Abstract
Andrographis paniculata (Burm.f) Nees is one of the most popular and important medicinal plant of the Orient, and South East Asia. It finds mention in various forms in Indian, Chinese, Malay, Thai, Unani, and Japanese systems of medicine. The plant exhibits anti-cancer, anti-inflammatory, anti-diabetic, anti-hypertensive, anti-venom, cholestatic, hepatoprotective, anti-thrombotic, anti-retroviral, anti-microbial, anti-pyretic, anti-malarial, anti-oxidant, immunomodulatory, and cardioprotective effects. The major active principles contributing to biological activity are diterpene lactones, but flavonoids, xanthones and caffeic acid derivatives also contribute to anti-oxidant, anti-proliferative, anti-atherosclerotic, and anti-malarial effects. As a result of its wide spectrum of pharmacological activity, almost impeccable safety profile, being a widely cultivated medicinal plant, we have collected and compiled various facets of this plant. Extensive datamining of the phytochemistry and pharmacology of Andrographis paniculata revealed more than 50 diterpene lactones, 30 flavonoids, 8 quinic acid derivatives, and 4 xanthones. This review contains information on around 80 isolated compounds, out of which more than half of the compounds have no reported pharmacological activity. Though there are some good reviews available on Andrographis paniculata, the authors of the earlier reviews focused on one or two aspects of the plant and none have attempted to integrate the available information on this plant. This provided us the much needed impetus, warranting a full-fledged and complete review on Andrographis paniculata, one of the most popular and important Oriental medicinal plant.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Approximately 90% people in developing countries rely on traditional or alternative and complementary medicines, based largely on different species of plants, and plant products which is their basic medical aid (Fabricant and Farnsworth 2001). This is especially true for Asian, African or South American countries where the majority of the population still depends on plant as source of medicines. Of the more than 120 pure pharmaceutical chemicals isolated from about 100 plant species, currently in use as drugs, 40 are obtained from tropical species (Farnsworth and Soejarto 1991). However, fewer than 5% of tropical forest plant species have been examined for chemical compounds and medicinal values (Zakrzewski 2002).
From the last decade or so, rapid strides in high throughput screening, laboratory techniques, molecular, pharmacological, tissue culture techniques, and germplasm conservation initiatives etc. have resulted in a huge number of publications of Andrographis paniculata. A recent review of literature with the keywords Andrographis paniculata on “PubMed”, returned around 250 results. Of research describing pharmacological aspects in laboratory animals the majority of citations are from the year 2000 onwards. It was observed that research articles published from years 1951 to 1989 on Andrographis paniculata and andrographolides numbered around 13, whereas from the years 1990 to 1999, research publications on this plant was around 36. In the next decade (2000–2010) the number of publications jumped to 169 from 36. The objective of this review is to generate a strong opinion on Andrographis paniculata encouraging the scientific community to unravel further potential uses of this medicinal plant for various diseases. It is expected that with a gradual shift to sophisticated techniques of extraction, the discovery of hitherto unknown phytochemicals and extension of pharmacological spectrum of activity of this plant is a distinct possibility, throwing up exciting challenges to researchers.
Mishra et al. (2007) has written a commentary with their focus on the phytochemistry and pharmacological aspects of Andrographis paniculata. They have also thrown light on the safety issues of the plant extract with mention of some studies. The authors have not given information on the agricultural advancements, neither have they listed out the clinical studies. So the end result is whether the said review will serve its purpose to researchers, agriculturists, small scale herbal industries and it is hard to justify it. Chao and Lin (2010) has also performed an excellent review on the isolation and identification of Andrographis paniculata. The review article’s thrust is on phytochemistry and occasionally has explained the pharmacological part also. A brief information is also available on the preclinical studies of various extracts of the plant. Fractionation of some bioactive extracts especially the ethyl acetate has been dealt with elaborately. It is worthwhile to mention here that the review did not mention the safety issues, the agronomical practices and clinical trials of the plant and hence may serve only academic purpose interests and may not generate an interest to diverse set of readers. Hence it is hoped that this review would serve as a cornerstone to the researchers, agronomists, and even small scale herbal industries and integrate the available information on Andrographis paniculata in order to realize the full pharmacological, agricultural and industrial potentials of this exciting medicinal plant.
Botanical description, habitat and parts used
Andrographis paniculata (Burm.f.) Nees is commonly known as “Hempedu Bumi”, which literally means “bile of the earth”, and “pokok cerita” in Malaysia, “Sambiloto” in Indonesia belonging to Family Acanthaceae. It is also known as “The Creat” in English and “King of bitters”. It is an annual, branched, herbaceous plant with a tap root, and grows to a height of around 90–110 cm. The leaves are 3–7 × 1–2.5 cm in size, green, simple, opposite, lanceolate and glabrous with a short petiole. Flowers are white in colour with reddish-purple spots on the petals. The stems are dark green in colour 0.5–1 m height and 2–6 mm in diameter with presence of longitudinal furrows and wings on the angles of young plants. Seeds are small, numerous, abundant and yellowish brown in color. Generally the aerial parts of the plant [including leaves and stems] (WHO, Geneva 2002) are used as traditional medicine since the major active principle andrographolide is present in higher quantities in the aerial parts, but roots (Dua et al. 2004) have also been used but with limited success.
The plant can be abundantly found in South India, North India, Sri Lanka, Islands of Java and Borneo, Malaysia, Brunei, Thailand. The popularity, medicinal use and economic feasibility prompted farmers to cultivate extensively in China, Thailand, and Malaysia of late. In Malaysia, Forest Research Institute Malaysia (FRIM) and Malaysian Agriculture Development Institute (MARDI) in a concerted approach have made cultivation of dedicated nurseries a great success by maintaining different cultivars.
Cultivation and harvesting
Andrographis paniculata is mainly cultivated as rainy season crop. With the onset of monsoon, plant grows luxuriantly and starts flowering with the moderation in temperature. Flowering and fruiting starts from around 2–3 months from planting. Maximum herb biomass can be obtained in 90–100 days beyond which leaves start shedding. At the time of flowering, the active principle andrographolide is high in leaves. Since the whole plant contains active principles, entire harvested material is dried in shade and powdered.
About 400 gm. seed are sufficient for one hectare. Application of sufficient quantities of nitrogen, and phosphorus (Chauhan and Gyanendra 2003) per hectare will increase the herb yield. Addition of 5–7 tons of vermicompost and manure are necessary for raising nursery. The plants at flowering stage (around 90–120 days after sowing) are cut at the base leaving 10–15 cm stem for plant regeneration. About 50–60 days after first harvest, final harvest (Maheshwari et al. 2002) is performed. The yield varies between 3 and 4 tons dry herb/hectare (Seema et al. 2003).
Recent developments in agronomic techniques for mass production, maximising yield of active principles, conservation and management
Tissue culture techniques
The increase in demand for andrographolide by the pharmaceutical industries is putting undue pressure on the existing wild populations. However, the commercial exploitation of this plant is hampered due to its limited availability (Kanjilal et al. 2002). The increasing demand of andrographolide in Indian as well as international markets has motivated Indian farmers to start commercial cultivation of this medicinal plant (Kanjilal et al. 2002), but still there is a gap between supply and demand which can only be bridged by using non conventional methods or tissue culture related techniques. Micropropagation is the proven method for efficient in vitro propagation of medicinal and aromatic plants and for commercial exploitation of valuable plant-derived pharmaceuticals (Bajaj et al. 1988; Purohit et al. 1994; Pattnaik and Chand 1996; Rout 2002; Faisal et al. 2005). Purkayastha et al. 2008, reported that in about 90 days, 60–70 shoots were obtained from a single nodal explant and the nodal explants from primary shoots further regenerated equivalent number of shoots, demonstrating high frequency regeneration potential. The plantlets were successfully transferred to soil after hardening with a 92% survival rate. The process from the initiation of shoot buds to the transplantation of regenerants to soil is complete within 8–9 weeks.
Newer techniques of sustainable agriculture is being advocated strongly these days, using reduced fertilizers and pesticides which in turn help preserve and conserve soil health, fertility, a pollution free environment have to be given maximum importance in developing countries. One of the methods of maximizing yield under sustainable modes of agriculture is through the use of microbial inoculants. It was shown that the seeds of Andrographis paniculata when raised in polythene bags containing soil inoculated with isolates of Glomus leptotichum and Glomus intraradices species of arbuscular mycorrhizal fungi, showed an increase in plant growth and andrographolide concentration (Chiramel et al. 2006) over those grown in the absence of the inoculation of soil with the fungi. The main effect of mycorrhizal fungi in improving plant growth (Marulanda et al. 2003; Nowak 2004; Chen et al. 2005; Khalafallah and Abo-Ghalia 2008) is thought to be due to improved uptake of nutrients. This is especially true for phosphorus (Smith et al. 2003) due to the exploration by the external hyphae of the soil beyond root hair zone when phosphorus is depleted. Increased phosphorus uptake has been attributed not only to increased surface area of absorption but also to enhanced hyphal translocation (Turk et al. 2006). Enhanced plant biomass and phosphorus uptake (Chen et al. 2005) due to arbuscular mycorrhizal fungal inoculation has been reported by earlier workers in forest tree species and a few medicinal plants (Vasanthakrishna et al. 1995; Sailo and Bagyaraj 2005).
The conventional vegetative propagation of Andrographis paniculata is a tedious process and too slow to meet the commercial quantities required. Moreover, a high degree of variability among the seed-derived progenies and a scanty and delayed rooting of seedlings curb its propagation via seeds. Hence a study on the in vitro propagation of Andrographis paniculata (Burm. f.) Wallichi ex Nees through somatic embryogenesis, and influence of 2,4-dichlorophenoxyacetic acid (2,4-D) on induction, maturation, and conversion of somatic embryos was investigated by Martin (2004). This study reported an̄ efficient protocol for plant regeneration from callus of Andrographis paniculata via somatic embryogenesis. Using this protocol, more than 1,800 plantlets can be produced from 1 g of callus within 200 days, facilitating rapid clonal propagation of this important medicinal plant.
Praveen et al. (2009) demonstrated the use of a cell culture technique to increase the yield of andrographolide from young leaves of Andrographis paniculata. Adventitious roots were induced directly from leaf explants using Murashige Skoog liquid medium with napththalene acetic acid and sucrose. The root cultures showed higher accumulation of biomass (fresh and dry weight) and andrographolide within 4 weeks. Seven-fold increment of fresh biomass was evident in suspension cultures along with 3.5-fold higher andrographolide compared to natural plants. These results demonstrated a great potentiality of adventitious root cultures for the production of andrographolides for commercial purposes on a large scale using bioreactor cultures.
The plant hormones or growth regulators influence their growth, differentiation and development (Aguiló et al. 2005). These growth regulatory compounds play a crucial role in the regulation and coordination of plant growth, morphogenesis, metabolism and role in biosynthesis of secondary metabolites (Jaleel et al. 2006). In a study performed by Anuradha et al. (2010), growth regulators like Abscisic acid and Gibberellic acid were used to study the effect on the andrographolide content and antioxidant potentials of Andrographis paniculata. These were applied by means of foliar spray during morning hours. A significant enhancement in nonenzymatic antioxidant contents was observed in all the plants under growth regulator treatments. Ascorbic acid and α-tocopherol content was increased significantly under the growth regulator treatments in leaves, stem and roots of Andrographis paniculata. HPLC analysis used to quantify the andrographolide content showed an increase in andrographolide content of growth regulator treated plants compared to control.
A few studies have focused on the agronomic traits of this plant. A study undertaken by Bhan et al. (2006), with the objective of evaluating Andrographis paniculata for growth behaviour, yield and andrographolide content for optimization of yield among different collections from ten locations in India. The parameters of interest that were recorded were total andrographolide concentration in leaves, dry herbage yield per plant, and leaf/stem ratio. The results demonstrate that the plant achieved highest growth at Jammu adapting to the subtropical conditions and was significantly superior with respect to the production of total andrographolides and dry herbage yield. Harvesting at 100 days after transplantation (DAT) was recommended.
Prathanturarug et al. (2007) has reported the variation in growth and diterpene lactone content among field-cultivated Andrographis paniculata. They isolated the elite individual plants containing high amounts of andrographolide and 14-deoxy-11,12-didehydroandrographolide which would serve as a favorable measure in selection of appropriate plant material for the phytomedicine production, standardized compounds and phytomass. They also identified and selected superior plants for further breeding experiments. Micropropagation or Clonal propagation techniques using shoot tip and nodal segments (Sharma et al. 2010) are beneficial for mass-scale multiplication and conservation of an endangered or threatened and medicinally important species within short period and limited space.
For the regeneration of a whole plant from a cell or from a callus mass cytodifferentiation is not enough and there should be differentiation leading to organogenesis. This may occur through shoot bud differentiation (organogenesis) or through somatic embryogenesis. In the former, shoot buds (monopolar structures) were formed while in the latter, somatic embryos (bipolar structures) were formed both leading to regeneration of whole plant (Sharma et al. 2010).
It is useful to preserve the natural germplasm and also apply modern tissue culture techniques in mass cultivation and commercial feasibility. It would also be beneficial to develop suitable genetic markers for propagation and breeding programs to support natural conservation of various species of this plant. Propagation through tissue culture techniques provides sustainable development solutions for mass propagation of plants in general and endangered plants in particular. The powerful techniques of plant cell and tissue culture, recombinant DNA and bioprocessing technologies have offered mankind a great opportunity to exploit the medicinal plants under in vitro conditions.
Genetic markers and gene based techniques
Genetic markers are useful for identifying plant at genomic level (Srivastava and Mishra 2009) and establish new standards in standardization and quality control of botanicals. These markers are highly polymorphic in nature, show codominant inheritance, and occur frequently in genome. They are unbiased to environmental conditions or management practices and at the same time easily available, highly reproducible and allows easy exchange of information between laboratories. Gene-based initiatives for authentic identification of this plant and identify it from the other 28 species of Andrographis paniculata (Lattoo et al. 2008) will go a long way in differentiating the medically useful from the other less medically useful ones.
DNA-based molecular markers (Srivastava and Mishra 2009) have been used extensively for a wide range of applications. These applications include study of genetic variation, cultivar identification, genotyping, cross-breeding studies, identification of disease-resistant genes, identification of quantitative-trait loci, diversity analysis of exotic germplasms, sex identification of dioeceous plants, phylogenetic analysis, etc. This is especially useful in case of those that are frequently substituted or adulterated with other species or varieties that are morphologically and/or phytochemically indistinguishable.
DNA sequencing (Pereira et al. 2008) can also be used as a definitive means for identifying species of medicinal importance and can used for correct botanical identification and authentification of crude plant material as part of standardization and quality control procedures (WHO 2002). Variations due to transversion, insertion or deletion can be assessed directly and information on a defined locus can be obtained.
DNA microarray was developed in response to the need for a high-throughput, efficient and comprehensive strategy that can simultaneously measure all the genes or a large defined subset, encoded by a genome. DNA microarray can also be used for studying herb-drug interactions (Berman 2000), for discovery of new diagnostic indicators (Izuka et al. 2003), investigating the mechanism of action (DeFeudis et al. 2003) and the mechanisms underlying these interactions in terms of molecular pharmacology (Coldren et al. 2003), identification of genes involved in drug sensitivity or resistance, correct botanical identification and authentification of crude plant drugs etc.
Marker assisted breeding involves the use of DNA markers linked with DNA sequences of interest. Inheritance pattern of sequences/traits can be confirmed even prior to expression using Restriction Fragment Length Polymorphism (RFLP), RAPD, Amplified Fragment Length Polymorphism (AFLP) and minisatellite markers (Yu et al. 1991; Ma et al. 1994). Thus marker assisted breeding could be of use in producing a higher content of the principle marker andrographolide, or an increase in herb biomass.
Random Amplification of Polymorphic DNA (RAPD) analysis is a sophisticated and effective technique to measure the magnitude of diversity and discriminate between genotypes. A dominant marker, Random Amplification of Polymorphic DNA-based molecular markers have been found to be useful in differentiating different accessions of Andrographis paniculata (Lattoo et al. 2008) collected from different geographical regions. Germplasm analysis to study genetic diversity and effective conservation and management of germplasm is another important area in which a lot of efforts have been put in (Lattoo et al. 2008). The congruence of RAPD markers with the morphological descriptors provides a viable alternative to characterize the germplasm of Andrographis paniculata for optimum genetic improvement and effective conservation of its genetic resources. A better understanding of distribution of genetic variation at the intraspecific level would help identify superior genotypes for cultivar upgradation as well as to evolve strategies for the establishment of effective in situ and ex situ conservation programmes (Padmesh et al. 1999).
Fingerprinting of medicinal plants is also being carried out extensively. This information has potential in strategic planning of future breeding towards crop sustainability. DNA-based techniques have been widely used for authentication of plant species of medicinal importance.
Extraction techniques
Extraction procedure plays an important role in preserving the active constituents of plant extract and hence the pharmacological activity. The selection of extraction procedure ultimately depends on the nature of the active constituents of interest to be extracted. So it is very important to choose a proper extraction technique such that the active constituents of interest are maintained in the natural state, but especially, in herbal industries timing, extraction efficiency, automation etc. are valid points for consideration. Conventional extraction of active constituents from Andrographis paniculata has been performed by maceration, Soxhlet extraction and ultrasonic extraction (Mukherjee 2002) and their large scale application has been severely limited by several disadvantages.
A dynamic microwave-assisted extraction with on line coupling with HPLC using flow injection interface to analyse andrographolide and dehydroandrographolide has been reported by Chen et al. (2007a). This approach offers several advantages and bypasses many of the problems associated with more traditional approaches. By tweaking several experimental parameters, the optimized extraction conditions of extraction were found to yield mean recoveries of 97.8 and 98.7% for andrographolide and dehydroandrographolide respectively. This technique was demonstrated to generate a higher extraction yield in a shorter time when compared with ultrasonic extraction official in the Chinese Pharmacopoeia. In addition, only small quantities of solvent (5 ml) and sample (10 mg) were required.The authors reported rapid analysis time, economy in solvent use, and reduced cost of analysis. The reliability and repeatability of the analysis were improved, with analysis and sample extraction taking place in a closed automated system, the associated risks of sample loss and contamination were reported to be minimal as well.
Presently supercritical fluid extraction (Lang and Wai 2001) is fast gaining momentum and has the most important application in the extraction of natural products. With increasing trends of herbal product use (Tonthubthimthong et al. 2001; Song et al. 1992; Ambrosino et al. 1999), this technique may well become the gold standard for extraction in the future.
A few studies have been carried out to determine the extraction efficiency of supercritical fluid extraction technique on the yield of andrographolides. Ge et al. (2002) reported the use of supercritical carbondioxide, and carbondioxide-ethanol mixture to extract andrographolide in a custom made extractor unit. He used orthogonal experiments to determine optimum conditions however the study had several short comings. The study failed to explain the effect of particle size and the supercritical solvent flow rate. The study also could not explain the effect of each operating parameter. Moreover, the geometry of the extractor, the supercritical solvent flow pattern, and the ethanol concentration in the supercritical solvent were not reported. The experiments conducted proved that at high temperature and pressure, the extraction rate and yield were higher, but the andrographolide content in the extracts was very low. But an enhanced extract yield and the andrographolide content with addition of ethanol as co-solvent was reported.
Kumoro and Hasan (2007), examined the effect of the solvent flow rate, pressure, and temperature on the supercritical carbondioxide extraction of andrographolide. During the course of experiments, the operating pressures were varied from 7.5 to 20 MPa, temperatures varied from 30 to 60°C, and the flow rates were varied from 0.5 to 4 ml/min. The highest yield within a set of experimental conditions occurred at 10 MPa, 40°C, and a flow rate of 2 ml/min for a 3 g sample of Andrographis paniculata ground-dried leaves. The measured extraction rate was found to be about 0.0174 g of andrographolide per gram of andrographolide present in the leaves per hour of operation.
Cui et al. (2009) compared extracts of different sample preparation procedures like microwave-assisted, marinated extraction, reflux, ultrasonic extraction of Andrographis paniculata using andrographolide and dehydroandrographolide as reference compounds. The authors reported a simple and timesaving extraction method i.e microwave-assisted extraction with a more effective fingerprint containing additional spectral information namely an enhanced fingerprint. According to the author enhanced fingerprint in comparison with conventional fingerprint with additional spectral information provides more advantages. Enhanced fingerprint displays more peaks which cannot be detected at a fixed wavelength but have UV responses at other wavelengths; Secondly, in quality assessment, conventional fingerprint might give different chromatographic patterns at different wavelengths, whereas enhanced fingerprint technique is more objective and credible.
Analytical techniques
Several methods have been reported for the quantitative determination of lactones in Andrographis paniculata, including TLC, HPTLC, Liquid chromatography, micellar electrokinetic capillary chromatography (MECC), high speed counter current chromatography (Du et al. 2003), Ultra Performance Liquid Chromatography (UPLC).
A High-performance liquid chromatography coupled with on-line solid phase extraction and ultraviolet detection was developed by Chen et al. (2007b) for determining andrographolide and dehydroandrographolide in rabbit plasma. Plasma samples (100 μl) were injected directly into a C18 column and the mobile phase consisted of methanol:acetonitrile:water (50:10:40; v/v). The UV detection was performed at 225 nm. The calibration curves showed excellent linear relationship (R ≥ 0.9993). The inter- and intra-day precisions of two analytes were in the range of 1.2–6.5% and the accuracies were between 92.0 and 102.1%. Their recoveries were all greater than 94%. The limits of detection were 0.019 μg/ml for andrographolide and 0.022 μg/ml for dehydroandrographolide.
Cui et al. (2009) reported on the comparison of conventional and enhanced fingerpoint profile of andrographolide and dehydroandrographolide by HPLC DAD. The precision, stability and repeatability of method were evaluated by the analysis of five successive injections of the same sample solution and were found to be good. It was seen that compared with conventional fingerprints, enhanced fingerprint contains more peaks, especially in the retention time of 15–25. Moreover, the signals in enhanced fingerprint are much stronger than those in conventional fingerprints. Hence compared with enhanced fingerprint, the classification ability and discrimination power of conventional chromatographic fingerprint is limited to perform further quality control. This study underlines the fact that the most relevant factor on the quality of Andrographis paniculata was the collecting location and the harvesting time of the plant. In order to get consistent raw materials of Andrographis paniculata, the collecting location and harvest time should be fixed.
Kavuri et al. (2010) reported a simple, rapid, precise, accurate, robust and stability indicative Ultra Performance Liquid Chromatography method with UV detection for the quantitative estimation of andrographolide and 14-deoxy 11, 12-didehydroandrographolide from Andographis paniculata. The method for quantification of andrographolide and 14-deoxy, 11-12 didehydoandrographolide was validated with regard to its selectivity, linearity, precision, accuracy, stability, robustness, limit of detection and quantification. The ultra fast LC method showed good linearity, accuracy and satisfactory precision with run time of less than 3 min. The developed and validated method was found to be suitable to analyze lower as well as higher amounts of andrographolide and 14-deoxy 11, 12-didehydroandrographolide in plant raw material, crude plant extracts and various dosage forms.
A summary of reported methods is presented in Table 1. Simple HPLC and HPTLC methods are also used for estimating amount of andrographolide, 14-deoxy-11,12 didehydroandrographolide, and neoandrographolide using reverse phase technique with UV detection at 230 nm have been used.
Ethnopharmacology
Andrographis paniculata has been used for centuries together in Asia to treat fevers, sore throat, gastric infections, upper respiratory tract infections, and many other chronic and infectious disorders. The plant which is known as “Kalmegh” in Ayurvedic texts is an important constituent in majority of Ayurvedic preparations and is official in the Ayurvedic Pharmacopoeia. More importantly, Andrographis paniculata has also been included in “WHO monographs on selected medicinal plants” (WHO 2002), which serves as an authority and ready reckoner for health officials, scientists, doctors and pharmacists and will also be appreciated by the lay man. The monographs play an essential role in promoting the safe and proper use of plants as medicines globally.
In Southern India, the whole plant of Andrographis paniculata is widely employed either orally or applied locally, or both as an antidote against snake bites (Russell 1980) of cobra, Russell’s viper, Banded Krait etc. and this has verified experimentally by Samy et al. (2008). The expressed juice of the leaves of Andrographis paniculata alone or together with cardamom, cloves and cinnamon made into small globules and prescribed as a common household remedy for griping, irregular stools, loss of appetite, flatulence, and diarrhoea of children. In Eastern India, the tribals of Santal, Kheria and Moora, Khatra region of Bankura district, West Bengal, India, use the infusion of the whole plant to recover from fever (Mandal et al. 2001). Decoction or infusion of the leaves has been used with satisfactory results in sluggish liver, neuralgia, certain forms of dyspepsia associated with gaseous distension of the bowels, in general debility, as a blood purifier (Girach et al. 1994), in convalescence after fevers and in advanced stages of dysentery. During the influenza epidemic in India around 1919, the plant was claimed to be highly efficacious in arresting the progress of the disease. The leaves and roots are also used as febrifuge, tonic, stomachic, cholagogue and anthelminthic. It is used indigenously in medicine particularly as bitter tonic, curing fevers, dysentery and eliminating intestinal worms (Singh et al. 2010). The plant is used to relieve griping, irregular stools and loss of appetite in case of infants (Kirtikar and Basu 1935). It is also reported to heal peptic ulcer (Kirtikar and Basu 1935).
Ancient Chinese physicians used it to treat inflammatory conditions, fever, cold, laryngitis and have been described as a cold property herb (Huang and Wu 2002) to get rid of body heat and dispose toxins from body. In Malaysian folk medicine the decoction of fresh leaves has been used for centuries as an antidiabetic and antihypertensive (Burkill 1966). In Scandinavian countries, it is commonly used for the prevention and relief of cold (Cáceres et al. 1999) (Spasov et al. 2004) and sore throat. In Thailand, it is included as one of the important medicinal plant used in the primary healthcare system.
The most surprising and useful feature of this plant is a broad pharmacological activity ranging from antidiabetic (Rammohan et al. 2006, 2008a, b, c) anticancer (Kumar 2003), immunostimulatory (Xu et al. 2007), antihypertensive (Zhang et al. 2006), anti-inflammatory (Shen et al. 2002), antiviral including anti HIV (Reddy et al. 2005; Wiart et al. 2005), anti malarial (Dua et al. 2004), cardioprotective (Ojha et al. 2009), renoprotective (Singha et al. 2007), hepatoprotective (Singha et al. 2007) and anti thrombotic effects (Thisoda et al. 2006).
Phytochemistry
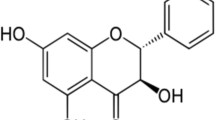
The use of aerial parts of Andrographis paniculata has been described for its myriad uses. There are wide differences in the composition of phytochemicals with respect to part used, geography, season and time of harvesting etc. Andrographolides are present in high levels just before the flowering season (after 90 days) which gradually falls. The aerial parts (leaves and stems) contain the highest proportion of available diterpene lactones of which the major one contributing to pharmacological activity is andrographolide, and roots the lowest. The other lactones are 14-deoxy-11-andrographolide, 14-deoxyandrographolide, neoandrographolide, 14-deoxy-11,12 didehydroandrographolide, andrographon, andragraphan, deoxyandrographiside, andrographiside, andrographosterol etc. It is worthy to note here that a majority of the active constituents of Andrographis paniculata represent the ent labdane class occurring together with some flavonoids also. Quinic acid derivatives and xanthones (only roots) are also present as minor constituents of Andrographis paniculata. A few of the reported works on phytochemistry are mentioned here.
A study by Matsuda et al. (1994) on the ethyl acetate fraction of methanol extract of Andrographis paniculata was found to contain a new series of six ent-labdane diterpenoids, two diterpene glucosides, and four diterpene dimers along with already known compounds. They were found to be potent cell differentiation inducers (Table 2) and may be used in the treatment of cancer.
Reddy et al. (2003), reported the isolation of a flavone, 5-hydroxy-7′,2′,6′-trimethoxyflavone and a 23-carbon terpenoid, 14-deoxy-15-isopropylidene-11,12-didehydroandrographolide together with five known flavonoids (7-Omethyldihydrowogonin, 7-O-methylwogonin, skullcapflavone I 2′-methyl ether, 7-O-methylwogonin 5-O-glucoside,and skullcapflavone I 2′-O-glucoside) and four known diterpenoids (14-deoxy-11,12-didehydroandrographolide, andrographolide, isoandrographolide and neoandrographolide). Rao et al. (2004), isolated and identified 5,7,20,30-tetramethoxyflavanone and 5-hydroxy-7,20,30-trimethoxyflavone.
Reddy et al. (2005), reported a novel bis-andrographolide ether along with five known known flavonoids (7-Omethyldihydrowogonin, 7-O-methylwogonin, skullcapflavone I 20-methyl ether, 7-O-methylwogonin 5-O-glucoside, skullcapflavone I 20-O-glucoside and four known labdane type diterpenoids (14-deoxy-11,12-didehydroandrographolide, andrographolide, isoandrographolide, and neoandrographolide) isolated from the methanolic extract of aerial parts of Andrographis paniculata and structure was established by spectral data.
Pramanick et al. (2006), reported the isolation of new labdane type diterpenoid, andropanolide, from the methanolic extract of leaves of Andrographis paniculata, along with seven known diterpenoids (andrographolide, andrograpanin,isoandrographolide, 14-deoxy-11,12-didehydroandrographolide,14-deoxyandrographolide, isoandrographolide, neoandrographolide). Another new diterpene, andrographolactone, isolated from the aerial parts of ethanolic extracts was found to have cytotoxic effects on two human cancer cell lines (Wang et al. 2009). Recently Xu et al. (2010) reported the yield of one novel diterpene (13R, 14R) 3, 13, 14, 19-tetrahydroxy-ent-labda-8, 11-dien-16, 15-olide from the ethanol extract of the leaves of Andrographis paniculata, which has a cis-diol group in the lactone moiety, and 3, 19-isopropylidene-14-deoxy-ent-labda-8, 13-dien-16,15-olide, together with eight known diterpenoids (andrographolide, neoandrographolide, andrograpanin, 14-deoxyandrographolide, 14-deoxy-11,12-didehydroandrographolide, 14-deoxyandrographoside, andrographoside and 8-methylandrographolide).
The isolated compounds from Andrographis paniculata and their activity are listed in Table 2.
Pharmacology
Andrographis paniculata is having wide range of pharmacological effects. It is also interesting to note that from 1982 to 1999 the pharmacological effects reported centered on antiretroviral (Basak et al. 1999), antioxidative (Das et al. 2009; Lin et al. 2009; Akowuah et al. 2009), antimalarial (Najila et al. 2002), anticancer (Singh et al. 2001; Rajagopal et al. 2003; Kumar et al. 2004; Madamanchi et al. 2008; Lee et al. 2010c), immunomodulatory (Sheeja and Kuttan 2007b), antihypertensive (Zhang and Tan, 1999, Zhang and Tan1998), anti-psychotic (Mandal et al. 2001), anti-inflammatory (Xia et al. 2004; Chandrasekaran et al. 2010; Chao et al. 2009a, b, Qin et al. 2006), anti-thrombotic (Thisoda et al. 2006), antidiabetic (Reyes et al. 2006; Yu et al. 2008, Verma and Vinayak 2008), antidiabetic (Lee et al. 2010a), anti-atherosclerotic (Chen et al. 2004), cholestatic (Lee et al. 2010a), respiratory conditions (Roxas and Jurenka 2007), analgesic, antipyretic and antiulcerogenic (Madav et al. 1995) studies. The screening techniques used in the previous decades were primal and involved simple methodologies. But with the advent of molecular biology and receptor pharmacology techniques, better isolation and separation techniques, sophisticated analytical equipments, enzyme inhibition studies, cutting edge research methods were employed resulting in discovery or elucidation of various mechanisms of action of Andrographis paniculata extract for a variety of conditions and disorders.
Preclinical pharmacology
A few studies on preclinical pharmacology of Andrographis paniculata on laboratory animals by various researchers are described here:
Kumar et al. (2004) reported an account of anti-cancer and immunomodulatory effects of dichloromethane fraction of the methanolic extract of the herb on in vitro cell growth assay at concentrations ranging from 0.1 to 100 μg/ml. The active constituents responsible for these effects were found to be andrographolide, 14-deoxyandrographolide and 14-deoxy-11,12-didehydroandrographolide.
The protective activity of andrographolide and arabinogalactan proteins against alcohol-induced hepatic and renal toxicity was studied in mice by Singha et al. (2007). It was observed that pretreatment of mice with andrographolide, arabinogalactan proteins and silymarin as a positive control at doses of 62.5, 125, 250 and 500 mg/kg intraperitoneally administered every alternate day for 7 days reduced the toxicity in comparison to ethanol treated group. Andrographolide and arabinogalactan proteins were found to be the bioactive compounds responsible for protection against alcohol-induced hepatic and renal toxicity.
A study was carried out to evaluate the immunomodulatory activities of an ethanolic extract of Andrographis paniculata and andrographolide by Xu et al. (2007). The extract and andrographolide was administered at 25, 50 mg/kg body weight and 1, 4 mg/kg body weight respectively by oral route in mice immunised with an inactivated Salmonella typhimurium vaccine. Mice vaccinated with either one or two doses of killed Salmonella typhimurium vaccine were administered two different quantities of ethanolic extract of Andrographis paniculata and andrographolide for 14 days in mice immunised with one dose of the vaccine, and for 28 days in mice immunised with two doses of vaccine. The results showed enhanced Immunoglobulin G antibody levels against S. typhimurium by both ethanolic extract of Andrographis paniculata and andrographolide. There was also a demonstrated increase in the production of Interferon-γ following stimulation with the bacterial lysate, indicating an induction of Salmonella-specific cell-mediated response/immune response.
Sheeja et al. (2007) performed a study on the anti-angiogenic activity of ethanolic extract of Andrographis paniculata and andrographolide on mice. The ip administration of extract and andrographolide at 10 mg/dose/animal and 500 μg/dose/animals respectively significantly inhibited the B16F-10 melanoma cell line induced capillary formation in C57BL6 mice. The serum cytokine profile showed an upward trend in levels of the proinflammatory cytokines such as InterLeukin-1β, InterLeukin-6, Tumour Necrosis Factor-α, Granulocyte Macrophage-Colony Stimulating Factor and the most potent angiogenic factor Vascular Endothelial Growth Factor in angiogenesis induced animals. Treatment with extract and andrographolide significantly reduced these elevated levels. The results demonstrate that crude ethanolic extract of Andrographis paniculata and andrographolide inhibited the tumor specific angiogenesis by regulating the production of various pro and anti-angiogenic factors such as pro-inflammatory cytokines, nitric oxide, Vascular Endothelial Growth Factor, InterLeukin-2 and Tissue Inhibitor of Metallo Proteinases-1.
The protective properties of andrographolide, and aqueous extract of Andrographis paniculata were investigated against nicotine induced liver, kidney, heart, lung and spleen toxicity by Neogy et al. (2008). Wistar rats administered nicotine intraperitoneally 1 mg/kg body weight/day), nicotine + andrographolide 250 mg/kg, nicotine + aqueous extract 250 mg/kg for a period of 7 days showed a significant increase in levels of lipid peroxidation, protein oxidation and the decreased antioxidant enzyme status noted in nicotine treated group as compared to vehicle treated group. The aqueous extract and andrographolide significantly reduced the lipid peroxidation, protein oxidation and increased the antioxidant enzyme status. This indicates that the herb and active constituent, andrographolide may act as protective agents against nicotine induced tissue injury.
Iruretagoyena et al. (2005) and coresearchers reported that andrographolide interferes with T cell activation and reduces experimental autoimmune encephalomyelitis. In a mouse model of autoimmune encephalomyelitis, andrographolide significantly reduces the disease symptoms in by inhibiting T cell and antibody responses directed towards myelin antigens. Andrographolide prevented ovalbumin-pulsed dendritic cells from activating either CD4+ or CD8+ T cell hybridomas. Thus treatment with andrographolide prevented processing and presentation of ovalbumin peptides on MHC molecules to ovalbumin-specific T cells. Moreover, andrographolide reduced the efficiency of dendritic cell maturation in response to lipopolysaccharide mediated inflammation. Andrographolide also caused in vitro inhibition of T cell activation leading to the suppression of the immune response in the experimental systems. Thus, IgG secretion against T cell-dependent antigen NP17-BSA was significantly reduced by andrographolide treatment. Additionally andrographolide treatment significantly reduced both the incidence and clinical severity of experimental autoimmune encephalitis in C57BL/6 mice during early phase of disease. This ability of andrographolide to impair T cell activation could be the most probable reason. Observation of lymph node cellular suspensions from andrographolide treated mice showed reduced IFN-γ and IL-2 release in response to administration of myelin oligodendrocyte glycoprotein. Andrographolide-treated mice showed reduction in vivo T cell priming, which is probably responsible for the decreased delayed type hypersensitivity reactions and autoimmune encephalitis which may stem from the impairment on dendritic cell maturation and generation of peptide-MHC complexes. Hence andrographolide can be used to block T cell activation in vitro and in vivo which may be used as an immunosuppressive in the treatment of autoimmune diseases. Andrographolide could also interfere with neutrophils and microglial functions and these have already been implicated in the pathogenesis of inflammatory conditions. Antiapoptotic activity could also contribute to a reduction in the severity of autoimmune encephalitis by increasing neuronal resistance to cell death induced by local inflammation. Thus, andrographolide most likely interferes with autoimmune encephalitis by preventing activation of autoreactive T cells and also by reducing inflammatory damage.
Xia et al. (2004) and colleagues reported the potent inhibition of Nuclear Factor Kappa B (NF-κB) by andrographolide. Andrographolide was found to act as a small molecular weight antagonist for NF-κB activation by covalently modifying reduced cysteine 62 of p50. A significant reduction in the peritoneal deposition of neutrophils induced by cytokines and LPS was observed in this study. Moreover a complete reversal of the mortality or increase in survival time from endotoxin shock was seen. A remarkable inhibition of leukocyte infiltration into bronchial airway lavage was also reported. Andrographolide covalently conjugates reduced cysteine 62 of p50, thus preventing NF-κB oligonucleotide binding to p50, inhibiting NF-κB transcriptional activity, and attenuating inflammation in various in vitro assays and in vivo models (Xia et al. 2004). Interference of andrographolide with reduced cysteine 62 evaluated against oxidized cysteine 62 shows that, cysteine 62 of p50 is less reduced in the cytoplasm, when there is a tendency for stronger reduction by redox factor-1 in the nucleus. This has now been proposed as essential for the NF-κB activation. Andrographolide mainly targets this critical regulatory site of p50, the reduced cysteine 62 of p50, for attenuation of nuclear NF-κB transcriptional activity. Hence the redox modulation of cysteine 62 for the interaction of p50 with NF-κB oligonucleotide follows the inhibition of NF-κB activation by covalent conjugation to reduced cysteine 62 of p50 by andrographolide. In addition, andrographolide also suppresses NO production and down-regulates leukocyte integrin Mac-1 causing inhibition of neutrophil adhesion and transmigration. Taken together the results of the animal models examined in this study have collectively demonstrated the therapeutic efficacy of andrographolide for treating various inflammatory disorders.
Andrographolide was analysed for the activation of NF-κB induced by platelet-activating factor and N-formyl-methionyl-leucyl-phenylalanine (fMLP) in HL-60 cells differentiated to neutrophils (Hidalgo et al. 2005). In this study a neutrophil-like dimethylsulfoxide was used as a cellular model of neutrophils. The results have shown that NF-κB activation by PAF and fMLP are strongly inhibited by andrographolide, an effect that is mediated by blocking the binding of NF-κB to DNA. The final step in NF-κB activation is its binding to DNA which was inhibited by andrographolide. It is well known now andrographolide interferes with a transcription factor, and also affects the expression of target genes controlled by NF-κB during inflammatory processes. Expression of COX2 is reduced by NF-KB inhibitors in endothelial cells stimulated by PAF (Marrache et al. 2002) and in microglial cells andrographolide decreased COX2 expression induced by LPS (Wang et al. 2004). Our results of decreased COX2 expression after PAF or fMLP stimulation are in agreement with previous findings and demonstrate further that the inhibition of NF-κB binding to DNA, was able to decrease protein expression. Hence it was concluded that andrographolide exerts its anti-inflammatory effects by inhibiting NF-κB binding to DNA, thereby reducing the expression of proinflammatory proteins, such as COX2.
The neuroprotective effect of andrographolide was investigated by Chan et al. (2010). It was found that andrographolide at a dose of 0.1 mg/kg administered i.p. 1 h after the surgical procedure, reduced infarct volume with a maximum reduction of approximately 50%. In addition, andrographolide suppressed the translocation of p65 from cytosol to nucleus, indicating reduced NF-κB activation. Andrographolide exhibited neuroprotective effects, with accompanying suppression of NF-κB and microglial activation, and reduction in the production of tumour necrosis factor-α and interleukin-1β, and pro-inflammatory factors such as prostaglandin E2. These findings indicate the possible use of andrographolide in the treatment of cerebral stroke.
Hsieh et al. (2011) and coresearchers investigated the mechanism involved in andrographolide suppression of NF-κB signaling. They exposed the rat vascular smooth muscle cells (VSMCs) to various proinflammatory stimuli like Lipopolysaccharide, and IFN-γ. Andrographolide was shown to suppress LPS/IFN-γ-induced inducible nitric-oxide synthase and matrix metalloprotease 9 expression in rat VSMCs. Andrographolide also inhibited LPS/IFN-γ-induced p65 nuclear translocation, DNA binding activity, p65 Ser536 phosphorylation, and NF-κB reporter activity. These results demonstrate the protective anti-inflammatory actions of andrographolide which may be mediated through inhibition of transcription factor nuclear factor NF-κB.
Some of the reported pharmacological actions of Andrographis paniculata on commonly used laboratory animals are mentioned in Table 3.
Preclinical safety pharmacology
Numerous studies have been performed on the occurrence/presence of toxicity of Andrographis paniculata on rats, rabbits, and mice, but generally have been proved to be safe.
Akbarsha and Murugaian (2000) reported on the male reproductive toxicity on administering andrographolide at 25 and 50 mg/kg doses orally to Wistar albino rats for a period of 48 days. It was observed that sperm counts decreased, spermatozoa were not motile, and possessed abnormalities. The seminiferous epithelium was totally destroyed and in the seminiferous tubules fully differentiated spermatozoa were too limited. Thus a possible male reproductive toxicity effect could be considered. However a different study performed by Allan et al. (2009) demonstrated contrasting results. The researchers determined the effect of Andrographis paniculata extract Nees standardized to ≥10% andrographolide on male fertility in albino Wistar rats. The extract was orally administered at doses of 20, 200, and 1,000 mg/kg daily for 65 days prior to mating and 21 days during mating. The testosterone levels and fertility indices in treatment groups were found to be comparable with that of the control indicating no effect on fertility. Total sperm count and sperm motility were not affected. The testes and epididymies did not show any gross and histopathological changes. Hence the no-observed adverse effect level (NOAEL) of extract of Andrographis paniculata (≥10% andrographolide) was found to be more than 1,000 mg/kg per day.
A recent study by Chandrasekaran et al. (2009) on the genotoxic potential and acute oral toxicity of standardized extract of Andrographis paniculata has concluded that at a highest concentration of 5,000 μg/ml the standardized extract did not induce mutations both in the presence and absence of S9 in Salmonella typhimurium mutant strains of TA98 and TAMix. In chromosome aberration and micronucleus studies the extract did not induce clastogenicity in CHO-K1 cells in vitro. Based on these results, it is evident that the extract is genotoxically safe. In the acute oral toxicity study, female rats treated at the highest dose of 5,000 mg/kg of the standardised extract failed to produce any signs of toxicity after 14 days.
In a study performed by Sattayasai et al. (2010), the effects of andrographolide from Andrographis paniculata on sexual functions, vascular reactivity and serum testosterone level in experimental animals were observed. Andrographolide was administered orally in 5% DMSO at a dose of 50 mg/kg to male ICR mice and were mated with female mice. The mating behaviors, mounting latency and mounting frequency, were determined and compared with the standard reference drug sildenafil citrate. Administration of andrographolide significantly decreased the mounting latency at 120 and 180 min and increased mounting frequency at 180 min after treatment. Administration of 50 mg/kg andrographolide orally to male mice once daily for 2, 4, 6 or 8 weeks had no significant effects on sperm morphology and motility. Interestingly, at week 4, serum testosterone level in mice treated with andrographolide was significantly increased when compared to the control. It was concluded that the effects of andrographolide on vascular response to norepinephrine and testosterone level observed in this study might contribute to the sexual enhancing properties.
Guo et al. (1988) proved the lack of toxicity, on administering 500 mg/kg of extract and andrographolide for 20 days continuosly, for 10 days to mice. No effects on their appetite, growth or any untoward appearance on stool was found. The animals behaved normally and no altered blood counts were seen.
Rats and rabbits orally receiving 1 g/kg andrographolide or neoandrographolide for 7 days did not alter body weight, blood count, hepatic or renal functioning or functioning of other vital organs, demonstrating a clean toxicity record.
Cytochrome P450 plays an important role in the pharmacology of drugs and toxicology of xenobiotics. Understanding how drugs or xenobiotics induce or inhibit P450 activity is biologically relevant and ultimately leads to better models for predicting the actions of these agents. There was also an observed induction of of expression of CYP1A1 mRNA by andrographolide itself in a concentration-dependent manner in mouse hepatocyte primary culture (Jaruchotikamol et al. 2007).
Jarukamjorn et al. (2006) studied the impact of Andrographis paniculata crude extract on mouse hepatic cytochrome P450 enzymes by determining P450 content and P450-associated activities by treating ICR male mice with 3-methylcholanthrene at 100 mg/kg/day and phenobarbital at 100 mg/kg/day intraperitoneally in normal saline consecutively daily for 3 days. Andrographis paniculata in distilled water (equivalent to 5 mg/kg/day andrographolide) was administered orally consecutively daily for 7, 14, 21, and 30 days respectively. Assessment of hepatic microsomal P450 activities by alkoxyresorufin O-dealkylations showed that both the aqueous and alcoholic extracts of Andrographis paniculata significantly increased ethoxyresorufin O-dealkylase and pentoxyresorufin O-dealkylase activities, while those of ethoxyresorufin O-dealkylase and pentoxyresorufin O-dealkylase activities suggested that Andrographis paniculata might affect hepatic cytochrome P450 enzymes of which CYP1A1 and CYP2B are the most responsive P450 isoforms. This study may serve as an invaluable guideline of rational administration and precaution for using of herbal medicines.
In another study by Pekthong et al. (2008), the inhibitory effect of Andrographis paniculata extract and andrographolide in the extract on hepatic cytochrome P450s (CYPs) activities was examined using rat and human liver microsomes. It was found that the extract inhibited the catalytic activities of rat and human liver microsomal CYP1A2, CYP2C and human liver microsomal CYP3A4. These results suggest that the extract could act as an anticarcinogen in humans because of its specific inhibitory effect on CYP1A2 activity. The inhibitory effect of extract on CYP3A and 2C9 activities cannot be ruled out to cause drug–drug interactions, especially for CYP2C9 due to its low expression in human liver and because it is known to metabolize several narrow therapeutic index drugs.
Kondo et al. (2011) examined the effect of changes caused by andrographolide to intracellular GSH levels during the β-naphthoflavone-induced expression of CYP1A1 mRNA using mouse hepatocytes in primary culture. It was observed that the modification of inducible CYP1A1 mRNA expression by andrographolide was bimodal depending on treatment period and the modulation was retrieved by changing the intracellular GSH content. These results suggest that GSH status might be involved β-NF-induced CYP1A1 mRNA expression, and interaction of andrographolide with GSH might modulate the expression.
Another study carried out by Ooi et al. (2011) evaluated the effects of andrographolide and 14-deoxy-11, 12-didehydroandrographolide on CYP1A2, CYP2D6, and CYP3A4 expressions in HepG2 hepatoma cells. The mRNA and protein expression of the CYP.enzymes were performed by Quantitative RT-PCR and Western blot techniques. It was found that both diterpenoid active principles inhibited the mRNA and protein expressions of CYP1A2, CYP2D6, and CYP3A4. These findings suggest that patients known to take herbal preparation containing Andrographis paniculata Nees should be advised and cautioned of potential drug-herb interactions.
Clinical pharmacology
The standardized extract of Andrographis paniculata either alone or in combination with other plant extracts have been used in several clinical trials in recent years to ascertain its safety and efficacy on children, adult healthy humans, or adults with specific conditions/diseases. The studies of Andrographis paniculata either alone or in combination with other herbs are presented in Table 4. The standardized extract have been used in humans with specific conditions like ulcerative colitis, AIDS, common cold, upper respiratory tract infection with sinusitis, and arthritis and the safety and efficacy results have been promising. Few cases of adverse effects on humans were found to be self-limiting with nausea, diarrhea, metallic taste, allergic reaction, fatigue, headache, lymph node pain and lymphadenopathy. The adverse reports on human use were usually mild and infrequent. But then, most clinical trials were of short duration (less than 2 weeks), so the safety of the plant in chronic use could not be gauged. Furthermore, a systematic review undertaken by the Natural Standard Research Collaboration (Kligler et al. 2006) which compiled safety and efficacy results from 2 reviews and 7 clinical trials showed that out of 879 subjects exposed to the plant in any dosage form, only one case of anaphylactic shock was reported.
Saxena et al. (2010) evaluated the effect of an extract of Andrographis paniculata, in patients with uncomplicated upper respiratory tract infection (URTI). A total of 223 patients of both sexes were randomized in two groups which received either KalmCold (200 mg/day) or placebo in a double blind manner. The mean scores of all symptoms showed a decreasing trend from day 1 to day 3 but from day 3 to day 5 most of the symptoms in placebo treated group either remained unchanged (cough, headache, and earache) or got aggravated(sore throat and sleep disturbance) whereas in KalmCold TM treated group all symptoms showed a decreasing trend.
A prospective, randomized, double blind, and placebo-controlled study in patients with rheumatoid arthritis was performed by Burgos et al. (2009). The extract of Andrographis paniculata formulated as tablets (30% total andrographolides) were administered three times a day for 14 weeks, after a 2-week washout period to 60 patients with arthritis. It was observed that the intensity of joint pain decreased in the active vs placebo group at the end of treatment, although these differences were not statistically significant. A significant diminishing in tender joints, number of swollen joints, total grade of swollen joint, number of tender joints, total grade of swollen joints, total grade of tender joints was seen within the group with the active drug treatment. These improvements were also associated with a reduction of rheumatoid factor, IgA, and C4. These findings suggest that Andrographis paniculata could be a useful herbal treatment in the treatment of arthritis.
Moreover, very few reports on the incidences and propensity of the plant to cause frequent drug–drug interactions (Pekthong et al. 2008) are available which indicates its safety on administration in humans. It is hoped that with the demonstration of safety of Andrographis paniculata extracts in humans, the determination of effects of the extract in various diseases and condition is now possible, if the proof of concept preclinical studies demonstrate significant activity.
Conclusion
In promoting the proper use of medicinal products, a comprehensive road map must be charted supporting R&D, cultivation, production, trade and appropriate use in the community and healthcare sectors. Thus a joint, coordinated and holistic effort among scientists, farmers, manufacturers, traders, health care professionals and regulatory authorities is essential to propel the industry to comply with consumer expectations of quality, safety and efficacy in herbal products.
Although Ge et al. (2002) was the first to perform extraction of andrographolide using supercritical carbon dioxide as well as carbon dioxide and ethanol mixture, the technique itself is not new. But there is a need to suitably integrate this technique into research programs which will help reap rich dividends. It was shown that using supercritical fluids at high temperature and pressure, the extraction rate and yield were higher, but the yield of andrographolide in the extracts was very low and the simultaneous addition of ethanol as co-solvent enhanced the extract yield and the andrographolide content.
Previous works on the phytochemistry of Andrographis paniculata have established around 50 labdane diterpenoids and 30 flavonoids (Xu et al. 2010). More than 30 novel phytochemicals from Andrographis paniculata have been isolated, their structures elucidated using spectral and chromatographic techniques and found to have anticancer activity. It is interesting to note that in the isolation and identification of novel phytochemicals from Andrographis paniculata, the primary choice of extraction has been the conventional liquid–solid extraction technique of which Soxhlet extraction has been particularly favored. It is expected that with a gradual shift to the state of art, supercritical fluid extraction, the discovery of hitherto unknown phytochemicals and extension of pharmacological spectrum of activity of little known compounds of this plant is a distinct possibility, throwing up exciting challenges to researchers.
Datamining of available literature on Andrographis paniculata clearly shows an overwhelming therapeutic potential and can be safely regarded as one of the “modern world’s panacea”. Hence this high volume of research, keenly displays the interest of pharmacologists, medicinal chemists, and natural product chemists hot in pursuit to unravel the mysteries of this important medicinal plant. Moreover, quality control, maintaining batch to batch consistency and regulatory approval needs suitable means of redressal. Vital to the continuing research interests in Andrographis paniculata is a shift of focus from animal to humans. This final frontier can also be conquered given the fact that more than nine clinical trials on the investigation of Andrographis paniculata for a variety of conditions have already been successfully completed with no report of serious adverse effects incidents or fatalities.
So with the increase of interest in this plant the demand for active principles is skyrocketing and hence the dependence on wild populations is increasing, putting pressure on the already fragile ecosystem. Hence tissue culture techniques for mass production, and gene based initiatives for maximizing the active principles are assuming massive thrusts. Ultimately there is a positive scenario for a tremendous increase in organized cultivation and farming serving as a definite source of income. With the increase in popularity of the plant, there is a demand for this plant in the market thus sustaining the small scale, medium scale herbal industries employing a steady workforce.
Hence future prospects of Andrographis paniculata will be clearly on an upswing as research groups world over will be looking to exploit this herb for the treatment of a variety of disorders, industries thriving on this plant, and eventually the agriculturist also reaping his benefits. It is imperative that detailed information presented as a review here might fill the vacuum and paucity of information on this plant, providing the much required stimulus to utilize this plant in generating and sustaining a possible means of livelihood. But still it is far from reaching the “extraordinary league” of select group of phytochemicals like, paclitaxel, docetaxel, vinblastine, vincristine, taxols, podophyllotoxin, cannabinoids, digitoxin, camptothecins, etc. which are already established drugs forming an important part of current drug therapy for various disorders.
References
Aguiló A, Tauler P, Fuentespina E, Tur JA, Córdova A, Pons A (2005) Antioxidant response to oxidative stress induced by exhaustive exercise. Physiol Behav 84:1–7
Akbarsha MA, Murugaian P (2000) Aspects of the male reproductive toxicity/male antifertility property of andrographolide in albino rats: effect on the testis and the cauda epididymidal spermatozoa. Phyto Res 14:432–435
Akowuah GA, Zhari I, Norhayati I, Mariam A (2006) HPLC and HPTLC densitometric determination of andrographolides and anti-oxidant potential of Andrographis paniculata. J Food Comp Anal 19:118–126
Akowuah GA, Zhari I, Mariam A, Yam MF (2009) Absorption of andrographolides from Andrographis paniculata and its effect on CCl4-induced oxidative stress in rats. Food ChemToxicol 47:2321–2326
Allan JJ, Pore MP, Deepak M, Murali B, Mayachari AS, Agarwal A (2009) Reproductive and fertility effects of an extract of Andrographis paniculata in male Wistar rats. Int J Toxicol 28:308–317
Ambrosino P, Fresa R, Fogliano V, Monti SM, Ritieni A (1999) Extraction of Azadirachtin A from neem seed kernels by supercritical fluid and its evaluation by HPLC and LC/MS. J Agri Food Chem 47:5252–5256
Anuradha VE, Jaleel CA, Salem MA, Gomathinayagam M, Panneerselvam R (2010) Plant growth regulators induced changes in antioxidant potential and andrographolide content in Andrographis paniculata Wall.ex Nees. Pest Biochem Physiol 98:312–316
Bajaj YPS, Furmanowa M, Olszowska O (1988) Biotechnology of the micropropagation of medicinal and aromatic plants. In: Bajaj YPS (ed) Biotech agri forest. Medicinal and aromatic plants. Springer, Berlin, Germany, pp 60–103
Bao Z, Guan S, Cheng C, Wu S, Wong SH, Kemeny DM, Leung BP, Wong WS (2009) A novel antiinflammatory role for andrographolide in asthma via inhibition of the nuclear factor-ΚB pathway. Am J Resp Crit Care Med 179:657–665
Basak A, Cooper S, Roberge AG, Banik UK, Chrétien M, Seidah NG (1999) Inhibition of proprotein convertases-1,-7 and furin by diterpines of Andrographis paniculata and their succinoyl esters. Biochem J 338:107–113
Batkhuu J, Hattori K, Takano F, Fushiya S, Oshiman K, Fujimiya Y (2002) Suppression of NO production in activated macrophages in vitro and ex vivo by neoandrographolide isolated from Andrographis paniculata. Biol Pharm Bull 25:1169–1174
Berman AF (2000) Herb-drug interactions. Lancet 355:134–138
Bhan MK, Dhar AK, Khan S, Lattoo SK, Gupta KK, Choudhary DK (2006) Screening and optimization of Andrographis paniculata (Burm.f.) Nees for total andrographolide content, yield and its components. Sci Hort 107:386–391
Burgos RA, Loyola M, Hidalgo MA, Labranche TP, Hancke JL (2003) Effect of 14-deoxyandrographolide on calcium-mediated rat uterine smooth muscle contractility. Phyto Res 17:1011–1015
Burgos RA, Hidalgo MA, Monsalve J, LaBranche TP, Eyre P, Hancke JL (2005a) 14-deoxyandrographolide as a platelet activating factor antagonist in bovine neutrophils. Planta Med 71:604–608
Burgos RA, Seguel K, Perez M, Meneses A, Ortega M, Guarda MI, Loaiza A, Hancke JL (2005b) Andrographolide inhibits IFN-gamma and IL-2 cytokine production and protects against cell apoptosis. Planta Med 71:429–434
Burgos RA, Hancke JL, Bertoglio JC, Aguirre V, Arriagada S, Calvo M, Cáceres DD (2009) Efficacy of an Andrographis paniculata composition for the relief of rheumatoid arthritis symptoms: a prospective randomized placebo-controlled trial. Clin Rheumatol 28:931–946
Burkill IH (1966) A dictionary of the economic products of the Malay Peninsula, vol I (A–H). Ministry of Agriculture and Co-operatives, Kuala Lumpur, Malaysia, pp 45–67
Cáceres DD, Hancke JL, Burgos RA, Sandberg F, Wikman GK (1999) Use of visual analogue scale measurements (VAS) to assess the effectiveness of standardized Andrographis paniculata extract SHA-10 in reducing the symptoms of common cold. A randomized double blind-placebo study. Phytomedicine 6:217–223
Calabrese C, Berman SH, Babish JB, Ma X, Shinto L, Dorr M, Wells K, Wenner CQ, Standish LA (2000) A Phase I trial of andrographolide in HIV positive patients and normal volunteers. Phyto Res 14:333–338
Carretta MD, Alarcón P, Jara E, Solis L, Hancke JL, Concha II, Hidalgo MA, Burgos RA (2009) Andrographolide reduces IL-2 production in T-cells by interfering with NFAT and MAPK activation. Eur J Pharmacol 602:413–421
Chan SJ, Wong WSF, Wong PTH, Bian JS (2010) Neuroprotective effects of andrographolide in a rat model of permanent cerebral ischaemia. Brit J Pharmacol 161:668–679
Chander R, Srivastava V, Tandon JS, Kapoor NK (1995) Anti-hepatotoxic activity of diterpenes of Andrographis paniculata (Kalmegh) against Plasmodium berghei-induced hepatic damage in Mastomys natalensis. Int J Pharmacog 33:135–138
Chandrasekaran CV, Thiyagarajan P, Sundarajan K, Goudar KS, Deepak M, Murali B, Allan JJ, Agarwal A (2009) Evaluation of the genotoxic potential and acute oral toxicity of standardized extract of Andrographis paniculata Kalm Cold™. Food Chem Toxicol 47:1892–1902
Chandrasekaran CV, Gupta A, Agarwal A (2010) Effect of an extract of Andrographis paniculata leaves on inflammatory and allergic mediators in vitro. J Ethnopharmacol 129:203–207
Chao WW, Lin BF (2010) Isolation and identification of bioactive compounds in Andrographis paniculata (Chuanxinlian). Chin Med 17:1–15
Chao WW, Kuo YH, Li WC, Lin BF (2009a) The production of nitric oxide and prostaglandin E2 in peritoneal macrophages is inhibited by Andrographis paniculata, Angelica sinensis and Morus alba ethyl acetate fractions. J Ethnopharmacol 122:68–75
Chao WW, Kuo YH, Hsieh SL, Lin BF (2009b) Inhibitory effects of ethyl acetate extract of Andrographis paniculata on NF-κB trans-activation activity and LPS-induced acute inflammation in mice. Evid Based Compl Alt Med 6:5–17
Chauhan SK, Gyanendra T (2003) Effect of sources and levels of nitrogen on partitioning of andrographolide in Kalmegh (Andrographis paniculata Nees). Ind J Plant Physiol 8:60–62
Chen JH, Hsiao G, Lee AR, Wu CC, Yen MH (2004) Andrographolide suppresses endothelial cell apoptosis via activation of phosphatidyl inositol-3-kinase/Akt pathway. Biochem Pharmacology 67:1337–1345
Chen X, Tang JJ, Zhi GY, Hu SJ (2005) Arbuscular mycorrhizal colonization and phosphorus acquisition of plants: effects of coexisting plant species. Appl Soil Ecol 28:259–269
Chen LX, Qiu F, Wei H, Qu GX, Yao XS (2006) Nine new ent-Labdane diterpenoids from the aerial parts of Andrographis paniculata. Helv Chim Acta 89:2654–2664
Chen L, Jin H, Ding L, Zhang H, Wang X, Wang Z, Li J, Qu C, Wang Y, Zhang H (2007a) On-line coupling of dynamic microwave-assisted extraction with high-performance liquid chromatography for determination of andrographolide and dehydroandrographolide in Andrographis paniculata Nees. J Chrom A 1140:71–77
Chen L, Yu A, Zhuang X, Zhang K, Wang X, Ding L, Zhang H (2007b) Determination of andrographolide and dehydroandrographolide in rabbit plasma by on-line solid phase extraction of high-performance liquid chromatography. Talanta 74:146–152
Chen L, Zhu H, Wang R, Zhou K, Jing Y, Qiu F (2008) ent-Labdane diterpenoid lactone stereoisomers from Andrographis paniculata. J Nat Prod 71:852–855
Chen JX, Xue HJ, Ye WC, Fang BH, Liu YH, Yuan SH, Yu P, Wang YQ (2009) Activity of andrographolide and its derivatives against influenza virus in vivo and in vitro. Biol Pharm Bull 32:1385–1391
Cheung HY, Cheung CS, Kong CK (2001) Determination of bioactive diterpenoids from Andrographis paniculata by micellar electrokinetic chromatography. J Chromato A 930:171–176
Cheung HY, Cheung SH, Li J, Cheung CS, Lai WP, Fong WF, Leung FM (2005) Andrographolide isolated from Andrographis paniculata induces cell cycle arrest and mitochondrial-mediated apoptosis in human leukemic HL-60 cells. Planta Med 71:1106–1111
Chiramel T, Bagyaraj DJ, Patil CSP (2006) Response of Andrographis paniculata to different arbuscular mycorrhizal fungi. J Agri Tech 2:221–228
Coldren CD, Hshim P, Ali JM, Oh SY, Sinskey AJ, Rha CK (2003) Gene expression changes in the human fibroblast induced by Centella asiatica triterpenoids. Planta Med 69:725–732
Cui Y, Wang Y, Ouyang X, Han Y, Zhu H, Chen Q (2009) Fingerprint profile of active components for Andrographis paniculata Nees by HPLC-DAD. Sens Instr Food Qual 3:165–179
Das S, Gautam N, Dey SK, Maiti T, Roy S (2009) Oxidative stress in the brain of nicotine-induced toxicity: protective role of Andrographis paniculata Nees and vitamin E. Appl Physiol Nutr Metab 34:125–135
DeFeudis F, Papadopoulos V, Drieu K (2003) Ginkgo biloba extracts and cancer: a research area in its infancy. Fund Clin Pharmacol 17:405–417
Du Q, Jerz G, Winterhalter P (2003) Separation of andrographolide and neoandrographolide from the leaves of Andrographis paniculata using high-speed counter-current chromatography. J Chromato A 984:147–151
Dua VK, Ojha VP, Roy R, Joshi BC, Valecha N, Usha Devi C, Bhatnagar MC, Sharma VP, Subbarao SK (2004) Anti-malarial activity of some xanthones isolated from the roots of Andrographis paniculata. J Ethnopharmacol 95:247–251
Fabricant DS, Farnsworth NR (2001) The value of plants used in traditional medicine for drug discovery. Environ Health Persp 109:69–75
Faisal M, Ahmad N, Anis M (2005) Shoot multiplication in Rauvolfia tetraphylla L. using thidiazuron. Plant Cell Tiss Org Cult 80:187–190
Farnsworth NR, Soejarto DD (1991) Global importance of medicinal plants. In: the conservation of medicinal plants. Proceedings of an international consultation. Akerele O, Heywood V, Synge H (eds) Chiang Mai, Thailand. Cambridge University Press, Cambridge, pp 25–51
Fujita T, Fujitani R, Takeda Y, Takaishi Y, Yamada T, Kido M, Miura I (1984) On the diterpenoids of Andrographis paniculata: x-ray crystallographic analysis of andrographolide and structure determination of new minor diterpenoids. Chem Pharm Bull 32:2117–2125
Gabrielian ES, Shukarian AK, Goukasova GI, Chandanian GL, Panossian AG, Wikman G, Wagner H (2002) A double blind, placebo-controlled study of Andrographis paniculata fixed combination Kan Jang in the treatment of acute upper respiratory tract infections including sinusitis. Phytomedicine 9:589–597
Ge FH, Lin XX, Huan XF, Shi Q, Liang B, Li J, Zhong G (2002) Study on extraction of active ingredients from Andrographis paniculata using orthogonal experiment with supercritical carbon dioxide. J Chin Med Mat 25:101–102
WHO Geneva (2002) WHO monographs on selected medicinal plants, 2: 12–24
Girach RD, Khan A, Khan S (1994) Andrographis paniculata—a possible substitute for Swertia chirata in Southeastern India. Pharm Biol 32:95–97
Gu Y, Ma J, Liu Y, Chen B, Yao S (2007) Determination of andrographolide in human plasma by high-performance liquid chromatography/mass spectrometry. J Chromat B 854:328–331
Guo SY, Li DZ, Li WS, Fu AH, Zhang LF (1988) Study of the toxicity of andrographolide in rabbits. J Beijing Med Univ 5:422–428
Han G, Liu L, Yu QT, Xie SQ (2006). Antiinflammatory and anticancer. Chinese Patent, CN 1785177A
He X, Zeng X, Hu H, Wu Y (2010) Cytotoxic biotransformed products from andrographolide by Rhizopus stolonifer ATCC 1939. J Molec Catal Enzy 62:242–247
Hidalgo M, Romero A, Figueroa J, Corte′s P, Concha II, Hancke JL, Burgos RA (2005) Andrographolide interferes with binding of NF-κB to DNA in HL-60-derived neutrophilic cells. Brit J Pharmacol 144: 680–686
Hsieh CY, Hsu MJ, Hsiao G, Wang YH, Huang CW, Chen SW, Jayakumar T, Chiu PT, Chiu YH, Sheu JR (2011) Andrographolide enhances nuclear factor-κB subunit p65 Ser536 dephosphorylation through activation of protein phosphatase 2A in vascular smooth muscle cells. J Biol Chem 286:5942–5955
Huang CJ, Wu MC (2002) Differential effects of foods traditionally regarded as ‘heating’ and ‘cooling’ on prostaglandin E2 production by a macrophage cell line. J Biomed Sci 9:596–606
Hung SK, Hung LC, Kuo CD, Lee KY, Lee MS, Lin HY, Chen YJ, Fu SL (2010) Andrographolide sensitizes Ras-transformed cells to radiation in vitro and in vivo. Int J Rad Oncol Biol Phy 77:1232–1239
Iruretagoyena MI, Tobar JA, González PA, Sepúlveda SE, Figueroa CA, Burgos RA, Hancke JL, Kalergis AM (2005) Andrographolide interferes with T cell activation and reduces experimental autoimmune encephalomyelitis in the mouse. J Pharmacol Exp Therap 312:366–372
Izuka N, Oka M, Yamamoto K, Tangoku A, Miyamoto K, Miyamoto T, Uchimura S, Hamamoto Y, Okita K (2003) Identification of common or distinct genes related to antitumor activities of a medicinal herb and its major component by oligonucleotide microarray. Int J Cancer 107:666–672
Jain DC, Gupta MM, Saxena S, Kumar S (2000) LC analysis of hepatoprotective diterpenoids from Andrographis paniculata. J Pharm Biomed Anal 22:705–709
Jaleel CA, Gopi R, Lakshmanan GMA, Panneerselvam R (2006) Triadimefon induced changes in the antioxidant metabolism and ajmalicine production in Catharanthus roseus (L.) G. Don. Plant Sci 171:271–276
Jaruchotikamol A, Jarukamjorn K, Sirisangtrakul W, Sakuma T, Kawasaki Y, Nemoto N (2007) Strong synergistic induction of CYP1A1 expression by andrographolide plus typical CYP1A inducers in mouse hepatocytes. Toxicol Appl Pharmacol 224:156–162
Jarukamjorn K, Don-in K, Pearaksa P, Sripanidkulchai B (2006) Impact of Andrographis paniculata crude extract on mouse hepatic cytochrome P450 enzymes. J Ethnopharmacol 105:464–467
Ji LL, Wang Z, Dong F, Zhang WB, Wang ZT (2005) Andrograpanin, a compound isolated from anti-inflammatory traditional Chinese medicine Andrographis paniculata, enhances chemokine SDF-1α-induced leukocytes chemotaxis. J Cell Biochem 95:970–978
Ji L, Liu T, Liu J, Chen Y, Wang Z (2007) Andrographolide inhibits human hepatoma-derived Hep3B cell growth through the activation of c-Jun N-terminal kinase. Planta Med 73:1397–1401
Jiang CG, Li JB, Liu FR, Wu T, Yu M, Xu HM (2007) Andrographolide inhibits the adhesion of gastric cancer cells to endothelial cells by blocking E-selectin expression. Anticancer Res 27:2439–2448
Kamdem RE, Sang S, Ho CT (2002) Mechanism of the superoxide scavenging activity of neoandrographolide-a natural product from Andrographis paniculata Nees. J Agric Food Chem 50:4662–4665
Kanjilal PB, Bordoloi S, Kalita R, Burman P, Singh RS (2002) Cultivation practices for Kalmegh (Andtrographis paniculata) and Spiderling. In: Govil JN, Kumar PA, Singh VK (eds) Recent progress in medicinal plants, vol 5. Stadium Press LLC, USA, pp 175–180
Kapil A, Koul IB, Banerjee SK, Gupta BD (1993) Antihepatotoxic effects of major diterpenoid constituents of Andrographis paniculata. Biochem Pharmacol 46:182–185
Kavuri SR, Mukkamala SB, Putheti R (2010) Quantitative determination of two bioactive compounds in Andrographis paniculata (Burm.f) Nees by ultra performance liquid chromatography. J Pharm Res 3:1682–1684
Khalafallah AA, Abo-Ghalia HH (2008) Effect of arbuscular mycorrhizal fungi on the metabolic products and activity of antioxidant system in wheat plants subjected to short-term water stress, followed by recovery at different growth stages. J Appl Sci Res 4:559–569
Kirtikar KR, Basu BD (1935). Indian medicinal plants vol 3. Publishers M/s Bishen Singh Mahendrapal, New Delhi, India pp 45–59
Kligler B, Ulbrich C, Basch E, Kirkwoo CD, Abrams TR, Miranda M, Khalsa KS, Giles M, Boon H, Woods T (2006) Andrographis paniculata for the treatment of upper respiratory infection: a systematic review by the natural standard research collaboration. Explore 1:25–29
Ko HC, Wei BL, Chiou WF (2006) The effect of medicinal plants used in Chinese folk medicine on RANTES secretion by virus-infected human epithelial cells. J Ethnopharmacol 107:205–210
Kondo Chatuphonprasert W, Jaruchotikamol A, Sakuma T, Nemoto N (2011) Cellular glutathione content modulates the effect of andrographolide on β-naphthoflavone-induced CYP1A1 mRNA expression in mouse hepatocytes. Toxicol 280:18–23
Kulyal P, Tiwari UK, Shukla A, Gaur AK (2010) Chemical constituents isolated from Andrographis paniculata. Ind J Chem 49B:356–359
Kumar RA, Sridevi K, Kumar NV, Nanduri S, Rajagopal S (2004) Anticancer and immunostimulatory compounds from Andrographis paniculata. J Ethnopharmacol 92:291–295
Kumaran KS, Thirugnanasambantham P, Viswanathan S, Ramamurthy MS (2003) An HPLC method for the estimation of andrographolide in rabbit serum. Ind J Pharmacol 35:109–112
Kumoro AC, Hasan M (2007) Supercritical carbon dioxide extraction of andrographolide from Andrographis paniculata: effect of the solvent flow rate, pressure, and temperature. Chin J Chem Eng 15:877–883
Kuroyanagi M, Sata M, Ueno A, Nishi K (1987) Flavonoids from Andrographis paniculata. Chem Pharm Bull 35:4429–4435
Lala S, Nandy AK, Mahato SB, Basu MK (2003) Delivery in vivo of 14-deoxy-11-oxo andrographolide an antileishmanial agent, by different drug carriers. Ind J Biochem Biophys 40:169–174
Lang A, Wai CM (2001) Supercritical fluid extraction in herbal and natural product studies -a practical review. Talanta 53:771–782
Lattoo SK, Dhar RS, Khan S, Bamotra S, Bhan MK, Dhar AK, Gupta KK (2008) Comparative analysis of genetic diversity using molecular and morphometric markers in Andrographis paniculata (Burm. f.) Nees. Gen Resour Crop Evol 55:33–43
Lee MJ, Rao YK, Chen K, Lee YC, Chung YS, Tzeng YM (2010a) Andrographolide and 14-deoxy-11, 12-didehydroandrographolide from Andrographis paniculata attenuate high glucose-induced fibrosis and apoptosis in murine renal mesangeal cell lines. J Ethnopharmacol 132:497–505
Lee TY, Lee KC, Chang HH (2010b) Modulation of the cannabinoid receptors by andrographolide attenuates hepatic apoptosis following bile duct ligation in rats with fibrosis. Apoptosis 15:904–914
Lee YC, Lin HH, Hsu CH, Wang CJ, Chiang TA, Chen JH (2010c) Inhibitory effects of andrographolide on migration and invasion in human non-small cell lung cancer A549 cells via down-regulation of PI3K/Akt signaling pathway. Eur J Pharmacol 632:23–32
Li WK, Fitzloff JF (2002) Determination of andrographolide in commercial Andrographis (Andrographis paniculata) products using HPLC with evaporative light scattering detection. J Liq Chromato Rel Technol 25:1335–1343
Li WK, Fitzloff JF (2004) HPLC-PDA determination of bioactive diterpenoids from plant materials and commercial products of Andrographis paniculata. J Liq Chromato Rel Technol 27:2407–2420
Li W, Xu X, Zhang H, Ma C, Fong H, Van Breemen R, Fitzloff J (2007) Secondary metabolites from Andrographis paniculata. Chem Pharm Bull 55:455–458
Li J, Luo L, Wang X, Liao B, Li G (2009) Inhibition of NF-κB expression and allergen-induced airway inflammation in a mouse allergic asthma model by andrographolide. Cell Mol Immunol 6:381–385
Liang FP, Lin CH, Kuo CD, Chao HP, Fu SL (2008) Suppression of v-Src transformation by andrographolide via degradation of the v-Src protein and attenuation of the Erk signaling pathway. J Biol Chem 283:5023–5033
Lin TP, Chen SY, Duh PD, Chang LK, Liu YN (2008) Inhibition of the Epstein-Barr virus lytic cycle by andrographolide. Biol Pharm Bull 31:2018–2023
Lin FL, Wu SJ, Lee SC, Ng LT (2009) Antioxidant, antioedema and analgesic activities of Andrographis paniculata extracts and their active constituent andrographolide. Phyto Res 23:958–964
Liu J, Wang ZT, Ji LL, Ge BG (2007) Inhibitory effects of neoandrographolide on nitric oxide and prostaglandin E2 production in LPS-stimulated murine macrophage. Mol Cell Biochem 298:49–57
Liu J, Wang ZT, Ge BX (2008) Andrograpanin, isolated from Andrographis paniculata, exhibits anti-inflammatory property in lipopolysaccharide-induced macrophage cells through down-regulating the p38 MAPKs signaling pathways. Int Immunopharmacol 8:951–958
Ma ZQ, Sorrells ME, Tanksley SD (1994) RFLP markers linked to powdery mildew resistance genes Pm1, Pm2, Pm3, and Pm4 in wheat. Genome 37:871–875
Ma XC, Gou ZP, Wang CY, Yao JH, Xin XL, Lin Y, Liu KX (2010) A new ent-labdane diterpenoid lactone from Andrographis paniculata. Chin Chem Lett 71:852–855
Madamanchi G, Rao YK, Fang SH, Tzeng YM (2008) Cytotoxic constituents from Andrographis paniculata induce cell cycle arrest in jurkat cells. Phyto Res 22:136–1341
Madav S, Tripathi C, Tandon S, Mishra SK (1995) Analgesic, anti-pyretic and anti-ulcerogenic effects of andrographolide. Ind J Pharm Sci 57:121–125
Maheshwari SK, Sharma RK, Mishra PK, Gangrade SH (2002) Response of Kalmegh (Andrographis paniculata) to dates of planting and harvesting in a shallow back soil. J Med Arom Plant Sci 24:969–971
Mandal SC, Dhara AK, Maiti BC (2001) Studies on psychopharmacological Activity of Andrographis paniculata Extract. Phyto Res 15:253–256
Manikam SD, Stanslas J (2009) Andrographolide inhibits growth of acute promyelocytic leukaemia cells by inducing retinoic acid receptor-independent cell differentiation and apoptosis. J Pharm Pharmacol 61:69–78
Marrache AM, Gobeil Jr F, Bernier SG, Stankova J, Rola-Pleszczynski M, Choufani S, Bkaily G, Bourdeau A, Sirois MG, Vazquez-Tello A, Fan L, Joyal JS, Filep JG, Varma DR, Ribeiro-Da-Silva A, Chemtob S (2002) Proinflammatory gene induction by platelet-activating factor mediated via its cognate nuclear receptor. J Immunol 169:6474–6481
Martin KP (2004) Plant regeneration protocol of medicinally important Andrographis paniculata (Burm. F.) Wallich ex nees via somatic embryogenesis. In vitro Cell Develop Biol-Plant 40:204–209
Marulanda A, Azcon R, Luiz-Lozano JM (2003) Contribution of six arbuscular mycorrhizal fungal isolates to water uptake by Lactuca sativa plants under drought stress. Physiol Planta 119:526–533
Matsuda T, Kuroyanagi M, Sugiyama S, Umehara K, Ueno A, Nishi K (1994) Cell differentiation inducing diterpenes from Andrographis paniculata. Chem Pharm Bull 42:1216–1225
Mishra SK, Sangwan NS, Sangwan RS (2007) Andrographis paniculata (Kalmegh): a review. Pharmacog Rev 1:283–298
Misra P, Pal NL, Guru PY, Katiyar JC, Srivastava V, Tandon JS (1992) Antimalarial activity of Andrographis paniculata (Kalmegh) against plasmodium berghei NK65 in Mastomys natalensis. Int J Pharmacog 30:263–274
Mkrtchyan A, Panosyan V, Panossian A, Wikman G, Wagner H (2005) A phase I clinical study of Andrographis paniculata fixed combination Kan Jang™ versus ginseng and valerian on the semen quality of healthy male subjects. Phytomedicine 12:403–409
Mukherjee PK (2002). Analytical profiles of selected medicinal plants. Quality control of herbal drugs. An approach to evaluation of botanicals (1st edn). Business Horizons, New Delhi India, pp 704
Naik SR, Hule A (2009) Evaluation of immunomodulatory activity of an extract of andrographolides from Andographis paniculata. Planta Med 75:785–791
Najila SMJ, Noor Rain A, Mohamad Kamel AG, Syed Zahir SI, Khozirah S, Lokman Hakim S, Zakiah I, Azizol AK (2002) The screening of extracts from Goniothalamus scortechinii, Aralidium pinnatifidum and Andrographis paniculata for anti-malarial activity using the lactate dehydrogenase assay. J Ethnopharmacol 82:239–242
Neogy S, Das S, Mahapatra SK, Mandal N, Roy S (2008) Amelioratory effect of Andrographis paniculata Nees on liver, kidney, heart, lung and spleen during nicotine induced oxidative stress. Env Toxicol Pharmacol 25:321–328
Nowak J (2004). Effects of arbuscular mycorrhizal fungi and organic fertilization on growth, lowering, nutrient uptake, photosynthesis and transpiration of geranium (Pelargonium hortorum) LH Bailey “Tango Orange”. Symbiosis 37: 259–266
Ojha SK, Nandave M, Kumari S, Arya DS (2009) Antioxidant activity of Andrographis paniculata in ischemic myocardium of rats. Global J Pharmacol 3:154–157
Ooi JP, Kuroyanagi M, Sulaiman SF, Muhammad TST, Tan ML (2011) Andrographolide and 14-Deoxy-11, 12-Didehydroandrographolide inhibit cytochrome P450 s in HepG2 hepatoma cells. Life Sci 88:447–454
Padmesh P, Sabu KK, Seeni S, Pushpangadan P (1999) The use of RAPD in assessing genetic variability in Andrographis paniculata Nees, a hepatoprotective drug. Curr Sci 76:833–835
Panossian A, Davtyan T, Gukassyan N, Gukasova G, Mamikonyan G, Gabrielian E, Wikman G (2002) Effect of andrographolide and Kan Jang-fixed combination of extract SHA-10 and extract SHE-3 on proliferation of human lymphocytes, production of cytokines and immune activation markers in the whole blood cells culture. Phytomedicine 9:598–605
Parichatikanond W, Suthisisang C, Dhepakson P, Herunsalee A (2010) A study of anti-inflammatory activities of the pure compounds from Andrographis paniculata (burm.f.) Nees and their effects on gene expression. Int Immunopharmacol 10:1361–1373
Pattnaik SK, Chand PK (1996). In vitro propagation of the medicinal herbs Ocimum americanum L. syn. Ocimum canum Sims (hoary basil) Ocimum sanctum (holy basil). Plant Cell Reports 15: 846–850
Pekthong D, Martin H, Abadie C, Bonet A, Heyd B, Mantion G, Richert L (2008) Differential inhibition of rat and human hepatic cytochrome P450 by Andrographis paniculata extract and andrographolide. J Ethnopharmacol 115:432–440
Pereira F, Joao Carneiro, Amorim A (2008) Identification of species with DNA-based technology: current progress and challenges. Rec Pat DNA Gene Seq 2:187–200
Pfisterer P, Rollinger J, Schyschka L, Rudy A, Vollmar A, Stuppner H (2010) Neoandrographolide from Andrographis paniculata as a potential natural chemosensitizer. Planta Med 76:1698–1700
Pholphana N, Rangkadilok N, Thongnest S, Ruchirawat M, Jutamaad S (2004) Determination and variation of three active diterpenoids in Andrographis paniculata (Burm.f.) Nees. Phytochem Anal 15:365–371
Pramanick S, Banerjee S, Achari B, Das B, Sen AK, Mukhopadhyay S, Neuman A, Prange T (2006) Andropanolide and Isoandrographolide, minor diterpenoids from Andrographis paniculata: structure and X-ray crystallographic analysis. J Nat Prod 69:403–405
Prathanturarug S, Soonthornchareonnon N, Chuakul W, Saralamp P (2007) Variation in growth and diterpene lactones among field-cultivated Andrographis paniculata. J Nat Med 61:159–163
Praveen N, Manohar SH, Naik PM, Nayeem A, Jeong JH, Murthy HN (2009) Production of andrographolide from adventitious root cultures of Andrographis paniculata. Curr Sci 96:694–697
Purkayastha J, Sugla T, Paul A, Solleti S, Sahoo L (2008) Rapid in vitro multiplication and plant regeneration from nodal explants of Andrographis paniculata: a valuable medicinal plant. In vitro Cell Develop Biol-Plant 44:442–447
Purohit SD, Dave A, Kukda D (1994) Micropropagation of safed musli (Chlorophytum borivilianum), a rare medicinal herb. Plant Cell Tiss Org Cult 39:93–96
Qin LH, Kong L, Shi GJ, Wang ZT, Ge BX (2006) Andrographolide inhibits the production of TNF-alpha and interleukin-12 in lipopolysaccharide-stimulated macrophages: role of mitogen-activated protein kinases. Biol Pharm Bull 29:220–224
Rahman NNNA, Furuta T, Kojima S, Takane K, Mohd MA (1999) Antimalarial activity of extracts of Malaysian medicinal plants. J Ethnopharmacol 64:249–254
Rajagopal S, Kumar RA, Deevi DS, Satyanarayana C, Rajagopalan R (2003) Andrographolide, a potential cancer therapeutic agent isolated from Andrographis paniculata. J Exp Therap Oncol 3:147–158
Rammohan S, Asmawi MZ (2006) Inhibition of a-glucosidase by Andrographis paniculata ethanol extract in rats. Pharm Biol 44:600–606
Rammohan S, Asmawi MZ, Sadikun A (2008a) Effect of ethanolic extract of Andrographis paniculata Nees on a combination of fat-fed diet and low dose streptozotocin induced chronic insulin resistance in rats. Diabetolog Croat 37:13–22
Rammohan S, Asmawi MZ, Sadikun A (2008b) Effect of Andrographolide and ethanol extract of Andrographis paniculata on liver glycolytic, gluconeogenic, and lipogenic enzymes in a Type 2 diabetic rat model. Pharm Biol 46:1–11
Rammohan S, Asmawi MZ, Sadikun A (2008c) In vitro alpha glucosidase and alpha amylase enzyme inhibitory effects of Andrographis paniculata extract and andrographolide. Acta Biochim Pol 55:391–398
Rao YK, Vimalamma G, Rao CV, Tzeng YM (2004) Flavonoids and andrographolides from Andrographis paniculata. Phytochem 65:2317–2321
Reddy MK, Reddy MVB, Gunasekar D, Murthy MM, Caux C, Bodo B (2003) A flavone and an unusual 23-carbon terpenoid from Andrographis paniculata. Phytochem 62:1271–1275
Reddy VLN, Reddy SM, Ravikantha V, Krishnaiah P, Goud TV, Rao TP, Sivaram T, Gonnade RG, Bhadbhade M, Venkateswarlu Y (2005) A new BIS-andrographolide ether from Andrographis paniculata Nees and evaluation of anti-HIV activity. Nat Prod Res 19:223–230
Reyes BAS, Bautista ND, Tanquilut NC, Anunciado RV, Leung AB, Sanchez GC, Magtoto RL, Castronuevo P, Tsukamura H, Maeda KI (2006) Anti-diabetic potentials of Momordica charantia and Andrographis paniculata and their effects on estrous cyclicity of alloxan-induced diabetic rats. J Ethnopharmacol 105:196–200
Rout GR (2002) Direct plant regeneration from leaf explants of Plambago species and its genetic fidelity through RAPD markers. Ann Appl Biol 140:305–313
Roxas MND, Jurenka JMT (2007) Colds and influenza: a review of diagnosis and conventional, botanical, and nutritional considerations. Alt Med Rev 12:25–48
Roy DN, Mandal S, Sen G, Mukhopadhyay S, Biswas T (2010) 14-Deoxyandrographolide desensitizes hepatocytes to tumour necrosis factor-alpha-induced apoptosis through calcium-dependent tumour necrosis factor receptor superfamily member 1A release via the NO/cGMP pathway. Brit J Pharmacol 160:1823–1843
Ruengsitagoon W, Anuntakarun K, Aromdee C (2006) Flow injection spectrophotometric determination of andrographolide from Andrographis paniculata. Talanta 69:900–905
Russell FE (1980) Snake venom poisoning, vol 562. J.B. Lippincott Company, Philadelphia, USA, pp 165–166
Sailo GL, Bagyaraj DJ (2005) Influence of different AM fungi on growth, nutrition and forskholin content of Coleus forskohlii. Mycol Res 109:795–798
Samy RP, Thwin MM, Gopalakrishnakone P, Ignacimuthu S (2008) Ethnobotanical survey of folk plants for the treatment of snakebites in Southern part of Tamilnadu, India. J Ethnopharmacol 115:302–312
Sandborn W, Targan S, Byers V, Yan X, Tang T (2009) Double-blind, placebo-controlled phase IIB trial of HMPL-004 (Andrographis paniculata Extract) in active mild-to-moderate ulcerative colitis (UC). Gastroenterol 138(Supplement 1):S115–S116
Sattayasai J, Srisuwan S, Arkaravichien T, Aromdee C (2010) Effects of andrographolide on sexual functions, vascular reactivity and serum testosterone level in rodents. Food ChemToxicol 48:1934–1938
Saxena RC, Singh R, Kumar P, Yadav SC, Negi MPS, Saxena VS, Joshua AJ, Vijayabalaji V, Goudar KS, Venkateshwarlu K, Amit A (2010) A randomized double blind placebo controlled clinical evaluation of extract of Andrographis paniculata (KalmCold™) in patients with uncomplicated upper respiratory tract infection. Phytomedicine 17:178–185
Seema N, Mohod NB, Wankhade SG, Paturde JT (2003) Effects of planting and harvesting dates on yield and quality of Kalmegh (Andrographis paniculata). J Med Aroma Plant Sci 25:981–983
Seth SK, Banerjee S, Kar T (2010) Crystal structure and DFT calculations of andrographiside. J Mol Struc 965:45–49
Sharma S, Rathi N, Kamal N, Pundir D, Kaur B, Arya A (2010) Conservation of biodiversity of highly important medicinal plants of India through tissue culture technology- a review. Agric Biol J North Am 1:827–833
Sheeja K, Kuttan G (2006) Protective effect of Andrographis paniculata and andrographolide on cyclophosphamide-induced urothelial toxicity. Integr Canc Therap 5:244–251
Sheeja K, Kuttan G (2007a) Activation of cytotoxic T lymphocyte responses and attenuation of tumor growth in vivo by Andrographis paniculata extract and andrographolide. Immunopharmacol Immunotoxicol 29:81–93
Sheeja K, Kuttan G (2007b) Modulation of natural killer cell activity, antibody-dependent cellular cytotoxicity, and antibody-dependent complement-mediated cytotoxicity by andrographolide in normal and Ehrlich ascites carcinoma-bearing mice. Integ Canc Therap 6:66–73
Sheeja K, Guruvayoorappan C, Kuttan G (2007) Anti-angiogenic activity of Andrographis paniculata extract and andrographolide. Int Immunopharmacol 7:211–221
Shen YC, Chen CF, Chiou WF (2002) Andrographolide prevents oxygen radical production by human neutrophils: possible mechanism(s) involved in its anti-inflammatory effect. Brit J Pharmacol 135:399–406
Shen YS, Li RT, Xiao WL, Xu G, Lin ZW, Zhao QS, Sun HD (2006) ent-Labdane diterpenoids from Andrographis paniculata. J Nat Prod 69:319–322
Shi MD, Lin HH, Lee YC, Chao JK, Lin RA, Chen JH (2008) Inhibition of cell-cycle progression in human colorectal carcinoma Lovo cells by andrographolide. Chem Biol Interac 174:201–210
Shi MD, Lin HH, Chiang TA, Tsai LY, Tsai SM, Lee YC, Chen JH (2009) Andrographolide could inhibit human colorectal carcinoma Lovo cells migration and invasion via down-regulation of MMP-7 expression. Chem Biol Interac 180:344–352
Singh RP, Banerjee S, Rao AR (2001) Modulatory influence of Andrographis paniculata on mouse hepatic and extrahepatic carcinogen metabolizing enzymes and antioxidant status. Phytotherap Res 15:382–390
Singh S, Mehta A, John J, Mehta P (2010) Anthelmintic potential of Andrographis paniculata, Cajanus cajan and Silybum marianum. Pharm J 2:5–9
Singha PK, Roy S, Dey S (2007) Protective activity of andrographolide and arabinogalactan proteins from Andrographis paniculata Nees. against ethanol-induced toxicity in mice. J Ethnopharmacol 111:13–21
Sinha J, Mukhopadhyay S, Das N, Basu MK (2000) Targeting of liposomal andrographolide to Leishmania donovani-infected macrophages in vivo. Drug Delivery 7:209–213
Smith SE, Smith FA, Jakobsen I (2003) Mycorrhizal fungi can dominate phosphate supply to plants irrespective of growth responses. Plant Physiol 133:16–20
Smith PL, Maloney KN, Pothen RG, Clardy J, Clapham DE (2006) Bisandrographolide from Andrographis paniculata activates TRPV-4 channels. J Biol Chem 281:29897–29904
Song KM, Park SW, Hong WH, Lee H, Kwak SS, Liu JR (1992) Isolation of vindoline from Catharanthus roseus by supercritical fluid extraction. Biotech Prog 8:583–586
Spasov AA, Ostrovskij OV, Chernikov MV, Wikman G (2004) Comparative controlled study of Andrographis paniculata fixed combination, Kan Jang® and an Echinacea preparation as adjuvant, in the treatment of uncomplicated respiratory disease in children. Phytother Res 18:47–53
Srivastava S, Mishra N (2009) Genetic markers-A cutting-edge technology in herbal drug research. J Chem Pharm Res 1:1–18
Srivastava A, Misra H, Verma RK, Gupta MM (2004) Chemical fingerprinting of Andrographis paniculata using HPLC, HPTLC and densitometry. Phytochem Anal 15:280–285
Suo XB, Zhang H, Wang YQ (2007) HPLC determination of andrographolide in rat whole blood: study on the pharmacokinetics of andrographolide incorporated in liposomes and tablets. Biomed Chromatogr 21:730–734
Tan ML, Kuroyanagi M, Sulaiman SF, Najimudin N, Muhammad TTS (2005) Cytotoxic activities of major diterpenoid constituents of Andrographis paniculata in a panel of human tumor cell lines. Pharm Biol 43:501–508
Thamlikitkul V, Dechatiwongse T, Theerapong S, Chantrakul C, Boonroj P, Punkrut W, Ekpalakorn W, Boontaeng N, Taechaiya S, Petcharoen S (1991) Efficacy of Andrographis paniculata (Nees) for pharyngotonsillitis in adults. J Med Assoc Thai 74:437–442
Thisoda P, Rangkadilok N, Pholphana N, Worasuttayangkurn L, Ruchirawat S, Satayavivad J (2006) Inhibitory effect of Andrographis paniculata extract and its active diterpenoids on platelet aggregation. Eur J Pharmacol 553:39–45
Tonthubthimthong P, Chuaprasert S, Douglas P, Luewisutthichat W (2001) Supercritical CO2 extraction of nimbin from neem seeds-an experimental study. J Food Eng 47:289–293
Turk MA, Assaf TA, Hameed KM, Tawaha ALA (2006) Significance of Mycorrhizae. World J Agric Sci 2:16–20
Vasanthakrishna M, Bagyaraj DJ, Nirmalnath JP (1995) Selection of efficient VA mycorrhizal fungi for Casuarina equisetifolia- second screening. New Forests 9:157–162
Verma N, Vinayak M (2008) Antioxidant action of Andrographis paniculata on lymphoma. Mol Biol Rep 35:535–540
Wang T, Liu B, Zhang W, Wilson B, Hong JS (2004) Andrographolide reduces inflammation-mediated dopaminergic neurodegeneration in mesencephalic neuron glia cultures by inhibiting microglial activation. J Pharmacol ExpTherap 308:975–983
Wang GC, Wang Y, Williams ID, Sung HHY, Zhang XQ, Zhang DM, Jiang RW, Yao XS, Ye WC (2009) Andrographolactone, a unique diterpene from Andrographis paniculata. Tetrahed Lett 50:4824–4826
Wang W, Wang J, Dong SF, Liu CH, Italiani P, Sun SH, Xu J, Boraschi D, Ma SP, Qu D (2010) Immunomodulatory activity of andrographolide on macrophage activation and specific antibody response. Acta Pharmacol Sin 31:191–201
Wiart C, Kumar K, Yusof MY, Hamimah H, Fauzi ZM, Sulaiman M (2005) Antiviral properties of ent-labdene diterpenes of Andrographis paniculata nees, inhibitors of herpes simplex virus type 1. Phytother Res 19:1069–1070
Wongkittipong R, Prat L, Damronglerd S, Gourdon C (2004) Solid-liquid extraction of andrographolide from plants-experimental study, kinetic reaction and model. Sep Purifi Technol 40:147–154
Woo AY, Waye MM, Tsui SK, Yeung ST, Cheng CH (2008) Andrographolide up-regulates cellular-reduced glutathione level and protects cardiomyocytes against hypoxia/reoxygenation. J Pharmacol Exp Therap 325:226–235
Wu TS, Chern HJ, Damu AG, Kuo PC, Su C, Lee EJ, Teng CM (2008) Flavonoids and ent-labdane diterpenoids from Andrographis paniculata and their antiplatelet aggregatory and vasorelaxing effects. J Asian Nat Prod Res 10:17–24
Xia YF, Ye BQ, Li YD, Wang JG, He XJ, Lin XF, Yao X, Ma D, Slungaard A, Hebbel RP, Key NS, Geng JG (2004) Andrographolide attenuates inflammation by inhibition of NF- κB activation through covalent modification of reduced cysteine 62 of p50. J Immunol 173:4207–4217
Xu Y, Chen A, Fry S, Barrow RA, Marshall RL, Mukkur TKS (2007) Modulation of immune response in mice immunised with an inactivated Salmonella vaccine and gavaged with Andrographis paniculata extract or andrographolide. Int Immunopharmacol 7:515–523
Xu C, Chou GX, Wang ZT (2010) A new diterpene from the leaves of Andrographis paniculata Nees. Fitoterapia 81:610–613
Yang S, Evans AM, Prachand S, Singh AT, Bhalla S, David K, Gordon LI (2010) Mitochondrial-mediated apoptosis in lymphoma cells by the diterpenoid lactone Andrographolide, the active component of Andrographis paniculata. Clin Canc Res 16:4755–4768
Yoopan N, Thisoda P, Rangkadilok N, Sahasitiwat S, Pholphana N, Ruchirawat S, Satayavivad J (2007) Cardiovascular effects of 14-deoxy-11, 12-didehydroandrographolide and Andrographis paniculata extracts. Planta Med 73:503–511
Yu ZH, Mackill DJ, Bonman JM, Tanksley SD (1991) Tagging genes for blast resistance in rice via linkage to RFLP markers. Theor Appl Genet 81:471–476
Yu BC, Hung CR, Chen WC, Cheng JT (2003) Anti-hyperglycaemic effect of andrographolide in streptozotocin-induced diabetic rats. Planta Med 69:1075–1079
Yu BC, Chang CK, Su CF, Cheng JT (2008) Mediation of beta-endorphin in andrographolide-induced plasma glucose-lowering action in type I diabetes-like animals. Naunyn Schmiedebergs Arch Pharmacol 377:529–540
Zakrzewski PA (2002) Bioprospecting or biopiracy? The pharmaceutical industry’s use of indigenous medicinal plants as a source of potential drug candidates. Univ Toronto Med J 9:252–254
Zhang CY, Tan BKH (1996) Hypotensive activity of aqueous extract Andrographis paniculata in rats. Clin Exp Pharmacol Physiol 23:675–678
Zhang CY, Tan BK (1998) Vasorelaxation of rat thoracic aorta caused by 14-deoxyandrographolide. Clin Exp Pharmacol Physiol 25:424–429
Zhang CY, Tan BK (1999) Effects of 14-deoxyandrographolide and 14-deoxy-11, 12-didehydroandrographolide on nitric oxide production in cultured human endothelial cells. Phytother Res 13:157–159
Zhang XF, Tan BK (2000) Anti-hyperglycaemic and anti-oxidant properties of Andrographis paniculata in normal and diabetic rats. Clin Exp Physiol Pharmacol 27:358–363
Zhang C, Kuroyangi M, Tan BK (1998) Cardiovascular activity of 14-deoxy-11, 12-didehydroandrographolide in the anaesthetised rat and isolated right atria. Pharmacol Res 38:413–417
Zhang XQ, Wang GC, Ye WC, Li Q, Zhou GX, Yao XS (2006) New diterpenoids from Andrographis paniculata. J Integ Plant Biol 48:1122–1125
Zhao J, Yang G, Liu H, Wang D, Song X, Chen Y (2002) Determination of andrographolide, deoxyandrographolide and neoandrographolide in the Chinese Herb Andrographis paniculata by Micellar Electrokinetic Capillary Chromatography. Phytochem Anal 13:222–227
Zhou J, Zhang S, Ong CN, Shen HM (2006) Critical role of pro-apoptotic Bcl-2 family members in andrographolide-induced apoptosis in human cancer cells. Biochem Pharmacol 72:132–144
Zhou J, Lu GD, Ong CS, Ong CN, Shen HM (2008a) Andrographolide sensitizes cancer cells to TRAIL-induced apoptosis via p53-mediated death receptor 4 up-regulation. Mol Canc Therap 7:2170–2180
Zhou KL, Chen LX, Zhuang YL, Wang NL, Yao XS, Qiu F (2008b) Two new ent-labdane diterpenoid glycosides from the aerial parts of Andrographis paniculata. J Asian Nat Prod Res 10:939–943
Zhou J, Ong CN, Hur GM, Shen HM (2010) Inhibition of the JAK-STAT3 pathway by andrographolide enhances chemosensitivity of cancer cells to doxorubicin. Biochem Pharmacol 79:1242–1250
Zou QY, Li N, Dan C, Deng WL, Peng SL, Ding LS (2010) A new ent-labdane diterpenoid from Andrographis paniculata. Chin Chem Lett 21:1091–1093
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Subramanian, R., Zaini Asmawi, M. & Sadikun, A. A bitter plant with a sweet future? A comprehensive review of an oriental medicinal plant: Andrographis paniculata . Phytochem Rev 11, 39–75 (2012). https://doi.org/10.1007/s11101-011-9219-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11101-011-9219-z