Abstract
We aimed to observe the therapeutic effects of lithium on inhalational anesthetic sevoflurane-induced apoptosis in immature brain hippocampus. From postnatal day 5 (P5) to P28, male Sprague–Dawley pups were intraperitoneally injected with lithium chloride or 0.9 % sodium chloride. On P7 after the injection, pups were exposed to 2.3 % sevoflurane or air for 6 h. Brain tissues were harvested 12 h and 3 weeks after exposure. Cleaved caspase-3, nNOS protein, GSK-3β,p-GSK-3β were assessed by Western blot, and histopathological changes were assessed using Nissl stain and TUNEL stain. From P28, we used the eight-arm radial maze test and step-through test to evaluate the influence of sevoflurane exposure on the learning and memory of juvenile rats. The results showed that neonatal sevoflurane exposure induced caspase-3 activation and histopathological changes in hippocampus can be attenuated by lithium chloride. Sevoflurane increased GSK-3β activity while pretreatment of lithium decreased GSK-3β activity. Moreover, sevoflurane showed possibly slight but temporal influence on the spatial learning and the memory of juvenile rats, and chronic use of lithium chloride might have the therapeutic effect. Our current study suggests that lithium attenuates sevoflurane induced neonatal hippocampual damage by GSK-3β pathway and might improve learning and memory deficits in rats after neonatal exposure.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sevoflurane is a widely used inhalational anesthetic agent in pediatric anesthesia [1]. However, growing evidence has indicated that neonatal exposure to sevoflurane can lead to neurotoxicity and learning deficits [2–5]. It has been proved that neuronal apoptosis and necrosis are greatly involved in anesthetic-induced developing brain neurotoxicity [6]. Our previous work reported that single exposure to sevoflurane causes neuronal apoptosis with pathological changes in the brain hippocampus of infant rats, and the level of neuronal nitric oxide synthase (nNOS) protein simultaneously decreased within 24 h after sevoflurane exposure in a time-dependent manner [7]. But so far, little research has been done to investigate the prevention and treatment of the lesion caused by sevoflurane exposure.
Lithium, a well-known mood-stabilizing medicine, has been used in patients with manic-depressive illness for a long time. Recent studies reported the neuroprotective effect of lithium in preventing apoptosis-dependent cellular death [8, 9]. A previous study performed by Zhong et al. [10] showed that short-term lithium therapy can prevent ethanol-induced developing brain apoptosis. Subsequently, Straiko et al. [11] proved lithium’s neuroprotective effect against ketamine and propofol anesthesia induced infant mice brain apoptosis. Lithium’s anti-neuroapoptotic mechanism involved inactivation of N-methyl-D- aspartate (NMDA) receptors through different mechanisms including the induction of neurotrophic/neuroprotective proteins [12]. Among them, Glycogen synthase kinase-3β (GSK-3β) is a pro-apoptotic protein that serves as an intersection point for apoptotic pathways and plays a crucial role in lithium’s neuroprotective capabilities [13]. GSK-3β has the potential to be phosphorylated at Ser9. Phosphorylation of Ser9 induces the inactivation of GSK-3β and plays a critical role in the neuroprotection [14].
Chronic lithium treatment has been used in other in vivo studies. Zhu et al. reported that the long-term usage of lithium might be a helpful therapeutic strategy for brain injury with multiple beneficial effects [15]. Another research indicated that chronic lithium administration, at therapeutic doses, has a neuroprotective properties [16]. So, we have undertaken this study to evaulate whether sevoflurane supress nNOS and/or GSK-3β phosphorylation, and whether short-term lithium administration counteracts this action. More importantly, we test for the first time whether chronic lithium, at therapeutically concentrations, has a protective effect and beneficial cognitive outcome against anesthetic exposure in the juvenile rats’ brain.
In this study, we found out lithium would play a positive role in preventing and treating neurotoxicity caused by sevoflurane in developing brains and its possible mechanism involved nNOS and GSK-3β phosphorylation. In order to do that, we assessed the changes in the protein expression of cleaved caspase-3, nNOS and GSK-3β following the short-term and long-term lithium administration after sevoflurane exposure as well as the histological and behavioral effects of lithium in young rodent models of sevoflurane-induced neuropathology. Our current study emphasized that neonatal sevoflurane exposure can induce histopathological damages in hippocampus and show its possibly slight but temporal influence on the spatial learning and the memory of juvenile rats. And the use of lithium chloride effectively inhibited apoptosis and improved the histopathological changes caused by sevoflurane.
Materials and Methods
Animals and Treatment
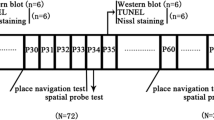
Male Sprague–Dawley (SD) rats were obtained from the Experimental Animal Center of Sun Yat-sen University. All animal studies were approved by the Institutional Animal Care and Use Committee at Sun Yat-sen University (Guangzhou, Guangdong, China; No. [2014]A-073) and kept in a temperature- and light-controlled environment with a 12 h light–dark cycle. On P5, the SD pups were randomly divided into the air-treated control group (Control group, n = 33 rats), the single lithium group (Lithium group, n = 33 rats), the Sevoflurane group (Sevoflurane group, n = 33 rats) and a combination of sevoflurane and lithium-treated group (Sevo + Li group, n = 33 rats).
We injected i.p. 3 % LiCl (Sigma-Aldrich, St. Louis, MO, USA), dissolved in normal saline, into pups in the Lithium group and the Sevo + Li group daily, starting from P5 to P28. On P5, LiCl was injected at a dose of 2 mmol.kg−1 followed by 1 mmol.kg−1 at 24 h intervals. The dose and the time of LiCl were selected based on published data [8] as well as our pilot experiments. The Control and the Sevoflurane group received the same amount volume of NaCl.
On P7 after the injection, the rats in the Sevoflurane group and Sevo + Li group were placed in a plastic container and exposed continuously to 2.3 % sevoflurane for 6 h. The pups in the control group and the Lithium group were placed in the same chamber and treated in the same fashion as those in the Sevoflurane group, except that they were exposed only to air for 6 h. Sevoflurane vaporized using air as a carrier; the total gas flow was 2l min−1. During exposure, the temperature inside the container was maintained at 30 °C by using a heating device (NPS-A3 heated device, Midea Co., Guangdong, China) outside and a hot-water bag on the bottom of the container with a constant temperature maintained between 30 and 35 °C. A gas monitor (Detex-Ohmeda, Louisville, CO, USA) was used to regulate the sevoflurane, oxygen and carbon dioxide levels in the chamber. An investigator monitored the rats’ respiratory frequency and skin colors every 5 min to detect for apnea or hypoxia. Once these symptoms were detected, the rats were exposed to air immediately and excluded from the experiment. After a 6 h exposure, sevoflurane was ceased, and the rats were exposed only to air. They returned to the mother’s cage when the pups had awakened.
Arterial Blood Gas Analysis
We performed arterial blood analysis in order to exclude other factors that might contribute to apoptosis [2, 17]. The arterial blood samples from both the sevoflurane-treated and air-treated groups were obtained from the left cardiac ventricle immediately after removal from the maternal cage (0 h, n = 3 in each subgroup) or at the end of anesthesia (6 h, n = 3 in each subgroup). They were analyzed immediately after collection using a blood gas analyzer (Gem premier 3000, US). We analyzed the pH, arterial carbon dioxide tension, arterial oxygen tension, and blood glucose levels of the arterial blood samples. The analysis of each sample was repeated at least three times.
Western Blot Analysis
Western blots were performed as previously described [7]. The samples were extracted from the hippocampus. The rats were sacrificed separately by decapitation 12 h after the 6 h exposure and on P28, with 3 rats at each time point in each of the four groups. The dissected hippocampus was homogenized and the supernatant-containing protein was separated. The proteins were separated by 10 and 12 % gel electrophoresis and transferred to polyvinylidene fluoride membranes (Pall Co. USA). The blots were incubated with an antibody against cleaved caspase-3 (1:1000, rabbit polyclonal, Asp175, Cell Signaling Technology, Inc.), nNOS (1:500, mouse monoclonal, Santa Cruz Biotechnology), anti-phosphor-GSK-3β(Ser9) (1:1000, rabbit polyclonal; Cell Signaling Technology) or anti-GSK-3β (1:1000, rabbit polyclonal; Santa Cruz). The β-actin protein (1:2000, mouse monoclonal, Santa Cruz Biotechnology) was used as a loading control. The changes of protein levels were examined using the ECL-PLUS system and photographed. We captured the scanned images on films and transformed them to optical density, measuring and analyzing them using the Image J software (NIH, USA). GSK-3β activity was represented as the radio of GSK-3β(total) control and compared to phosphor-GSK-3β(Ser9). The other data were represented as the ratio of β-actin loading control and compared among the treatments. All of the studies were performed a minimum of three times for each hippocampus sample from each rat.
Histopathological Examination
The four groups were sacrificed for the Nissl staining(n = 3/group) and TUNEL staning(n = 3/group) 12 h after the 6 h exposure and on P28. The animals were anesthetized with a lethal dose of 10 % chloral hydrate and perfused transcardially with 0.9 % saline followed by an aldehyde fixative that lasted for 15–25 min with a concentration of 4 % paraformaldehyde in 0.1 M PB (pH 7.4). The brains were removed and kept overnight in 4 % paraformaldehyde. Paraffin blocks, which included different levels of the hippocampus along the associated areas, were prepared for Nissl staining and TUNEL staning and sliced coronally into 5 μm thickness. The paraffin sections were examined under light microscopy (Nikon ECLIPSE, 50i, Japan).
Nissl staining The paraffin sections were performed with Nissl staining to study the morphological changes of the pyramidal neurons, in part, of the CA1 and CA3 regions. The neurons in 3 sections per animal (n = 3 for each group) were counted. In Nissl stained slides, the Nissl-positive neurons were demonstrated only if the structures of the appropriate size and shape of the neurons, with Nissl-positive nucleus and cytoplasmic Nissl-positive particles. The number of the Nissl-positive neurons in the pyramidal cell layers of the bilateral CA1 and CA3 regions was counted at 400× magnification by two persons in a blinded fashion [18]. Questionable structures were examined under 1000× magnification and were not counted if the identification remained uncertain.
In situ terminal deoxynucleotidyl transferase UTP nick end labeling (TUNEL) staining TUNEL staining was performed to detect DNA fragmentation with an In Situ Cell Death Detection kit following the manufacturer’s protocol (Roche, Mannheim, Germany). Brain sections were permeabilized with proteinase K solution (20 g/mL) for 20 min, then intubated with terminal deoxynucleotidyl transferase(TdT) and dUTP in a humidified chamber at 37 °C for 2 h. The reaction was then stopped and followed by colorization with Hematoxylin Staining Solution (Beyotime, China). The apoptotic cells and dying cells were identified as those with dark brown color and small, dense nuclei. The number of apoptotic cells in the hippocampal pyramidal cells was counted under a high light microscope at 400× magnification by two persons in a blinded fashion. The TUNEL-positive cells ratio = (number of TUNEL-positive cells/total cells) × 100 %. Questionable structures were examined under 1000× magnification and were not counted if the identification remained uncertain.
Behavioral Studies
In this study, two measures were used to observe the neurological function of rats exposed to sevoflurane and the lithium treatment. Both tests started from P28.
Eight-Arm Radial Maze Test The eight-arm radial maze test was used to evaluate the working memory and reference memory of the Control group, the Sevoflurane group, the Lithium group and the Sevo + Li groupin P28–P39 rats (n = 8/group). The procedures were followed according to the method previously described [19]. The radial maze was constructed with eight arms (67 cm long, 20 cm deep, 10 cm wide), which radiated from a central octagonal area. The maze was surrounded by extra maze cues, and food cups were placed at the end of each arm, which was invisible from the central area. The rats were deprived of food but were given ad libitum access to water. At the acquisition phase, the animals were treated to eat food (20 mg) that was placed at the end of all arms at the first entry to each arm. Once the rats were habituated to their environment, the memory test began. For the 10-day test, each rat had its own specific four-baited arms placed with food such that two of four arms were adjacent, and the other arms were 90° apart from these arms (e.g., arms 1, 2, 4, and 7). Each rat was placed in the central area of the radial arm maze and was watched as it consumed its food in 10 min. The routine for each rat was recorded. Entry into an unbaited arm was scored as a reference memory error. Re-entry into a baited arm from which the food pellet had already been consumed within a trial was scored as a working memory error. The animals, which entered less than three arms or more than 20 arms in the first 2 min of the habituation phase, were considered inactive or excessively active and were excluded from the test.
Step-through test The step-through test was performed based on the protocol performed elsewhere [20]. The apparatus used was consisted of six separated rooms. Every room (15 × 10 × 11 cm) could be divided into an illuminated compartment and a dark compartment with an interconnecting semicircular door (3 cm in diameter). The copper grid floor under the dark compartment was connected to a shocker-maker which could deliver an electric current (36 V). For training trials, each animal was gently placed in the illuminated compartment with its back facing the door and allowed to enter the dark compartment freely for 3 min. When entering the dark compartment, the rat would immediately receive an electric shock to the paws and went back to the illuminated space through the door. After adaptation, formal training started and lasted for 5 min as mentioned above. The tests were repeated 24 and 48 h later. During retention trials, the number of mistakes and the latency to the initial entry into the dark compartment were recorded within 5 min, respectively. If the rat did not receive a shock within 5 min, it was assigned 300 s for the retention latency value (n = 10/each group).
Statistical Analysis
All of the data are expressed as the mean ± standard deviation(S.D.). We conducted the statistical tests using SPSS 15.0 software. We performed the arterial blood analyses using Student’s t test. The data from the Nissl staining, TUNEL staining and western blot were analyzed using one-way ANOVA and individual post hoc comparisons (Bonferroni post hoc test). We compared the data between the two groups using independent-samples t tests. We analyzed the data from the radial eight-arm maze and step-through test between groups using the repeated measures two-way ANOVA; for a detailed statistical analysis, we used the post hoc test. We analyzed the data between testing days within the group using Friedman ANOVA for repeated measurements, followed by Wilcoxon’s matched pairs test when appropriate; the data between groups on a given testing day were analyzed using the Mann–Whitney U test. p < 0.05 was considered to be statistically significant.
Results
Neonatal Sevoflurane Exposure does not Cause Respiratory or Metabolic Disorder
During the exposure to sevoflurane for 6 h on P7, there were no signs of respiratory or metabolic disorder. No significant differences were found between the air-exposed rats and the sevoflurane-exposed rats in the pH, PaCO2, PaO2 and arterial blood levels before and after exposure (Table 1). There was no animal excluded from the experiment during the exposure period.
Pretreatment of Lithium Attenuated the Activity of Cleaved Caspase-3 Caused by Sevoflurane Exoposure in Neonatal Rat Hippocampus
On P7 (12 h after exposure), there was no significant difference between the Control group and the Lithium group with respect to the level of the cleaved caspase-3 protein (p > 0.05; Fig. 1a). The expression level in Sevoflurane group at the same time point was significantly higher than the Control group (p < 0.05; Fig. 1a). In the Sevo + Li group, LiCl showed its preventive effect. The expression level of the cleaved caspase-3 protein in the Sevo + Li group was significantly lower than in the Sevoflurane group (p < 0.05; Fig. 1a), although it was still higher compared with the Control group (p < 0.05; Fig. 1a). On P28, the expression of the cleaved caspase-3 showed no significant difference among the four groups (p > 0.05; Fig. 1d).
Assessment of the expression level of cleaved caspase-3 protein and nNOS protein and the activity of GSK-3β after sevoflurane exposure and lithium intervention in neonatal rat hippocampus. The samples were obtained from the hippocampus of rats subjected to the Control group (control, lane 1 in a–f), the Lithium group (Lithium, lane 2 in a–f), the Sevoflurane group (Sevoflurane, lane 3 in a–f) and the Sevo + Li group (Sevo + Li, lane 4 in a–f) on P7 (a–c) and P28 (d–f) (n = 3 per each group). a, d: the expression changes of cleaved caspase-3 on P7 after exposure (a) and P28 (d). b, e: the activity change of GSK-3β on P7 (b) after exposure and P28(e). c, f: the expression changes of nNOS protein on P7 after sevoflurane and P28. a–f: # p < 0.05 versus the Control group; *p < 0.05 versus the Sevoflurane group
The Effect of Lithium on the Expression of p-GSK-3β-Ser 9 and GSK-3β(total)
As the data shows, on P7, p-GSK-3β-Ser 9/GSK-3β(total) in Sevoflurane group was significantly lower than the Control group and the Lithium group (p < 0.05; Fig. 1b), but p-GSK-3β-Ser 9/GSK-3β (total) in Sevo + Li group was significantly higher than the Sevofluanre group (p < 0.05; Fig. 1b). It means sevoflurane increased the GSK-3β activity and short-term lithium decreased the GSK-3β activity.
On P28, chronic lithium treatment significantly increased the p-GSK-3β-Ser 9/GSK-3β (total) in Lithium group and Sevo + Li group (p < 0.05; Fig. 1e). Lithium consistently decreased the GSK-3β activity (Fig. 1e). But there is no difference in p-GSK-3β-Ser 9/GSK-3β (total) between the Control group and Sevoflurane group (p > 0.05; Fig. 1e).
Chronic Lithium Treatment Upgrades the Expression Level of the nNOS Protein Gradually
We assessed simultaneously the expression level of nNOS protein. The results demonstrated that there were no significant differences between the Control group and the Lithium group on the level of the nNOS protein on P7 and P28 (p > 0.05; Fig. 1c, f). At each time point, the nNOS protein in the Sevoflurane group was significantly lower than the Control group or the Lithium group (p < 0.05; Fig. 1 c, f). On P7, the nNOS protein expression level in the hippocampus of the Sevo + Li group was the same as that expressed in the Sevoflurane group (p > 0.05), but was significantly lower than the Control group or the Lithium group (p < 0.05; Fig. 1c). Interestingly, on P28, the nNOS protein expressed in the Sevo + Li group was higher than the Sevoflurane group (p < 0.05) and there was no significant difference between the Sevo + Li group and the Control group (p > 0.05; Fig. 1f).
Lithium Ameliorates Histopathological Changes Caused by Sevoflurane in Neonatal Rat Hippocampus
On P7 (12 h after exposure), the hippocampal neurons in the Control group and the Lithium group were ranked in neat order (Fig. 2a–h). In the CA1 subfield of the Sevoflurane group, the neurons were ranked disorganized with fewer layers (Fig. 2i–j). In the CA3 subfield of the Sevoflurane group, there were morphology changes and disorganized of the neurons (Fig. 2k–l). In the Sevo + Li group, the loss of neurons and the neuronal morphology changes were not so obvious (Fig. 2m–p). At the same time point, the number of the neurons in CA1 and CA3 regions was analyzed. We found a significant decrease in the density of healthy pyramidal neurons in both CA1 and CA3 subfields in Sevoflurane group compared with the Control group and the Lithium group (Fig. 2q). In the Sevo + Li group, the number of the neurons in CA1 or CA3 regions was not so much decreased as the Sevoflurane group (Fig. 2q), indicating that lithium pre-treatment for 3 days ameliorated histopathological changes caused by a 6 h sevoflurane exposure.
Lithium ameliorated the histopathological changes caused by sevoflurane in the neonatal rat hippocampus on P7. Representative photomicrographs of the coronal sections of the hippocampus of P7 rats, showing Nissl-stained normal pyramidal neurons. a–d: In the CA1 (a–b) and CA3 (c–d) regions at 12 h after the 6 h air treatment (open bar). e–h: In the CA1 (e–f) and CA3 (g–h) regions of the lithium treatment group (striped bar). i–l: In the CA1 (i–j) and CA3 (k–l) regions at 12 h after the 6 h sevoflurane treatment (closed bar). m–p: In the CA1 (m–n) and CA3 (o–p) regions of the Sevo + Li group (dotted bar). (Magnification, 100×, Scale bar, 50 μm in a, c, e, g; magnification, 400×, Scale bar, 200 μm in b, d, f, h) i: Quantification of the Nissl-positive pyramidal neuron numbers in CA1 and CA3 regions of the hippocampus. Each value represents the mean ± SD (n = 3 rats per group). # p < 0.05 versus the Control group; *p < 0.05 versus the Sevoflurane group
On P28, in the Control group and the Lithium group, the neurons were ranked in good order (Fig. 3a–h). However, in the Sevoflurane group, the cells were not ranked so closely with fewer layers (Fig. 3i–l). Especially in the CA3 region, there were typical neuropathological changes of neurons, including irregular neuronal morphology changes and cytoplasmic Nissl body losses (Fig. 3k–l). Our statistical analysis showed, on P28, that the numbers of neurons in the Sevoflurane group were smaller than the Control group or the Lithium group (p < 0.05; Fig. 3q). The hippocampal pyramidal neurons in the Sevo + Li group were morphologically normal (Fig. 3m–p). There was a significant difference between the Sevoflurane group and the Sevo + Li group on the number of the pyramidal neurons on P28 (p < 0.05; Fig. 3q).
Lithium ameliorated the histopathological changes caused by sevoflurane in the neonatal rat hippocampus on P28. Representative photomicrographs of the coronal sections of the hippocampus of P7 rats, showing the Nissl-stained normal pyramidal neurons. a–d: In the CA1 (a–b) and CA3 (c–d) regions on P28 of the Control group (open bar). e–h: In the CA1 (e–f) and CA3 (g–h) regions on P28 of the lithium treatment group (striped bar). i–l: In the CA1 (i–j) and CA3 (k–l) regions on P28 of the Sevoflurane group (closed bar). m–p: In the CA1 (m–n) and CA3 (o–p) regions on P28 of the Sevo + Li group (dotted bar). (Magnification, 100×, Scale bar, 50 μm in a, c, e, g; magnification, 400×, Scale bar, 200 μm in b, d, f, h) i: Quantification of the Nissl-positive pyramidal neuron numbers in CA1 and CA3 regions of the hippocampus. Each value represents the mean ± SD (n = 3 rats per group). # p < 0.05 versus the Control group; *p < 0.05 versus the Sevoflurane group
To further evaluate the morphological changes and prove neuronal death, we performed TUNEL staing on P7 (Fig. 4a–d) and P28 (Fig. 4e–h). On P7, TUNEL-positive cells were detected in the Control group and Lithium group in small number. However, increases in the percentages of TUNEL positive cells were observed in the sevoflurane groups as compared with the Control group and Lithium group (p < 0.05; Fig. 4i). Pretreatment of lithium could obviously decrease the percentages of TUNEL positive cells as showed in Sevo + Li group compared with Sevoflurane group (p < 0.05; Fig. 4i). On P28, no significant differences in the percentages of TUNEL positive cells were observed among the four groups (p > 0.05; Fig. 4i).
TUNEL stain performed on the hippocampal pyramidal neurons on P7 (Fig. 4 a–d) and P28 (Fig. 4 e–h). a, e: Control group (open bar), showing normal pyramidal neurons; b, f: Litium-treated group (striped bar); c, g: Sevoflurane group (closed bar), showing apoptotic cells (dark cells). d, h: Sevo + Li group (dotted bar), lithium treatment protected neurons against apoptosis induced by sevoflurane. (Magnification, 400×, Scale bar, 200 μm in a–h) i: percentages of TUNEL positive cells in hippocampus on P7 and P28. Each value represents the mean ± SD (n = 3 rats per group). # p < 0.05 versus the Control group; *p < 0.05 versus the Sevoflurane group
Treatment Effect of Lithium Chloride on Spatial Learning and Memory of Rats After Sevoflurane exposure
We used the radial eight-arm maze tests from P28 to P39. There was no difference in the average reference memory errors among the four groups (Fig. 5a). The average working memory errors in the Sevoflurane group were significantly higher than those in the Control group, the Lithium group, and the Sevo + Li group on the first day of formal training (Fig. 5b). However, on the following days, we observed no difference in the average working memory errors (Fig. 5b).
Treatment effect of lithium chloride on spatial learning and memory after sevoflurane exposure. Data are expressed as the mean ± SD (n = 8/each group).The average working memory errors in the Sevoflurane group was significantly higher than those in the Control group, the Lithium group, and the Sevo + Li group on the first day of formal training (a, b: Control group: diamond; lithium group: rectangle; Sevoflurane group: open triangle; Sevo + Li group: circle) *p < 0.05 versus the air control group; # p < 0.05 versus the Lithium group; ▲p < 0.05 versus the Sevo + Li group
Treatment Effect of Lithium Chloride on the Passive Avoidance Response in the Step-Through Test After Sevoflurane Exposure
Significant effect of sevoflurane on the latency to the initial entry into the dark compartment was observed on the first testing day (Fig. 6a). Exposure to sevoflurane in very young age significantly decreased the latencies during the training and retention trials, and these differences only exist shortly on the first day. Lithium chloride showed its treatment effect in this trial (p > 0.05, Fig. 6a). During the next retention trials, no significant difference between the four groups on the latency to the initial entry into the dark compartment was found (p > 0.05, Fig. 6a). No significant difference was found in the number of mistakes among the four groups (p > 0.05, Fig. 6b).
Treatment effect of lithium chloride on the the latency to the initial entry into the dark compartment (a) and the number of mistakes of passive avoidance response (b) in the step-through test after sevoflurane exposure. Data are expressed as mean ± s.e.m (n = 10/each group). Exposure to sevoflurane in very young age significantly decreased the average latencies during the training and retention trials, and these differences only exist shortly on the first day (a). No significant difference was found in the average number of mistakes among the four groups (b). *p < 0.05 versus the air control group; # p < 0.05 versus the Lithium group; ▲p < 0.05 versus the Sevo + Li group
Discussion
Recent clinical data from several retrospective cohort studies indicated that early anesthesia exposure increases the risks for later learning disabilities, atypical behavior or developmental disorder [21–24]. For the rodent brain, there is a comparable peak developmental period in the first 3 weeks after birth, during which period the brain seems to be more susceptible to neurodegenerative influences [25]. In this study we aimed to find out the long-term harmful effect caused by sevoflurane on 1 week old rats and lithium’s therapeutic action as well as benefits. We chose the following time points to observe the changes in neonatal rat hippocampus, and they were 12 h after a 6 h sevoflurane exposure and 3 weeks after sevoflurane exposure.
In the current investigation, we found out that 12 h after a 6 h clinical relevant concentration of sevoflurane exposure lead to apoptosis and caused a decreased number of neurons in the hippocampus in the developing brain. It is consist with other researches which suggested that the developing brain exposure to sevoflurane could cause neuronal apoptosis [3–5] as well as memory impairment [26]. Besides that, for the first time in this paper, we concluded that this lesion caused by sevoflurane can be prolonged for 3 weeks in histopathological level. Additionally, the nNOS protein decreased constantly for 3 weeks after sevoflurane exposure and we assumed that this long-lasting effect on nNOS protein might be caused by the death of hippocampal neuronal cell.
Growing evidence implied that lithium might be useful for treating acute brain damage and several chronic neurodegenerative diseases [8, 27]. It might because of its neuroprotective effects in preventing apoptosis-dependent cellular death [13]. Lithium can protect against neuroapoptosis caused by ethanol and anesthetic agents, such as ketamine, propofol, isoflurane and chloral hydrate in the developing brain [10, 11]. However, there are still no published data showing the neuroprotective effect of lithium in preventing neuroapoptosis caused by sevoflurane. The neuroprotective effects of lithium reported required pretreatment because the protection becomes apparent only after treatment for more than 2 ± 3 days; therefore, we gave lithium chloride 2 days before sevoflurane exposure [28]. So, we gave rats the first dose, 2 mmol.kg−1 of lithium chloride injected i.p. on P5; subsequently, we administered 1 mmol.kg−1 i.p. of lithium chloride at 24 h intervals to maintain a steady plasma concentration. We chose the dose and method of administering lithium chloride similar to the dose that Li et al. [8] had used. Li et al. [8] demonstrated that this dose can prevent neuronal apoptosis after brain hypoxia–ischemia. The current data showed that the pretreatment with lithium for 2 days could effectively down-regulate the activated caspase-3 12 h after sevoflurane exposure. Lithium could also simultaneously ameliorate the histopathological changes. We observed that 12 h after sevoflurane exposure, the number of hippocampal pyramidal neurons in the Sevo + Li group decreased not as much as the single sevoflurane inhalation group. It indicated that pretreatment of lithium exerted its neuroprotective effect against apoptosis induced by sevoflurane and reduces the number of damaged neurons. The molecular mechanisms of lithium protection involve activation of antiapoptotic cell signaling pathway and leading to the inhibition of GSK-3β [29]. To find out the possible mechanism of lithium’s neuroapoptosis effect, we assessed the expression level of GSK-3β and pGSK-3β(Ser9). In our study, we found the sevoflurane increased GSK-3β activity while lithium did decreased GSK-3β activity, which indicated that GSK-3β plays a crucial role in lithium’s neuroprotective capabilities. GSK-3β is a pro-apoptotic protein that acts as an intersection factor for apoptotic pathways and plays an important role in lithium’s neuroprotective activities. It has been reported that a wide variety of systems and kinases regulate GSK-3β activity, including protein kinase A (PKA), protein kinase C(PKC), Akt, MAP kinases and the Wnt pathway. And inhibition of GSK-3β regulates numerous pathways, proteins and transcription factors, including β-catenin, HSF-1, AP-1 and CREB [13]. In our research, pretreatment with lithium affects GSK-3β and prevents apoptosis. This action correlates with lithium’s neuroprotective effects.
We continued to give lithium chloride for 3 weeks in order to observe the effects of chronic treatment. At P28, neuronal shapes of CA3 region seem to be more damaged. This is the long-term effect caused by neonatal exposure to sevoflurane. However, on P28, sevoflurane could no longer lead to apoptosis according to the TUNEL stain as well as Western-blot analysis. The effect is an after effect, which caused the morphological changes and even functional changes. This explains why the number of neurons in sevoflurane group of P28 was decreased less than that of P7. Interestingly, we found that the numbers of neurons in the Sevo + Li group on P28 were slightly increased compared to the numbers on P7. It seems that chronic lithium usage promoted the hippocampal neurogenesis in the injured brain. Lithium reportedly has the capability to enhance the hippocampal neurogenesis [30]. The neurogenesis occurs in several brain regions where the pluripotent progenitor cells localized including dentate gyrus (DG) circuitry of hippocampus [30, 31]. Published reports have shown that the activated endogenous progenitors after brain injury might directly migrate toward the damaged areas, which lead to regeneration of the neurons in other areas of the hippocampus [31]. This information about brain injury explains why the number of pyramidal neurons in a Lithium group did not increase significantly. However, additional and further studies are needed to validate the specific reason.
On P7, sevoflurane could down-regulate the nNOS protein and induce apoptosis. At the same time point, lithium exerts a neuroprotective effect against apoptosis induced by sevoflurane without changing the nNOS protein. This action may indicate that nNOS played a role in cellular survival or death pathway after sevoflurane exposure but may not play a role in lithium-caused cellular survival pathway. Interestingly, chronic lithium treatment after sevoflurane exposure did up-regulate the expression of nNOS protein on P28. We assume that the neurogenesis caused by lithium may contribute to the up-regulated expression of the nNOS protein in developing the brain hippocampus. The nNOS protein has the function as a retrograde messenger during long-term potentiation (LTP) and is important for memory formation [32]. nNOS knock-out mice show impaired cognitive performance [32, 33].
To assess the learning and memory of juvenile rats, we used two different tests from P28. The first test was radial eight-arm maze test which can evaluate the working memory and reference memory. On the first day of the test, the rats in the Sevoflurane group performed worse than the other groups in their working memory; however, this influence was transient and disappeared on subsequent days. In the step-through test, the rats after sevoflurane exposure showed shorter step-through latency than the other three groups only on the first day. Our results are consistent with previous studies which showed that sevoflurane exposure induced only short-term dysfunctions of learning and memory [34, 35]. Additionally, lithium seems to demonstrate its treatment efficacy in this test. Considering its important role in learning and memory, the nNOS protein elevated by lithium may contribute to this effect. But we still have to notice that, due to the sample size limitaion, there were still doubts about whether they are functionally meaningful data. So, more behavior tests are needed to prove the effects of sevoflurane and lithium in the further studies.
In summary, this study demonstrated that lithium inhibited the apoptosis caused by sevoflurane 12 h after exposure, whereas chronic lithium treatment up-regulated the nNOS protein. Lithium also ameliorated the histopathological changes caused by sevoflurane in neonatal rat hippocampus. Moreover, sevoflurane caused possibly slight effects on learning and memory abilities of treated rats of juvenile age, and lithium might have some treatment effect. Additional investigations are required to determine the treatment effects of lithium after sevoflurane exposure on learning and memory abilities in early life.
References
Goa KL, Noble S, Spencer CM (1999) Sevoflurane in paediatric anaesthesia: a review. Arch Neurol 1(2):127–153
Satomoto M, Satoh Y, Terui K, Miyao H, Takishima K, Ito M, Imaki J (2009) Neonatal exposure to sevoflurane induces abnormal social behaviors and deficits in fear conditioning in mice. Anesthesiology 110(3):628–637. doi:10.1097/ALN.0b013e3181974fa2
Piehl E, Foley L, Barron M, D’Ardenne C, Guillod P, Wise-Faberowski L (2010) The effect of sevoflurane on neuronal degeneration and GABAA subunit composition in a developing rat model of organotypic hippocampal slice cultures. J Neurosurg Anesthesiol 22(3):220–229. doi:10.1097/ANA.0b013e3181e16c89
Zhang X, Xue Z, Sun A (2008) Subclinical concentration of sevoflurane potentiates neuronal apoptosis in the developing C57BL/6 mouse brain. Neurosci Lett 447(2–3):109–114. doi:10.1016/j.neulet.2008.09.083
Dong Y, Zhang G, Zhang B, Moir RD, Xia W, Marcantonio ER, Culley DJ, Crosby G, Tanzi RE, Xie Z (2009) The common inhalational anesthetic sevoflurane induces apoptosis and increases beta-amyloid protein levels. Arch Neurol 66(5):620–631. doi:10.1001/archneurol.2009.48
Sun L (2010) Early childhood general anaesthesia exposure and neurocognitive development. Br J Anaesth 105(Suppl 1):i61–i68. doi:10.1093/bja/aeq302
Feng X, Liu JJ, Zhou X, Song FH, Yang XY, Chen XS, Huang WQ, Zhou LH, Ye JH (2012) Single sevoflurane exposure decreases neuronal nitric oxide synthase levels in the hippocampus of developing rats. Br J Anaesth 109(2):225–233. doi:10.1093/bja/aes121
Li Q, Li H, Roughton K, Wang X, Kroemer G, Blomgren K, Zhu C (2010) Lithium reduces apoptosis and autophagy after neonatal hypoxia-ischemia. Cell Death Dis 1:e56. doi:10.1038/cddis.2010.33
Caldero J, Brunet N, Tarabal O, Piedrafita L, Hereu M, Ayala V, Esquerda JE (2010) Lithium prevents excitotoxic cell death of motoneurons in organotypic slice cultures of spinal cord. Neuroscience 165(4):1353–1369. doi:10.1016/j.neuroscience.2009.11.034
Zhong J, Yang X, Yao W, Lee W (2006) Lithium protects ethanol-induced neuronal apoptosis. Biochem Biophys Res Commun 350(4):905–910. doi:10.1016/j.bbrc.2006.09.138
Straiko MM, Young C, Cattano D, Creeley CE, Wang H, Smith DJ, Johnson SA, Li ES, Olney JW (2009) Lithium protects against anesthesia-induced developmental neuroapoptosis. Anesthesiology 110(4):862–868. doi:10.1097/ALN.0b013e31819b5eab
Chuang DM (2005) The antiapoptotic actions of mood stabilizers: molecular mechanisms and therapeutic potentials. Ann N Y Acad Sci 1053:195–204. doi:10.1196/annals.1344.018
Rowe MK, Chuang DM (2004) Lithium neuroprotection: molecular mechanisms and clinical implications. Expert Rev Mol Med 6(21):1–18. doi:10.1017/S1462399404008385
Jope RS, Yuskaitis CJ, Beurel E (2007) Glycogen synthase kinase-3 (GSK3): inflammation, diseases, and therapeutics. Neurochem Res 32(4–5):577–595. doi:10.1007/s11064-006-9128-5
Zhu ZF, Wang QG, Han BJ, William CP (2010) Neuroprotective effect and cognitive outcome of chronic lithium on traumatic brain injury in mice. Brain Res Bull 83(5):272–277. doi:10.1016/j.brainresbull.2010.07.008
Riadh N, Allagui MS, Bourogaa E, Vincent C, Croute F, Elfeki A (2011) Neuroprotective and neurotrophic effects of long term lithium treatment in mouse brain. Biometals 24(4):747–757. doi:10.1007/s10534-011-9433-6
Feng X, Liu JJ, Zhou X, Song FH, Yang XY, Chen XS, Huang WQ, Zhou LH, Ye JH (2012) Single sevoflurane exposure decreases neuronal nitric oxide synthase levels in the hippocampus of developing rats. Br J Anaesth. doi:10.1093/bja/aes121
He FQ, Qiu BY, Zhang XH, Li TK, Xie Q, Cui DJ, Huang XL, Gan HT (2011) Tetrandrine attenuates spatial memory impairment and hippocampal neuroinflammation via inhibiting NF-kappaB activation in a rat model of Alzheimer’s disease induced by amyloid-beta(1-42). Brain Res 1384:89–96. doi:10.1016/j.brainres.2011.01.103
Ren WJ, Liu Y, Zhou LJ, Li W, Zhong Y, Pang RP, Xin WJ, Wei XH, Wang J, Zhu HQ, Wu CY, Qin ZH, Liu G, Liu XG (2011) Peripheral nerve injury leads to working memory deficits and dysfunction of the hippocampus by upregulation of TNF-alpha in rodents. Neuropsychopharmacol: official publication of the American College of Neuropsychopharmacology 36(5):979–992. doi:10.1038/npp.2010.236
Liu Y, Zhuang X, Gou L, Ling X, Tian X, Liu L, Zheng Y, Zhang L, Yin X (2013) Protective effects of nizofenone administration on the cognitive impairments induced by chronic restraint stress in mice. Pharmacol Biochem Behav 103(3):474–480. doi:10.1016/j.pbb.2012.09.009
Wilder RT, Flick RP, Sprung J, Katusic SK, Barbaresi WJ, Mickelson C, Gleich SJ, Schroeder DR, Weaver AL, Warner DO (2009) Early exposure to anesthesia and learning disabilities in a population-based birth cohort. Anesthesiology 110(4):796–804. doi:10.1097/01.anes.0000344728.34332.5d
DiMaggio C, Sun LS, Kakavouli A, Byrne MW, Li G (2009) A retrospective cohort study of the association of anesthesia and hernia repair surgery with behavioral and developmental disorders in young children. J Neurosurg Anesthesiol 21(4):286–291. doi:10.1097/ANA.0b013e3181a71f11
Kalkman CJ, Peelen L, Moons KG, Veenhuizen M, Bruens M, Sinnema G, de Jong TP (2009) Behavior and development in children and age at the time of first anesthetic exposure. Anesthesiology 110(4):805–812. doi:10.1097/ALN.0b013e31819c7124
Flick RP, Katusic SK, Colligan RC, Wilder RT, Voigt RG, Olson MD, Sprung J, Weaver AL, Schroeder DR, Warner DO (2011) Cognitive and behavioral outcomes after early exposure to anesthesia and surgery. Pediatrics 128(5):e1053–e1061. doi:10.1542/peds.2011-0351
Berns M, Zacharias R, Seeberg L, Schmidt M, Kerner T (2009) Effects of sevoflurane on primary neuronal cultures of embryonic rats. Eur J Anaesthesiol 26(7):597–602
Pan Z, Lu XF, Shao C, Zhang C, Yang J, Ma T, Zhang LC, Cao JL (2011) The effects of sevoflurane anesthesia on rat hippocampus: a genomic expression analysis. Brain Res 1381:124–133. doi:10.1016/j.brainres.2011.01.020
Wada A, Yokoo H, Yanagita T, Kobayashi H (2005) Lithium: potential therapeutics against acute brain injuries and chronic neurodegenerative diseases. J Pharmacol Sci 99(4):307–321
Chuang DM, Chen RW, Chalecka-Franaszek E, Ren M, Hashimoto R, Senatorov V, Kanai H, Hough C, Hiroi T, Leeds P (2002) Neuroprotective effects of lithium in cultured cells and animal models of diseases. Bipolar Disord 4(2):129–136
Jope RS (2003) Lithium and GSK-3: one inhibitor, two inhibitory actions, multiple outcomes. Trends Pharmacol Sci 24(9):441–443. doi:10.1016/S0165-6147(03)00206-2
Chen G, Rajkowska G, Du F, Seraji-Bozorgzad N, Manji HK (2000) Enhancement of hippocampal neurogenesis by lithium. J Neurochem 75(4):1729–1734
Luo CX, Zhu XJ, Zhou QG, Wang B, Wang W, Cai HH, Sun YJ, Hu M, Jiang J, Hua Y, Han X, Zhu DY (2007) Reduced neuronal nitric oxide synthase is involved in ischemia-induced hippocampal neurogenesis by up-regulating inducible nitric oxide synthase expression. J Neurochem 103(5):1872–1882. doi:10.1111/j.1471-4159.2007.04915.x
Weitzdoerfer R, Hoeger H, Engidawork E, Engelmann M, Singewald N, Lubec G, Lubec B (2004) Neuronal nitric oxide synthase knock-out mice show impaired cognitive performance. Nitric Oxide 10(3):130–140. doi:10.1016/j.niox.2004.03.007S1089860304000588
Tanda K, Nishi A, Matsuo N, Nakanishi K, Yamasaki N, Sugimoto T, Toyama K, Takao K, Miyakawa T (2009) Abnormal social behavior, hyperactivity, impaired remote spatial memory, and increased D1-mediated dopaminergic signaling in neuronal nitric oxide synthase knockout mice. Mol Brain 2:19. doi:10.1186/1756-6606-2-19
Liu XS, Xue QS, Zeng QW, Li QA, Liu JA, Feng XM, Yu BW (2010) Sevoflurane impairs memory consolidation in rats, possibly through inhibiting phosphorylation of glycogen synthase kinase-3 beta in the hippocampus. Neurobiol Learn Mem 94(4):461–467. doi:10.1016/j.nlm.2010.08.011
Wiklund A, Granon S, Faure P, Sundman E, Changeux JP, Eriksson LI (2009) Object memory in young and aged mice after sevoflurane anaesthesia. NeuroReport 20(16):1419–1423. doi:10.1097/Wnr.0b013e328330cd2b
Acknowledgments
This work was funded by National Natural Science Foundation of China, Grant No. 81571032 and Guangdong Science Technology Planning Project, Grant No. 2013B051000045.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interests
There is no conflict of interest in this work.
Ethical approval
All applicable international, national, and Institutional Animal Care and Use Committee at Sun Yat-sen University (Guangzhou, Guangdong, China; No. [2014]A-073) guidelines for the care and use of animals were followed.
Additional information
Xue Zhou and Wen-da Li contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zhou, X., da Li, W., Yuan, BL. et al. Lithium Treatment Prevents Apoptosis in Neonatal Rat Hippocampus Resulting from Sevoflurane Exposure. Neurochem Res 41, 1993–2005 (2016). https://doi.org/10.1007/s11064-016-1909-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-016-1909-x