Abstract
Tyrosinemia type II, also known as Richner–Hanhart syndrome, is an autosomal recessive inborn error of metabolism caused by a deficiency of hepatic cytosolic tyrosine aminotransferase, and is associated with neurologic and development difficulties in numerous patients. Considering that the mechanisms underlying the neurological dysfunction in hypertyrosinemic patients are poorly known and that studies demonstrated that high concentrations of tyrosine provoke oxidative stress in vitro and in vivo in the cerebral cortex of rats, in the present study we investigate the oxidative stress parameters (enzymatic antioxidant defenses, thiobarbituric acid-reactive substances and protein carbonyl content) in cerebellum, hippocampus and striatum of 30-old-day rats after acute administration of l-tyrosine. Our results demonstrated that the acute administration of l-tyrosine increased the thiobarbituric acid reactive species levels in hippocampus and the carbonyl levels in cerebellum, hippocampus and striatum. In addition, acute administration of l-tyrosine significantly decreased superoxide dismutase activity in cerebellum, hippocampus and striatum, while catalase was increased in striatum. In conclusion, the oxidative stress may contribute, along with other mechanisms, to the neurological dysfunction characteristic of hypertyrosinemia and the administration of antioxidants may be considered as a potential adjuvant therapy for tyrosinemia, especially type II.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Tyrosinemia type II, also known as Richner–Hanhart syndrome (RHS), is an autosomal recessive inborn error of metabolism caused by a deficiency of hepatic cytosolic tyrosine aminotransferase (TAT; l-tyrosine:2-oxoglutarate aminotransferase, EC 2.6.1.5) with a world frequency estimated in approximately 1 in 250,000 newborns. This blockage causes the accumulation of tyrosine, as well as tyrosine derivatives (4-hydroxyphenylpyruvic acid, 4-hydroxyphenyllactic acid and 4-hydroxyphenylacetic acid) in tissues, cerebrospinal fluid, blood and urine [1]. Interestingly, plasma tyrosine levels in TAT deficient patients range from 370 to 3,420 μM (normal < 90 μM) in untreated patients [1]. Individuals with Tyrosinemia type II usually present bilateral pseud dendritic keratitis, painful palm plantar hyperkeratotic lesions, mental retardation, and variable manifestations of central nervous system involvement [2–4].
In spite neurological sequelae are common in Tyrosinemia type II patients, but the mechanisms of brain damage are still poorly understood. However, current data do not eliminate the possibility that elevated levels of tyrosine and/or its derivatives may have noxious effects on central nervous system (CNS) development in these patients [5]. In this context, it has been recently reported that high concentrations of tyrosine induces oxidative stress by increasing free radical formation and decreasing brain antioxidant defenses in the cerebral cortex of rats [6, 7]. In addition, studies have reported that tyrosine reduces in vitro the activities of cytosolic and mitochondrial creatine kinase and pyruvate kinase in the cerebral cortex of rats and that this inhibition was prevented by reduced glutathione and creatine, suggesting that the enzymes inhibition occurred by alteration of sulfhydryl groups of the enzymes [8, 9].
Free radicals and oxidative stress have been associated with a large number of diseases including various neurodegenerative disorders, epileptic seizures, demyelination and dementia [10–14]. This may occur because the CNS is highly susceptible to oxidative damage due of its higher oxygen consumption and unique membrane lipid composition, and for the immature brain, susceptibility to oxidative stress is augmented by the increased metabolic demand associated with growth as well as the lower reserves of protective enzymes and antioxidants. In addition, relative to the adult brain, the developing nervous system is deficient in glia, which protects neurons by scavenging reactive oxidative species [15].
Oxidative stress can result from increased production of reactive oxygen species (ROS), decreased antioxidant defense, or failure to repair oxidative damage. ROS are free radicals or reactive anions/molecules containing oxygen atoms such as hydroxyl radical, superoxide, hydrogen peroxide, and peroxynitrite and can cause cell damage by enzyme inactivation, lipid peroxidation, and DNA modification [16]. Superoxide dismutase (SOD) and catalase (CAT) are the most important antioxidant enzymes [17, 18]. SOD is a protective enzyme that can selectively scavenge the superoxide anion radical (O2−) by catalyzing its dismutation to hydrogen peroxide (H2O2) and CAT metabolizes the excess of H2O2 producing O2 + H2O. Elevated SOD/CAT activities results in an increase of H2O2 concentration, or increased rate of H2O2 production and may lead to lipid and protein oxidation, resulting in increased neuronal damage [17, 19].
Considering that the mechanisms underlying the neurological dysfunction in hypertyrosinemic patients are poorly known and that studies demonstrated that high concentrations of tyrosine provoke oxidative stress in vitro and in vivo in the cerebral cortex of rats [6, 7], in the present study we investigate the oxidative stress parameters (enzymatic antioxidant defenses, thiobarbituric acid-reactive substances and protein carbonyl content) in cerebellum, hippocampus and striatum of 30-old-day rats after acute administration of l-tyrosine.
Materials and Methods
Animals
Male Wistar rats with 30-day-old (100–150 g) were obtained from the Central Animal House of Universidade do Extremo Sul Catarinense. The rats were caged in groups of five with free access to food and water. The rats were maintained on a 12-h light–dark cycle (lights on at 7:00 am) at a temperature of 23 ± 1 °C. All of the experimental procedures were carried out in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals and the Brazilian Society for Neuroscience and Behavior recomendations for animal care with the approval of the Ethics Committee from the Universidade do Extremo Sul Catarinense (protocol number 42/2010).
Administration of l-Tyrosine
l-Tyrosine was dissolved (25 °C) in saline solution (pH was adjusted to 7.4) and 500 mg/kg body weight of free l-tyrosine was administered intraperitoneally, in volumes of 10 μL/g of body weight. Control group received the same volume of saline solution (0.9 %) [6]. This dose was chosen in order to obtain tyrosine concentrations about 10 times the normal levels within 1 h after administration [20, 21], which are similar variations of plasma tyrosine levels observed in patients affected by tyrosinemia type II [1, 22]. For acute administration, l-tyrosine or saline solution given to rats on postnatal day (PD) PD 30 (n = 6). One hour after injections, rats were killed by decapitation without anesthesia. The cerebellum, hippocampus and striatum were rapidly removed and kept on an ice-plate for the measurements of the oxidative stress parameters.
Tissue and Homogenate Preparation
Cerebellum, hippocampus and striatum were homogenized (1:10, w/v) in SETH buffer, pH 7.4 (250-mM sucrose, 2-mM EDTA, 10-mM Trizma base, 50 IU/ml heparin). The homogenates were centrifuged at 800×g for 10 min at 4 °C, and the supernatants were kept at −70 °C until use as enzymes activity determination. The maximal period between homogenate preparation and enzyme analysis was always 5 days. Protein content was determined by the method described by Lowry and colleagues [23] using bovine serum albumin as a standard.
Thiobarbituric Acid Reactive Species Content in Tissue
To determine oxidative damage in lipid, we measured the formation of thiobarbituric acid reactive species (TBARS) during an acid-heating reaction, as previously described by Draper and Hadley [24]. The samples were mixed with 1 ml of trichloroacetic acid 10 % and 1 ml of thiobarbituric acid 0.67 %, and then heated in a boiling water bath for 30 min. Malondialdehyde equivalents were determined in tissue and in submitochondrial particles of the rat brain spectrophotometrically by the absorbance at 532 nm.
Carbonyls Protein Formation
The oxidative damage to proteins was assessed by the determination of carbonyl groups content based on the reaction with dinitrophenylhydrazine (DNPH), as previously described by Levine and colleagues [25]. Proteins were precipitated by the addition of 20 % trichloroacetic acid and were redissolved in DNPH. The absorbance was monitored spectrophotometrically at 370 nm.
Superoxide Dismutase Activity
This method for the assay of SOD activity is based on the capacity of pyrogallol to autoxidize, a process highly dependent on O2–2; a substrate for SOD [26]. The inhibition of autoxidation of this compound thus occurs when SOD is present, and the enzymatic activity can be then indirectly assayed spectrophotometrically at 420 nm, using a double-beam spectrophotometer with temperature control. A calibration curve was performed using purified SOD as the standard, in order to calculate the specific activity of SOD present in the samples. A 50 % inhibition of pyrogallol autoxidation is defined as 1 unit of SOD, and the specific activity is represented as units per mg of protein.
Catalase Activity
The CAT activity was assayed using a double-beam spectrophotometer with temperature control. This method is based on the disappearance of H2O2 at 240 nm in a reaction medium containing 20 mM H2O2, 0.1 % Triton X-100, 10 mM potassium phosphate buffer, pH 7.0, and 0.1–0.3 mg protein/ml [27]. One CAT unit is defined as 1 mol of hydrogen peroxide consumed per minute, and the specific activity is reported as units per mg protein.
Statistical Analysis
The results are presented as the mean ± standard deviation. All of the assays were performed in duplicate, and the mean was used for the statistical analysis. Student’s t test was used for the comparison of two means. Differences between the groups were rated significant at p < 0.05. All of the analyses were carried out with an IBM-compatible PC computer using the Statistical Package for the Social Sciences (SPSS) software.
Results
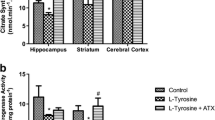
The present study evaluated oxidative stress parameters by measuring TBARS and protein carbonyl levels, as well as enzymatic activity of SOD and CAT in cerebellum, hippocampus and striatum of 30-day-old rats after acute administration of l-tyrosine. Our results demonstrated that TBARS levels were significantly increased only in hippocampus after acute administration of l-tyrosine, when compared to the control group (Fig. 1). However, protein carbonyl content was significantly increased in cerebellum, hippocampus and striatum of rats (Fig. 2). SOD activity was significantly decreased in cerebellum, hippocampus and striatum; CAT activity was increased in cerebellum after acute administration of l-tyrosine in rats (Figs. 3 and 4, respectively).
Effect of acute administration of l-tyrosine on thiobarbituric acid reactive species levels in the brain of 30-day-old rats. Data were analyzed by a one-way analysis of variance (ANOVA) followed by Student’s t test when F was significant. Values are expressed as nmol/min mg protein, mean ± S.D. (n = 7). *Different from control; p < 0.05
Effect of acute administration of l-tyrosine on carbonyls protein formation levels in the brain of 30-day-old rats. Data were analyzed by a one-way analysis of variance (ANOVA) followed by Student’s t test when F was significant. Values are expressed as nmol/min mg protein, mean ± S.D. (n = 7). *Different from control; p < 0.05
Effect of acute administration of l-tyrosine on superoxide dismutase activity in the brain of 30-day-old rats. Data were analyzed by a one-way analysis of variance (ANOVA) followed by Student’s t test when F was significant. Values are expressed as nmol/min mg protein, mean ± S.D. (n = 7). *Different from control; p < 0.05
Effect of acute administration of l-tyrosine on catalase activity in the brain of 30-day-old rats. Data were analyzed by a one-way analysis of variance (ANOVA) followed by Student’s t test when F was significant. Values are expressed as nmol/min mg protein, mean ± S.D. (n = 7). *Different from control; p < 0.05
Discussion
Inborn errors of tyrosine catabolism lead to hypertyrosinemia, especially tyrosinemia type II or TAT deficiency, in which tyrosine levels are highly elevated in tissues and physiological fluids of these patients [1]. Although brain tyrosine concentration in type II patients is not known, it has been reported plasma tyrosine levels exceeding 1,000 μM in untreated type II patients with results ranging from 370 to 3,420 μM (normal < 90 μM) along with some tyrosine derivatives accumulation (4-hydroxyphenylpyruvic acid, 4-hydroxyphenyllactic acid and 4-hydroxyphenylacetic acid) [1]. Though the molecular deficiency and symptoms of TAT deficiency are well described, the mechanisms responsible for the neuropathophysiology of this metabolic disorder are largely unknown, but it is well established that high concentrations of tyrosine and/or its derivatives were associated with the appearance of the neurological symptoms and these compounds seem to be the main neurotoxic metabolite in the illness [5].
Tissue damage caused by oxidative stress, mediated by excessive free radicals, is involved in a diversity of biological phenomena, including damage to proteins, lipids and DNA [28]. ROS are abundantly produced in the brain because neurons consume a large amount of oxygen, neuronal mitochondria generate superoxide anion, and the brain readily retains bio-available irons. Moreover, the adverse consequences of oxidative stress have been implicated in a variety of central nervous system diseases, including inherited metabolic disorders [29–35]. In this context, the main objective of the present work was to evaluate the in vivo effects of l-tyrosine on important parameters of oxidative stress cerebellum, striatum and hippocampus of young rats, in order to contribute to the understanding of the pathophysiology of hypertyrosinemia.
In the present study, we demonstrated that the acute administration of l-tyrosine increased the TBARS levels in hippocampus and the carbonyl levels in cerebellum, hippocampus and striatum. TBARS reflects the content of malondialdehyde, the most abundant individual aldehyde resulting from lipid breakdown due to lipid peroxidation process. On the other hand, the level of carbonyl groups in proteins is widely used as a marker of oxidative protein damage [36]. Therefore, these results suggest that acute administration of l-tyrosine induces oxidative damage to lipids in hippocampus, and oxidative damage to proteins in striatum, hippocampus and cerebellum. Increased protein carbonylation may facilitate the formation of protein aggregates, as a result of protein cross-links, and this is very likely to culminate in widespread cellular dysfunction. Additionally, increased oxidative damage to proteins might result in increased free iron, due to its release from damaged ferritin and other iron-containing proteins, favoring the maintenance of the pro-oxidative state by facilitates the production OH, the most powerful oxidant molecule, through a reaction with iron or copper (Fenton chemistry) [15, 36]. Regarding to the antioxidant defense system, l-tyrosine markedly decreased the SOD activity in cerebellum, hippocampus and striatum. In contrast, the activity of CAT, enzyme responsible for catalyses the decomposition of hydrogen peroxide into water and molecular oxygen was increased in striatum by acute administration of l-tyrosine.
The metabolism of catecholamines, such as dopamine and norepinephrine is probably associated with free radical formation and conditions associated with increased catecholamine metabolism may increase the free radical burden. In this context, studies have shown that the brain has different responses to dopamine syntheses in accordance with the administration of tyrosine since the tyrosine is responsible for syntheses of catecholamines (epinephrine and dopamine) [37]. Stoerner et al. [38] have demonstrated that a high level of tyrosine in serum can affect metabolism of dopamine and serotonin in the brain. Tyrosine hydroxylase catalyzes the enzymatic conversion of l-tyrosine to catecholamines and is present in both noradrenergic and dopaminergic terminals of the hippocampus and striatum [39], but its sub-regional distribution is not well characterized. It is known that endogenously available dopamine in nigral neurons can undergo autoxidation and form reactive quinones that attack and potentially inhibit the function of intracellular proteins. In addition to dopamine autoxidation and metabolism of dopamine by monoamine oxidase can increase H2O2 production and iron-dependent ROS production [40]. Considering that the enhanced generation of reactive species and/or impaired antioxidant detoxification functions contributes to an imbalance between oxidative and reductive reactions, leading to oxidative stress [41], we suggest that our results are parameter indicative of oxidative stress in hippocampus and striatum as a consequence of increased dopamine synthesis.
In summary, l-tyrosine induces oxidative stress in cerebellum, hippocampus and striatum of young rats, suggesting the oxidative stress may contribute, along with other mechanisms, to the neurological dysfunction observed in hypertyrosinemias and the administration of antioxidants may be considered as a potential adjuvant therapy for tyrosinemias, especially type II. Unfortunately, it is almost impossible to indicate which l-tyrosine is the responsible for the observed effects, given the vast number of tyrosine metabolites (4-hydroxyphenylpyruvic acid, 4-hydroxyphenyllatic acid, 4-hydroxyphenylacetic acid, N-acetyltyrosine, and 4-tyramine) observed in hypertyrosinemic patients [1, 21].
References
Mitchell GA, Grompe M, Lambert M, Tanguay RM (2001) Hypertyrosinemia. In: Scriver CR, Beaudet AL, Sly WS, Valle D (eds) The metabolic and molecular bases of inherited disease. McGraw-Hill, New York, pp 1977–1982
Buist NR, Kennaway NG, Fellman JH (1995) Tyrosinaemia type II. In: Bickel H, Wachtel V (eds) Inherited diseases of aminoacid metabolism, 1st edn. Georg Thieme Verlag, Stuttgart, pp 203–235
Light IJ, Sutherland JM, Berry HK (1973) Clinical significance of tyrosinemia of prematurity. J Inherit Metab Dis 12:13–22
Mamunes P, Prince PE, Thornton NH, Hunt PA, Hitchcock ES (1976) Intellectual deficits after transient Mediators Inflamm 7:239–255
Sener RN (2005) Tyrosinemia-computed tomography, magnetic resonance imaging, diffusion magnetic resonance imaging, and proton spectroscopy findings in the brain. J Comput Assist Tomogr 29:323–325
Sgaravatti AM, Magnusson AS, de Oliveira AS, Rosa AP, Mescka CP, Zanin FR, Pederzolli CD, Wyse AT, Wannmacher CM, Wajner M, Dutra-Filho CS (2009) Tyrosine administration decreases glutathione and stimulates lipid and protein oxidation in rat cerebral cortex. Metab Brain Dis 24:415–425
Sgaravatti AM, Vargas BA, Zandoná BR, Deckmann KB, Rockenback FJ, Moraes TB, Monserrat JM, Sgarbi MB, Pederzolli CD, Wyse ATS, Wannmacher CMD, Wajner M, Dutra-Filho CS (2008) Tyrosine promotes oxidative stress in cerebral cortex of young rats. Int J Dev Neurosci 26:553–559
de Andrade RB, Gemelli T, Rojas DB, Funchal C, Dutra-Filho CS, Wannmacher CM (2011) Tyrosine inhibits creatine kinase activity in cerebral cortex of young rats. Metab Brain Dis 26:221–227
de Andrade RB, Gemelli T, Rojas DB, Funchal C, Dutra-Filho CS, Wannmacher CM (2012) Tyrosine impairs enzymes of energy metabolism in cerebral cortex of rats. Mol Cell Biochem 364:253–261
Halliwell B (1994) Free radicals, antioxidants and human disease: curiosity, cause or consequence? Lancet 344:721–724
Halliwell B (2001) Role of free radicals in the neurodegenerative diseases: therapeutic implications for antioxidant treatment. Drugs Aging 18:685–716
Reznick AZ, Parker L (1993) Free radicals and antioxidants in muscular neurological diseases and disorders. In: Poli G, Albano E, Dianzani MU (eds) Free radicals: from basic science to medicine. Birkäuser Verlag, Basel, pp 425–437
Przedborski S, Donaldson DBS, Jakowec M, Kish SJ, Guttman M, Rosoklija G, Hays AP (1996) Brain superoxide dismutase, catalase and glutathione peroxidase activities in amyotrophic lateral sclerosis. Ann Neurol 39:158–165
Ben-Menachem E, Killerman R, Markleind S (2000) Superoxide dismutase and glutathione peroxidase function in progressive myoclonus epilepsies. Epilepsy Res 40:29–33
Slotkin TA, Oliver CA, Seidler FJ (2005) Critical periods for the role of oxidative stress in the developmental neurotoxicity of chlorpyrifos and terbutaline, alone or in combination. Brain Res Dev Brain Res 157:172–180
Halliwell B, Gutteridge JMC (2007) Measurement of reactive species. In: Halliwell B, Gutteridge JMC (eds) Free radicals in biology and medicine, 4th edn. Oxford University Press, Oxford, pp 268–340
Halliwell B (1991) Reactive oxygen species in living systems: source, biochemistry, and role in human disease. Am J Med 91:14S–22S
Yu BP (1994) Cellular defenses against damage from reactive oxygen species. Physiol Rev 74:139–162
Bannister JV, Bannister WH, Rotilio G (1987) Aspects of the structure, function and applications of superoxide dismutase. CRC Crit Rev Biochem 22:111–180
Bongiovanni R, Yamamoto BK, Simpson C, Jaskiw GE (2003) Pharmacokinetics of systemically administered tyrosine: a comparison of serum, brain tissue and in vivo microdialysate levels in the rat. J Neurochem 87:310–317
Morre MC, Hefti F, Wurtman RJ (1980) Regional tyrosine levels in rat brain after tyrosine administration. J Neural Transm 49:45–50
Held PK (2006) Disorders of tyrosine catabolism. Mol Genet Metab 88:103–106
Lowry OH, Rosebough NG, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Draper HH, Hadley M (1990) Malondialdehyde determination as index of lipid peroxidation. Methods Enzymol 186:421–431
Levine RL, Williams JA, Stadtman ER, Shacter E (1994) Carbonyl assays for determination of oxidatively modified proteins. Methods Enzymol 233:346–357
Bannister JV, Calabrese L (1987) Assays for superoxide dismutase. Methods Biochem Anal 32:279–312
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126
Marklund SL (1985) Pyrogallol autoxidation. In: Greenwald RA (ed) Handbook of Methods for Oxygen Radical Research, CRC Press Inc, pp 243–247
Bird S, Miller NJ, Collins JE, Rice-Evans A (1995) Plasma antioxidant capacity in two cases of tirosinaemia type 1: one case treated with NTBC. J Inherit Metab Dis 18:123–126
Colome C, Sierra C, Vilaseca MA (2000) Congenital errors of metabolism: cause of oxidative stress? Med Clin 115:111–117
Latini A, Ferreira CG, Scussiato K, Schuck PF, Dutra-Filho CS, Vargas CR, Wajner M (2007) Induction of oxidative stress by chronic and acute glutaric acid administration to rats. Cell Mol Neurobiol 27:423–438
Sitta A, Barschak AG, Deon M, Terroso T, Pires R, Giugliani R, Dutra-Filho CS, Wajner M, Vargas CR (2006) Investigation of oxidative stress parameters in treated phenylketonuric patients. Metab Brain Dis 20:287–296
Streck EL, Vieira PS, Wanmacher CM, Dutra-Filho CS, Wajner M, Wyse AT (2003) In vitro effect of homocysteine on some parameters of oxidative stress in rat hippocampus. Metab Brain Dis 18:147–154
Wajner M, Latini A, Wyse ATS, Dutra-Filho CS (2004) The role of oxidative damage in the neuropathology of organic acidurias: insights from animal studies. J Inherit Metab Dis 27:427–448
Barschak AG, Sitta A, Deon M, Oliveira MH, Haeser A, Dutra-Filho CS, Wajner M, Vargas CR (2006) Evidence that oxidative stress is increased in plasma from patients with maple syrup urine disease. Metab Brain Dis 21:279–286
Stadtman ER, Levine RL (2003) Free radical-mediated oxidation of free amino acids and amino acid residues in proteins. Amino Acids 25:207–218
Scott CR (2006) The genetic tyrosinemias. Am J Med Genet C Med Genet 142:121–126
Stoerner JW, Butler IJ, Morriss FH Jr, Howell RR Jr, Seifert WE Jr, Caprioli RM, Adcock EW, Denson SE (1980) CSF neurotransmitter studies. An infant with ascorbic acid-responsive tyrosinemia. Am J Dis Child 134:492–494
Schmidt RH, Bhatnagar RK (1979) Assessment of the effects of neonatal subcutaneous 6-hydroxydopamine on noradrenergic and dopaminergic innervation of the cerebral cortex. Brain Res 166:309–319
Jana S, Sinha M, Chanda D, Roy T, Banerjee K, Munshi S, Patro BS, Chakrabarti S (2011) Mitochondrial dysfunction mediated by quinone oxidation products of dopamine: implications in dopamine cytotoxicity and pathogenesis of Parkinson’s disease. Biochim Biophys Acta 1812:663–673
Moriarty-Craige SE, Jones DP (2004) Extracellular thiols and thiol/disulfide redox in metabolism. Annu Rev Nutr 24:481–509
Acknowledgments
This study was supported by grants from Conselho Nacional de Pesquisa e Desenvolvimento (CNPq), Fundação de Apoio à Pesquisa Científica e Tecnológica do Estado de Santa Catarina (FAPESC) and Universidade do Extremo Sul Catarinense (UNESC).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Macêdo, L.G.R.P., Carvalho-Silva, M., Ferreira, G.K. et al. Effect of Acute Administration of l-Tyrosine on Oxidative Stress Parameters in Brain of Young Rats. Neurochem Res 38, 2625–2630 (2013). https://doi.org/10.1007/s11064-013-1180-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-013-1180-3