. Physiol Plant 77:457–464. doi:10.1111/j.1399-3054.1989.tb05667.x
Amsellem Z, Jansen MAK, Driesenaar ARJ, Gressel J (1993) Developmental variability of photooxidative stress tolerance in paraquat-resistant Conyza. Plant Physiol 103:1097–1106
An G (1987) Binary Ti vectors for plant transformation and promoter analysis. Methods Enzymol 153:292–305. doi:10.1016/0076-6879(87)53060-9
Anderson JV, Chevone B, Hess JL (1992) Seasonal variation in the antioxidant system of eastern white pine needles. Plant Physiol 98:501–508
Aono M, Kubo A, Saji H, Tanaka KK, Kondo N (1993) Enhanced tolerance to photooxidative stress of transgenic Nicotiana tabacum with chloroplastic glutathione reductase activity. Plant Cell Physiol 34:129–135
Aono M, Saji H, Fujiyama K, Sugita M, Kondo N, Tanaka K (1995) Decrease in activity of glutathione reductase enhanced paraquat sensitivity in transgenic Nicotiana tabacum. Plant Physiol 107:645–648
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol 24:1–15
Arrigoni O, Dipierro S, Borraccino G (1981) Ascorbate free radical reductase: a key enzyme of the ascorbic acid system. FEBS Lett 125:242–244. doi:10.1016/0014-5793(81)80729-6
Asada K (1999) The water–water cycle in chloroplasts: scavenging active oxygen species and dissipation of excess photons. Annu Rev Plant Physiol Mol Biol 50:601–639. doi:10.1146/annurev.arplant.50.1.601
Bestwick CS, Brown IR, Bennett MH, Mansfield JW (1997) Localization of hydrogen peroxide accumulation during the hypersensitive reaction of lettuce cells to Pseudomonas syringae pv phaseolicola. Plant Cell 9:209–221
Beyer WF, Fridovich I (1987) Assaying for superoxide dismutase activity: some large consequences of minor changes in condition. Anal Biochem 72:248–254
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein using the principal of protein-dye binding. Anal Biochem 72:248–254. doi:10.1016/0003-2697(76)90527-3
Broadbent P, Creissen GP, Kular B, Wellburn AR, Mullineaux PM (1995) Oxidative stress responses in transgenic tobacco containing altered levels of glutathione reductase activity. Plant J 8:247–255. doi:10.1046/j.1365-313X.1995.08020247.x
Chang CC, Ball L, Fryer MJ, Baker NR, Karpinski S, Mullineaux PM (2004) Induction of ASCORBATE PEROXIDASE 2 expression in wounded Arabidopsis leaves does not involve known wound-signalling pathways but is associated with changes in photosynthesis. Plant J 38:499–511. doi:10.1111/j.1365-313X.2004.02066.x
Creissen GP, Mullineaux PM (1995) Cloning and characterization of glutathione reductase cDNAs and identification of two genes encoding the tobacco enzyme. Planta 197:422–425. doi:10.1007/BF00202667
Creissen GP, Edwards EA, Enard C, Wellburn A, Mullineaux PM (1992) Molecular characterization of glutathione reductase cDNAs from pea (Pisum sativum L.). Plant J 2:129–131
Creissen GP, Reynolds H, Xue Y, Mullineaux PM (1995) Simultaneous targeting of pea glutathione reductase and of a bacterial fusion protein to chloroplasts and mitochondria in transgenic tobacco. Plant J 8:167–175. doi:10.1046/j.1365-313X.1995.08020167.x
Dodge AD (1994) Herbicide action and effects on detoxification processes. In: Foyer CH, Mullineaux PM (eds) Causes of photooxidative stress and amelioration of defense systems in plants. CRC Press, Boca Raton, pp 219–236
Edwards EA, Rawsthorne S, Mullineaux PM (1990) Subcellular distribution of multiple forms of glutathione reductase in leaves of pea (Pisum sativum L.). Planta 180:278–284. doi:10.1007/BF00194008
Edwards EA, Enard C, Creissen GP, Mullineaux PM (1994) Synthesis and properties of glutathione reductase in stressed peas. Planta 192:137–143
Foyer CH (1997) Oxygen metabolism and electron transport in photosynthesis. In: Scandalios J (ed) Oxidative stress and the molecular biology of antioxidant defenses. Cold Spring Harbor Laboratory Press, New York, pp 587–621
Foyer CH, Halliwell B (1976) The presence of glutathione and glutathione reductase in chloroplast: a proposed role in ascorbic acid metabolism. Planta 133:21–25. doi:10.1007/BF00386001
Foyer CH, Noctor G (2005) Redox homeostasis and antioxidant signalling: a metabolic interface between stress perception and physiological responses. Plant Cell 17:1866–1875. doi:10.1105/tpc.105.033589
Foyer CH, Lelandais M, Galap C, Kunert KJ (1991) Effects of elevated cytosolic glutathione reductase activity on the cellular glutathione pool and photosynthesis in leaves under normal and stress conditions. Plant Physiol 97:863–872
Foyer CH, Souriau N, Perret S, Lelandais M, Kunert KJ, Pruvost C, Jouanin L (1995) Overexpression of the glutathione reductase but not glutathione synthetase leads to increases in antioxidant capacity and resistance to photoinhibition in poplar trees. Plant Physiol 109:1047–1057. doi:10.1104/pp.109.3.1047
Foyer CH, Lopez-Delgado H, Dat JF, Scott IM (1997) Hydrogen peroxide- and glutathione-associated mechanisms of acclamatory stress tolerance and signalling. Physiol Plant 100:241–254. doi:10.1111/j.1399-3054.1997.tb04780.x
Fryer MJ, Ball L, Oxborough K, Karpinski S, Mullineaux PM, Baker NR (2003) Control of ascorbate peroxidase 2 expression by hydrogen peroxide and leaf water status during excess light stress reveals a functional organisation of Arabidopsis leaves. Plant J 33:691–705. doi:10.1046/j.1365-313X.2003.01656.x
Gallogly MM, Mieyal JJ (2007) Mechanisms of reversible protein glutathionylation in redox signalling and oxidative stress. Curr Opin Pharmacol 7:381–391. doi:10.1016/j.coph.2007.06.003
Gomez LD, Noctor G, Knight MR, Foyer CH (2004) Regulation of calcium signalling and gene expression by glutathione. J Exp Bot 55:1851–1859. doi:10.1093/jxb/erh202
González A, Steffen KL, Lynch JP (1998) Light and excess manganese. Plant Physiol 118:493–504. doi:10.1104/pp.118.2.493
Hausladen A, Alscher RG (1994) Cold-hardness-specific glutathione reductase isoforms in red spruce. Plant Physiol 105:215–223. doi:10.1104/pp.105.1.215
Hell R (1997) Molecular physiology of plant sulfur metabolism. Planta 202:138–148. doi:10.1007/s004250050112
Hodges DM, De Long JM, Forney CF, Prange RK (1999) Improving the thiobarbituric acid-reactive-substances assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta 270:604–611. doi:10.1007/s004250050524
Horsh RB, Fry JE, Hoffman HL, Eicholts D, Rogers SG, Fraley RT (1985) Simple and general method for transferring genes into plants. Science 277:1229–1231
Huang Z, Pinto JT, Deng H, Richie JP Jr (2008) Inhibition of caspase-3 activity and activation by protein glutathionylation. Biochem Pharmacol 75:2234–2244. doi:10.1016/j.bcp.2008.02.026
Jiménez A, Hernandez JA, del Rio LA, Sevilla F (1997) Evidence for the presence of the ascorbate-glutathione cycle in mitochondria and peroxisomes of pea leaves. Plant Physiol 114:275–284
Karpinski S, Escobar C, Karpinska B, Creissen G, Mullineaux PM (1997) Photosynthetic electron transport regulates the expression of cytosolic ascorbate peroxidase genes in excess light stress. Plant Cell 9:627–640
Karpinski S, Reynolds H, Karpinska B, Wingsle G, Creissen G, Mullineaux P (1999) Systemic signalling and acclimation in response to excess excitation energy in Arabidopsis. Science 23:654–657. doi:10.1126/science.284.5414.654
Karpinski S, Gabrys H, Mateo A, Karpinska B, Mullineaux PM (2003) Light perception in plant disease defence signalling. Curr Opin Plant Biol 6:390–396. doi:10.1016/S1369-5266(03)00061-X
Knörzer OC, Durner J, Böger P (1996) Alterations in the antioxidative system of suspension-cultured soybean cells (Glycine max) induced by oxidative stress. Physiol Plant 97:388–396. doi:10.1034/j.1399-3054.1996.970225.x
Kocsy G, Brunner M, Rüegsegger A, Stamp P, Brunold C (1996) Glutathione synthesis in maize genotypes with different sensitivities to chilling. Planta 198:365–370. doi:10.1007/BF00620052
Kocsy G, Szalai G, Vágújfalvi A, Stéhli L, Orosz G, Galiba G (2000) Genetic study of glutathione accumulation during cold hardening in wheat. Planta 210:295–301. doi:10.1007/PL00008137
Krause GH, Weis E (1991) Chlorophyll fluorescence and photosynthesis: the basics. Annu Rev Plant Physiol Plant Mol Biol 42:313–349. doi:10.1146/annurev.pp.42.060191.001525
Kunert KJ, Foyer CH (1993) Thiol/disulphide exchange in plants. In: De Kok LJ (ed) Sulfur nutrition and assimilation in higher plants. SPB Academic Publishing, The Hague, The Netherlands, pp 139–151
Laemmli HK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685. doi:10.1038/227680a0
Lim CE, Choil JN, Kim IA, Lee SA, Hwang YS, Lee CH, Lim J (2008) Improved resistance to oxidative stress by a loss-of-function mutation in the Arabidopsis UGT71C1 Gene. Mol Cells 25:368–375
Maciejewska U, Kacperska A (1987) Changes in the levels of oxidized and reduced pyridine nucleotides during cold acclimation of winter grape plants. Physiol Plant 69:687–691. doi:10.1111/j.1399-3054.1987.tb01985.x
Marrs K (1996) The functions and regulation of gluthathione S-transferases in plants. Annu Rev Plant Physiol Plant Mol Biol 48:127–158. doi:10.1146/annurev.arplant.47.1.127
Maughan S, Foyer CH (2006) Engineering and genetic approaches to modulating the glutathione network in plants. Physiol Plant 126:382–397. doi:10.1111/j.1399-3054.2006.00684.x
May MJ, Vernoux T, Leaver C, Montagu MV, Inzé D (1998) Glutathione homeostasis in plants: implications for environmental sensing and plant development. J Exp Bot 49:649–667. doi:10.1093/jexbot/49.321.649
Mittova V, Theodoulou FL, Kiddle G, Gómez L, Volokita M, Tal M, Foyer CH, Guy M (2003) Coordinate induction of glutathione biosynthesis and glutathione metabolizing enzymes is correlated with salt tolerance in tomato. FEBS Lett 554:417–421. doi:10.1016/S0014-5793(03)01214-6
Mullineaux P, Karpinski S (2002) Signal transduction in response to excess light: getting out of the chloroplast. Curr Opin Plant Biol 5:43–48. doi:10.1016/S1369-5266(01)00226-6
Mullineaux PM, Karpinski S, Baker NR (2006) Spatial dependence for hydrogen peroxide-directed signalling in light stressed plants. Plant Physiol 141:346–350. doi:10.1104/pp.106.078162
Nakano Y, Asada K (1980) Spinach chloroplasts scavenge hydron perodide on illumination. Plant Cell Physiol 21:1295–1307
Noctor G, Foyer CH (1998) Ascorbate and glutathione: keeping active oxygen under control. Annu Rev Plant Physiol Plant Mol Biol 49:249–279. doi:10.1146/annurev.arplant.49.1.249
Noctor G, Arisi AM, Jouanin L, Kunert KJ, Rennenberg H, Foyer CH (1998) Glutathione: biosynthesis, metabolism and relationship to stress tolerance explored in transformed plants. J Exp Bot 49:623–647. doi:10.1093/jexbot/49.321.623
Noctor G, Gomez L, Vanacker H, Foyer CH (2002) Interactions between biosynthesis, compartmentation and transport in the control of glutathione homeostasis and signalling. J Exp Bot 53:1283–1304. doi:10.1093/jexbot/53.372.1283
Pilon-Smit EAH, Zhu YL, Sears T, Terry N (2000) Overexpression of glutathione reductase in Brassica juncea: effects on cadmium accumulation and tolerance. Physiol Plant 110:455–460. doi:10.1111/j.1399-3054.2000.1100405.x
Queval G, Hager J, Gakière B, Noctor G (2008) Why are literature data for H2O2 contents so variable? A discussion of potential difficulties in the quantitative assay of leaf extracts. J Exp Bot 59:135–146. doi:10.1093/jxb/erm193
Robinson GP, Walker DA (1979) Rapid separation of the chloroplast and cytoplasmic fractions from intact leaf protoplasts. Arch Biochem Biophys 196:319–323. doi:10.1016/0003-9861(79)90584-8
Sanchez-Fernández R, Fricker M, Corben LB, White NS, Sheard N, Leaver CJ, Van Montagu M, Inzé D, Nay NJ (1997) Cell proliferation and hair tip growth in the Arabidopsis root are under mechanistically different forms of redox control. Proc Natl Acad Sci USA 94:2745–2750. doi:10.1073/pnas.94.6.2745
Stevens RG, Creissen GP, Mullineaux PM (1997) Cloning and characterisation of a cytosolic glutathione reductase cDNA from pea (Pisum sativum L.) and its expression in response to stress. Plant Mol Biol 35:641–654. doi:10.1023/A:1005858120232
Stevens RG, Creissen GP, Mullineaux PM (2000) Characterization of pea cytosolic glutathione reductase expressed in transgenic tobacco. Planta 211:537–545. doi:10.1007/s004250000304
Veljovic-Jovanovic S, Noctor G, Foyer CH (2002) Are leaf hydrogen peroxide concentrations commonly overestimated? The potential influence of artefactual interference by tissue phenolics and ascorbate. Plant Physiol Biochem 40:501–507. doi:10.1016/S0981-9428(02)01417-1
Wesley SV, Helliwell CA, Smith NA, Wang MB, Rouse DT, Liu Q, Gooding PS, Singh SP, Abbott D, Stoutjesdijk PA, Robinson SP, Gleave AP, Green AG, Waterhouse PM (2001) Construct design for efficient, effective and high throughput gene silencing in plants. Plant J 27:581–590. doi:10.1046/j.1365-313X.2001.01105.x
Wingate VPM, Lawton MA, Lamb CJ (1988) Glutathione causes a massive and selective induction of plant defense genes. Plant Physiol 31:205–211
Xiong Y, Contento AL, Nguyen PQ, Bassham DC (2007) Degradation of oxidized proteins by autophagy during oxidative stress in Arabidopsis. Plant Physiol 143:291–299. doi:10.1104/pp.106.092106
Yang X, Liang Z, Lu C (2005) Genetic engineering of the biosynthesis of glycinebetaine enhances photosynthesis against high temperature stress in transgenic tobacco plants. Plant Physiol 138:2299–2309. doi:10.1104/pp.105.063164
Yoshimura K, Miyao K, Gaber A, Takeda T, Kanaboshi H, Miyasaka H, Shigeoka S (2004) Enhancement of stress tolerance in transgenic tobacco plants overexpressing Chlamydomonas glutathione peroxidase in chloroplasts or cytosol. Plant J 37:21–33. doi:10.1046/j.1365-313X.2003.01930.x
Acknowledgments
We thank CSIRO Plant Industry for providing the RNAi vectors and Prof. Jinxin Lin for help with cytochemical analysis. This work was supported by the Frontier Project of the Knowledge Innovation Engineering of Chinese Academy of Sciences (KSCX2-YW-N-042), the National Natural Science Foundation of China (30725024), and the Major Key Basic Research and Development Plan of China (2009CB118500).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ding, S., Lu, Q., Zhang, Y. et al. Enhanced sensitivity to oxidative stress in transgenic tobacco plants with decreased glutathione reductase activity leads to a decrease in ascorbate pool and ascorbate redox state. Plant Mol Biol 69, 577–592 (2009). https://doi.org/10.1007/s11103-008-9440-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-008-9440-3