Abstract
Several experimental and epidemiological reports have associated manganese exposure with induction of oxidative stress and locomotor dysfunctions. Diphenyl diselenide (DPDS) is widely reported to exhibit antioxidant, anti-inflammatory and neuroprotective effects in in vitro and in vivo studies via multiple biochemical mechanisms. The present study investigated the protective effect of DPDS on manganese-induced toxicity in Drosophila melanogaster. The flies were exposed, in a dietary regimen, to manganese alone (30 mmol per kg) or in combination with DPDS (10 and 20 µmol per kg) for 7 consecutive days. Exposure to manganese significantly (p < 0.05) increased flies mortality, whereas the survivors exhibited significant locomotor deficits with increased acetylcholinesterase (AChE) activity. However, dietary supplementation with DPDS caused a significant decrease in mortality, improvement in locomotor activity and restoration of AChE activity in manganese-exposed flies. Additionally, the significant decreases in the total thiol level, activities of catalase and glutathione-S-transferase were accompanied with significant increases in the generation of reactive oxygen and nitrogen species and thiobarbituric acid reactive substances in flies exposed to manganese alone. Dietary supplementation with DPDS significantly augmented the antioxidant status and prevented manganese-induced oxidative stress in the treated flies. Collectively, the present data highlight that DPDS may be a promising chemopreventive drug candidate against neurotoxicity resulting from acute manganese exposure.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Manganese (Mn) is a naturally occurring trace element widely distributed in the earth crust. Besides, manganese compounds are components of some fertilizers, pesticides and gasoline additive [1, 2]. These compounds are reportedly released to the environment as combustion products from automobiles and industrial processes. The two major routes of exposure to manganese are inhalation and ingestion. Absorption of manganese following exposure is often affected by exposure route, the chemical form, the age of individual and dietary factors. Although manganese is an essential biometal which is required in trace amounts in animals and humans, several experimental and epidemiological reports have demonstrated serious health deficits associated with elevated manganese level [3–6].
The brain is the principal target during manganese toxicity. Several investigations with different animal models have associated manganese exposure with loss of cognitive and motor functions [7–9]. Similarly, numerous reports from occupational studies have associated elevated manganese exposure with permanent damage including impaired neurological and neuromuscular control, mental disorders, as well as difficulties with breathing or swallowing [10]. Moreover, several previous investigations have shown that induction of oxidative stress, diminution of endogenous antioxidants, alteration in calcium metabolism and dopaminergic regulation of neuronal activity represent the molecular mechanisms involved in the neurotoxicity of manganese [4, 7, 11].
Selenium is an essential trace element with a long history of health benefits in several living organisms, including humans. Selenium is necessary for the expression of about 25 selenium-dependent proteins (selenoproteins) which modulate the cellular redox status [12, 13]. Most of the biological functions of selenium are associated with the antioxidant properties of selenoenzymes. These enzymes play a significant role in the protection of brain tissues against oxidative damage. Furthermore, the interest in organoselenium compounds has increased due to their pharmacological potential. Diphenyl diselenide (DPDS) is a highly lipophilic organic selenium compound widely reported to exhibit antioxidant, anti-inflammatory, anti-nociceptive, hepatoprotective and neuroprotective effects in vitro and in vivo [14–19]. However, there is dearth of information on the possible modulatory effect of DPDS on manganese-induced neurotoxicity in experimental animals.
One of the major concerns of researchers is the reduction in the number of higher laboratory animals for research and testing due to ethical issues. The fruit fly, Drosophila melanogaster, has been recommended by the European Centre for the Validation of Alternative Methods (ECVAM) for promoting the 3Rs (reduction, refinement and replacement) of laboratory animal usage in toxicity studies [20]. Drosophila possesses systems which control nutrient uptake, storage and metabolism and these systems have been reported to be analogous to those of humans [21–24]. Drosophila is well known for its high sensitivity to toxic substances and is being considered as a useful model for toxicity studies as well as evaluating the biological action of pharmacological agents [25]. Indeed, Drosophila has been previously demonstrated to be a useful model for elucidating the mechanisms underlying manganese-induced neurotoxicity [26, 27].
The present study assessed the neuroprotective effects of DPDS on manganese-induced toxicity in newly emerged flies, by evaluating the mortality and locomotor deficit following short-term dietary regimen. Furthermore, using the head region of flies, we evaluated some biochemical markers of toxicity such as acetylcholinesterase (AChE) activity, antioxidant enzymes activities, reactive oxygen and nitrogen species (RONS) level, as well as levels of total thiol and thiobarbituric acid reactive substances (TBARS), subsequent to manganese exposure in the flies, in order to understand the protective mechanism of DPDS.
Materials and Methods
Chemicals
Manganese chloride (MnCl2), diphenyl diselenide (DPDS), acetylthiocholine iodide, glutathione (GSH), 2′,7′-dichlorofluorescein diacetate (DCFH-DA), thiobarbituric acid (TBA), 5′,5′-dithiobis (2-nitrobenzoic acid) (DTNB) and 1-chloro-2,4-dinitrobenzene (CDNB) were purchased from Sigma Aldrich (St. Louis, MO, USA).
Drosophila melanogaster Stock and Culture
Drosophila melanogaster (Harwich strain) used in the present investigation were obtained from the National Species Stock Center (Bowling Green, OH, USA). The flies were maintained and reared on corn meal medium containing 2 % w/v sucrose, 1 % w/v brewer’s yeast, 1 % w/v agar, 1 % w/v powdered milk and 0.08 % v/w nipagin at constant temperature and humidity (23 ± 1 °C; 60 % relative humidity, respectively) under 12 h light/dark cycle conditions. All the experiments were performed using the same fly strain.
Experimental Design
The assessment of the effects of DPDS on manganese-induced mortality, locomotor deficits, neurotoxicity and oxidative stress was carried out by employing a short-term dietary regimen. Newly emerged flies (2- to 3-day-old) were divided into five groups consisting of 50 flies each in three replicates per group were exposed to manganese (30 mmol per kg) with or without DPDS (10 and 20 µmol per kg) for 7 consecutive days. The doses of manganese and DPDS as well as the duration for the present study were selected based on the pilot study and previously published data [25, 27]. The average life span of Drosophila melanogaster in optimal temperatures is 60–70 days [25, 26]. The newly emerged flies were used for the present study because it represents an important stage in which most organisms undergo dramatic changes in shape as they grow and mature as well as when they exhibit a variety of ontogenetic changes in morphology and physiology. Thus, the developing organism is commonly more susceptible to toxic insult than the adult. The 7 days treatment period chosen for this model organism would correspond with young age of humans.
-
Group I Control flies fed with normal diet.
-
Group II Flies fed with diet containing DPDS (20 µmol per kg).
-
Group III Flies fed with diet containing manganese (30 mmol per kg).
-
Group IV Flies fed with diet containing manganese (30 mmol per kg) and DPDS (10 µmol per kg).
-
Group V Flies fed with diet containing manganese (30 mmol per kg) and DPDS (20 µmol per kg).
Lethality Response
The flies were observed daily for the incidence of mortality. The survival rate was determined by counting the number of dead flies, while the survivors were transferred to freshly prepared diet. The data were subsequently analyzed and plotted as cumulative mortality and percentage of live flies after the treatment period.
Measurement of Locomotor Performance by Negative Geotaxis Assay
Evaluation of locomotor performance of the control and treated flies was carried out after the treatment period using the negative geotaxis assay [28]. Briefly, ten flies from control and the treated groups were separately immobilized in ice anesthesia using labeled vertical glass columns (15 cm in length × 1.5 cm in diameter). Following a 10 min of recovery, the flies were tapped at the bottom of the column and the number of flies that crossed the 6 cm line within 6 s was recorded. Normally, flies without locomotor deficit move very fast to the top, whereas those with motor defect are slow in movement and may remain near the bottom. The climbing scores denote the average percentage of flies that crossed the 6 cm line among the total number of flies per experiment. The results are expressed as percentage of flies that escaped beyond a minimum distance of 6 cm in 6 s during three independent experiments.
Preparation of Sample for Biochemical Assays
Following the negative geotaxis assay, the flies from control and the treatment groups were anaesthetized in ice and weighed. The head region was carefully separated, homogenized in 10 volumes 0.1 M phosphate buffer, pH 7.4, and centrifuged at 6000g for 10 min at 4 °C in a Biofuge Sorvall Fresco centrifuge (Kendro Laboratory Products, Germany). The supernatant was collected and subsequently used for the determination of biochemical parameters. The protein concentration of head homogenates was determined using the Lowry method [29]. All biochemical determinations were performed in duplicates in three independent experiments.
Estimation of Acetylcholinesterase (AChE) Activity
AChE activity was determined according to the method of Ellman et al. [30]. Briefly, the assay medium consisted of 135 µL of distilled water, 20 µL of 100 mM potassium phosphate buffer (pH 7.4), 20 µL of 10 mM DTNB, 5 µL of sample, and 20 µL of 8 mM acetylthiocholine as substrate. The degradation of acetylthiocholine iodide was analyzed for 5 min (30 s intervals) at 412 nm using a SpectraMax plate reader (Molecular Devices, CA, USA) and the results were expressed as µmol/min/mg protein.
Measurement of Reactive Oxygen and Nitrogen Species (RONS) Generation
The level of intracellular RONS generation as an index of oxidative stress was determined by quantifying 2′,7′-dichlorofluorescein (DCFH) oxidation according to established procedure [31]. The assay reaction mixture contained 150 µL of 0.1 M potassium phosphate buffer (pH 7.4), 40 µL of distilled water, 5 µL of DCFH-DA (200 µM, final concentration 5 µM), and 5 µL of the sample (1:10 dilution). The fluorescence emission of DCF resulting from DCFH oxidation was analyzed for 10 min (30 s intervals) at 488 and 525 nm, excitation and emission wavelengths, respectively, using a SpectraMax plate reader (Molecular Devices, CA, USA). The rate of DCF formation was expressed as percentage of control group.
Determination of Thiobarbituric Acid Reactive Substances
The lipid peroxidation end products were quantified as thiobarbituric acid reactive substances (TBARS) according to established procedure [32]. Briefly, the stock reagent consisted of equal volumes of trichloroacetic acid (10 %, w/v) and 2-thiobarbituric acid (0.75 %, w/v) in 0.1 M HCl. One volume (100 µL) of tissue supernatant and two volumes (200 µL) of stock reagent were incubated at 95 °C for 60 min for color development. The reaction mixture was subsequently centrifuged at 8000×g for 10 min and the absorbance of the supernatant measured at 532 nm using a SpectraMax plate reader (Molecular Devices, CA, USA). The TBARS values were expressed as nMol MDA/mg protein.
Estimation of the Total Thiol Level
The total thiol level was determined in the control and treated flies according to the method previously described by Ellman [33]. The reaction system was made up of 170 µL of 0.1 M potassium phosphate buffer (pH 7.4), 20 µL of sample, and 10 µL of 10 mM DTNB. At the end of 30 min incubation at room temperature (25 °C), the absorbance was measured at 412 nm. A standard curve was plotted for each measurement using GSH as a standard and the results expressed as µMol/mg protein.
Estimation of Catalase (CAT) Activity
Catalase activity was assayed spectrophotometrically according to the method of Aebi [34] by monitoring the disappearance of H2O2. Briefly, the reaction medium contained 1800 µL of 50 mM phosphate buffer (pH 7.0), 180 µL of 300 mM H2O2, and 20 µL of sample (1:10 dilution). The reaction was analyzed for 2 min (10 s intervals) at 240 nm with a UV–visible spectrophotometer (Shimadzu, Japan). Activity of CAT is expressed as µmol of H2O2 consumed/min/mg protein.
Estimation of Glutathione-S-Transferase (GST) Activity
Glutathione-S-transferase activity was assayed according to the method of Habig and Jakoby [35] with slight modifications [36] using 1-chloro-2,4-dinitrobenzene (CDNB) as a substrate. The reaction mixture consisted of 270 µL of a solution containing (20 mL of 0.25 M potassium phosphate buffer, pH 7.0, 10.5 mL of distilled water, and 500 µL of 0.1 M GSH at 25 °C), 20 µL of sample (1:5 dilution), and 10 µL of 25 mM CDNB. The reaction was analyzed for 5 min (30 s intervals) at 340 nm in a SpectraMax plate reader (Molecular Devices, CA, USA) and GST activity expressed as µMol/min/mg protein using the molar extinction coefficient (ε) of 9.6 mM−1 cm−1 for CDNB conjugate.
Statistical Analyses
Statistical analyses were carried out using one-way analysis of variance (ANOVA) to compare the experimental groups followed by Bonferroni’s post hoc test to identify significantly different groups (SPSS for Windows, version 17; SPSS Inc., Chicago, IL). The level of statistical significance was set as p < 0.05.
Results
Mortality of Flies During Co-Exposure to Manganese and DPDS
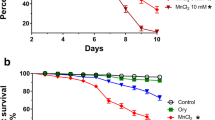
The cumulative mortality and percent live flies in the control and flies exposed to manganese alone or in combination with DPDS for 7 consecutive days are presented in Fig. 1. There was no mortality of flies in control and those treated with 20 µmol per kg of DPDS alone. There was a significant (p < 0.05) increase in the mortality of flies exposed to manganese when compared with the control. Conversely, co-exposure with DPDS significantly decreased flies mortality when compared with the manganese-treated flies. The cumulative percent live flies at the end of 7 days was 100 % for both control and flies exposed to DPDS alone. Acute exposure to manganese significantly decreased the percent live flies to 72 %. Interestingly, co-exposure with DPDS significantly increased the percentage of live flies when compared with the group treated with manganese alone. The percentages of live flies were 90 and 92 % for groups co-treated with 10 and 20 µmol per kg DPDS respectively.
Cumulative mortality expressed as number of dead flies and the percent live flies expressed as survival (%) of flies during a 7-day co-exposure period to manganese and DPDS. n = 50 flies/three replicates. Values are mean + SD. a p < 0.05 against control; b p < 0.05 against 20 µmol DPDS; c p < 0.05 against manganese alone
Locomotor Performance and Acetylcholinesterase Activity
The locomotor performance and AChE activity of control and flies exposed to manganese alone or in combination with DPDS for 7 consecutive days are presented in Fig. 2. The climbing behavior of control and flies treated with DPDS alone were not significantly different from each other. The percentages of flies that crossed the 6 cm mark within 6 s were 94 and 95 % for control and flies treated with DPDS alone at 20 µmol per kg respectively. Conversely, the flies exposed to manganese alone showed significant impairment in the climbing behavior with 47 % of flies crossing the 6 cm mark within 6 s when compared with control. Interestingly, the flies co-exposed to manganese and DPDS demonstrated significant improvement in the locomotor performance when compared with those inmanganese alone group. The percentage of flies that crossed the 6 cm mark within 6 s were 76 and 82 % for groups co-treated with DPDS at 10 and 20 µmol per kg respectively. However, a contrary trend of result was observed in the AChE activity in the control and treated flies. There was a significant increase in AChE activity in flies exposed to manganese alone, whereas co-treatment with DPDS resulted in a significant decrease in AChE activity when compared with flies exposed to manganese alone.
Incidence of locomotor impairment (expressed as percentage of climbing 6 cm in 6 s) determined in a negative geotaxis assay and acetylcholinesterase (AChE) activity in control and treated flies during a 7-day co-exposure period to manganese and DPDS. n = 50 flies/three replicates. Values are mean + SD. a p < 0.05 against control; b p < 0.05 against 20 µmol DPDS; c p < 0.05 against manganese alone
Biomarkers of Oxidative Stress in Control and Treated Flies
The levels of oxidative stress indices determined in control and flies exposed to manganese alone or in combination with DPDS for 7 consecutive days are presented in Fig. 3. There were no treatment-related effects on the oxidative stress indices in flies exposed to DPDS alone when compared with the control. However, manganese exposure alone significantly elevated the levels of TBARS in the treated flies by 300 % when compared with control group. Co-treatment with DPDS at 10 and 20 µmol per kg significantly decreased the levels of TBARS production by 80 and 98 % respectively, when compared with flies treated with manganese alone group. Similarly, manganese exposure caused a significant elevation in RONS level, whereas it decreased the total thiol level in the treated flies by 110 and 60 % respectively, when compared with the control group. However, co-treatment with DPDS at 10 and 20 µmol per kg significantly decreased the level of RONS production by 92 and 109 %, whereas it increased the total thiol level by 85 and 91 % respectively, when compared with flies in manganese alone group.
Levels of total thiol, reactive oxygen and nitrogen species (RONS) and thiobarbituric acid reactive substances (TBARS) in control and treated flies after a 7-day co-exposure period to manganese and DPDS. n = 50 flies/three replicates. Values are mean + SD. a p < 0.05 against control; b p < 0.05 against 20 µmol DPDS; c p < 0.05 against manganese alone
Antioxidant Enzymes Activities
The antioxidant activities of CAT and GST determined in control and flies exposed to manganese alone or in combination with DPDS for 7 consecutive days are presented in Fig. 4. There were no treatment-related effects on CAT and GST activities in flies exposed to DPDS alone when compared with the control. However, manganese exposure alone significantly decreased CAT and GST activities in the treated flies by 50 and 57 % respectively, when compared with the control group. Co-treatment with DPDS at 10 and 20 µmol per kg significantly increased CAT activity by 88 and 96 % while GST activity was increased by 86 and 90 % respectively, when compared with flies in manganese alone group.
Discussion
Manganese is an essential trace element for the regulation of many biochemical processes including the development and maintenance of the central nervous system [8, 37, 38]. However, excessive exposure to manganese is well-known to cause manganism, a neurological disorder characterized by psychological and neurological abnormalities [39]. This is the first report to describe the protective role of DPDS in manganese induced toxicity in Drosophila melanogaster. This study demonstrated the neuroprotective potential of DPDS against manganese induced motor dysfunction and mortality in flies which is attributable to the previously reported beneficial effects in different experimental animals. In addition, we report herein the ameliorative effect of DPDS against manganese induced oxidative stress in the flies at exposure regimen of 10 and 20 µmol per kg.
The present study demonstrated that dietary exposure to manganese at 30 mmol per kg caused a significant increase in the cumulative number of dead flies and consequently, a significant reduction in the percentage of live Drosophila melanogaster following 7 days of treatment regimen. This observation could be attributed to the cytotoxic effect of manganese which has been previously reported. Exposure to manganese significantly and dose-dependently reduced cell viability in astrocyte cultures from cerebral cortices of newborn Sprague–Dawley rats [38]. In addition, manganese reportedly decreased cell viability through increased ROS production and apoptotic cell death in rat astrocytoma C6 cells [40]. However, dietary supplementation of DPDS significantly prevented the manganese mediated toxicity by decreasing the mortality and consequently increased the percent live flies. The protective effect of DPDS against mortality induced by insecticide chloropyrifos in Drosophila melanogaster has been reported [25]. It is worthy of note that the relative safety of DPDS at 0.3, 3.0 and 30 p.p.m during 8 months administration to rabbits has been reported [41]. Moreover, chronic administration of DPDS to diabetic rats reportedly abolished mortality rate without obvious sign of toxicity [42]. Thus, the lack of statistically significant difference in the cumulative number of dead flies and the percent live flies following DPDS treatment in the present investigation possibly suggests its safety benefit at these doses.
Acetylcholinesterase (AChE) is a serine protease that hydrolyses acetylcholine, a neurotransmitter which regulates motor function and locomotion [43]. Acute exposure of newly emerged flies to manganese caused significant decrease in climbing activity with concomitant increase in AChE activity in the present study. These findings are in agreement with previous observations in rodents. Short-term administrations of high-doses of manganese were reported to significantly increased brain AChE activity in rat [44, 45]. Also, exposure to manganese induced a significant increase in AChE activity in both the rat serum and brain [46]. However, the contradictory results from Santos et al. [47] has been attributed to the use of pentobarbital for euthanizing the rats rather than decapitation. Pentobarbital reportedly decreased rat brain AChE activity [48–50]. Thus, the increase in the AChE activity following dietary exposure to manganese in the present study could lead to a decrease in acetylcholine levels in the synaptic cleft and consequently reduce cholinergic neurotransmission efficiency and impair climbing activity in the flies. However, dietary supplementation of DPDS was associated with decreased AChE activity and improved climbing performance in flies exposed to manganese, thus indicating the protective role of DPDS in Drosophila melanogaster model of manganese neurotoxicity.
Total thiol level is a well-known indirect oxidative stress biomarker, which is indicative of chemical changes in thiol groups of proteins and peptides [36, 51, 52]. Catalase and GST are antioxidant enzymes which protect cellular macromolecules by neutralizing intracellular peroxides and electrophilic oxidants, respectively. The present study demonstrated a state of oxidative stress in flies treated with manganese alone as evidenced by the significant elevation in RONS and TBARS production with concomitant significant decrease in total thiol level as well as CAT and GST activities. The reduction in these antioxidant enzymes activities could increase the RONS levels and consequently lead to cell death. These findings are in accordance with several previous reports which showed that increased AChE activity is associated with increased neuronal oxidative and nitrosative stress, alterations in energy metabolism in invertebrates and vertebrates [53]. Interestingly, dietary supplementation with DPDS significantly decreased RONS and TBARS production levels, augmented total thiol level and increased antioxidant enzymes activities in the treated flies, thus suggesting the antioxidant effect of DPDS against manganese-induced oxidative stress in the treated flies. The protective mechanism of DPDS on manganese-induced cellular damage is attributed to its ability to maintain antioxidant enzymes, scavenge RONS and consequently inhibit oxidative damage in the treated flies.
Taken together, the findings from this study revealed that DPDS protected against manganese-induced toxicity in flies via reduction of mortality, inhibition of oxidative stress indices and maintenance of endogenous antioxidants. Moreover, dietary DPDS supplementation was associated with neuroprotection characterized by improvement in the AChE activity and locomotor function in Drosophila melanogaster. These observations highlight that DPDS may be a promising chemopreventive drug candidate against neurotoxicity resulting from acute manganese exposure.
References
US EPA (2003) Health effects support document for manganese. U.S. Environmental Protection Agency, Office of Water, EPA. EPA-822-R-03-003, Washington, D.C
Dobson AW, Erikson KM, Aschner M (2004) Manganese neurotoxicity. Ann N Y Acad Sci 1012:115–128
Claus Henn B, Schnaas L, Ettinger AS, Schwartz J, Lamadrid-Figueroa H, Hernández-Avila M, Amarasiriwardena C, Hu H, Bellinger DC, Wright RO, Téllez-Rojo MM (2012) Associations of early childhood manganese and lead coexposure with neurodevelopment. Environ Health Perspect 120:126–131
Avila DS, Puntel RL, Aschner M (2013) Manganese in health and disease. Met Ions Life Sci 13:199–227
Zoni S, Lucchini RG (2013) Manganese exposure: cognitive, motor and behavioral effects on children: a review of recent findings. Curr Opin Pediatr 25:255–260
Kim G, Lee HS, Seok Bang J, Kim B, Ko D, Yang M (2015) A current review for biological monitoring of manganese with exposure, susceptibility, and response biomarkers. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev 33:229–254
Burton NC, Guilarte TR (2009) Manganese neurotoxicity: lessons learned from longitudinal studies in nonhuman primates. Environ Health Perspect 117:325–332
Roels HA, Bowler RM, Kim Y, Claus Henn B, Mergler D, Hoet P, Gocheva VV, Bellinger DC, Wright RO, Harris MG, Chang Y, Bouchard MF, Riojas-Rodriguez H, Menezes-Filho JA, Tellez-Rojo MM (2012) Manganese exposure and cognitive deficits: a growing concern for manganese neurotoxicity. Neurotoxicology 33:872–880
Guilarte TR (2013) Manganese neurotoxicity: new perspectives from behavioral, neuroimaging, and neuropathological studies in humans and non-human primates. Front Aging Neurosci 5:23–31
Santos D, Batoreu C, Mateus L, Marreilha Dos Santos AP, Aschner M (2014) Manganese in human parenteral nutrition: considerations for toxicity and biomonitoring. Neurotoxicology 43:36–45
Aschner M, Guilarte TR, Schneider JS, Zheng W (2007) Manganese: recent advances in understanding its transport and neurotoxicity. Toxicol Appl Pharmacol 221:131–147
Bellinger FP, Raman AV, Reeves MA, Berry MJ (2009) Regulation and function of selenoproteins in human disease. Biochem J 422:11–22
Sunde RA, Raines AM (2011) Selenium regulation of the selenoprotein and nonselenoprotein transcriptomes in rodents. Adv Nutr 2:138–150
Rosa RM, Roesler R, Braga AL, Saffi J, Henriques JA (2007) Pharmacology and toxicology of diphenyl diselenide in several biological models. Braz J Med Biol Res 40:1287–1304
de Bem AF, Farina M, Portella RL, Nogueira CW, Dinis TCP, Laranjinha JAN, Almeida LM, Rocha JBT (2008) Diphenyldiselenide, a simple glutathione peroxidase mimetic, inhibits human LDL oxidation in vitro. Atherosclerosis 201:92–100
Kade IJ, Borges VC, Savegnago L, Ibukun EO, Zeni G, Nogueira CW, Rocha JB (2009) Effect of oral administration of diphenyl diselenide on antioxidant status, and activity of delta aminolevulinic acid dehydratase and isoforms of lactate dehydrogenase, in streptozotocin-induced diabetic rats. Cell Biol Toxicol 25:415–424
Posser T, de Paula MT, Franco JL, Leal RB, Rocha JBT (2010) Diphenyl diselenide induces apoptotic cell death and modulates ERK1/2 phosphorylation in human neuroblastoma SH-SY5Y cells. Arch Toxicol 117:645–651
Corte CLD, Soares FAA, Aschner M, Rocha JBT (2012) Diphenyl diselenide prevents methylmercury-induced mitochondrial dysfunction in rat liver slices. Tetrahedron 68:10437–10443
Avila DS, Benedetto A, Au C, Manarin F, Erikson K, Soares FA, Rocha JB, Aschner M (2012) Organotellurium and organoselenium compounds attenuate Mn-induced toxicity in Caenorhabditis elegans by preventing oxidative stress. Free Radic Biol Med 52:1903–1910
Benford DJ, Hanley AB, Bottrill K, Oehlschlager S, Balls M, Brance F, Castegnara JJ, Descotes J, Hemminiky K, Lindsay D, Schilter B (2000) Biomarkers as predictive tools in toxicity testing. The report and recommendations of ECVAM workshop 40. Alt Lab Anim 28:119–131
Baker KD, Thummel CS (2007) Diabetic larvae and obese flies-emerging studies of metabolism in Drosophila. Cell Metab 6:257–266
Jafari M (2010) Drosophila melanogaster as a model system for the evaluation of anti-aging compounds. Fly (Austin) 4:253–257
Riemensperger T, Issa AR, Pech U, Coulom H, Nguyễn MV, Cassar M, Jacquet M, Fiala A, Birman S (2013) A single dopamine pathway underlies progressive locomotor deficits in a Drosophila model of Parkinson disease. Cell Rep 5:952–960
Nguyen TB, Ida H, Shimamura M, Kitazawa D, Akao S, Yoshida H, Inoue YH, Yamaguchi M (2014) Role of SCOX in determination of Drosophila melanogaster lifespan. Am J Cancer Res 4:325–336
Adedara IA, Klimaczewski CV, Barbosa NB, Farombi EO, Souza DO, Rocha JBT (2015) Influence of diphenyl diselenide on chlorpyrifos-induced toxicity in Drosophila melanogaster. J Trace Elem Med Biol 32:52–59
Bonilla E, Contreras R, Medina-Leendertz S, Mora M, Villalobos V, Bravo Y (2012) Minocycline increases the life span and motor activity and decreases lipid peroxidation in manganese treated Drosophila melanogaster. Toxicology 294:50–53
Mora M, Bonilla E, Medina-Leendertz S, Bravo Y, Arcaya JL (2014) Minocycline increases the activity of superoxide dismutase and reduces the concentration of nitric oxide, hydrogen peroxide and mitochondrial malondialdehyde in manganese treated Drosophila melanogaster. Neurochem Res 39:1270–1278
Le Bourg E, Lints FA (1992) Hypergravity and aging in Drosophila melanogaster. 4. Climbing activity. Gerontology 38:59–64
Lowry OH, Rosenbrough NJ, Farr AL, Randall RJ (1951) Protein measurement with folin phenol reagent. J Biol Chem 193:265–275
Ellman GL, Courtney KD, Andres V Jr, Feather-Stone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95
Pérez-Severiano F, Santamaría A, Pedraza-Chaverri J, Medina-Campos ON, Ríos C, Segovia J (2004) Increased formation of reactive oxygen species, but no changes in glutathione peroxidase activity, in striata of mice transgenic for the Huntington’s disease mutation. Neurochem Res 29:729–733
Puntel RL, Roos DH, Grotto D, Garcia SC, Nogueira CW, Rocha JB (2007) Antioxidant properties of Krebs cycle intermediates against malonate pro-oxidant activity in vitro: a comparative study using the colorimetric method and HPLC analysis to determine malondialdehyde in rat brain homogenates. Life Sci 81:51–62
Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82:70–77
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126
Habig WH, Jakoby WB (1981) Assays for differentiation of glutathione S-transferases. Methods Enzymol 77:398–405
Abolaji OA, Kamdem JP, Lugokenski TH, Nascimento TK, Waczuk EP, Farombi EO, Loreto ÉL, Rocha JBT (2014) Involvement of oxidative stress in 4-vinylcyclohexene-induced toxicity in Drosophila melanogaster. Free Radic Biol Med 71:99–108
Aschner M, Dorman DC (2006) Manganese: pharmacokinetics and molecular mechanisms of brain uptake. Toxicol Rev 25:147–154
Evren V, Apaydin M, Khalilnezhad A, Erbas O, Taskiran D (2015) Protective effect of edaravone against manganese-induced toxicity in cultured rat astrocytes. Environ Toxicol Pharmacol 40:563–567
Farina M, Avila DS, da Rocha JB, Aschner M (2013) Metals, oxidative stress and neurodegeneration: a focus on iron, manganese and mercury. Neurochem Int 62:575–594
Gorojod RM, Alaimo A, Porte Alcon S, Pomilio C, Saravia F, Kotler ML (2015) The autophagic-lysosomal pathway determines the fate of glial cells under manganese- induced oxidative stress conditions. Free Radic Biol Med 87:237–251
de Bem AF, de Lima Portella R, Farina M, Perottoni J, Paixão MW, Nogueira CW, Teixeira Rocha JB (2007) Low toxicity of diphenyldiselenide in rabbits: a long-term study. Basic Clin Pharmacol Toxicol 101:47–55
Barbosa NB, Rocha JB, Soares JC, Wondracek DC, Gonçalves JF, Schetinger MR, Nogueira CW (2008) Dietary diphenyl diselenide reduces the STZ-induced toxicity. Food Chem Toxicol 46:186–194
Day J, Damsma G, Fibiger HC (1991) Cholinergic activity in the rat hippocampus, cortex and striatum correlates with locomotor activity: an in vivo microdialysis study. Pharmacol Biochem Behav 38:723–729
Liapi C, Zarros A, Galanopoulou P, Theocharis S, Skandali N, Al-Humadi H, Anifantaki F, Gkrouzman E, Mellios Z, Tsakiris S (2008) Effects of short-term exposure to manganese on the adult rat brain antioxidant status and the activities of acetylcholinesterase, (Na+, K+)-ATPase and Mg2+-ATPase: modulation by l-cysteine. Basic Clin Pharmacol Toxicol 103:171–175
Chtourou Y, Fetoui H, Garoui EM, Zeghal N (2012) Improvement of cerebellum redox states and cholinergic functions contribute to the beneficial effects of silymarin against manganese-induced neurotoxicity. Neurochem Res 37:469–479
Lebda MA, El-Newwshy MS, El-Sayed YS (2012) Neurohepatic toxicity of subacute manganese chloride exposure and potential chemoprotective effects of lycopene. Neurotoxicology 33:98–104
Santos D, Milatovic D, Andrade V, Batoreu MC, Aschner M, Marreilha dos Santos AP (2012) The inhibitory effect of manganese on acetylcholinesterase activity enhances oxidative stress and neuroinflammation in the rat brain. Toxicology 292:90–98
Micic D, Micic J, Kaltzo I, Spatz M (1978) Effect of pentobarbital on the synaptosomal activity of acetylcholinesterase in Mongolian gerbils. Experientia 34:169–170
Vernadakis A, Rutledge CO (1973) Effects of ether and pentobarbital anaesthesia on the activities of brain acetylcholinesterase and butyrylcholinesterase in young adult rats. J Neurochem 20:1503–1504
Santos D, Milatovic D, Andrade V, Batoreu MC, Aschner M, Marreilha dos Santos AP (2012) Comments to the Editor concerning the cholinergic response to manganese-induced neurotoxicity, based on the paper entitled “The inhibitory effect of manganese on acetylcholinesterase activity enhances oxidative stress and neuroinflammation in the brain” by Santos et al. Toxicology 298:59–60
Adedara IA, Rosemberg DB, Souza DO, Kamdem JP, Farombi EO, Aschner M, Rocha JBT (2015) Biochemical and behavioral deficits in lobster cockroach Nauphoeta cinerea model of methylmercury exposure. Toxicol Res 4:442–451
Adedara IA, Rosemberg DB, de Souza D, Farombi EO, Aschner M, Souza DO, Rocha JBT (2016) Neurobehavioral and biochemical changes in Nauphoeta cinerea following dietary exposure to chlorpyrifos. Pesticide Biochem Physiol. doi:10.1016/j.pestbp.2015.12.004
Milatovic D, Gupta RC, Aschner M (2006) Anticholinesterase toxicity and oxidative stress. Sci World J 6:295–310
Acknowledgments
This work was supported in part by the TWAS-CNPq 2013 Postdoctoral Fellowship (FR number: 3240274252) awarded to IAA.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Rights and permissions
About this article
Cite this article
Adedara, I.A., Abolaji, A.O., Rocha, J.B.T. et al. Diphenyl Diselenide Protects Against Mortality, Locomotor Deficits and Oxidative Stress in Drosophila melanogaster Model of Manganese-Induced Neurotoxicity. Neurochem Res 41, 1430–1438 (2016). https://doi.org/10.1007/s11064-016-1852-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-016-1852-x