Abstract
Purpose
Patients suffering from craniopharyngiomas currently have good survival rates, but long-term sequelae, such as development of obesity, worsen their quality of life. Optimal treatment is still controversial and changed during the decades, becoming less aggressive. Transcranial (TC) surgery was the first approach to be used, followed by extended transsphenoidal (eTNS) access. This study aims to compare the two approaches in terms of risk of hypothalamic damage leading to obesity.
Methods
This is a monocentric retrospective analysis of post-puberal patients treated for primary craniopharyngioma. Postoperative obesity and percentual postsurgical BMI variation were considered proxy for hypothalamic function and used to fit regression models with basal BMI, type of surgery, tumor volume and hypothalamic involvement (anterior vs. anteroposterior).
Results
No difference in radicality was observed between the two approaches; eTNS was more effective in ameliorating visual function but was significantly associated with CSF leaks. The TC approach was associated with a higher incidence of diabetes insipidus. Regression analysis showed only tumor volume and basal BMI resulted as independent predictors for both postoperative obesity (respectively, OR 1.15, P = 0.041, and OR 1.57, P < 0.001) and percentual BMI variation (respectively, + 0.92%, P = 0.005, and − 1.49%, P = 0.001).
Conclusions
Larger lesions portend a higher risk to develop postoperative obesity, independently of hypothalamic involvement. Interestingly, basal BMI is independent of lesional volume and is associated with postoperative obesity, but lesser postoperative BMI variation. The surgical approach does not influence the obesity risk. However, eTNS proves valid in managing large tumors with important hypothalamic invasion.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Craniopharyngioma is a histologically benign tumor arising in the sellar and suprasellar region along the path of the craniopharyngeal duct. It occurs at a rate of 1.3 per million person per year [1] and accounts for 2–5% of all primary intracranial tumors, and 3–5.6% of intracranial neoplasms in the pediatric population [2, 3]. Despite its benign appearance, it can portend an unfavorable prognosis due to the relationships with deep diencephalic structures [4]. The quality of life should be considered a principal endpoint, since patients currently experience good overall survival rates, as high as 85–93% at 10 years, regardless of the degree of surgical resection [5]. Long-term sequelae worsen the quality of life in 50% of long-term survivors, especially obesity and neurobehavioral impairment caused by hypothalamic involvement or treatment-related lesions [3].
The optimal treatment of patients with craniopharyngioma is still controversial and has changed during the decades, shifting from attempting total removal to a less aggressive surgery [6,7,8,9,10,11]. Historically, transcranial approaches were the first to be used to address this pathology [5, 12, 13]. Transsphenoidal surgery (TNS), which allows access to the sellar region without brain retraction [14], was reserved only for lesions with a predominant intrasellar component [5, 15]. However, with the development of the extended transsphenoidal access [16, 17], by means of removal of the tuberculum sellae and the posterior planum sphenoidale, asportation of purely suprasellar pathologies became possible [18, 19], so that in the last decades this technique has increasingly been reported for such lesions [8, 20,21,22].
Therefore, we intended to assess whether the extended transsphenoidal (eTNS) approach is more advantageous than the transcranial (TC) one to reduce the risk of hypothalamic surgical damage and particularly of obesity.
Materials and methods
Study design
This is a cohort study consisting of a retrospective analysis of patients treated for primary craniopharyngioma at the Department of Neurosurgery of our institution, between 1994 and 2019. Increasing experience and comfort with eTNS technique, led to treat more patients through this approach over the years. As a matter of fact, before 2010, the eTNS approach was reserved for lesions with important sellar invasion and enlargement, while afterwards the main criterion used to select this approach has become the MRI evidence of a direct sagittal corridor to prevalently cystic tumors, mostly independent of sellar dimension. Consequently, after 2010 eTNS has been progressively adopted even for tumors with predominant suprasellar extension and normal-size pituitary fossa, while the TC approach has remained the first choice for very asymmetrical lesions, with predominant lateral or subfrontal extension, reaching up to the foramina of Monro and beyond. Therefore, the groups of interest are the eTNS and TC surgical cohorts with hypothalamic involvement, as assessed by preoperative magnetic resonance imaging (MRI). Exclusion criteria consisted of previous surgery, no hypothalamic involvement according to Müller’s classification [23], prepuberal stage (age < 14 years), and missing data about body mass index (BMI) or tumor volume.
Data collection
Clinical and tumor characteristics at baseline and during long-term follow-up were collected in a prospectively maintained database that included general information (age, sex, date of surgery, height, weight, BMI), clinical presentation (pituitary function, visual deficits), tumor characteristics (location, volume, hypothalamic involvement, histology), treatment outcome (radicality of resection at early postoperative MRI, as assessed by the two senior authors, which was dichotomized in radical resection vs. macroscopic evidence of residue), and long-term follow-up information (further treatments, either surgical or radiant, complications, date of last follow-up, and BMI at last follow-up).
Surgical complications were considered major when resulted in permanent disabling sequelae or required further surgical intervention, otherwise were classified as minor. Infections of the central nervous system were considered separately from other infective events and were classified as major when they occurred in the presence of cerebrospinal fluid (CSF) leak, and minor in the absence of overt fistulae. CSF fistulae were dichotomized in major, when requiring surgical repair, and minor when autonomously recovered following single lumbar puncture or a short course of continuous external lumbar drainage.
Preoperative neuroradiological evaluation
DICOM images were analyzed with OsiriX software (version 8.5.2) and craniopharyngioma was delimited as a region of interest (ROI), for which the volume was automatically provided in cubic centimeters. Hypothalamic involvement was graded into anterior and anteroposterior according to Müller’s classification [23]. We also considered the T2 hyperintensity of hypothalamic structure [24].
Clinical evaluation
Pituitary function was assessed as detailed elsewhere [4]. Briefly, GH secretion deficit was diagnosed in pubertal children with growth arrest and in adults with an insufficient GH response to stimulation tests. Hypogonadotropic hypogonadism was diagnosed in premenopausal women with amenorrhea and in men with subnormal testosterone levels, and low or normal gonadotropin levels in both sexes. Secondary hypoadrenalism was diagnosed in patients with low morning cortisol levels and/or clinical symptoms of hypoadrenalism which improved after replacement therapy with glucocorticoids. Secondary hypothyroidism was detected in patients with low free-thyroxine levels and normal or suppressed TSH concentrations. Diabetes insipidus was diagnosed, before or after surgery, in patients who had hypotonic polyuria (> 40 ml/kg body weight daily).
Pre- and postoperative visual function was investigated, both in relation to acuity and perimetry, and their evolution after surgical treatment in terms of amelioration, deterioration, or development of new deficit.
Hypothalamic function evaluation
Anthropometric data were used as a proxy for hypothalamic control over body composition both preoperatively and at last follow-up. BMI was calculated as the body weight divided by squared height (kg/m2). Postoperative BMI and the percentual BMI variation with respect to preoperative BMI were considered. Obesity was defined as a BMI > 30 kg/m2.
Follow-up and recurrence
Patients were followed with serial contrast-enhanced MRI examinations, the first postoperative study being within 3 months of surgery, and subsequent surveillance imaging recommended at 6-month intervals for 2 years and then yearly or at increasing intervals, depending on the radiological and clinical findings. All patients also underwent a complete postoperative neuro-ophthalmologic evaluation with visual field testing.
Length of follow-up was measured in months and was separated into clinical follow-up (from the day of surgery to the last clinical update) and radiological follow-up (from surgery date to the date of last MRI).
Recurrence (the appearance of pathological tissue that had not previously been detected on MRI) and progression (new growth of pre-existing residual disease) were considered together. In such case, time to recurrence/progression was calculated as the interval between date of surgery and the date of the MRI showing recurrence/progression.
Statistical analysis
Continuous data were examined for homogeneity of variance and normal distribution. Mean ± standard deviation (SD) and median and interquartile range (IQR) were reported, as appropriate. Statistics of means were carried out using the unpaired Student t-test for normally distributed variables and Wilcoxon rank sum tests for not normally distributed variables. Categorical variables were described by means of crosstabulation and were compared with the Chi-square test or Fisher exact test as appropriate. Postoperative control over body weight was investigated by means of two models. In the former, the categorical variable of postoperative obesity (defined as a postoperative BMI > 30 kg/m2) was analyzed by means of simple and multiple logistic regression. In the latter model, percentual BMI variation with respect to baseline was examined by means of simple and multiple linear regression analysis. Estimates of the cumulative event rate for recurrence/progression of the tumor were calculated with the Kaplan–Meier method and statistically significant difference between patient subgroups were investigated with the Log Rank test. Data for patients who were lost at follow-up, died, or refused further controls were censored at the time of the last radiological study. A P value < 0.050 was considered to indicate statistical significance and all reported probability values were two-tailed. All analyses were performed using R (version 3.6.3) and RStudio (version 1.2.1335).
Results
Patient population
From 1994 to 2019, a total of 201 patients with craniopharyngioma have been treated at our institution. However, we were able to systematically review the images of patients whose studies were carried out at our hospital after the installment of the new digital image archive system or of those who sent us their preoperative MRI when contacted for the clinical follow-up. Additionally, more dated studies were almost exclusively in non-digital format, thus not valuable for software volumetric analyses. Therefore, we selected 90 total patients that were subsequently cut down to 59 (20 eTNS and 39 TC) according to the exclusion criteria (Fig. 1). To ascertain that this selection had not introduced any unseen bias in our study population and that the two cohorts could be considered bona fide homogeneous, we evaluated a range of relevant basal and postoperative clinical, pathological, and radiological data, grouped according to the type of surgical approach, as reported in Tables 1 and 2. Notably, significant differences between the two groups were observed for the detection at MRI of hypothalamic involvement, as anteroposterior extension was consistently more represented in the TC group (69.2% vs 35%, P = 0.025). This imbalance is explained by the fact that the anteroposterior extension of the tumor was an important determinant in the choice of the surgical approach up until the more recent years. The TC cohort showed a slightly higher tumoral volume than eTNS, even if not significant, as well as a slightly more compromised basal pituitary function. However, only hypogonadism was significantly more frequent in TC patients (81.6% vs 42.1%, P = 0.007). Correlation between tumor volume and hormonal deficits was significant only for the thyroid ax (t-test and logistic regression P = 0.038), while not significant when considering basal gonadal (t-test P = 0.199, logistic regression P = 0.219) and adrenal (t-test P = 0.602, logistic regression P = 0.581) axes. The eTNS cohort comprised older patients (50 ± 16.4 vs. 40.2 ± 12.9 years, P = 0.017) and consisted mainly of males (80% vs 43.6%, P = 0.026). No difference in the surgical radicality was observed between the two groups (89.5% in both cohorts, P = 1.00), indicating that both techniques were equally effective. On the other hand, the eTNS cohort had significantly shorter clinical (38.4 ± 45.0 vs 80.8 ± 52.7 months, P = 0.001) and radiological (33.2 ± 31.3 vs 78.4 ± 49.2 months, P < 0.001) follow-up times, as this technique has been increasingly used in the more recent years.
Postsurgical visual and pituitary outcome
As outlined in Table 3, when considering population with basal preoperative visual defects, eTNS proved to be more effective than TC in ameliorating visual function (improved 64.3% vs. 59.3%, normalized 35.7% vs. 7.4%, P = 0.031). Conversely, there was no difference between the two approaches in the development of new onset visual deficits. The TC approach was associated with a higher incidence of new onset diabetes insipidus than the eTNS approach (89.7% vs. 62.5%, P = 0.050).
Postsurgical BMI and obesity
In the total study population, the mean postoperative BMI at last follow-up was 29.7 ± 6.0 kg/m2 (median 29.1 kg/m2, IQR 6.21) with a percentual variation of BMI of + 16.6 ± 20.0% with respect to the presurgical value. Overt obesity (BMI > 30 kg/m2) affected 26/59 (44.1%) of patients. Considering the two surgical cohorts, mean last follow-up BMI was 29.3 ± 5.2 kg/m2 (median 28.7 kg/m2, IQR 5.86) for the eTNS and 29.9 ± 6.3 kg/m2 (median 29.4 kg/m2, IQR 6.31) for the TC (t-test on means P = 0.723, Wilcoxon test on medians P = 0.861). Paired analysis of postoperative and basal BMI on the total population (paired t-test P < 0.001, paired Wilcoxon test P < 0.001) as well as on eTNS (paired t-test P < 0.001, paired Wilcoxon test P = 0.002) and TC groups (paired t-test P < 0.001, paired Wilcoxon test P < 0.001) showed significant increase of postoperative BMI. The percentual postoperative BMI variation was + 10.4 ± 11.0% in the eTNS group and + 20.0 ± 22.9% in the TC group (P = 0.174). Overt obesity occurred in 9/20 (45.0%) of eTNS patients and in 17/39 (43.6%) of the TC patients (P = 1.00). When considering only new onset postoperative obesity in those not preoperatively obese, 5/15 (33.3%) eTNS patients and 10/31 (32.3%) TC patients became obese (P = 1.00). These results indicate an invariable increase in BMI after surgery, independent of the approach utilized.
We then investigated postoperative body weight evolution by two different methods, i.e., considering both the occurrence of overt obesity (BMI > 30 kg/m2) and the percentual postoperative BMI variation. When we considered the dichotomous category of postoperative obesity by means of logistic regression, only tumor volume (OR 1.15, P = 0.041) and basal BMI (OR 1.57, P < 0.001) were independent predictors of postoperative obesity (Table 4).
When addressing the percentual postsurgical BMI change, we found a significant correlation with tumor volume, basal BMI, and anteroposterior hypothalamic involvement (Table 5). The eTNS approach was associated with a trend toward a reduced postoperative BMI variation, even though it did not reach statistical significance (Fig. 2). We found that tumor volume and basal BMI were independently associated with the percentual variation of BMI, as proven by multiple linear regression in this analysis, too (Table 5). The independence of these two variables is clearly reported in Fig. 3a. Remarkably, while tumor volume was positively associated with postoperative BMI variation (increase of 0.92%, P = 0.005, see also Fig. 3b), basal BMI was inversely associated with it (decrease of 1.49%, P = 0.001). This implies that patients with higher basal BMI have a proportionally lower further weight gain in the postsurgical setting, probably because hypothalamic control over body weight is already impaired from the outset and can hardly be further worsened (Supplementary Fig. 1).
Correlation between craniopharyngioma volume and BMI: a shows independency of tumor volume and basal BMI; b shows the correlation between lesional burden and postoperative BMI variation with respect to baseline. Black dots indicate eTNS cases, gray triangles TC cases. Black lines show the linear regression fit
Additional analyses to correlate basal hypogonadism, hypothyroidism and hypoadrenalism with baseline BMI (respectively P = 0.709, P = 0.240, P = 0.589) and baseline obesity (respectively P = 0.716, P = 0.722, P = 1) did not show significant correlation. The same was evident when considering postoperative hypogonadism, hypothyroidism and hypoadrenalism with postoperative BMI (respectively P = 0.442, P = 0.523, P = 0.422) and obesity (respectively P = 0.436, P = 0.430, P = 0.277).
Complications
Major surgical complications occurred in 5 patients (8.5%). No differences were seen between the two surgical groups (10.0% in the eTNS group vs. 7.7% in the TC group, P = 1.00). CSF leaks affected primarily eTNS patients (P = 0.020): in particular, minor cases, i.e., those not requiring repeated surgery, were recorded both in the eTNS (3/20, 15.0%) and in the TC (1/39, 2.6%) groups, whereas major CSF leak, requiring reintervention, occurred in the eTNS group only (2/20, 10.0%). Two cases of meningitis associated with CSF fistula (10.0%) and 1 not associated with CSF leak (5.0%) affected the eTNS cohort, while only one meningitis with no fistula (2.6%) was recorded in the TC group (P = 0.108).
Recurrence/progression-free survival analysis
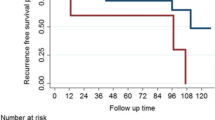
All patients were alive at the end of the study. Recurrence/progression occurred in 10 cases (17.2%), of whom 4 (20.0%) were in the eTNS group and 6 (15.8%) in the TC group (P = 0.724). Five years progression-free survival (5y-PFS) in the whole group of patients was 83.3 ± 6.0%. In particular 5y-PFS was of 59.7 ± 17.3% in the eTNS cohort and 89.9 ± 5.7% in the TC group. Figure 4 shows the Kaplan–Meier curves (Log-rank P = 0.095).
Discussion
Hypothalamic obesity
Hypothalamic obesity occurs when a tumor impinges on the infundibulum and injures the median eminence and arcuate nucleus; 12–19% of patients affected by craniopharyngioma are obese at diagnosis [25], which is in keeping with what observed in our study (20.3%).
There are two hypotheses for describing the genesis of hypothalamic obesity: either destruction of the hypothalamic appetite control center, leading to hyperphagia with resultant obesity and hyperinsulinism, or disinhibition of efferent fibers of the vagus nerve, leading to hypersecretion of insulin that promotes increased fat deposition and insulin resistance. In the latter situation, eating behavior is not different from age, sex, and BMI-matched controls. Therefore, obesity is probably explained more by a decreased energy expenditure rather than an increased appetite [25]. Patients with craniopharyngioma typically have a significant and rapid weight gain in the first 6–12 months after surgical resection with a prevalence of obesity between 22 and 75% [25, 26]. BMI then reaches a plateau, without further increase during long-term follow-up [27]. The consequences of hypothalamic obesity include an increased risk of developing metabolic syndrome, cardiovascular disease, respiratory problems, psychosocial complications, and excess mortality [28].
The transcranial approach for craniopharyngiomas may be responsible for direct hypothalamic damage or injury to small perforating arteries [29], with the subsequent major threat of hypothalamic syndrome, which almost always includes obesity [30,31,32]. Several studies have reported postsurgical hypothalamic damage rates ranging from 65 to 80%, while increase in body weight or obesity ranged from 39 to 55% during long-term follow-up [33]. Both hyperphagia and hypothalamic obesity have been suggested to occur more frequently when gross total removal of the tumor is attempted, especially in children, leading several authors to accept the trade-off between a less aggressive tumor removal with a decreased likelihood of hypothalamic damage [5, 32, 34].
In this study, we investigated whether the type of surgical approach is associated with the risk of developing postoperative obesity, because it is generally held that TC surgery is more damaging than the eTNS approach [8, 12, 21]. Our final multivariate models showed that only tumor volume and basal BMI were the main, independent predictors of postoperative weight gain, while the type of surgical approach did not have an independent association with the development of hypothalamic obesity. Accordingly, Hofmann et al. stated that preoperative hypothalamic functional disturbance, in the form of presurgical weight gain, is a risk factor for hypothalamic obesity; hence it should be considered a contraindication for aggressive surgery [35, 36]. Remarkably, our data confirm that while higher preoperative BMI was associated with increased risk of postoperative obesity, it was associated with an inferior BMI variation during follow-up. The possible explanation is that hypothalamic dysfunction in the preoperative setting is unlikely to reverse after surgical removal of the tumor, but it is also unlikely to deteriorate, an observation which is in line with other authors [37, 38].
The anatomical basis of hypothalamic obesity probably involves the dysfunction of various diencephalic nuclei, including the ventromedial hypothalamus, the lateral hypothalamic area, the paraventricular nuclei, and the arcuate nucleus. Moreover, the development of hypothalamic obesity is also affected by the interplay between the hypothalamus and other critical encephalic structures. In particular, the basolateral amygdala, the medial prefrontal cortex, and the lateral hypothalamic area form a critical neural network in the regulation of eating by learned, motivational cues [39]. In this context, the amygdala nuclei seed a complex network of direct and indirect projections to the hypothalamus, with the basolateral amygdala and lateral hypothalamic area as the essential components through which the learned cues can override satiety and increase eating [39]. Additionally, the hypothalamic regulation of food intake relies on the reward and motivational neural circuitry to modify eating behavior and is influenced by the frontal cortex including medial prefrontal cortex and nucleus accumbens, that have been implicated in motivational and emotional processing [39]. Hence, anteroposterior involvement of hypothalamic nuclei might affect this circuitry, possibly by eliminating a still unknown brake on the lateral hypothalamic “feeding center”.
The importance of hypothalamic invasion was reported by Bogusz et al., who showed a significant increase in BMI during follow-up in a cohort of patients, independent of different extent of hypothalamic involvement; yet, craniopharyngiomas with anteroposterior hypothalamic lesions presented higher BMI at each time point, when compared to patients with only anterior or without hypothalamic lesions [27]. Accordingly, other authors proved that neither preoperative anterior hypothalamic involvement nor anterior surgical hypothalamic lesions seemed to predispose for significant early weight gain during the first year after craniopharyngioma diagnosis, but specific posterior hypothalamic lesions were observed to have a causative impact [22, 38, 40]. In our series, anteroposterior hypothalamic involvement was significant only in univariate analysis. An imbalance in the hypothalamic involvement between our two surgical groups might have introduced an important bias in our study. However, even when we performed secondary analyses by further splitting the two cohorts according to the type of hypothalamic involvement, we did not find significant changes in BMI variation or obesity (see Supplementary Table 1). In fact, the difference in the percentual variation of BMI were identical in patients with anterior hypothalamic lesions (+ 9.5% in the eTNS group vs. + 10.1% in the TC group; P = 0.892) and was slightly but not significantly different in patients with anteroposterior hypothalamic tumors (+ 12.3% in the eTNS group vs. + 25.2% in the TC group; P = 0.233), even though this could suffer from an insufficient number of patients in these further subgroups.
Peritumoral hypothalamic edema, as observed on T2/FLAIR scan has been associated with preoperative hyperphagia and higher BMI and, most importantly, with postoperative obesity [25]. In the present study, T2 hyperintensity in the diencephalic region did not have an independent association with postoperative BMI variation or obesity.
Visual and pituitary outcome
Our series shows a better visual outcome in the eTNS cohort than in the TC group. This is in accordance with what reported by other authors [5, 41]. Anterior pituitary function worsened after surgery, regardless of the surgical approach. Notably, deficit of thyroid hormones and their suboptimal replacement favors deposition of fat and supraphysiological supplementation of cortisol might further induce deposition of adipose tissue. Therefore, timely screening and correct treatment of endocrinopathies might improve the metabolic state, and possibly diminish the degree of obesity [38]. The only statistically significant deficit in the anterior pituitary hormonal axes between the eTNS and TC groups is the baseline hypogonadism, while no clear differences were recorded in new or worsened hypopituitarism. As for the interplay between hormonal deficits and weight control, our results showed independence of these occurrences, suggesting that anterior pituitary insufficiencies may be another epiphenomenon of a common, underlying dysfunction (i.e., hypothalamic derangement) causing both hormonal impairment and obesity, without a strong, direct contribution of hormone defects on the actual BMI increment and obesity.
Newly postoperative diabetes insipidus was more frequent in the TC cohort than in the eTNS group, in keeping with what reported in the literature [1, 5]. This may be explained by more proximal lesions of axons coming from the supraoptic nucleus in the TC rather than in the eTNS approach
Complications
Overall, we found a low occurrence of surgical complications. In particular, no significant differences in the frequency of complications was evident between the eTNS and TC groups, except for a significantly higher incidence of CSF leaks in the eTNS group, a trend that was described in the eTNS endoscopic cohort by Wannemuehler and collaborators, even if their series was too small to detect a statistical significance [41].
Extent of resection and recurrence
MRI-proven surgical radicality was identical in the two surgical techniques, and recurrence of disease very similar, in line with what reported by other authors [41]. Van Iersel et al. correlated partial resection of the tumor with a higher recurrence rate, especially in patients who did not receive adjuvant radiotherapy after initial surgery. The role and timing of radiotherapy, however, is still controversial, taking into account the option of an initial wait- and-scan policy followed by adjuvant radiotherapy in the case of tumor progression [38].
Study limitations
Several limitations of our study should be noted. First, the availability of preoperative MRI led to select only a subset of the total patient population, which was the main limiting factor, rather than a subsequent elimination due to the exclusion criteria adopted (see Fig. 1). Still, we believe that this subselection has not significantly impaired the results of our study. Additionally, the two groups of patients showed some imbalances in clinical characteristics, such as gender and age. However, we do not expect that such variables might impact on the postoperative functional hypothalamic outcome.
Baseline anteroposterior hypothalamic involvement was more prevalent in the TC cohort than in the eTNS group, thus introducing a possible bias in the postoperative evaluation of the BMI variation. However, in the supplemental material, we performed a separate analysis by splitting the two groups of patients according to the baseline degree of hypothalamic involvement and found no difference as compared with the results of the main analysis.
The longer follow-up in the TC group than in the eTNS patients should not affect our findings, as it is well documented in the literature that the development of hypothalamic obesity usually occurs in the first 6–18 months after surgery [27], a period that is well covered in both the eTNS and TC groups. This disparity is simply explained by the fact that the eTNS technique has been increasingly used instead of the TC approach in the last few years and consequently the follow up is accordingly shorter.
Finally, we only used two simple neuroradiological indicators for grading hypothalamic involvement (T2 hyperintense modifications and distortion of hypothalamic structures based on mammillary bodies demarcation), as evaluated by a single author. Estimates of tumor volumes were carried out by means of software calculations, whenever possible.
Conclusions
We found that the eTNS approach was associated with a better outcome of visual function and a lower incidence of postoperative diabetes insipidus. However, the eTNS group had an increased risk of postoperative CSF leakage. Multivariate analysis showed that only basal BMI and tumor volume were significantly associated with the risk of postoperative obesity and percentual BMI variation, suggesting that a larger lesion portends a higher risk of developing postoperative obesity, independent of the surgical approach and of the type of hypothalamic involvement (at least according to gross criteria, such as T2 signal and mammillary bodies demarcation). Interestingly, basal BMI is independent of lesional volume and independently concurs in defining the risk of postoperative obesity. The choice of the approach apparently does not influence the obesity risk, notwithstanding the abovementioned limitations. However, our study points out how eTNS is a valid approach in managing also larger tumors with important hypothalamic invasion.
Data availability
The datasets generated and analyzed during the current study are secured in the institution archive and are available from the corresponding author on reasonable request.
Code availability
The R code scripts used for the current study analyses are available from the corresponding author on reasonable request.
References
Honegger J, Tatagiba M (2008) Craniopharyngioma surgery. Pituitary 11:361–373. https://doi.org/10.1007/s11102-008-0137-z
Rickert CH, Paulus W (2001) Epidemiology of central nervous system tumors in childhood and adolescence based on the new WHO classification. Child’s Nerv Syst 17:503–511. https://doi.org/10.1007/s003810100496
Mortini P (2017) Craniopharyngiomas: a life-changing tumor. Endocrine 57:191–192. https://doi.org/10.1007/s12020-016-1192-2
Mortini P, Losa M, Pozzobon G et al (2011) Neurosurgical treatment of craniopharyngioma in adults and children: early and long-term results in a large case series—clinical article. J Neurosurg 114:1350–1359. https://doi.org/10.3171/2010.11.JNS10670
Buchfelder M, Schlaffer SM, Lin F, Kleindienst A (2013) Surgery for craniopharyngioma. Pituitary 16:18–25. https://doi.org/10.1007/s11102-012-0414-8
Mortini P, Gagliardi F, Boari N, Losa M (2013) Surgical strategies and modern therapeutic options in the treatment of craniopharyngiomas. Crit Rev Oncol Hematol 88:514–529. https://doi.org/10.1016/j.critrevonc.2013.07.013
Alli S, Isik S, Rutka JT (2016) Microsurgical removal of craniopharyngioma: endoscopic and transcranial techniques for complication avoidance. J Neurooncol 130:299–307. https://doi.org/10.1007/s11060-016-2147-4
Chakrabarti I, Amar AP, Couldwell W, Weiss MH (2005) Long-term neurological, visual, and endocrine outcomes following transnasal resection of craniopharyngioma. J Neurosurg 102:650–657. https://doi.org/10.3171/jns.2005.102.4.0650
Zacharia BE, Amine M, Anand V, Schwartz TH (2016) Endoscopic endonasal management of craniopharyngioma. Otolaryngol Clin North Am 49:201–212. https://doi.org/10.1016/j.otc.2015.09.013
Jeon C, Kim S, Shin HJ et al (2011) The therapeutic efficacy of fractionated radiotherapy and gamma-knife radiosurgery for craniopharyngiomas. J Clin Neurosci 18:1621–1625. https://doi.org/10.1016/j.jocn.2011.03.028
Yang I, Sughrue ME, Rutkowski MJ et al (2010) Craniopharyngioma: a comparison of tumor control with various treatment strategies. Neurosurg Focus 28:1–11. https://doi.org/10.3171/2010.1.FOCUS09307
Maira G, Anile C, Rossi GF, Colosimo C (1995) Surgical treatment of craniopharyngiomas: an evaluation of the transsphenoidal and pterional approaches. Neurosurgery 36:715–724
Evans J, Kenning T (2014) Craniopharyngiomas: comprehensive diagnosis, treatment and outcome, 6th edn. Elsevier, Oxford
Flitsch J, Aberle J, Burkhardt T (2015) Surgery for pediatric craniopharyngiomas: is less more? J Pediatr Endocrinol Metab 28:27–33. https://doi.org/10.1515/jpem-2014-0417
Abe T, Lüdecke DK (1999) Transnasal surgery for infradiaphragmatic craniopharyngiomas in pediatric patients. Neurosurgery 44:957–964
Weiss MH, Apuzzo MJL (1987) The transnasal transsphenoidal approach. Williams & Wilkins, Baltimore
Kato T, Sawamura Y, Abe H, Nagashima M (1998) Transsphenoidal-transtuberculum sellae approach for supradiaphragmatic tumours: technical note. Acta Neurochir (Wien) 140:715–719. https://doi.org/10.1007/s007010050167
Laufer I, Anand VK, Schwartz TH (2007) Endoscopic, endonasal extended transsphenoidal, transplanum transtuberculum approach for resection of suprasellar lesions. J Neurosurg 106:400–406. https://doi.org/10.3171/jns.2007.106.3.400
Gardner PA, Kassam AB, Snyderman CH et al (2008) Outcomes following endoscopic, expanded endonasal resection of suprasellar craniopharyngiomas: a case series. J Neurosurg 109:6–16. https://doi.org/10.3171/JNS/2008/109/7/0006
Couldwell WT, Weiss MH, Rabb C et al (2004) Variations on the standard transsphenoidal approach to the sellar region, with emphasis on the extended approaches and parasellar approaches: surgical experience in 105 cases. Neurosurgery 55:539–547. https://doi.org/10.1227/01.NEU.0000134287.19377.A2
Maira G, Anile C, Albanese A et al (2004) The role of transsphenoidal surgery in the treatment of craniopharyngiomas. J Neurosurg 100:445–451. https://doi.org/10.3171/jns.2004.100.3.0445
Müller HL, Reichel J, Boekhoff S et al (2018) Low concordance between surgical and radiological assessment of degree of resection and treatment-related hypothalamic damage: results of KRANIOPHARYNGEOM 2007. Pituitary 21:371–378. https://doi.org/10.1007/s11102-018-0883-5
Müller HL, Gebhardt U, Teske C et al (2011) Post-operative hypothalamic lesions and obesity in childhood craniopharyngioma: results of the multinational prospective trial KRANIOPHARYNGEOM 2000 after 3-year follow-up. Eur J Endocrinol 165:17–24. https://doi.org/10.1530/EJE-11-0158
Van Gompel JJ, Nippoldt TB, Higgins DM, Meyer FB (2010) Magnetic resonance imaging-graded hypothalamic compression in surgically treated adult craniopharyngiomas determining postoperative obesity. Neurosurg Focus 28:1–8. https://doi.org/10.3171/2010.1.FOCUS09303
Cohen LE (2016) Update on childhood craniopharyngiomas. Curr Opin Endocrinol Diabetes Obes 23:339–344. https://doi.org/10.1097/MED.0000000000000264
Müller HL (2015) Craniopharyngioma: long-term consequences of a chronic disease. Expert Rev Neurother 15:1241–1244. https://doi.org/10.1586/14737175.2015.1100078
Bogusz A, Boekhoff S, Warmuth-Metz M et al (2019) Posterior hypothalamus-sparing surgery improves outcome after childhood craniopharyngioma. Endocr Connect 8:481–492. https://doi.org/10.1530/ec-19-0074
Müller HL (2011) Consequences of craniopharyngioma surgery in children. J Clin Endocrinol Metab 96:1981–1991. https://doi.org/10.1210/jc.2011-0174
Fahlbusch R, Honegger J, Paulus W et al (1999) Surgical treatment of craniopharyngiomas: experience with 168 patients. J Neurosurg 90:237–250. https://doi.org/10.3171/jns.1999.90.2.0237
Di Rocco C, Caldarelli M, Tamburrini G, Massimi L (2006) Surgical management of craniopharyngiomas–experience with a pediatric series. J Pediatr Endocrinol Metab 19(Suppl):1
Van Effenterre R, Boch AL (2002) Craniopharyngioma in adults and children: a study of 122 surgical cases. J Neurosurg 97:3–11. https://doi.org/10.3171/jns.2002.97.1.0003
Pierre-Kahn A, Recassens C, Pinto G et al (2005) Social and psycho-intellectual outcome following radical removal of craniopharyngiomas in childhood. A prospective series. Child’s Nerv Syst 21:817–824. https://doi.org/10.1007/s00381-005-1205-6
Morisako H, Goto T, Goto H et al (2016) Aggressive surgery based on an anatomical subclassification of craniopharyngiomas. Neurosurg Focus 41:1–15. https://doi.org/10.3171/2016.9.FOCUS16211
Müller HL, Faldum A, Etavard-Gorris N et al (2003) Functional capacity, obesity and hypothalamic involvement: cross-sectional study on 212 patients with childhood craniopharyngioma. Klin Padiatr 215:310–314
Hofmann BM, Höllig A, Strauss C et al (2012) Results after treatment of craniopharyngiomas: further experiences with 73 patients since 1997. Clinical article. J Neurosurg 116:373–384. https://doi.org/10.3171/2011.6.JNS081451
Hoffmann A, Boekhoff S, Gebhardt U et al (2015) History before diagnosis in childhood craniopharyngioma: associations with initial presentation and long-term prognosis. Eur J Endocrinol 173:853–862. https://doi.org/10.1530/EJE-15-0709
Vinchon M, Weill J, Delestret I, Dhellemmes P (2009) Craniopharyngioma and hypothalamic obesity in children. Child’s Nerv Syst 25:347–352. https://doi.org/10.1007/s00381-008-0754-x
van Iersel L, Meijneke RWH, Schouten-van Meeteren AYN et al (2018) The development of hypothalamic obesity in craniopharyngioma patients: a risk factor analysis in a well-defined cohort. Pediatr Blood Cancer 65:1–9. https://doi.org/10.1002/pbc.26911
Zhang Y, Wang J, Zhang G et al (2015) The neurobiological drive for overeating implicated in Prader-Willi syndrome. Brain Res 1620:72–80. https://doi.org/10.1016/j.brainres.2015.05.008
Müller HL, Gebhardt U, Etavard-Gorris N et al (2004) Prognosis and sequela in patients with childhood craniopharyngioma—results of HIT-ENDO and update on KRANIOPHARYNGEOM 2000. Klin Padiatr 216:343–348
Wannemuehler TJ, Rubel KE, Hendricks BK et al (2016) Outcomes in transcranial microsurgery versus extended endoscopic endonasal approach for primary resection of adult craniopharyngiomas. Neurosurg Focus. https://doi.org/10.3171/2016.9.FOCUS16314
Funding
No funds, grants, or other support was received.
Author information
Authors and Affiliations
Contributions
Conceptualization: ALG, LRB, ML; Methodology: ALG, LRB, ML; Formal analysis and investigation: ALG; Writing—original draft preparation: ALG; Writing—review and editing: ALG, LRB, ML, MM, LA, FG, PM; Supervision: PM.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
This type of study does not require specific consent for data publication; however all participants signed a general consent to publish results obtained from studies involving their data.
Ethical approval
This is an observational study. The Vita-Salute University, I.R.C.C.S. San Raffaele Scientific Institute Research Ethics Committee has confirmed that no ethical approval is required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gallotti, A.L., Barzaghi, L.R., Albano, L. et al. Comparison between extended transsphenoidal and transcranial surgery for craniopharyngioma: focus on hypothalamic function and obesity. Pituitary 25, 74–84 (2022). https://doi.org/10.1007/s11102-021-01171-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11102-021-01171-2