No Heading
Purpose.
To assess the effects of polysorbates 80 and 60 on intestinal lipoprotein processing in vitro, using Caco-2 cells, and to compare the results with those obtained using an in vivo intestinal lymphatic cannulated rat model.
Methods.
Caco-2 monolayers were used to monitor changes in lipoprotein secretion following exposure to excipients. In vivo data was obtained by monitoring intestinal lymphatic triglyceride levels following intraduodenal administration of the excipient to an anesthetised mesenteric lymph cannulated rat.
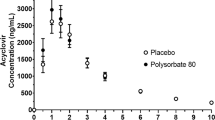
Results.
Caco-2 cells digested the polysorbate 80 to liberate oleic acid, which was used by the cells to enhance basolateral secretion of triglyceride-rich lipoproteins including chylomicrons. This response was not seen with polysorbate 60. Polysorbate 80 elicited a similar response in vivo in the rat model, stimulating enhanced triglyceride secretion in mesenteric lymph. Inhibition of lipoprotein secretion by Cremophor EL in Caco-2 cells was reversed by co-administration with polysorbate 80.
Conclusions.
Polysorbate 80 promoted chylomicron secretion in Caco-2 cells and counteracted the inhibitory effects of other surfactants. These properties, in tandem with its P-gp inhibitory activity, make polysorbate 80 an ideal excipient for lymphotrophic vehicles. The ability to predict the in vivo response to Polysorbate 80 implies that the Caco-2 model is useful for studying absorption mechanisms from oral lipid-based formulations.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
1. P. P. Constantinides. Lipid microemulsions for improving drug dissolution and oral absorption: physical and biopharmaceutical aspects. Pharm. Res. 12:1561–1572 (1995).
2. E. V. Batrakova, H. Y. Han, V. Y. Alakhov, D. W. Miller, and A. V. Kabanov. Effects of pluronic block copolymers on drug absorption in Caco-2 cell monolayers. Pharm. Res. 15:850–855 (1998).
3. D. M. Woodcock, M. E. Linsenmeyer, G. Chojnowski, A. B. Kriegler, V. Nink, and L. K. Webster. Reversal of multidrug resistance by surfactants. Br. J. Cancer 66:62–68 (1992).
4. L. Yu, A. Bridgers, J. Polli, A. Vickers, S. Long, A. Roy, R. Winnike, and M. Coffin. Vitamin E-TPGS increases absorption flux of an HIV protease inhibitor by enhancing its solubility and permeability. Pharm. Res. 16:1812–1817 (1999).
5. M. M. Nerurkar, P. S. Burton, and R. T. Borchardt. The use of surfactants to enhance the permeability of peptides through Caco-2 cells by inhibition of an apically polarized efflux system. Pharm. Res. 13:528–534 (1996).
6. P. Tso and S. R. Gollamudi. Pluronic L81: a potent inhibitor of intestinal chylomicrons. Am. J. Physiol. 247:G32–G36 (1984).
7. J. Luchoomun and M. M. Hussain. Assembly and secretion of chylomicrons by differentiated Caco-2 cells. Nascent triglycerides and preformed phospholipids are preferentially used for lipoprotein assembly. J. Biol. Chem. 274:19565–19572 (1999).
8. F. Seeballuck, M. B. Ashford, and C. M. O’Driscoll. The effects of pluronic block copolymers and cremophor EL on intestinal lipoprotein processing and the potential link with p-glycoprotein in Caco-2 cells. Pharm. Res. 20:1085–1092 (2003).
9. A. Seelig. A general pattern for substrate recognition by P-glycoprotein. Eur. J. Biochem. 251:252–261 (1998).
10. R. E. Counsell and R. C. Pohland. Lipoproteins as potential site-specific delivery systems for diagnostics and therapeutic agents. J. Med. Chem. 25:1115–1120 (1982).
11. C. M. O’Driscoll. Lipid-based formulations for intestinal lymphatic delivery. Eur. J. Pharm. Sci. 15:405–415 (2002).
12. M. M. van Greevenbroek, G. van Meer, D. W. Erkelens, and T. W. de Bruin. Effects of saturated, mono-, and polyunsaturated fatty acids on the secretion of apo B containing lipoproteins by Caco-2 cells. Atherosclerosis 121:139–150 (1996).
13. S. Saarinen-Savolainen, T. Järvinen, K. Araki-Sasaki, H. Watanabe, and A. Urtii. Evaluation of Cytotoxicity of various ophthalmic drugs, eye drop excipients and cyclodextrins in an immortalized human corneal epithelial cell line. Pharm. Res. 15:1275–1280 (1998).
14. M. Mehran, E. Levy, M. Bendayan, and E. Seidman. Lipid, apolipoprotein, and lipoprotein synthesis and secretion during cellular differentiation in Caco-2 cells. In Vitro Cell. Dev. Biol. 33:118–128 (1997).
15. J. Folch, M. Lees, and G. H. S. Stanley. A simple method for the isolation and purification of total lipids from animal tissues. J. Biol. Chem. 226:497–509 (1957).
16. A. Bakillah, Z. Zhou, J. Luchoomun, and M. M. Hussain. Measurement of apolipoprotein B in various cell lines: correlation between intracellular levels and rates of secretion. Lipids 32:1113–1118 (1997).
17. S. Hamilton, R. J. Hamilton, and P. A. Sewell. Extraction of lipids and derivative formation. In R. J. Hamilton and S. Hamilton (eds), Lipid Analysis: A Practical Approach, Oxford University Press, New York, 1992, pp. 13–64.
18. H. Roche. Postprandial lipaemia and haemostasis. PhD. thesis submitted to Trinity College, Dublin (1995).
19. C. M. O’Driscoll, R. A. Myers, and V. J. Stella. Blood and lymph transport of DDT after oral and parenteral administration to anaesthetised rats. Int. J. Pharm. 73:177–183 (1991).
20. M. J. Lawrence. Polyoxyethylene sorbitan fatty acid esters. In R. C. Rowe, P. J. Sheskey, and P. J. Weller (eds), Handbook of Pharmaceutical Excipients, American Pharmaceutical Association, Washington DC, 2003, pp. 479–483.
21. K. Kawakami, T. Yoshikawa, Y. Moroto, E. Kanaoka, K. Takahashi, Y. Nishihara, and K. Masuda. Microemulsion formulation for enhanced absorption of poorly soluble drugs I. Prescription design. J. Control. Rel. 81:65–74 (2002).
22. H. Saito, H. Tomioka, T. Watanabe, and T. Yoneyama. Mycobacteriocins produced by rapidly growing mycobacteria are Tween-hydrolyzing esterases. J. Bacteriol. 153:1294–1300 (1983).
23. V. Fernandez-Lopez, G. Serrero, R. Negrel, and G. Ailhaud. Esterolytic activities of rat intestinal mucosal. Eur. J. Biochem. 71:259–270 (1976).
24. A. D. Tipton, S. Frase, and C. M. Mansbach II. Isolation and characterization of a mucosal triacylglycerol pool undergoing hydrolysis. Am. J. Physiol. 20:G871–G878 (1989).
25. R. H. Rao and C. M. Mansbach. Alkaline lipase in rat intestinal mucosa: physiological parameters. Arch. Biochem. Biophys. 304:483–489 (1993).
26. J. T. Mahan, G. D. Heda, and R. H. Rao. and C. M. 2. Mansbach. The intestine expresses pancreatic triacylglycerol lipase: regulation by dietary lipid. Am. J. Physiol. 280:G1187–G1196 (2001).
27. R. Negrel, G. Serrero, G. Ailhaud, and J.-F. Rey. and J. Delmont. Glycerol-ester hydrolase from human small intestine and its possible role in the degradation of neutral lipids. Digestion 16:146–159 (1977).
28. C. M. Meaney and C. M. O’Driscoll. A comparison of the permeation enhancement potential of simple bile salt and mixed bile salt: fatty acid micellar systems using the Caco-2 cell culture model. Int. J. Pharm. 207:21–30 (2000).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Seeballuck, F., Lawless, E., Ashford, M. et al. Stimulation of Triglyceride-Rich Lipoprotein Secretion by Polysorbate 80: In Vitro and in Vivo Correlation Using Caco-2 Cells and a Cannulated Rat Intestinal Lymphatic Model. Pharm Res 21, 2320–2326 (2004). https://doi.org/10.1007/s11095-004-7684-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-004-7684-4