Abstract
Functional preservation is critical in glioma surgery, and the extent of resection influences survival outcome. Neoadjuvant chemotherapy is a promising option because of its potential to facilitate tumor shrinkage and maximum tumor resection. The object of this study was to assess the utility of the neoadjuvant strategy in a prospective series of gliomas with favorable molecular status. Twenty-six consecutive cases of diffuse gliomas of WHO grade II or III with either 1p19q codeletion or MGMT methylation were treated with upfront chemotherapy following maximal safe removal. In cases of incomplete initial surgery, second-look resection was intended after tumor volume decrease by chemotherapy. Among 22 evaluable cases, chemotherapy led to a median change in the sum of the product of perpendicular diameters of −35 %, and 14 out of the 22 cases (64 %) showed objective response. Second-look resection after tumor volume decrease was performed in 12 out of 19 cases of incomplete initial surgery (GTR/STR 9, removal of residual methionine PET uptake 3). The median progression-free survival among the 22 patients with grade II tumors was 57 months, with some cases showing durable progression-free survival after second-look resection. MIB-1 indices of the second-look resected tumors were lower than those of the initial tumors, and the methylation status of the MGMT gene was unchanged. Neoadjuvant chemotherapy based on molecular guidance often produces significant volume decrease of incompletely resected gliomas. Radical second-look resection is an optional advantage of upfront chemotherapy for chemosensitive gliomas compared with initial radiotherapy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Primary systemic chemotherapy, i.e. upfront chemotherapy, after surgical removal is being increasingly used as a treatment option not only for oligodendroglial tumors but also for low-grade astrocytic tumors [1–4]. Although histological distributions and the ratios of enhancing tumors vary between studies, the response rates of low-grade gliomas (LGGs) to either temozolomide or PCV (procarbazine, lomustine, vincristine) chemotherapy are reportedly between 30 and 75 % [2, 5–10]. Recent advance in translational research has provided molecular correlates with response to chemotherapy, and 1p19q codeletion, mutations in isocitrate dehydrogenase (IDH)1/2 genes, and possibly promoter methylation of the MGMT gene are predictive of chemotherapeutic response in grade II or III gliomas [7, 8, 11–14].
Neoadjuvant chemotherapy is a common treatment strategy for many cancers [15, 16]. The primary aim of this strategy is to reduce the size or extent of cancer before a main treatment, and thus decreasing the difficulty and morbidity of procedures with improved functional and/or survival outcomes. Although functional preservation is critical in glioma surgery, the neoadjuvant strategy has not been systematically used thus far.
Since February 2006, we have tailored treatment for diffuse gliomas according to molecular information. Specifically, after maximal safe removal, diffuse gliomas of grade II or III with favorable molecular status for chemosensitivity were treated with upfront chemotherapy regardless of histological diagnosis. In cases of incomplete initial surgery, a second surgery was advised after reducing tumor volume by chemotherapy. Because of invasion into eloquent areas, initial surgery was incomplete for the majority of the cases in the series, providing a unique opportunity to assess chemotherapeutic response and the utility of the neoadjuvant strategy in the consecutive cases selected by molecular profile.
Materials and methods
Study design and patient population
This is a prospective, observational study of the consecutive patients treated at Keio University Hospital between 2006 and 2013 who fulfilled the following criteria: (a) age > 18 years, (b) supratentorial tumor location, (c) histopathological diagnosis of diffuse glioma of WHO grade II or III, (d) no prior chemotherapy or radiotherapy, (e) 1p19q codeletion or promoter methylation of the MGMT gene, (f) written informed consent for molecular analysis of the tumor and for translational research approved by the Institutional Review Board at Keio University (Approval Number 20050002).
Treatment
The initial surgery was performed using intraoperative mapping technique including awake craniotomy with the aim of more than subtotal removal of abnormal high intensity area on T2-weighted or fluid-attenuated inversion recovery (FLAIR) MRI, or, as the second best, removal of increased methionine-uptake on positron emission tomography with 11C-methionine (11C-methionine PET) (Online Resource 1). For the cases in which these radical resections were considered high risk, biopsy or debulking surgery only to reduce mass effect was performed. After initial surgery, the patients with potentially chemosensitive gliomas based on molecular status were treated with upfront chemotherapy either by temozolomide, ACNU monotherapy, or PAV combination chemotherapy. Temozolomide was orally administered between days 1 and 5 in a 28-day cycle at a dose of 150–200 mg/m2/day. ACNU monotherapy was administered every 6–8 weeks at a dose of 80 mg/m2/day on day 1. PAV chemotherapy was administered every 8 weeks as follows: 70 mg/m2 intravenous nimustine on day 1, 60 mg/m2 oral procarbazine on days 8–21, and 1.4 mg/m2 vincristine (maximum dose, 2 mg/body) intravenously on days 8 and 29. The patients chose chemotherapy at their discretion after considering its potential efficacy, toxicity, and cost, according to information given by their primary neuro-oncologist (HS or TY). The upfront chemotherapy was given at progression in several patients who preferred watchful wait to immediate chemotherapy following initial resection. In cases of incomplete initial resection, second-look radical resection was intended after maximal response to chemotherapy, and grade III tumors were irradiated after chemotherapy or second-look resection. Grade II tumors, including those with focal anaplastic changes, were observed after chemotherapy or second-look resection. The present treatment strategy was approved by the Institutional Review Board at Keio University (Approval Number 20130250). Radiotherapy was administered at the time of recurrence in the majority of recurrent cases.
Neuroimaging
Imaging follow-up was performed by MRI (1.5 or 3T) every 3 cycles of temozolomide or 1–2 courses of nitrosourea-based chemotherapy. Tumor volume was assessed on T2-weighted or FLAIR MRI, and maximal chemotherapeutic response was defined in the absence of signs of improvement on next MRI. Response assessment in neuro-oncology (RANO) criteria was used with some modification, and percent change in tumor volume following chemotherapy was calculated by the sum of the product of perpendicular diameters (SPD) [17]. In cases of slowly growing non-enhancing areas that were indistinguishable from treatment-related changes, the date of progression was defined by any of the following: (1) unequivocal extension of the non-enhancing area, (2) development of enhancement, 3) clinical deterioration, or (4) increased methionine-uptake on 11C-methionine PET. Stable disease (SD) was defined as any other clinical status that does not meet the criteria for complete response (CR), partial response (PR), minor response (MR), or progressive disease (PD), lasting at least 6 months [8, 17, 18]. In selected cases, 11C-methionine PET was performed once a year or when tumor recurrence was doubted on MRI.
Histopathological and molecular-genetic examinations
Histopathological diagnoses were made according to the WHO classification [18]. The codeletion of 1p19q was assessed using comparative genomic hybridization [19, 20], promoter methylation of the MGMT gene was assessed using methylation-specific PCR [21], and mutations of IDH1/2 genes were assessed using immunohistochemistry with an anti-mutant IDH1 antibody (Dianova, Hamburg, Germany) or by sequencing [22, 23].
Statistical analysis
Progression-free survival (PFS) and overall survival (OS) were calculated from the date of initiation of upfront chemotherapy. Survival curves were estimated using the Kaplan–Meier method with JMP version 8.0.1 (SAS Institute, Cary, NC).
Results
Clinical characteristics
Among 26 patients who fulfilled the inclusion criteria, 22 had grade II tumors and 4 had grade III tumors (Online Resource 2, Table 1). Nineteen out of the 26 cases were newly diagnosed, and seven were progressive disease after wait and see following initial surgery. Presenting symptoms included seizures in 15 patients, subjective symptoms such as headache and vertigo in 3, and focal symptoms such as aphasia, hemianopsia, and apraxia in 4. The other 4 tumors were found either during medical check-ups or incidentally. Because most of the patients with gliomas referred to our hospital had invasions into eloquent areas, initial surgery was incomplete in the majority of cases, with biopsy in 11, partial removal in 8, subtotal removal (STR, > 95 %) in 4, and gross total removal (GTR, total removal of T2/FLAIR abnormality) in 3.
Molecular diagnoses and upfront chemotherapy
Clinical and molecular characteristics of the study population are presented in Table 1 (Online Resource 2). These 26 tumors were deemed chemosensitive according to the presence of 1p19q codeletion and/or MGMT promoter methylation. Seventeen tumors showed both 1p19q codeletion and MGMT methylation, 5 showed codeletion and unmethylated MGMT, 2 had codeletion with an unknown MGMT status, and 2 had no codeletion with methylated MGMT. Mutations of IDH genes were found in all 24 tumors that were successfully analyzed, including 22 with IDH1 mutations and 2 with IDH2 mutations. Upfront chemotherapy was immediately administered after initial surgery in 19 cases and at tumor progression following the observation period in 7 cases. Chemotherapy regimens comprised temozolomide in 20 patients and nitrosourea-based chemotherapy in 6. The median number of courses of temozolomide and nitorosourea-based chemotherapy was 12.5 (range, 6–40) and 3.5 (range, 3–6), respectively. One of the primary neuro-oncologists preferred long prescriptions of temozolomide. The regimens were well tolerated, although a patient (case 22) suffered from grade 4 myelosuppression after the 4th course of PAV therapy, and another patient (case 13) suffered from liver dysfunction after the 1st course of PAV therapy and was subsequently administered ACNU monotherapy. In the majority of cases, treatment was discontinued following 3–6 (for temozolomide) or 1–2 (for nitrosourea-based chemotherapy) additional courses after maximal response. Maximal tumor responses to chemotherapy were noted after a median of 6 courses of temozolomide (3–15) or 3 courses of nitrosourea-based chemotherapy (2–4; Table 1, Online Resource 2).
Chemotherapeutic response and second-look resection following tumor volume reduction
Of the 22 evaluable cases, objective response (CR + PR + MR) was noted in 14 (64 %), and no PD was observed within 6 months of chemotherapy initiation (Table 1, Online Resource 2). In 21 out of the 22 cases, percent change in tumor volume was calculated using SPD. The median SPD change following chemotherapy was −35 % (−100 to +2.1), and all but one case showed volume decrease after upfront chemotherapy, as estimated according to T2/FLAIR abnormality (Fig. 1). No clear differences in percentage tumor volume decrease were observed between non-codeleted or MGMT-unmethylated cases and cases with both codeletion and methylation (Fig. 1). Importantly, upfront chemotherapy separated T2/FLAIR abnormality from vital structures in several cases. Moreover, in some other cases, area of methionine uptake contracted to resectable size, despite that T2/FLAIR abnormality remained unresectable. These reduction in T2/FLAIR volume or methionine uptake enabled second-look radical resection, which was considered high risk at presentation or before chemotherapy. As a result, the second-look resection following tumor volume decrease was performed in 13 out of the 22 evaluable cases, i.e., in 12 out of the 19 cases with incomplete initial surgery: GTR/STR in 10 cases and removal of the area of methionine uptake in 3 [Fig. 1; Table (Online Resource 2)].
Maximum decrease of tumor volume (T2/FLAIR) from the baseline according to the sum of the products of tumor diameters (SPD) in 21 evaluable cases. The vertical axis indicates percent changes from the baseline to the minimum SPD after the beginning of upfront chemotherapy. Patients are indicated on the horizontal axis. Negative values in the vertical axis indicate tumor volume decrease from the baseline; all but one patient showed tumor volume decrease. Slashed boxes indicate cases that underwent second-look resection after upfront chemotherapy. Arrow codeleted but MGMT unmethylated case. Arrow head: MGMT methylated but non-codeleted case
Case illustration
Case 1 (Fig. 2)
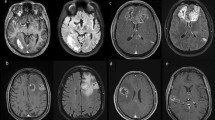
A 52-year-old man was diagnosed with a tumor in the right motor cortex. Biopsy revealed oligodendroglioma (1p19q codeletion, MGMT methylation, IDH1 mutation). Upfront temozolomide (14 courses in total) contributed to separation from the pyramidal tract and hand motor cortex, resulting in restriction of the tumor to the face motor cortex (PR, 56 % decrease by SPD). Subsequently, GTR was performed with an awake procedure, resulting in the cessation of seizures and no evidence of recurrence for 5 years and 5 months (5y 5 m) after the initiation of the upfront chemotherapy (4 years 3 months after second-look resection; KPS 100). Radiotherapy was not performed.
Serial MR images (FLAIR) of a case of second-look resection (case 1) a–c, e–g, i–k axial images. d, h Coronal images. a–c Before chemotherapy (before biopsy). The tumor was mainly located in the right precentral gyrus with invasion into the hand motor cortex and corona radiata. d–g After 12 courses of temozolomide. SPD of the tumor was decreased by 56 %. Upfront temozolomide separated the tumor from the corona radiata, and the tumor became restricted within the face area of the motor cortex. h–k After second-look removal (1 POD). Gross total removal of FLAIR abnormality was achieved
Case 7 (Fig. 3)
A 43-year-old man had initially undergone a partial removal of his right frontal tumor, and histopathological diagnosis was oligodendroglioma (1p19q codeletion, MGMT methylation, IDH1 mutation). After observation, the tumor progressed (5 years 6 months after the initial operation). Because he suffered from depression at that time, extensive resection of the frontal lobe was evaded, and 3 courses of upfront PAV were prescribed. The tumor showed a partial response (61 % decrease by SPD), and tumor invasion into the insular cortex disappeared. Subsequently, STR of the T2/FLAIR abnormality was performed with the indication that the frontal margin was incomplete. Tumor recurrence was noted at the frontal area 4 years 9 months after the initiation of PAV (2 years 8 months after second-look resection). However, tumor recurrence was not demonstrated in the insular cortex, where the T2/FLAIR abnormality was initially observed. The patient subsequently underwent right frontal lobectomy and radiotherapy, and is alive and tumor free at 7 years 5 months after the initiation of PAV (KPS 90).
Serial MR images (FLAIR) of a case of second-look resection (case 7). a–c At progression. The histopathological diagnosis of the initial resection was oligodendroglioma. A diffuse right frontal tumor is shown invading the insular cortex; d–f after 3 courses of PAV. SPD of the tumor was decreased by 61 %. No tumor invasion into the insular cortex is apparent; g–i after second-look removal (1 POD). Subtotal removal was achieved with some residual FLAIR abnormality at the anteromedial margin (arrow); j–l at recurrence (4 years and 9 months after the initiation of PAV). Despite diffuse tumor recurrence in the right frontal area, there is no recurrence in the insular area where the FLAIR abnormality was initially shown (arrowhead)
Case 26 (Fig. 4)
A 56-year-old woman had initially undergone a partial removal of her right frontal tumor, and histopathological diagnosis was diffuse astrocytoma (non-codeletion, MGMT methylation, IDH1 mutation). After observation (8years 7 months after initial operation), the tumor progressed and was treated with upfront temozolomide (15 courses in total). The tumor showed MR (30 % decrease by SPD), and partial frontal lobectomy, including the residual methionine-uptake area, was performed. The patient is free of recurrence at 5years 6 months after the initiation of upfront TMZ (4 years after second-look resection; KPS100). Radiotherapy was not performed.
Serial MR images (FLAIR) and 11C-methionine PET images of a case that underwent second-look resection (case 26) a–d at progression; the histopathological diagnosis of the initial resection was diffuse astrocytoma. After observation, a diffuse right frontal tumor invading the basal ganglia is shown; e–i after 15 courses of temozolomide. SPD of the tumor was decreased by 30 %. On the PET/CT image (e), some residual methionine uptake is shown in the right frontal lobe; j–n after second-look resection (2 months after resection). Partial frontal lobectomy, including the methionine uptake area, was performed
Histopathological diagnosis and molecular characteristics of the second-look resected tumors
The histopathological diagnosis of the tumors resected following tumor volume decrease was similar to that of the initial tumors, except for a progressive oligodendroglioma with malignant transformation (case 8; Table 2, Online Resource 2). Proliferative indices (MIB-1, Dako, Glostrup, Denmark) of the second-look resected tumors were generally lower than those of the initial tumors, except for case 8 (Table 2, Online Resource 2), suggesting that the second resection was performed under chemotherapeutic control. The promoter methylation status of the MGMT gene in the second-look resected tumors was unchanged as compared to those in the initial tumors.
Survival analysis
During the median 57-month follow-up period (12–89 months), recurrence was noted in 12 out of the 26 cases, and 4 patients died. The median PFS among the 22 patients with grade II gliomas who were treated with the neoadjuvant strategy, including those with focal anaplasia, was 57 months, and OS remained less than the median (Fig. 5).
Pattern of recurrence after second-look resection
Six out of the 13 patients who underwent second-look resection showed recurrence afterwards. In all but one case, tumor regrowth occurred around incomplete surgical margin based on T2 or FLAIR MRI (case 7; Fig. 3). In case 23, tumor dissemination on the contralateral ventricular wall was missed before upfront chemotherapy, and recurrence occurred there despite no evidence of recurrence around the surgical cavity of the second-look resection. Case 18 showed an aggressive clinical course after recurrence at the incomplete surgical margin.
Discussion
Some recent studies suggested the efficacy of neoadjuvant chemotherapy for gliomas of WHO grades II–III [24–26]. One large study reported 17 cases of LGGs that were successfully treated with a combination of neoadjuvant temozolomide and surgical removal after tumor volume reduction from among 170 recipients of upfront temozolomide [25, 26]. This study, however, reported selected cases in which temozolomide-induced tumor volume reduction enabled subsequent resection, and the original population in which the same strategy was attempted, is unknown. To the best of our knowledge, the present study is the first to report a prospective series of gliomas treated with a neoadjuvant strategy involving second-look resection after tumor volume decrease in cases of incomplete initial resection. Moreover, in contrast with the previous study of universally prescribed upfront temozolomide for LGGs [25, 26], upfront chemotherapy was applied only for cases with favorable molecular profiles in the present study. The median SPD change following chemotherapy in the present cohort was −35 % (−41 % in 19 evaluable codeleted tumors), with all but one case showing volume decrease of T2/FLAIR abnormality. Although previous studies suggest that LGGs responsive to chemotherapy may include those without favorable molecular profiles [26, 27], the volume changes from baseline in the present study were comparable to those of selected responsive cases in a previous study [26]. Thus, the molecular-guided approach can be used to select cases with a favorable response. Because the IDH mutation also predicts benefit from PCV chemotherapy in anaplastic oligodendroglial tumors [14] and temozolomide in LGGs [28], the analyses of IDH mutations may replace those of 1p19q codeletion and MGMT methylation for the selection of patients.
The median PFS in the present study was 57 months among the 22 grade II gliomas with either 1p19q codeletion or MGMT methylation, including the patients with focal anaplasia. Thus, upfront chemotherapy coupled with second-look resection for the cases with incomplete initial surgery might compare favorably with reported outcomes of chemotherapy only for 1p19q codeleted grade II tumors (e.g., median PFS: 42 months) [8]. Indeed, some of the present cases that received second-look resection (cases 1, 9, 12, 17, and 26) showed durable PFS for more than 5 years from the initiation of chemotherapy, despite initial biopsy or partial resection. In cases of incomplete initial resection, second-look resection following tumor volume decrease may improve survival outcome and offer an advantage of upfront chemotherapy over initial radiotherapy for chemosensitive LGGs. Moreover, durable PFS in several cases after second-look resections as well as the observation in case 7; recurrence was not demonstrated in the insular cortex where T2/FLAIR abnormality was initially shown (Fig. 3), may suggest that invasive glioma fronts can be reduced using chemotherapy.
Of the 13 second-look resections, removal of the residual 11C-methionine uptake was performed in three cases with extensive gliomas. Two of the three cases were progression free despite residual FLAIR abnormality (cases 5 and 26 in Table 1; Fig. 4), and radiotherapy was deferred, avoiding the potential for significant neurocognitive deterioration, especially in patients with large tumors. Adequate treatment (removal or radiotherapy) of areas with 11C-methionine PET uptake is associated with better outcome in gliomas [29–31]. Although further studies are warranted, the neoadjuvant strategy to remove residual methionine uptake following chemotherapy might delay tumor recurrence and/or malignant transformation, and could be among the therapeutic options for extensive gliomas.
Six of the 13 cases that received second-look resection showed subsequent recurrence, and all but one of these occurred at incomplete surgical margins on postoperative MRI, reiterating the importance of local tumor control. It should be noted that recurrence may occur even after second-look resection if the surgical margin is incomplete. Thus, second-look resection after chemotherapy should be indicated with discretion after consideration of anticipated resection volume and survival/functional benefit.
In conclusion, the present study demonstrates the utility of molecular-guided upfront chemotherapy with the intention of second-look resection for incomplete initial resections. Further studies are warranted, and optimization of the molecular criteria, delineation of the methionine uptake to be resected, and preoperative molecular diagnosis by imaging [32] would help improve the utility of the neoadjuvant strategy for gliomas. In clinical trials with a chemotherapy-alone arm, second-look resection after tumor shrinkage may be considered for incomplete initial resection.
References
Lassman AB, Iwamoto FM, Cloughesy TF, Aldape KD, Rivera AL, Eichler AF, Louis DN, Paleologos NA, Fisher BJ, Ashby LS, Cairncross JG, Roldan GB, Wen PY, Ligon KL, Schiff D, Robins HI, Rocque BG, Chamberlain MC, Mason WP, Weaver SA, Green RM, Kamar FG, Abrey LE, DeAngelis LM, Jhanwar SC, Rosenblum MK, Panageas KS (2011) International retrospective study of over 1000 adults with anaplastic oligodendroglial tumors. Neuro-oncology 13:649–659. doi:10.1093/neuonc/nor040
Viaccoz A, Lekoubou A, Ducray F (2012) Chemotherapy in low-grade gliomas. Curr Opin Oncol 24:694–701. doi:10.1097/CCO.0b013e328357f503
Soffietti R, Baumert BG, Bello L, von Deimling A, Duffau H, Frenay M, Grisold W, Grant R, Graus F, Hoang-Xuan K, Klein M, Melin B, Rees J, Siegal T, Smits A, Stupp R, Wick W (2010) Guidelines on management of low-grade gliomas: report of an EFNS-EANO Task Force. Eur J Neurol 17:1124–1133. doi:10.1111/j.1468-1331.2010.03151.x
Wen PY, DeAngelis LM (2007) Chemotherapy for low-grade gliomas: emerging consensus on its benefits. Neurology 68:1762–1763. doi:10.1212/01.wnl.0000266866.13748.a9
Brada M, Viviers L, Abson C, Hines F, Britton J, Ashley S, Sardell S, Traish D, Gonsalves A, Wilkins P, Westbury C (2003) Phase II study of primary temozolomide chemotherapy in patients with WHO grade II gliomas. Ann Oncol 14:1715–1721
Hoang-Xuan K, Capelle L, Kujas M, Taillibert S, Duffau H, Lejeune J, Polivka M, Criniere E, Marie Y, Mokhtari K, Carpentier AF, Laigle F, Simon JM, Cornu P, Broet P, Sanson M, Delattre JY (2004) Temozolomide as initial treatment for adults with low-grade oligodendrogliomas or oligoastrocytomas and correlation with chromosome 1p deletions. J Clin Oncol 22:3133–3138. doi:10.1200/jco.2004.10.169
Levin N, Lavon I, Zelikovitsh B, Fuchs D, Bokstein F, Fellig Y, Siegal T (2006) Progressive low-grade oligodendrogliomas: response to temozolomide and correlation between genetic profile and O6-methylguanine DNA methyltransferase protein expression. Cancer 106:1759–1765. doi:10.1002/cncr.21809
Kaloshi G, Benouaich-Amiel A, Diakite F, Taillibert S, Lejeune J, Laigle-Donadey F, Renard MA, Iraqi W, Idbaih A, Paris S, Capelle L, Duffau H, Cornu P, Simon JM, Mokhtari K, Polivka M, Omuro A, Carpentier A, Sanson M, Delattre JY, Hoang-Xuan K (2007) Temozolomide for low-grade gliomas: predictive impact of 1p/19q loss on response and outcome. Neurology 68:1831–1836. doi:10.1212/01.wnl.0000262034.26310.a2
Kesari S, Schiff D, Drappatz J, LaFrankie D, Doherty L, Macklin EA, Muzikansky A, Santagata S, Ligon KL, Norden AD, Ciampa A, Bradshaw J, Levy B, Radakovic G, Ramakrishna N, Black PM, Wen PY (2009) Phase II study of protracted daily temozolomide for low-grade gliomas in adults. Clin Cancer Res 15:330–337. doi:10.1158/1078-0432.ccr-08-0888
Peyre M, Cartalat-Carel S, Meyronet D, Ricard D, Jouvet A, Pallud J, Mokhtari K, Guyotat J, Jouanneau E, Sunyach MP, Frappaz D, Honnorat J, Ducray F (2010) Prolonged response without prolonged chemotherapy: a lesson from PCV chemotherapy in low-grade gliomas. Neuro-oncology 12:1078–1082. doi:10.1093/neuonc/noq055
Everhard S, Kaloshi G, Criniere E, Benouaich-Amiel A, Lejeune J, Marie Y, Sanson M, Kujas M, Mokhtari K, Hoang-Xuan K, Delattre JY, Thillet J (2006) MGMT methylation: a marker of response to temozolomide in low-grade gliomas. Ann Neurol 60:740–743. doi:10.1002/ana.21044
Cairncross G, Wang M, Shaw E, Jenkins R, Brachman D, Buckner J, Fink K, Souhami L, Laperriere N, Curran W, Mehta M (2013) Phase III trial of chemoradiotherapy for anaplastic oligodendroglioma: long-term results of RTOG 9402. J Clin Oncol 31:337–343. doi:10.1200/jco.2012.43.2674
van den Bent MJ, Jaeckle K, Baumert B, Wick W (2013) RTOG 9802: good wines need aging. J Clin Oncol 31:653–654. doi:10.1200/jco.2012.46.6896
Cairncross JG, Wang M, Jenkins RB, Shaw EG, Giannini C, Brachman DG, Buckner JC, Fink KL, Souhami L, Laperriere NJ, Huse JT, Mehta MP, Curran WJ Jr (2014) Benefit from procarbazine, lomustine, and vincristine in oligodendroglial tumors is associated with mutation of IDH. J Clin Oncol 32:783–790. doi:10.1200/jco.2013.49.3726
Bear HD, Anderson S, Smith RE, Geyer CE Jr, Mamounas EP, Fisher B, Brown AM, Robidoux A, Margolese R, Kahlenberg MS, Paik S, Soran A, Wickerham DL, Wolmark N (2006) Sequential preoperative or postoperative docetaxel added to preoperative doxorubicin plus cyclophosphamide for operable breast cancer: National Surgical Adjuvant Breast and Bowel Project Protocol B-27. J Clin Oncol 24:2019–2027. doi:10.1200/jco.2005.04.1665
Meeks JJ, Bellmunt J, Bochner BH, Clarke NW, Daneshmand S, Galsky MD, Hahn NM, Lerner SP, Mason M, Powles T, Sternberg CN, Sonpavde G (2012) A systematic review of neoadjuvant and adjuvant chemotherapy for muscle-invasive bladder cancer. Eur Urol 62:523–533. doi:10.1016/j.eururo.2012.05.048
van den Bent MJ, Wefel JS, Schiff D, Taphoorn MJ, Jaeckle K, Junck L, Armstrong T, Choucair A, Waldman AD, Gorlia T, Chamberlain M, Baumert BG, Vogelbaum MA, Macdonald DR, Reardon DA, Wen PY, Chang SM, Jacobs AH (2011) Response assessment in neuro-oncology (a report of the RANO group): assessment of outcome in trials of diffuse low-grade gliomas. Lancet Oncol 12:583–593. doi:10.1016/s1470-2045(11)70057-2
Louis DN, Ohgaki H, Wiestler OD, Cavenee WK (2007) WHO classification of tumours of the central nervous system. International agency for research on cancer, Lyon
Hirose Y, Aldape K, Takahashi M, Berger MS, Feuerstein BG (2001) Tissue microdissection and degenerate oligonucleotide primed-polymerase chain reaction (DOP-PCR) is an effective method to analyze genetic aberrations in invasive tumors. The Journal of molecular diagnostics : JMD 3:62–67. doi:10.1016/s1525-1578(10)60653-8
Miwa T, Hirose Y, Sasaki H, Ezaki T, Yoshida K, Kawase T (2011) Single-copy gain of chromosome 1q is a negative prognostic marker in pediatric nonependymal, nonpilocytic gliomas. Neurosurgery 68:206–212. doi:10.1227/NEU.0b013e3181fd2c2e
Ezaki T, Sasaki H, Hirose Y, Miwa T, Yoshida K, Kawase T (2011) Molecular characteristics of pediatric non-ependymal, nonpilocytic gliomas associated with resistance to temozolomide. Mol Med Rep 4:1101–1105. doi:10.3892/mmr.2011.573
Yan H, Parsons DW, Jin G, McLendon R, Rasheed BA, Yuan W, Kos I, Batinic-Haberle I, Jones S, Riggins GJ, Friedman H, Friedman A, Reardon D, Herndon J, Kinzler KW, Velculescu VE, Vogelstein B, Bigner DD (2009) IDH1 and IDH2 mutations in gliomas. N Engl J Med 360:765–773. doi:10.1056/NEJMoa0808710
Capper D, Zentgraf H, Balss J, Hartmann C, von Deimling A (2009) Monoclonal antibody specific for IDH1 R132H mutation. Acta Neuropathol 118:599–601. doi:10.1007/s00401-009-0595-z
Voloschin AD, Louis DN, Cosgrove GR, Batchelor TT (2005) Neoadjuvant temozolomide followed by complete resection of a 1p- and 19q-deleted anaplastic oligoastrocytoma: case study. Neuro-oncology 7:97–100. doi:10.1215/s1152851704000560
Blonski M, Taillandier L, Herbet G, Maldonado IL, Beauchesne P, Fabbro M, Campello C, Goze C, Rigau V, Moritz-Gasser S, Kerr C, Ruda R, Soffietti R, Bauchet L, Duffau H (2012) Combination of neoadjuvant chemotherapy followed by surgical resection as a new strategy for WHO grade II gliomas: a study of cognitive status and quality of life. J Neurooncol 106:353–366. doi:10.1007/s11060-011-0670-x
Blonski M, Pallud J, Goze C, Mandonnet E, Rigau V, Bauchet L, Fabbro M, Beauchesne P, Baron MH, Fontaine D, Peruzzi P, Darlix A, Duffau H, Taillandier L (2013) Neoadjuvant chemotherapy may optimize the extent of resection of World Health Organization grade II gliomas: a case series of 17 patients. J Neurooncol 113:267–275. doi:10.1007/s11060-013-1106-6
Jo J, Williams B, Smolkin M, Wintermark M, Shaffrey ME, Lopes MB, Schiff D (2014) Effect of neoadjuvant temozolomide upon volume reduction and resection of diffuse low-grade glioma. J Neurooncol 120:155–161. doi:10.1007/s11060-014-1538-7
Houillier C, Wang X, Kaloshi G, Mokhtari K, Guillevin R, Laffaire J, Paris S, Boisselier B, Idbaih A, Laigle-Donadey F, Hoang-Xuan K, Sanson M, Delattre JY (2010) IDH1 or IDH2 mutations predict longer survival and response to temozolomide in low-grade gliomas. Neurology 75:1560–1566. doi:10.1212/WNL.0b013e3181f96282
Pirotte BJ, Levivier M, Goldman S, Massager N, Wikler D, Dewitte O, Bruneau M, Rorive S, David P, Brotchi J (2009) Positron emission tomography-guided volumetric resection of supratentorial high-grade gliomas: a survival analysis in 66 consecutive patients. Neurosurgery 64:471–481. doi:10.1227/01.neu.0000338949.94496.85 (Discussion 481)
Tanaka Y, Nariai T, Momose T, Aoyagi M, Maehara T, Tomori T, Yoshino Y, Nagaoka T, Ishiwata K, Ishii K, Ohno K (2009) Glioma surgery using a multimodal navigation system with integrated metabolic images. J Neurosurg 110:163–172. doi:10.3171/2008.4.17569
Lee IH, Piert M, Gomez-Hassan D, Junck L, Rogers L, Hayman J, Ten Haken RK, Lawrence TS, Cao Y, Tsien C (2009) Association of 11C-methionine PET uptake with site of failure after concurrent temozolomide and radiation for primary glioblastoma multiforme. Int J Radiat Oncol Biol Phys 73:479–485. doi:10.1016/j.ijrobp.2008.04.050
Nishiyama Y, Sasaki H, Nagahisa S, Adachi K, Hayashi T, Yoshida K, Kawase T, Hattori N, Murayama K, Abe M, Hasegawa M, Hirose Y (2014) Radiological features of supratentorial gliomas are associated with their genetic aberrations. Neurosurg Rev 37:291–299. doi:10.1007/s10143-013-0515-5 (Discussion 299–300)
Acknowledgments
The present study was supported by Grant-in-Aid for Scientific Research (KAKENHI) by The Ministry of Education, Culture, Sports, Science and Technology and The Japan Society for the Promotion of Science (Grant Number 20591721, 23592141, 25462278). We greatly thank Ms. Yuko Aikawa, Ms. Naoko Tsuzaki, and Ms. Kiyomi Koide from the Department of Neurosurgery at Keio University School of Medicine for their technical assistance. We thank Dr. Yoshio Uchino from the Department of Neurosurgery at Chiba Ryogo Center for performing the 11C-methionine PET analyses. We also thank Dr. Akira Miyajima from the Department of Urology and Dr. Yoshiyuki Ishii and Dr. Maiko Takahashi from the Department of Surgery at Keio University School of Medicine for instructions in updated treatments of their specialty areas.
Conflict of interest
The authors report no conflict of interest concerning this work.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sasaki, H., Hirose, Y., Yazaki, T. et al. Upfront chemotherapy and subsequent resection for molecularly defined gliomas. J Neurooncol 124, 127–135 (2015). https://doi.org/10.1007/s11060-015-1817-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-015-1817-y