Abstract
Re-irradiation has been shown to be a meaningful option for recurrent high-grade glioma (HGG) patients. Furthermore, bevacizumab exerts certain activity in combination with chemotherapy/as monotherapy and was safely tested in combination with radiotherapy in several previous studies. To our knowledge, this is the largest cohort of patients treated with both re-irradiation and bevacizumab to date. After receiving standard radiotherapy (with or without TMZ) patients with recurrent HGG were treated with bevacizumab (10 mg/kg intravenously at d1 and d15) during re-irradiation. Median prescribed radiation dose during re-treatment was 36 Gy, conventionally fractionated. Datasets of 71 re-irradiated patients were retrospectively analyzed. Patients either received bevacizumab (N = 57) or not (N = 14; other substances (N = 4) and sole radiation (N = 10)). In patients receiving bevacizumab, both post-recurrence survival (PRS) (median 8.6 vs. 5.7 months; p = 0.003, log-rank test) and post-recurrence progression-free survival (PR-PFS, 5.6 vs. 2.5 months; p = 0.005, log-rank test; PFS-6 42.1 % for the bevacizumab group) were significantly increased which was confirmed by multivariate analysis. KPS, re-surgery, MGMT methylation status, sex, WHO grade, tumor volume and age were no significant predictors for neither PR-PFS nor PRS (univariate analysis). Re-irradiation with bevacizumab remains a feasible and highly effective treatment schedule. Studies on further salvage strategies and timing of sequential treatment options versus observation are warranted.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In patients with high-grade glioma (HGG) a high rate of local failures has been observed after multimodal therapy [1]. The addition of temozolomide (TMZ) increased local control and survival whereas the 2-year survival rate remained 27.2 % [2].
In selected patients, a second course of radiotherapy (RT) seems to be a reasonable treatment option [3–5]. Contrarily, conventional cytotoxic approaches were found to be not adequately effective [6] so that molecularly targeted drugs either alone or in combination with cytotoxic agents are currently undergoing clinical testing.
Various groups have investigated the use of bevacizumab—a humanised monoclonal antibody against VEGF-A with an already established role in metastatic colon, breast, and lung cancer [7]—for patients with recurrent HGG [8] and several trials have documented its efficacy [9–13] which may be due to the presence of pronounced hypoxia as well as high levels of tumor driven angiogenesis in HGG [14].
In glioma patients it has been proven beneficial by improving clinical symptoms reducing the extent of tumor edema. Glioma cell regression can occur independently from vascular regression, suggesting that high doses of bevacizumab have indirect anticancer cell properties in vivo [15]. VEGF-A has also been postulated to be involved in glioma tumor cell migration [16]. Bevacizumab was also tested in combination with radiotherapy and TMZ as up-front treatment of malignant glioma [17–19]. A recent prospective phase III trial (AVAglio) was designed to prove the efficacy of TMZ based radiochemotherapy with bevacizumab as first-line therapy [20].
Since the efficacy of radiation-based re-treatment is limited, it is reasonable to test in how far the addition of a radiation response modulator would impact on the efficacy of re-treatment. In this regard, one group tested the sequential use of radiosurgery and bevacizumab with favorable outcome [21]. Alternatively, Gutin et al. [22] determined the safety and activity of RT and concomitant bevacizumab—for the GBM cohort, PFS-6 was 65 %. In a previous retrospective study on 30 patients, 20 being treated with bevacizumab and 10 without bevacizumab we could show that PFS-6 within the bevacizumab-treated cohort was 72 % and survival was significantly enhanced [23].
After the publication of latter initial results we extended the use in clinical practice. During the first period, safety and feasibility were the most relevant issues—going forward with this treatment option, efficacy became a more relevant topic. Thus, the value of this approach was determined retrospectively by comparing the outcomes of patients having received a bevacizumab based re-irradiation treatment with those being re-treated without bevacizumab with a higher case number and substantially longer follow-up.
Materials and methods
Patient selection
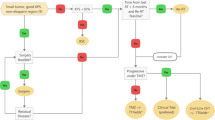
Only patients with histologically and/or FET-PET/MRI proven recurrence and macroscopic tumor (maximum diameter 5 cm with few exceptions, multifocality per se was no contraindication) were admitted to re-irradiation, the interval between first radiotherapy and re-irradiation had to be 6 months at minimum. Another precondition was the absence of meaningful alternative treatment options, e. g. complete resection by re-surgery, interstitial brachytherapy or systemic chemotherapy. In our hands, TMZ in combination with radiation in re-treatment settings has only been employed whenever initial TMZ use was limited. Historically, few patients did not receive any additional systemic therapy concurrently with the re-irradiation course.
Treatment schedule and follow-up
Before treatment, a gadolinium-enhanced brain MRI with gradient echo sequence and perfusion and/or a [18F]FET-PET. Patients treated with bevacizumab received 10 mg/kg at days 1 and 15 during radiotherapy. If applied in patients who had no previous progression after TMZ pre-treatment a dosage of 75 mg/m2 daily was chosen.
Treatment outcome was evaluated on a regular basis (every 3 months) by brain MRI [24] and/or FET-PET.
All MRI datasets were independently assessed by two experienced neuroradiologists (LE, JL) with regard to progressive disease (PD) or potential pseudoprogression according to the radiographic aspects of imaging criteria in the AVAglio protocol [25].
Adjuvant chemotherapy was prescribed on an individual base as no standard has been defined yet but was not defined as mandatory.
Radiotherapy
By analogy with Combs et al. [26]. patients received a total dose of 36 Gy in 18 fractions (2 Gy single doses) employing 3D conformal radiotherapy or IMRT if adjacent critical structures were present. Planning target volume (PTV) was defined as gross tumor volume (GTV) plus 10 mm margin at maximum. GTV included the contrast enhancing lesion in T1w + Gd MRI. To ensure reproducibility patients were immobilized with a thermoplastic mask system. Treatment planning was performed using the Oncentra® treatment planning system (OTP MasterPlan®, Nucletron, Solingen, Germany).
Toxicity evaluation
Adverse events and toxicity were determined retrospectively using the National Cancer Institute’s Common Toxicity Criteria, version 4.0 as reported before [23, 27]. Concerning adverse events of radiotherapy, focus was set on radiation necrosis as well as generalized leukoencephalopathy.
Statistics
Outcome measures of this retrospective analysis were overall survival for the total cohort from initial treatment, safety of bevacizumab given in combination with RT for recurrent HGG as well as post-recurrence and progression-free survival (PRS & PR-PFS) in patients treated with or without bevacizumab. Comparisons between groups were carried out using Fisher’s exact test or the Mann–Whitney U test. Survival analyses were based on Kaplan–Meier estimates, uni- and multivariate modelling was performed using a Cox proportional hazards analysis. For all patients, overall survival was measured from initial diagnosis, PRS from the first day of re-irradiation until death or last follow-up and progression-free survival until progressive disease or death (otherwise censored). A two-tailed p ≤ 0.05 was considered significant.
Results
Patient characteristics
Using the department’s database, 71 patients with recurrent HGG treated at our department from 5/2004 to 3/2012 were identified and retrospectively analyzed. All patient characteristics are shown in Table 1.
8.5 % of patients had a WHO grade II glioma at initial diagnosis, progressing to a secondary HGG at relapse, median age was 53 years (range 18–68 years) and median KPS was 80 (range 40–100). Thirty-four patients had a histologically proven relapse.
Before initial irradiation, 81.7 % of the patients received total or subtotal resection and 12.7 % upfront to re-irradiation (subtotal only). 78.9 % of patients were treated with TMZ during adjuvant/primary RT.
Because MGMT promoter methylation status was not systematically analyzed before 2006, it is only available in 61 out of 71 patients; a retrospective determination was not possible as no pathologic material was provided by external departments.
57 patients (14 WHO grade III, 43 WHO grade IV) received bevacizumab in addition to re-irradiation, 14 patients (5 WHO grade III, 9 WHO grade IV) were re-irradiated without bevacizumab.
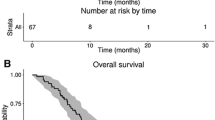
Median follow-up for all patients from the start of re-irradiation was 18 months (95 % CI 10–26 months).
Survival and toxicity data
Median overall survival (mOS) of the patient cohort was 32.5 months (95 % CI 26.7–38.3 months). As expected, patients with lower WHO grade had a longer survival history, median survival for WHO grade II patients was 196.1 months (95 % CI 53.3–338.9 months), for grade III 35.0 months (95 % CI 25.7–44.2 months; p = 0.003 compared to WHO grade II) and 27.5 months for grade IV (95 % CI 23.4–31.6 months; p = 0.16 compared to WHO grade III).
Considering now the course after re-irradiation, median post-recurrence progression-free survival (PR-PFS) was 4.9 months (95 % CI 4.0–5.8 months) and median post-recurrence survival 7.8 months, (95 % CI 6.0–9.5 months) for the entire patient population.
Re-irradiation with bevacizumab was generally well tolerated (three grade 2 toxicities, one grade 3, two grade 4 toxicity and one grade 5 toxicity) (Table 2).
Concerning the grade 5 toxicity, this patient suffered a perforation of the sigmoid colon the day after completed radiotherapy due to an existing diverticulosis with concomitant high-dose intake of steroids. Despite immediate surgical treatment, the patient died 4 days later.
Furthermore, imaging and histo-pathology revealed at maximum three cases with changes compatible with radiation necroses (see an example in Fig. 1).
Furthermore, three cases of grade 1 leukoencephalopathy, five cases of grade 2 and one case of grade 3 leukoencephalopathy were observed.
Comparison between re-irradiation with and without bevacizumab
When comparing both therapeutic subgroups (bevacizumab vs. no bevacizumab during re-irradiation), no statistically significant differences could be observed concerning WHO grade (p = 0.502), MGMT methylation status (p = 0.081), age (p = 0.131), sex (p = 0.223), tumor volume (p = 0.930), KPS (p = 0.128), re-surgery (p = 0.068) or adjuvant/salvage chemotherapy (p = 1.000)—so no relevant bias was present towards one of the subgroups (see Supplementary Table).
The results of this analysis show an association between increased PRS and PR-PFS rates and the combined treatment of re-irradiation and bevacizumab.
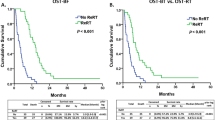
Median PR-PFS was 2.5 months in the group treated with radiotherapy alone compared to 5.6 months with re-irradiation plus bevacizumab (p = 0.005). PFS-6 was 42.1 % for re-irradiation and bevacizumab compared to re-irradiation alone with 14.3 % (Fig. 2). Median PRS after re-irradiation alone was 5.7 months, whereas median PRS after re-irradiation with additional bevacizumab increased to 8.6 months. This result was statistically significant (p = 0.003, Fig. 2).
Additionally performed early adjuvant (<8 weeks after the end of re-irradiation)/salvage systemic chemotherapy slightly increased median PRS (9.1 months with adjuvant therapy vs. 7.1 months without/with salvage therapy), however the difference between both groups was not significant (p = 0.274, log-rank test). Considering the subgroup of patients who received bevacizumab this difference seemed to be slightly more pronounced—median PRS 9.8 versus 7.5 months (p = 0.20, log-rank test).
When comparing PR-PFS for patients who did not receive adjuvant chemotherapy versus patients with adjuvant chemotherapy, no significant improvement in PR-PFS could be observed (median PR-PFS 4.2 vs. 4.4 months, p = 0.19); patients who received salvage chemotherapy had a trend towards longer PR-PFS before their relapse (median 7.9 vs. 4.2 months, p = 0.06).
Univariate and multivariate analyses
In order to define prognostic and/or predictive factors for PRS and PR-PFS univariate and multivariate testing were performed whereas for the latter significance/trend within univariate analysis and WHO grade were employed. The results are shown in Table 3.
Bevacizumab, chemotherapy post re-irradiation (adjuvant or as salvage treatment) and a total re-irradiation dose ≥36 Gy were the variables with statistically significant impact on survival in univariate testing (p = 0.003, p = 0.017 and p = 0.022, respectively). Concerning PR-PFS, only bevacizumab use remained significant (p = 0.005) whereas there was a trend for chemotherapy (p = 0.064).
Interestingly, WHO grade at relapse was non-significant regarding survival in the univariate analysis (p = 0.195), median PRS in patients with WHO IV was 9.1 months compared to 7.1 months in patients with WHO III.
Volume of the GTV (median 34.88 ml) showed a trend towards improved PRS and PR-PFS in case of a smaller volume (as continuous variable: p = 0.099, HR 0.62); patients with a volume smaller than the median volume (categorical variable) had a median PRS of 9.1 months versus 6.3 months (larger volumes), p = 0.096 whereas PR-PFS was 5.1 versus 3.8 months, p = 0.152.
Age, sex, MGMT methylation status, time interval between first percutaneous irradiation and re-irradiation as well as surgery before re-irradiation were found to be non-significant variables within the univariate analysis (p values see Table 3).
KPS was not statistically significant neither for PRS (p = 0.132), nor for PR-PFS (p = 0.104).
Concerning PRS and PR-PFS, the only factor that turned out to be a significant variable within the full multivariate Cox model as well in the univariate analysis was treatment with bevacizumab (PRS: p = 0.004, hazard ratio (HR) 3.27 (95 % CI 1.45–7.36; PR-PFS: p = 0.002, HR 3.56 (95 % CI 1.62–7.83).
Categorial GTV size turned out to be significant within multivariate analysis though being univariately not significant (PRS: p = 0.030, HR 0.49 (95 % CI 0.26–0.93); PR-PFS: p = 0.048, HR 0.56 (95 % CI 0.32–0.99)).
Applying chemotherapy in general (adjuvant/salvage) after re-irradiation showed a marginal influence in the multivariate analysis on PRS (p = 0.065), HR 1.77 (95 % CI 0.97–3.23) and KPS had significant influence on PR-PFS (p = 0.049, HR 1.96 (95 % CI 1.00–3.82), the remaining factors WHO grade at relapse, re-irradiation dose were non-significant according to PRS and PR-PFS, see hazard ratios in Table 3.
Subgroup analyses
The results for the entire patient cohort are shown together in Fig. 2 differentiated by subgroups including WHO grade at relapse, MGMT methylation status and the use of bevacizumab during re-irradiation. When performing a stratified subgroup analysis, especially patients with an MGMT promoter methylation had advanced PR-PRS and PRS rates (median PR-PFS 5.6 (bevacizumab) vs. 2.5 months (no bevacizumab); median PRS 7.8 vs. 2.8 months, p < 0.001 in both cases), for patients without MGMT methylation the difference was not significant whereas the case number was small. PRS was improved by bevacizumab in both WHO grade III and IV patients and again (p = 0.045, p = 0.039), the differences were non-significant for PR-PFS due to a small case number.
Another interesting aspect was stratification upon bevacizumab concerning tumor volume—taking the GTV cutoff of median 34.88 ml there was a strong significant difference in PRS in the relatively small group of patients without bevacizumab (7.8 vs. 2.5 months, p = 0.001) whereas this difference was not significant in patients treated with bevacizumab (9.6 vs. 7.7 months, p = 0.186). The same was true for PR-PFS: 3.3 versus 2.5 months (no bevacizumab, p = 0.027) and 5.6 versus 4.9 months (bevacizumab, p = 0.205).
Concerning the issue of patients with initial WHO II glioma (low-grade gliomas), an evaluation was performed where patients who did not have a stage III/IV histology at initial irradiation were excluded (N = 4). Results were comparable, especially bevacizumab remained a significant prognostic variable for improved PR-PFS (median 5.1 vs. 2.8 months, p = 0.011) and PRS (median 8.5 vs. 6.2 months, p = 0.012).
When focusing on GBM patients who had been treated according to the EORTC/NCIC regimen during their primary therapy (N = 45, 38 patients treated with bevacizumab additionally to re-irradiation), median PR-PFS was 5.1 months (bevacizumab) versus 3.4 months (no bevacizumab), p = 0.06. Median PRS was 9.3 months (bevacizumab) compared to 6.1 months (no bevacizumab), p = 0.27, so in principle comparable results were derived whereas significance was failed due to only seven patients in the arm without bevacizumab. When comparing the cohort (N = 30) which was examined in a previous evaluation of our group [23], there were no significant differences in patient characteristics concerning age, sex, tumor volume, time interval between previous and re-irradiation or histology but patients had a significantly better KPS (87.5 vs. 63.3 % ≥70, p = 0.02) and there was a trend towards a more frequent use of bevacizumab (87.8 vs. 70.0 %, p = 0.08). Nevertheless, PRS and PR-PFS were not significantly different (PRS: median 8.6 vs. 7.0 months, p = 0.75; PR-PFS: median 5.7 vs. 4.2 months, p = 0.52).
Discussion
For certain subgroups of recurrent HGG patients re-irradiation may be a strategy to prolong survival with acceptable toxicity. The aim of this study was to analyze whether the improvement caused by the use of concomitant bevacizumab in our previous analysis was still present within a larger patient cohort and with longer follow-up keeping in mind that this is the largest cohort to our knowledge uniformly treated with re-irradiation and bevacizumab in one center.
In this regard, the outcome of our trial compares nicely with data presented by Gutin and colleagues or those of Hundsberger et al. [28]. The results of Gutin et al. combining bevacizumab with radiation were superior than those of a matched cohort of patients who received re-irradiation only which is in line with our results [22]. But in contrast to Gutin, our updated study was monocentric and is to date the only one directly comparing survival rates of re-irradiated patients with or without bevacizumab; thus it may be regarded being less biased concerning institutional differences. A further difference was the fact that bevacizumab was applied until disease progression which was not regularly the case in our cohort. The survival rate of the combined treatment is promising and PFS-6 compares favorably with data found in the literature [3, 4, 29].
The combined treatment approach was well tolerated. Overall toxicity in our study was not higher than in the use of sole re-irradiation and bevacizumab alone or in combination with other agents in patients with HGG [30, 31].
Since all of the approaches in recurrent HGGs have a limited activity and are associated with relevant and sometimes severe toxicity we consider re-irradiation with bevacizumab to be a very effective and safe approach for those patients in whom a second course of radiotherapy is feasible. The survival data obtained for re-irradiation and bevacizumab are very promising and even in this retrospective unselected cohort, a long-term survival plateau of 13.2 %, a median PRS of 8.6 months and a 1y-PRS of 31.1 % could be achieved which is quite favorable compared to historical data of bevacizumab mono [32–34] or re-irradiation alone [29].
Nevertheless, we have to discuss several shortcomings and interesting aspects of this analysis.
Firstly, the patient cohort is very heterogeneous with a potential timing bias as some patients with initially low-grade tumors have been multimodally treated many years before re-irradiation—due to the introduction of bevacizumab and initial positive results this option became more frequently used so that results could be obtained for a more realistic patient cohort without selection bias. This explains why the historical group of patients who have only been re-irradiated is comparatively small. Though this control group was small, the power is adequate and the type II error negligible.
Secondly, our results from patients with sole re-irradiation are nominally inferior to other series employing stereotactic fractionated radiotherapy or radiosurgery. This might be due to a slightly more conservative fractionation caused by in average larger tumors and our in-house policy.
Thirdly, one further shortcoming was the fact that in ten cases the MGMT promoter methylation status was not known; in these cases no histopathologic material was available in our university and external departments did not provide relevant material. Though being one of the most important prognostic outcome markers in the primary setting, the influence of the MGMT status in re-treatment settings has not been validated and not in all cases a new status was obtained.
There does not seem to be a necessity to start adjuvant chemotherapy immediately after re-irradiation—there was no significant difference in PFS comparing patients with adjuvant chemotherapy and patients without chemotherapy—altogether adjuvant chemotherapy did not improve survival when compared to patients without maintenance chemotherapy and those who received salvage chemotherapy (to exclude the bias of salvage patients who lived long enough in order to receive salvage treatment).
Another interesting aspect was the relevance of tumor size—depending on the application of bevacizumab, this well-known prognostic factor lost its significance which leaves the speculation that potential neurotoxicity is compensated by bevacizumab.
Concerning our fractionation schedule, a more conservative approach was chosen. This is historically related to the seminal paper by Mayer and Sminia [35] who derived a cumulative NTD threshold of 100 Gy, but compared to other fractionation schedules [22, 36] 2/36 Gy is far more cautious—ex post one has to state that large tumor volumes were included (up to 158 ml) and margins were not too tight with up to 10 mm. Hypofractionation could have yielded a higher rate of radiation necroses than actually observed in our study and the high rate of marginal/distant recurrences during hypofractionated re-irradiation [37] might be a further reason not to shrink the PTV margin. A simultaneous integrated boost to the GTV would probably be most appropriate for further investigations.
In how far our approach is superior to other approaches is currently difficult to assess since randomized trials comparing different re-treatment options are lacking. At present, bevacizumab based systemic approaches resulted in PFS-6 rates from 29 % to 50.3 % [34, 38, 39]. The result of our series (42.1 %) is in this range.
In conclusion, the results of the randomized controlled trials on the use of bevacizumab concomitantly to irradiation are expected—treatment will probably become more diverse; especially in those patients who were treated with a temozolomide-based radiochemotherapy. Re-irradiation with bevacizumab may be an effective salvage option—many interesting questions on further salvage strategies have to be solved and studies are needed to find prognostic markers to identify those patients who would profit most from re-irradiation [40–42] or to optimize target volume delineation [43].
References
Niyazi M, Schnell O, Suchorska B, Schwarz SB, Ganswindt U, Geisler J, Bartenstein P, Kreth FW, Tonn JC, Eigenbrod S, Belka C, la Fougere C (2012) FET-PET assessed recurrence pattern after radio-chemotherapy in newly diagnosed patients with glioblastoma is influenced by MGMT methylation status. Radiother Oncol 104:78–82
Stupp R, Hegi ME, Mason WP, van den Bent MJ, Taphoorn MJ, Janzer RC, Ludwin SK, Allgeier A, Fisher B, Belanger K, Hau P, Brandes AA, Gijtenbeek J, Marosi C, Vecht CJ, Mokhtari K, Wesseling P, Villa S, Eisenhauer E, Gorlia T, Weller M, Lacombe D, Cairncross JG, Mirimanoff RO (2009) Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol 10:459–466
Henke G, Paulsen F, Steinbach JP, Ganswindt U, Isijanov H, Kortmann RD, Bamberg M, Belka C (2009) Hypofractionated reirradiation for recurrent malignant glioma. Strahlenther Onkol 185:113–119
Vordermark D, Kolbl O, Ruprecht K, Vince GH, Bratengeier K, Flentje M (2005) Hypofractionated stereotactic re-irradiation: treatment option in recurrent malignant glioma. BMC Cancer 5:55
Niyazi M, Siefert A, Schwarz SB, Ganswindt U, Kreth FW, Tonn JC, Belka C (2010) Therapeutic options for recurrent malignant glioma. Radiother Oncol 98:1–14
Kappelle AC, Postma TJ, Taphoorn MJ, Groeneveld GJ, van den Bent MJ, van Groeningen CJ, Zonnenberg BA, Sneeuw KC, Heimans JJ (2001) PCV chemotherapy for recurrent glioblastoma multiforme. Neurology 56:118–120
Jenab-Wolcott J, Giantonio BJ (2009) Bevacizumab: current indications and future development for management of solid tumors. Expert Opin Biol Ther 9:507–517
Beal K, Abrey LE, Gutin PH (2011) Antiangiogenic agents in the treatment of recurrent or newly diagnosed glioblastoma: analysis of single-agent and combined modality approaches. Radiat Oncol 6:2
Reardon DA, Desjardins A, Vredenburgh JJ, Gururangan S, Sampson JH, Sathornsumetee S, McLendon RE, Herndon JE 2nd, Marcello JE, Norfleet J, Friedman AH, Bigner DD, Friedman HS (2009) Metronomic chemotherapy with daily, oral etoposide plus bevacizumab for recurrent malignant glioma: a phase II study. Br J Cancer 101:1986–1994
Desjardins A, Reardon DA, Coan A, Marcello J, Herndon JE 2nd, Bailey L, Peters KB, Friedman HS, Vredenburgh JJ (2011) Bevacizumab and daily temozolomide for recurrent glioblastoma. Cancer 118:1302–1312
Reardon DA, Desjardins A, Peters K, Gururangan S, Sampson J, Rich JN, McLendon R, Herndon JE 2nd, Marcello J, Threatt S, Friedman AH, Vredenburgh JJ, Friedman HS (2011) Phase II study of metronomic chemotherapy with bevacizumab for recurrent glioblastoma after progression on bevacizumab therapy. J Neurooncol 103:371–379
Reardon DA, Desjardins A, Peters KB, Gururangan S, Sampson JH, McLendon RE, Herndon JE 2nd, Bulusu A, Threatt S, Friedman AH, Vredenburgh JJ, Friedman HS (2012) Phase II study of carboplatin, irinotecan, and bevacizumab for bevacizumab naive, recurrent glioblastoma. J Neurooncol 107:155–164
Lorgis V, Maura G, Coppa G, Hassani K, Taillandier L, Chauffert B, Apetoh L, Ladoire S, Ghiringhelli F (2012) Relation between bevacizumab dose intensity and high-grade glioma survival: a retrospective study in two large cohorts. J Neurooncol 107:351–358
Jain RK, Di Tomaso E, Duda DG, Loeffler JS, Sorensen AG, Batchelor TT (2007) Angiogenesis in brain tumours. Nat Rev Neurosci 8:610–622
von Baumgarten L, Brucker D, Tirniceru A, Kienast Y, Grau S, Burgold S, Herms J, Winkler F (2011) Bevacizumab has differential and dose-dependent effects on glioma blood vessels and tumor cells. Clin Cancer Res 17:6192–6205
Kil WJ, Tofilon PJ, Camphausen K (2012) Post-radiation increase in VEGF enhances glioma cell motility in vitro. Radiat Oncol 7:25
Lai A, Tran A, Nghiemphu PL, Pope WB, Solis OE, Selch M, Filka E, Yong WH, Mischel PS, Liau LM, Phuphanich S, Black K, Peak S, Green RM, Spier CE, Kolevska T, Polikoff J, Fehrenbacher L, Elashoff R, Cloughesy T (2010) Phase II study of bevacizumab plus temozolomide during and after radiation therapy for patients with newly diagnosed glioblastoma multiforme. J Clin Oncol 29:142–148
Narayana A, Gruber D, Kunnakkat S, Golfinos JG, Parker E, Raza S, Zagzag D, Eagan P, Gruber ML (2011) A clinical trial of bevacizumab, temozolomide, and radiation for newly diagnosed glioblastoma. J Neurosurg 116:341–345
Vredenburgh JJ, Desjardins A, Kirkpatrick JP, Reardon DA, Peters KB, Herndon JE 2nd, Marcello J, Bailey L, Threatt S, Sampson J, Friedman A, Friedman HS (2012) Addition of bevacizumab to standard radiation therapy and daily temozolomide is associated with minimal toxicity in newly diagnosed glioblastoma multiforme. Int J Radiat Oncol Biol Phys 82:58–66
Chinot OL, de La Motte Rouge T, Moore N, Zeaiter A, Das A, Phillips H, Modrusan Z, Cloughesy T (2011) AVAglio: phase 3 trial of bevacizumab plus temozolomide and radiotherapy in newly diagnosed glioblastoma multiforme. Adv Ther 28:334–340
Cuneo KC, Vredenburgh JJ, Sampson JH, Reardon DA, Desjardins A, Peters KB, Friedman HS, Willett CG, Kirkpatrick JP (2011) Safety and efficacy of stereotactic radiosurgery and adjuvant bevacizumab in patients with recurrent malignant gliomas. Int J Radiat Oncol Biol Phys 82:2018–2024
Gutin PH, Iwamoto FM, Beal K, Mohile NA, Karimi S, Hou BL, Lymberis S, Yamada Y, Chang J, Abrey LE (2009) Safety and efficacy of bevacizumab with hypofractionated stereotactic irradiation for recurrent malignant gliomas. Int J Radiat Oncol Biol Phys 75:156–163
Niyazi M, Ganswindt U, Schwarz SB, Kreth FW, Tonn JC, Geisler J, la Fougere C, Ertl L, Linn J, Siefert A, Belka C (2012) Irradiation and bevacizumab in high-grade glioma retreatment settings. Int J Radiat Oncol Biol Phys 82:67–76
Gallego PLJ, Lahutte M, Petrirena G, Reyes-Botero G, Gonzalez-Aguilar A, Houillier C, Guillevin R, Sanson M, Hoang-Xuan K, Delattre JY (2012) Response assessment in recurrent glioblastoma treated with irinotecan–bevacizumab: comparative analysis of the Macdonald, RECIST, RANO, and RECIST + F criteria. Neuro Oncol 14:667–673
Chinot OL, Macdonald DR, Abrey LE, Zahlmann G, Kerloeguen Y, Cloughesy TF (2013) Response assessment criteria for glioblastoma: practical adaptation and implementation in clinical trials of antiangiogenic therapy. Curr Neurol Neurosci Rep 13:347
Combs SE, Bischof M, Welzel T, Hof H, Oertel S, Debus J, Schulz-Ertner D (2008) Radiochemotherapy with temozolomide as re-irradiation using high precision fractionated stereotactic radiotherapy (FSRT) in patients with recurrent gliomas. J Neurooncol 89:205–210
Cohen MH, Shen YL, Keegan P, Pazdur R (2009) FDA drug approval summary: bevacizumab (Avastin) as treatment of recurrent glioblastoma multiforme. Oncologist 14:1131–1138
Hundsberger T, Brugge D, Putora PM, Weder P, Weber J, Plasswilm L (2013) Re-irradiation with and without bevacizumab as salvage therapy for recurrent or progressive high-grade gliomas. J Neurooncol 112:133–139
Combs SE, Gutwein S, Thilmann C, Huber P, Debus J, Schulz-Ertner D (2005) Stereotactically guided fractionated re-irradiation in recurrent glioblastoma multiforme. J Neurooncol 74:167–171
de Groot JF, Yung WK (2008) Bevacizumab and irinotecan in the treatment of recurrent malignant gliomas. Cancer J 14:279–285
Desjardins A, Vredenburgh JJ, Gururangan S, Peters KB, Friedman AH, Friedman H, Reardon DA (2009) A phase II study of bevacizumab plus etoposide among recurrent malignant glioma patients. Neurooncology 11:633
Raizer JJ, Grimm S, Chamberlain MC, Nicholas MK, Chandler JP, Muro K, Dubner S, Rademaker AW, Renfrow J, Bredel M (2010) A phase 2 trial of single-agent bevacizumab given in an every-3-week schedule for patients with recurrent high-grade gliomas. Cancer 116:5297–5305
Chamberlain MC, Johnston SK (2010) Salvage therapy with single agent bevacizumab for recurrent glioblastoma. J Neurooncol 96:259–269
Kreisl TN, Kim L, Moore K, Duic P, Royce C, Stroud I, Garren N, Mackey M, Butman JA, Camphausen K, Park J, Albert PS, Fine HA (2009) Phase II trial of single-agent bevacizumab followed by bevacizumab plus irinotecan at tumor progression in recurrent glioblastoma. J Clin Oncol 27:740–745
Mayer R, Sminia P (2008) Reirradiation tolerance of the human brain. Int J Radiat Oncol Biol Phys 70:1350–1360
Fogh SE, Andrews DW, Glass J, Curran W, Glass C, Champ C, Evans JJ, Hyslop T, Pequignot E, Downes B, Comber E, Maltenfort M, Dicker AP, Werner-Wasik M (2010) Hypofractionated stereotactic radiation therapy: an effective therapy for recurrent high-grade gliomas. J Clin Oncol 28:3048–3053
Shapiro LQ, Beal K, Goenka A, Karimi S, Iwamoto FM, Yamada Y, Zhang Z, Lassman AB, Abrey LE, Gutin PH (2013) Patterns of failure after concurrent bevacizumab and hypofractionated stereotactic radiation therapy for recurrent high-grade glioma. Int J Radiat Oncol Biol Phys 85:636–642
Friedman HS, Prados MD, Wen PY, Mikkelsen T, Schiff D, Abrey LE, Yung WK, Paleologos N, Nicholas MK, Jensen R, Vredenburgh J, Huang J, Zheng M, Cloughesy T (2009) Bevacizumab alone and in combination with irinotecan in recurrent glioblastoma. J Clin Oncol 27:4733–4740
Vredenburgh JJ, Desjardins A, Herndon JE 2nd, Dowell JM, Reardon DA, Quinn JA, Rich JN, Sathornsumetee S, Gururangan S, Wagner M, Bigner DD, Friedman AH, Friedman HS (2007) Phase II trial of bevacizumab and irinotecan in recurrent malignant glioma. Clin Cancer Res 13:1253–1259
Niyazi M, Jansen N, Ganswindt U, Schwarz SB, Geisler J, Schnell O, Busing K, Eigenbrod S, la Fougere C, Belka C (2012) Re-irradiation in recurrent malignant glioma: prognostic value of [(18)F]FET-PET. J Neurooncol 110:389–395
Niyazi M, Zehentmayr F, Niemoller OM, Eigenbrod S, Kretzschmar H, Schulze-Osthoff K, Tonn JC, Atkinson M, Mortl S, Belka C (2011) MiRNA expression patterns predict survival in glioblastoma. Radiat Oncol 6:153
Combs SE, Rieken S, Wick W, Abdollahi A, von Deimling A, Debus J, Hartmann C (2011) Prognostic significance of IDH-1 and MGMT in patients with glioblastoma: one step forward, and one step back? Radiat Oncol 6:115
Niyazi M, Geisler J, Siefert A, Schwarz SB, Ganswindt U, Garny S, Schnell O, Suchorska B, Kreth FW, Tonn JC, Bartenstein P, la Fougere C, Belka C (2011) FET-PET for malignant glioma treatment planning. Radiother Oncol 99:44–48
Conflict of interest
The authors declare that conflicts of interest do not exist.
Author information
Authors and Affiliations
Corresponding author
Additional information
All authors except UH are members of the CCC Neuro-Oncology, University of Munich.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Flieger, M., Ganswindt, U., Schwarz, S.B. et al. Re-irradiation and bevacizumab in recurrent high-grade glioma: an effective treatment option. J Neurooncol 117, 337–345 (2014). https://doi.org/10.1007/s11060-014-1394-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-014-1394-5