Abstract
Purpose
Since frameless stereotactic radiosurgery (SRS) techniques have been recently introduced, hypofractionated SRS (HF-SRS) for large brain metastases (BMs) is gradually increasing. To verify the efficacy and safety of HF-SRS for large BMs, we aimed to perform a systematic review and compared them with SF-SRS.
Methods
We systematically searched the studies regarding SF-SRS or HF-SRS for large (> 2 cm) BM from databases including PubMed, Embase, and the Cochrane Library on July 31, 2018. Biologically effective dose with the α/β ratio of 10 (BED10), 1-year local control (LC), and radiation necrosis (RN) were compared between the two groups, with the studies being weighted by the sample size.
Results
The 15 studies with 1049 BMs that described 1-year LC and RN were included. HF-SRS tended to be performed in larger tumors; however, higher mean BED10 (50.1 Gy10 versus 40.4 Gy10, p < 0.0001) was delivered in the HF-SRS group, which led to significantly improved 1-year LC (81.6 versus 69.0%, p < 0.0001) and 1-year overall survival (55.1 versus 47.2%, p < 0.0001) in the HF-SRS group compared to the SF-SRS group. In contrast, the incidence of radiation toxicity was significantly decreased in the HF-SRS group compared to the SF-SRS group (8.0 versus 15.6%, p < 0.0001).
Conclusion
HF-SRS results in better LC of large BMs while simultaneously reducing RN compared to SF-SRS. Thus, HF-SRS should be considered a priority for SF-SRS in patients with large BMs who are not suitable to undergo surgical resection.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Stereotacic radiosurgery (SRS) in brain metastases (BMs) has been performed for decades and is usually confined to small tumors. Large BMs have been hindered from SRS and treated with either surgical resection or conventional radiation therapy; however, recent advancements in frameless and image-guided SRS techniques have allowed fractionated SRS, and SRS for large BMs using fractionation has been increasingly performed for the last ten years [1].
The local control (LC) of single fraction SRS (SF-SRS) for BM has been well known to be linked to the prescription dose. Several authors reported that LC was significantly improved when the BMs were irradiated using SF-SRS with a minimal dose over 18 Gy [2, 3]. Additionally, a dose of ≥ 16 Gy is reportedly sufficient to achieve an acceptable LC, whereas a study that investigated the correlation between the prescription dose and the LC in BMs < 26 mm in a diameter (< 10 cm3 in a volume) recommended giving a dose ≥ 25 Gy to achieve a satisfactory LC [4]. The higher the radiation dose delivered to the BM, the better the LC attained, and overall, the 1-year LC of BMs after SF-SRS has been reported to roughly range from 80 to 90%: > 90% for small and non-small cell lung cancer with a median overall survival (OS) of 14 to 18 months [5, 6]; 86–94% for breast cancer with a median OS of 14.5 months [7]; 75–94% for melanoma [8]; and 94% for renal cell cancer [9]. Matsuo et al. reported that the minimal dose to the BMs (Dosemin) is the single most significant predictor of LC, and they recommended 25 Gy for BMs < 10 cm3 and 20 Gy for BMs ≥ 10 cm3 [4]. Shiau et al. also verified, from their study of the correlation between the prescription dose and LC, that the higher the dose delivered, the better the LC achieved [2]. Similarly, we are able to attain improved LC by radiation dose escalation; however, tumor size limits radiation dose escalation because the larger the tumor treated with SF-SRS is, the more likely local toxicity occurs, such as radiation-induced edema and radiation necrosis (RN). Therefore, to reduce the risk of RN to less than 3%, large tumors have generally been treated with a reduced radiation dose than the originally required dose, which frequently leads to local failure [2, 10,11,12,13]. According to a study of the relationship between the tumor volume, dose and local control performed by Richard et al., the stepwise reduction in marginal dose to tumor according to increasing tumor size, from 21–25 Gy, 18–20 Gy, to 12–15 Gy, caused a remarkable decline of 1-year LC from 82, 64, to 37%, respectively [13]; however, since the introduction of fractionated SRS, SRS practice has been shifting towards maintaining an adequate marginal dose through fractionation rather than dose reduction for large BMs, and favorable LC and toxicity have been reported by several institutes. Here, we aimed to perform a systematic review of SRS for large BMs over 2 cm in diameter (or 3 cm3 in volume) to compare the efficacy and safety of the two treatment modalities of SF-SRS and hypofractionated SRS (HF-SRS).
Methods
Search strategy
We systematically searched the studies regarding SF-SRS or HF-SRS for large (> 2 cm) BM from databases including PubMed, Embase, and the Cochrane Library on July 31, 2018. The detailed search strategy is presented in Supplementary Table 1. A manual search of reference lists from previous relevant studies and reviews was also conducted to further identify relevant literature.
Eligibility criteria
Both retrospective and prospective studies performed in humans were included in this analysis, with case reports, conference abstracts, and review articles being excluded. Studies written in a language other than English were excluded. If the contents of the abstract were considered compatible with this study, the full text of the study was entirely reviewed by two independent researchers (EJL and ESP). This review was focused on large metastatic brain tumors ≥ 2 cm in diameter (or 3 cm3 in volume) treated with either SF-SRS or HF-SRS. Hypofraction was defined as the number of fractions between 2 and 5. We considered radiation therapy as SRS if it was performed in a stereotactic manner with a safety margin within 3 mm even though it was described as “stereotactic radiotherapy”. The primary end point was 1-year LC after radiosurgery, and the secondary end point was RN. Only studies that reached a consensus between the two authors were included in the systematic review.
Data extraction
Two reviewers (EJL and ESP) independently reviewed the full text of each included study and extracted data using a standardized form. The abstracted data included the name of the author, publication year, sample size, tumor size (mean and range), study design, treatment modality, SRS dosimetry, 1-year LC, 1-year number at risk, 1-year OS, RN, and medical follow-up period. The biologically effective dose (BED) was estimated from the dose prescribed to the isocenter of the tumor using a linear-quadratic model assuming α/β to be 10 Gy [14].
Statistical analysis
After database searching and data extraction, we performed a systematic review in which the studies reported treatment results after SF-SRS or HF-SRS for large (> 2 cm) BMs. Selected study populations were divided into 2 categories by treatment modality, SF-SRS and HF-SRS. To compare the integrated data of the SF-SRS and HF-SRS groups, Mann–Whitney U tests [15] were performed for continuous nonparametric variables, with the studies being weighted by the sample size. Differences with p values < 0.05 were considered statistically significant. All statistical analyses were performed using SPSS v.22.0 (SPSS Inc., Chicago, IL).
Results
We found 289 articles from a search of PubMed, 240 from EMBASE, 22 from the Cochrane Library, and 1 additional article from manual searching. A total of 362 studies were identified after removing duplicates, and 347 potentially relevant articles were retrieved for full-text review. The 15 studies selected that described 1-year LC and RN for patients treated with SF-SRS or HF-SRS for BM ≥ 2 cm (or 3 cm3), and data on 1049 BMs were extracted (Fig. 1) [16,17,18,19,20,21,22,23,24,25,26,27,28,29,30]. Among these 15 publications, five studies covered only BMs treated with SF-SRS [26,27,28,29,30], seven studies dealt with only BMs treated with HF-SRS [16, 17, 19,20,21, 23, 24], and three studies included both BMs treated either SF-SRS or HF-SRS [18, 22, 25]. For the three studies that included both BMs treated with either SF- or HF-SRS, we collected data separately for SF- and HF-SRS groups.
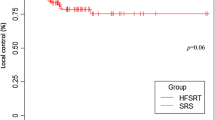
The studies that were finally included in this review are summarized in Tables 1, 2. All studies had mixed primary cancer pathologies, with lung cancer being the majority, except the study conducted by Flannery and colleagues that included only BM from prostate carcinoma [27]. In the SF-SRS group, eight studies were included, encompassing 463 composite BMs, and ten studies with a composite BM number of 586 in the HF-SRS group. HF-SRS was mostly delivered daily on consecutive days. Three studies directly compared the outcomes of SF-SRS and HF-SRS in large BMs [18, 22, 25]. Minniti et al. performed SF-SRS and HF-SRS as primary treatments for 179 and 164 BMs > 2.0 cm in diameter, respectively [22]. The tumor size of the HF-SRS group ranged from 4.1 to 47.9 cm3, which was significantly larger than that of the SF-SRS group (range, 3.1–24.1 cm3; p = 0.005). In the SF-SRS group, 18 Gy (BED10 = 50.4 Gy10) was delivered for BMs of 2–3 cm and 15–16 Gy (BED10 = 37.5–41.6 Gy10) for BMs ≥ 3 cm, whereas in the HF-SRS group, 27 Gy was delivered in 3 fractions (BED10 = 51.3 Gy10) at 80–90% isodose line. During a median radiologic follow-up of 10 months, BMs treated with SF-SRS recurred much more frequently than those treated with HF-SRS (16.6 vs 8%, p = 0.03), and the 1-year LC rate of SF-SRS was also significantly lower than that of HF-SRS (77 vs 91%, p = 0.01). On the other hand, RN occurred significantly more often in the SF-SRS group than in the HF-SRS group (20 vs 8%, p = 0.008). Propensity score matching analysis also demonstrated favorable outcomes in the HF-SRS group in terms of both LC (p = 0.01) and RN risk (p = 0.005). Feuvret et al. also reported the treatment results of SF-SRS and HF-SRS in BM > 3 cm in diameter [18]. In the SF-SRS group, 14 Gy (BED10 = 33.6 Gy10) was prescribed in 24 BMs with a GTV ranging from 4.5 to 33.1 cm3, whereas in the HF-SRS group, 7.7 Gy per day was delivered for 3 consecutive days (BED10 = 40.9 Gy10) in 12 BMs with a GTV ranging from 12.5 to 52.5 cm3. The size of tumors treated with HF-SRS was significantly larger than those treated with SF-SRS (p = 0.02). The 1-year LC rate tended to be superior in the HF-SRS group than in the SF-SRS group (100 vs 58%, p = 0.06). No RN was recorded in either SRS group during the 7-month median follow-up period. In contrast, another study that directly compared the SF- and HF-SRS for large BMs did not show a difference in either the 1-year LC (67 vs 75%, p = 0.27) or 1-year free from RN (85 vs 75%, p = 0.25) rates [25]. For 46 BMs, 15 Gy was delivered in a single fraction (BED10 = 37.5 Gy10), whereas 24 Gy was delivered in 3 fractions (BED10 = 43.2 Gy10) for 65 BMs. The composite results of the SF-SRS and HF-SRS groups are compared in Table 3. The mean BED10 (p = 0.021) was significantly higher in the composite HF-SRS group, which led to significantly improved 1-year LC (p = 0.031) by 17% and 1-year OS (p = 0.042) by 22% compared with the composite SF-SRS group; however, the incidence of radiation toxicity was comparable between the two groups. When analyzed by applying a weight to each study’s sample size, radiation toxicity occurred at a significantly lower rate in the HF-SRS group (8.0 versus 15.6%, p < 0.0001) with the higher mean BED10 (51.2 Gy10 versus 44.1 Gy10, p < 0.0001), while and 1-year LC (81.6 versus 69.0%, p < 0.0001) and 1-year OS (55.1 versus 47.2%, p < 0.0001) were significantly higher (Table 4).
Discussion
The prolonged survival of cancer patients due to advancements of molecular biology and targeted therapies has inevitably led to a gradual increase in the incidence of brain metastasis including large-sized tumors [31, 32]. Such chemotherapy developments have raised the expectation that even patients with advanced disease status can be treated, which subsequently increases the requirement to treat BM actively compared to the past. Meanwhile, as patients inappropriate for surgical resection increase due to age, comorbidities, multiple lesions, and the need for continuity of chemotherapy, demand for SRS as an attractive alternative to surgery is increasing. Additionally, SRS has become the preferred treatment over WBRT for patients with a limited number of BMs because it is less likely to cause neurocognitive decline as a late adverse effect of radiation [33]; however, SRS, in which a single high-dose radiation is delivered to the target in the conventional approach, can cause radiation toxicity by irradiating a correspondingly large amount of surrounding normal tissues for the treatment of a large BM [34,35,36]. The development of radiation necrosis is correlated with the volume of normal brain tissue irradiated with a dose ≥ 12 Gy [37, 38]. Therefore, before the era of hypofractionated SRS, SRS was performed using a reduced radiation dose to avoid radiation toxicity; however, it may lead to treatment failure. Irradiation in fractionation is a basic strategy of conventional radiation therapy. Fractionation allows for tumor cells to be redistributed in the cell cycle and reoxygenated to make them radiosensitive, which enhances therapeutic efficacy and permits injured normal tissues to be repaired and repopulated between treatments, decreasing radiation toxicity. Such a fractionation concept has also been applied to SRS in the form of HF-SRS in the hope of minimizing the risk of RN while maintaining BED to the tumors [33, 39]. Keeping a rigid frame on the head for several days was a considerable barrier to performing HF-SRS; however, since the frameless SRS system’s advent more than a decade ago, HF-SRS has become a popular treatment for large BM. HF-SRS started with the hypothesis that it would lessen RN risk while achieving LC comparable to SF-SRS in treating large BM [18, 23, 32, 40, 41]. To verify these assumptions, we performed a systematic review of the efficacy and safety of HF-SRS compared to SF-SRS. Fifteen studies of large BM SRS were selected in total, including three studies directly comparing SF-SRS and HF-SRS. The composite data estimated that higher BED10 was delivered in the HF-SRS group, leading to a significant improvement in 1-year LC and 1-year OS, whereas decreasing the radiation toxicity rate, compared to the SF-SRS group. Also, planning target volume expansion with the 1–3 mm margin in HF-SRS may be associated with better LC in the HF-SRS group than the SF-SRS group that mainly did not include a margin. Lehrer et al. have recently performed a meta-analysis to compare the efficacy and safety between SF-SRS and HF-SRS for large BMs > 2 cm [32]. Fifteen studies with 1157 large BMs were included to analyze 1-year LC, including two studies that directly compared SF-SRS and MF-SRS. The authors stratified the analysis of 1-year LC by dichotomizing the tumor size between 2–3 cm diameter (4–14 cm3) versus > 3 cm diameter (> 14 cm3). Overall, meta-analyses did not confirm the superiority of HF-SRS in tumor control compared to SF-SRS; however, in the tumor size group of 2–3 cm diameter, there was a trend of better 1-year LC in the HF-SRS group than the SF-SRS group (92.9 versus 77.1%, p = 0.18). On the other hand, HF-SRS significantly reduced RN risk compared to SF-SRS in the tumor size group of 2–3 cm diameter, while there was a trend of reduced RN incidence in HF-SRS groups in the tumor size group of > 3 cm diameter. Since the present study included only three studies that directly compared the results between SF-SRS and HF-SRS, we were concerned that meta-analysis with limited data would provide unsatisfactory or biased results. Instead, we integrated the data from the SF-SRS and HF-SRS groups and compared the two groups via the Mann–Whitney U tests, which weighted the study sample size, resulting in a significant improvement in 1-year LC and decreased RN in the HF-SRS group over the SF-SRS group. Meanwhile, a recent prospective study comparing SF-SRS (n = 67, median dose 20 Gy) and HF-SRS (n = 38, median cumulative dose 35 Gy in 5 fractions) for 2.5–3 cm sized BMs reported a significant decrease in the incidence of RN (29.9 vs. 5.3%, p < 0.001) and improvement in 1-year LC (66.6 vs. 92.4%, p = 0.028) in the HF-SRS groups with no difference in OS (median 13 months vs. 18 months, p = 0.239) between the two regimens [42].
Taken together, the results of SRS studies on large BMs indicate that HF-SRS may be a better treatment option for large BMs when considering efficacy and safety [43]. HF-SRS combines the advantages of SRS, which is precise administration of high-dose radiation to the target, and fractionation that allows repair, repopulation, redistribution, and reoxygenation [39, 44]. Whether it would be possible to lower the RN incidence even with a fraction number of 2–5 that is significantly smaller than that of conventional radiation therapy was unclear. Additionally, there was a concern that decreased dose per fraction in HF-SRS might lead to deteriorating tumor control in comparison with SF-SRS because high dose radiation in a single fraction is thought to be preferable to fractionated radiation therapy to control radioresistant tumors, such as BMs from hepatocellular carcinoma and melanoma. High-dose radiation is also believed to elicit severe damage in the tumor vessels, by which it exerts an additional antitumor effect [39]. Therefore, whether a decreased dose per fraction in HF-SRS would still preserve its influence on the vasculature was uncertain. However, a myriad of studies of HF-SRS has recently reported that fractionation with a limited number not only reduces RN risk even with administration of higher BED but also may improve tumor control despite decreased dose per fraction compared to SF-SRS. Radiation-induced DNA damage leads to accelerated senescence or cell death (apoptosis, autophagy, or necrosis) [45,46,47], depending on the radiation dose and radiosensitivity of the exposed tissue, with higher doses causing necrosis [48,49,50]. Unlike apoptosis, the necrosis process is associated with inflammation in surrounding normal tissue [51, 52]. The dose per fraction currently used in the HF-SRS seems to be high enough to treat malignant tumor cells but low enough not to induce the necrotic process well. Meanwhile, better LC in the HF-SRS group is generally considered to result from irradiation with higher BED. In addition to this point of view, we speculate that the acquisition of tumor cells radiosensitivity due to redistribution in the cell cycle and reoxygenation may play an important role in the local control of large BMs than expected [53, 54]. How large tumors can be safely and effectively treated using HF-SRS instead of surgical resection and whether HF-SRS will be more beneficial than SF-SRS even in small tumors remains unclear. Thus, the window of tumor size in which HF-SRS is helpful and the optimal dose per fraction and dose rate according to tumor size need to be established.
Meanwhile, as large peptide molecules such as monoclonal antibodies have recently been revealed to partially penetrate the blood–brain barrier in BM patients and exert tumor control effect, a combinational approach of SRS plus targeted or immunotherapy (TT/IT) has attracted attention [55]. Although data are limited and mostly retrospective, SRS may potentially synergistically affect LC and OS when combined with certain TT (in patients with EGFR-mutated NSCLC; HER 2-mutated breast cancer; and BRAFV600E-mutated melanoma) or IT, compared to SRS alone [56,57,58]. ITs are profitable to patients lacking a driver mutation who do not benefit from novel targeted therapies. Regarding the optimal timing of IT treatment in BM patients, several studies demonstrated that concurrent administration of IT with SRS or immediately after SRS conferred better LC or OS than IT prior to SRS [58,59,60]. There are conflicting results in the toxicity issue of combining SRS with TT/IT in BM, with symptomatic RN and intra-tumor hemorrhage being increased especially those with melanoma [57, 61]. Unfortunately, the studies included in this review did not specify adjuvant therapies, including TT/IT, except the study conducted by Flannery and colleagues [27]. Therefore, it was not possible to compare the effects of TT/IT on the treatment outcomes of SF-SRS and HF-SRS. The synergistic pros and cons of SRS with TT/IT need to be compared between the HF-SRS and SF-SRS groups in the future.
Limitations
The tumor α/β ratio may vary with the pathology: 10–30 Gy for squamous cell cancer and 4–5 Gy for breast cancer [62]. However, the studies except one included in this review had mixed primary cancer pathologies, with lung cancer being the majority. Given the complexity of calculating the BED with different α/β according to pathology, we used an α/β of 10 as the representative value for metastatic brain tumors to conceptually compare the difference in BED between the SF-SRS and HF-SRS groups. Moreover, we estimated the BED based on a linear-quadratic model, which does not sufficiently represent the complexity of the underlying biology, although it is widely used. Whether the linear-quadratic model is applicable to estimate the BED in HF-SRS has not been verified. The time between fractions in HF-SRS may influence BED; however, we considered the effect of tumor proliferation during the short fractionation schedule to be imperceptible in general and did not reflect it in the BED calculation formula. Due to these limitations of BED estimation, it is necessary to be cautious when comparing BED between the SF-SRS and the HF-SRS groups.
Conclusions
Hypofractionation in SRS enables higher BED10 administration to the target, resulting in better local control of large BMs, and simultaneously reduces radiation toxicity, compared to SF-SRS. Thus, HF-SRS should be considered a priority for SF-SRS in patients with large BMs who are not suitable to undergo surgical resection.
References
Jeong WJ, Park JH, Lee EJ, Kim JH, Kim CJ, Cho YH (2015) Efficacy and safety of fractionated stereotactic radiosurgery for large brain metastases. J Korean Neurosurg Soc 58:217–224. https://doi.org/10.3340/jkns.2015.58.3.217
Shiau CY, Sneed PK, Shu HKG, Lamborn KR, McDermott MW, Chang S, Nowak P, Petti PL, Smith V, Verhey LJ, Ho M, Park E, Wara WM, Gutin PH, Larson DA (1997) Radiosurgery for brain metastases: Relationship of dose and pattern of enhancement to local control. Int J Radiat Oncol Biol Phys 37:375–383. https://doi.org/10.1016/S0360-3016(96)00497-X
Breneman JC, Warnick RE, Albright RE Jr, Kukiatinant N, Shaw J, Armin D, Tew J Jr (1997) Stereotactic radiosurgery for the treatment of brain metastases. Results of a single institution series. Cancer 79:551–557
Matsuo T, Shibata S, Yasunaga A, Iwanaga M, Mori K, Shimizu T, Hayashi N, Ochi M, Hayashi K (1999) Dose optimization and indication of linac radiosurgery for brain metastases. Int J Radiat Oncol Biol Phys 45:931–939. https://doi.org/10.1016/S0360-3016(99)00271-0
Gerosa M, Nicolato A, Foroni R, Tomazzoli L, Bricolo A (2005) Analysis of long-term outcomes and prognostic factors in patients with non-small cell lung cancer brain metastases treated by gamma knife radiosurgery. J Neurosurg 102:75–80. https://doi.org/10.3171/jns.2005.102.s_supplement.0075
Sheehan J, Kondziolka D, Flickinger J, Lunsford LD (2005) Radiosurgery for patients with recurrent small cell lung carcinoma metastatic to the brain: outcomes and prognostic factors. J Neurosurg 102:247–254. https://doi.org/10.3171/jns.2005.102.s_supplement.0247
Muacevic A, Kreth FW, Tonn JC, Wowra B (2004) Stereotactic radiosurgery for multiple brain metastases from breast carcinoma—feasibility and outcome of a local treatment concept. Cancer 100:1705–1711. https://doi.org/10.1002/cncr.20167
Selek U, Chang EL, Hassenbusch SJ, Shiu AS, Lang FF, Allen P, Weinberg J, Sawaya R, Maor MH (2004) Stereotactic radiosurgical treatment in 103 patients for 153 cerebral melanoma metastases. Int J Radiat Oncol Biol Phys 59:1097–1106. https://doi.org/10.1016/j.ijrobp.2003.12.037
Muacevic A, Siebels M, Tonn JC, Wowra B (2005) Treatment of brain metastases in renal cell carcinoma: radiotherapy, radiosurgery, or surgery? World J Urol 23:180–184. https://doi.org/10.1007/s00345-004-0471-3
Alexander E 3rd, Moriarty TM, Davis RB, Wen PY, Fine HA, Black PM, Kooy HM, Loeffler JS (1995) Stereotactic radiosurgery for the definitive, noninvasive treatment of brain metastases. J Natl Cancer Inst 87:34–40. https://doi.org/10.1093/jnci/87.1.34
Mori Y, Kondziolka D, Flickinger JC, Kirkwood JM, Agarwala S, Lunsford LD (1998) Stereotactic radiosurgery for cerebral metastatic melanoma: Factors affecting local disease control and survival. Int J Radiat Oncol Biol Phys 42:581–589. https://doi.org/10.1016/S0360-3016(98)00272-7
Noel G, Simon JM, Valery CA, Cornu P, Boisserie G, Hasboun D, Ledu D, Tep B, Delattre JY, Marsault C, Baillet F, Mazeron JJ (2003) Radiosurgery for brain metastasis: impact of CTV on local control. Radiother Oncol 68:15–21. https://doi.org/10.1016/S0167-8140(03)00207-X
Molenaar R, Wiggenraad R, Verbeek-de Kanter A, Walchenbach R, Vecht C (2009) Relationship between volume, dose and local control in stereotactic radiosurgery of brain metastasis. Br J Neurosurg 23:170–178. https://doi.org/10.1080/02688690902755613
van Leeuwen CM, Oei AL, Crezee J, Bel A, Franken NAP, Stalpers LJA, Kok HP (2018) The alfa and beta of tumours: a review of parameters of the linear-quadratic model, derived from clinical radiotherapy studies. Radiat Oncol. https://doi.org/10.1186/s13014-018-1040-z
McClelland S 3rd, Ford B, Senatus PB, Frucht SJ, Winfield LM, Yu Q, Du YE, Pullman SL, McKhann GM 2nd, Goodman RR (2009) Typical variations of subthalamic electrode location do not predict limb motor function improvement in Parkinson’s disease. J Clin Neurosci 16:771–778. https://doi.org/10.1016/j.jocn.2008.10.011
Aoyama H, Shirato H, Onimaru R, Kagei K, Ikeda J, Ishii N, Sawamura Y, Miyasaka K (2003) Hypofractionated stereotactic radiotherapy alone without whole-brain irradiation for patients with solitary and oligo brain metastasis using noninvasive fixation of the skull. Int J Radiat Oncol Biol Phys 56:793–800. https://doi.org/10.1016/s0360-3016(03)00014-2
Ogura K, Mizowaki T, Ogura M, Sakanaka K, Arakawa Y, Miyamoto S, Hiraoka M (2012) Outcomes of hypofractionated stereotactic radiotherapy for metastatic brain tumors with high risk factors. J Neurooncol 109:425–432. https://doi.org/10.1007/s11060-012-0912-6
Feuvret L, Vinchon S, Martin V, Lamproglou I, Halley A, Calugaru V, Chea M, Valery CA, Simon JM, Mazeron JJ (2014) Stereotactic radiotherapy for large solitary brain metastases. Cancer Radiotherapie 18:97–106. https://doi.org/10.1016/j.canrad.2013.12.003
Murai T, Ogino H, Manabe Y, Iwabuchi M, Okumura T, Matsushita Y, Tsuji Y, Suzuki H, Shibamoto Y (2014) Fractionated stereotactic radiotherapy using cyberknife for the treatment of large brain metastases: a dose escalation study. Clin Oncol 26:151–158. https://doi.org/10.1016/j.clon.2013.11.027
Nagai A, Shibamoto Y, Yoshida M, Wakamatsu K, Kikuchi Y (2014) Treatment of single or multiple brain metastases by hypofractionated stereotactic radiotherapy using helical tomotherapy. Int J Mol Sci 15:6910–6924. https://doi.org/10.3390/ijms15046910
Kim JW, Park HR, Lee JM, Kim JW, Chung HT, Kim DG, Jung HW, Paek SH (2016) Fractionated stereotactic gamma knife radiosurgery for large brain metastases: a retrospective Single Center Study. PLoS ONE 11(9):e0163304. https://doi.org/10.1371/journal.pone.0163304
Minniti G, Scaringi C, Paolini S, Lanzetta G, Romano A, Cicone F, Osti M, Enrici RM, Esposito V (2016) Single-fraction versus multifraction (3 x 9 Gy) stereotactic radiosurgery for large (> 2 cm) brain metastases: a comparative analysis of local control and risk of radiation-induced brain necrosis. Int J Radiat Oncol Biol Phys 95:1142–1148. https://doi.org/10.1016/j.ijrobp.2016.03.013
Navarria P, Pessina F, Cozzi L, Ascolese AM, De Rose F, Fogliata A, Franzese C, Franceschini D, Tozzi A, D’Agostino G, Comito T, Iftode C, Maggi G, Reggiori G, Bello L, Scorsetti M (2016) Hypo-fractionated stereotactic radiotherapy alone using volumetric modulated arc therapy for patients with single, large brain metastases unsuitable for surgical resection. Radiat Oncol. https://doi.org/10.1186/s13014-016-0653-3
Marcrom SR, McDonald AM, Thompson JW, Popple RA, Riley KO, Markert JM, Willey CD, Bredel M, Fiveash JB (2017) Fractionated stereotactic radiation therapy for intact brain metastases. Adv Radiat Oncol 2:564–571. https://doi.org/10.1016/j.adro.2017.07.006
Wiggenraad R, Verbeek-de Kanter A, Mast M, Molenaar R, Kal HB, Lycklama A, Nijeholt G, Vecht C, Struikmans H (2012) Local progression and pseudo progression after single fraction or fractionated stereotactic radiotherapy for large brain metastases. A single centre study. Strahlenther Onkol 188:696–701. https://doi.org/10.1007/s00066-012-0122-3
Kano H, Niranjan A, Kondziolka D, Flickinger JC, Lunsford LD (2009) Stereotactic radiosurgery for pituitary metastases. Surg Neurol 72:248–255. https://doi.org/10.1016/j.surneu.2008.06.003
Flannery T, Kano H, Niranjan A, Monaco EA, Flickinger JC, Lunsford LD, Kondziolka D (2010) Stereotactic radiosurgery as a therapeutic strategy for intracranial metastatic prostate carcinoma. J Neurooncol 96:369–374. https://doi.org/10.1007/s11060-009-9966-5
Prabhu RS, Press RH, Patel KR, Boselli DM, Symanowski JT, Lankford SP, McCammon RJ, Moeller BJ, Heinzerling JH, Fasola CE, Asher AL, Sumrall AL, Buchwald ZS, Curran WJ Jr, Shu HG, Crocker I, Burri SH (2017) Single-fraction stereotactic radiosurgery (SRS) alone versus surgical resection and SRS for large brain metastases: a multi-institutional analysis. Int J Radiat Oncol Biol Phys 99:459–467. https://doi.org/10.1016/j.ijrobp.2017.04.006
Cummings M, Youn P, Bergsma DP, Usuki KY, Walter K, Sharma M, Okunieff P, Schell MC, Milano MT (2018) Single-fraction radiosurgery using conservative doses for brain metastases: durable responses in select primaries with limited toxicity. Neurosurgery 83:437–444. https://doi.org/10.1093/neuros/nyx427
Wolf A, Kvint S, Chachoua A, Pavlick A, Wilson M, Donahue B, Golfinos JG, Silverman J, Kondziolka D (2018) Toward the complete control of brain metastases using surveillance screening and stereotactic radiosurgery. J Neurosurg 128:23–31. https://doi.org/10.3171/2016.10.Jns161036
Kalkanis SN, Kondziolka D, Gaspar LE, Burri SH, Asher AL, Cobbs CS, Ammirati M, Robinson PD, Andrews DW, Loeffler JS, McDermott M, Mehta MP, Mikkelsen T, Olson JJ, Paleologos NA, Patchell RA, Ryken TC, Linskey ME (2010) The role of surgical resection in the management of newly diagnosed brain metastases: a systematic review and evidence-based clinical practice guideline. J Neurooncol 96:33–43. https://doi.org/10.1007/s11060-009-0061-8
Lehrer EJ, Peterson JL, Zaorsky NG, Brown PD, Sahgal A, Chiang VL, Chao ST, Sheehan JP, Trifiletti DM (2019) Single versus multifraction stereotactic radiosurgery for large brain metastases: an international meta-analysis of 24 trials. Int J Radiat Oncol Biol Phys 103:618–630. https://doi.org/10.1016/j.ijrobp.2018.10.038
Masucci GL (2018) Hypofractionated radiation therapy for large brain metastases. Front Oncol. https://doi.org/10.3389/fonc.2018.00379
Williams BJ, Suki D, Fox BD, Pelloski CE, Maldaun MV, Sawaya RE, Lang FF, Rao G (2009) Stereotactic radiosurgery for metastatic brain tumors: a comprehensive review of complications. J Neurosurg 111:439–448. https://doi.org/10.3171/2008.11.JNS08984
Blonigen BJ, Steinmetz RD, Levin L, Lamba MA, Warnick RE, Breneman JC (2010) Irradiated volume as a predictor of brain radionecrosis after linear accelerator stereotactic radiosurgery. Int J Radiat Oncol Biol Phys 77:996–1001. https://doi.org/10.1016/j.ijrobp.2009.06.006
Minniti G, Clarke E, Lanzetta G, Osti MF, Trasimeni G, Bozzao A, Romano A, Enrici RM (2011) Stereotactic radiosurgery for brain metastases: analysis of outcome and risk of brain radionecrosis. Radiat Oncol. https://doi.org/10.1186/1748-717x-6-48
Nedzi LA, Kooy H, Alexander E 3rd, Gelman RS, Loeffler JS (1991) Variables associated with the development of complications from radiosurgery of intracranial tumors. Int J Radiat Oncol Biol Phys 21:591–599. https://doi.org/10.1016/0360-3016(91)90675-t
Korytko T, Radivoyevitch T, Colussi V, Wessels BW, Pillai K, Maciunas RJ, Einstein DB (2006) 12 GY gamma knife radiosurgical volume is a predictor for radiation necrosis in non-AVM intracranial tumors. Int J Radiat Oncol Biol Phys 64:419–424. https://doi.org/10.1016/j.ijrobp.2005.07.980
Santacroce A, Kamp MA, Budach W, Hanggi D (2013) Radiobiology of radiosurgery for the central nervous system. Biomed Res Int 2013:362761. https://doi.org/10.1155/2013/362761
Jiang XS, Xiao JP, Zhang Y, Xu YJ, Li XP, Chen XJ, Huang XD, Yi JL, Gao L, Li YX (2012) Hypofractionated stereotactic radiotherapy for brain metastases larger than three centimeters. Radiat Oncol. https://doi.org/10.1186/1748-717x-7-36
Minniti G, D’Angelillo RM, Scaringi C, Trodella LE, Clarke E, Matteucci P, Osti MF, Ramella S, Enrici RM, Trodella L (2014) Fractionated stereotactic radiosurgery for patients with brain metastases. J Neurooncol 117:295–301. https://doi.org/10.1007/s11060-014-1388-3
Chon H, Yoon K, Lee D, Kwon DH, Cho YH (2019) Single-fraction versus hypofractionated stereotactic radiosurgery for medium-sized brain metastases of 2.5 to 3 cm. J Neurooncol 145:49–56. https://doi.org/10.1007/s11060-019-03265-1
Schlienger M, Nataf F, Huguet F, Pene F, Foulquier JN, Orthuon A, Roux FX, Touboul E (2010) Hypofractionated stereotactic radiotherapy for brain metastases. Cancer Radiother 14:119–127. https://doi.org/10.1016/j.canrad.2009.10.004
Larsson B, Leksell L, Rexed B, Sourander P, Mair W, Andersson B (1958) High-energy proton beam as a neurosurgical tool. Nature 182:1222–1223. https://doi.org/10.1038/1821222a0
Zhao W, Robbins ME (2009) Inflammation and chronic oxidative stress in radiation-induced late normal tissue injury: therapeutic implications. Curr Med Chem 16:130–143. https://doi.org/10.2174/092986709787002790
Panganiban RA, Mungunsukh O, Day RM (2013) X-irradiation induces ER stress, apoptosis, and senescence in pulmonary artery endothelial cells. Int J Radiat Biol 89:656–667. https://doi.org/10.3109/09553002.2012.711502
Panganiban RA, Snow AL, Day RM (2013) Mechanisms of radiation toxicity in transformed and non-transformed cells. Int J Mol Sci 14:15931–15958. https://doi.org/10.3390/ijms140815931
Rothkamm K, Lobrich M (2003) Evidence for a lack of DNA double-strand break repair in human cells exposed to very low x-ray doses. Proc Natl Acad Sci USA 100:5057–5062. https://doi.org/10.1073/pnas.0830918100
Lindsay KJ, Coates PJ, Lorimore SA, Wright EG (2007) The genetic basis of tissue responses to ionizing radiation. Br J Radiol 80:S2–S6. https://doi.org/10.1259/bjr/60507340
Surova O, Zhivotovsky B (2013) Various modes of cell death induced by DNA damage. Oncogene 32:3789–3797. https://doi.org/10.1038/onc.2012.556
Friedman EJ (2002) Immune modulation by ionizing radiation and its implications for cancer immunotherapy. Curr Pharm Des 8:1765–1780. https://doi.org/10.2174/1381612023394089
Mullins ME, Barest GD, Schaefer PW, Hochberg FH, Gonzalez RG, Lev MH (2005) Radiation necrosis versus glioma recurrence: conventional MR imaging clues to diagnosis. AJNR Am J Neuroradiol 26:1967–1972
Hall EJ, Brenner DJ (1993) The radiobiology of radiosurgery: rationale for different treatment regimes for AVMs and malignancies. Int J Radiat Oncol Biol Phys 25:381–385. https://doi.org/10.1016/0360-3016(93)90367-5
Ling CC, Lo YC, Larson DA (1995) Radiobiophysical aspects of stereotaxic radiation treatment of central nervous system diseases. Semin Radiat Oncol 5:192–196. https://doi.org/10.1054/SRAO00500192
Suh JH, Kotecha R, Chao ST, Ahluwalia MS, Sahgal A, Chang EL (2020) Current approaches to the management of brain metastases. Nat Rev Clin Oncol 17:279–299. https://doi.org/10.1038/s41571-019-0320-3
Jiang T, Min W, Li Y, Yue Z, Wu C, Zhou C (2016) Radiotherapy plus EGFR TKIs in non-small cell lung cancer patients with brain metastases: an update meta-analysis. Cancer Med 5:1055–1065. https://doi.org/10.1002/cam4.673
Tallet AV, Dhermain F, Le Rhun E, Noel G, Kirova YM (2017) Combined irradiation and targeted therapy or immune checkpoint blockade in brain metastases: toxicities and efficacy. Ann Oncol 28:2962–2976. https://doi.org/10.1093/annonc/mdx408
Kotecha R, Kim JM, Miller JA, Juloori A, Chao ST, Murphy ES, Peereboom DM, Mohammadi AM, Barnett GH, Vogelbaum MA, Angelov L, Suh JH, Ahluwalia MS (2019) The impact of sequencing PD-1/PD-L1 inhibitors and stereotactic radiosurgery for patients with brain metastasis. Neuro Oncol 21:1060–1068. https://doi.org/10.1093/neuonc/noz046
Knisely JP, Yu JB, Flanigan J, Sznol M, Kluger HM, Chiang VL (2012) Radiosurgery for melanoma brain metastases in the ipilimumab era and the possibility of longer survival. J Neurosurg 117:227–233. https://doi.org/10.3171/2012.5.JNS111929
Schoenfeld JD, Mahadevan A, Floyd SR, Dyer MA, Catalano PJ, Alexander BM, McDermott DF, Kaplan ID (2015) Ipilmumab and cranial radiation in metastatic melanoma patients: a case series and review. J Immunother Cancer 3:50. https://doi.org/10.1186/s40425-015-0095-8
Martin AM, Cagney DN, Catalano PJ, Alexander BM, Redig AJ, Schoenfeld JD, Aizer AA (2018) Immunotherapy and symptomatic radiation necrosis in patients with brain metastases treated with stereotactic radiation. JAMA Oncol 4:1123–1124. https://doi.org/10.1001/jamaoncol.2017.3993
Jones B, Dale RG, Deehan C, Hopkins KI, Morgan DA (2001) The role of biologically effective dose (BED) in clinical oncology. Clin Oncol R Coll Radiol 13:71–81. https://doi.org/10.1053/clon.2001.9221
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lee, E.J., Choi, KS., Park, E.S. et al. Single- and hypofractionated stereotactic radiosurgery for large (> 2 cm) brain metastases: a systematic review. J Neurooncol 154, 25–34 (2021). https://doi.org/10.1007/s11060-021-03805-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-021-03805-8